Sodium Arsenate: A Grounded Commentary
Historical Development
Factories and laboratories once saw sodium arsenate as a powerful tool. Centuries ago, arsenic compounds showed up in everything from pigments to pesticides, but sodium arsenate got its spotlight in the nineteenth century. Early chemists explored it for its high solubility and ability to work as a source of arsenic in experiments. Over decades, industry built a reputation for using sodium arsenate as an additive, a weed killer, and a wood preservative. Governments and health authorities kept a close watch as research uncovered serious health risks linked to all arsenic salts. Discovery of chronic toxicity led to stricter controls. European countries, the United States, and China all shifted regulations to protect water and food from arsenic pollution. Today, sodium arsenate mainly appears in research and narrow industrial processes, as safety and environmental pressures forced a step back from widespread use.
Product Overview
Sodium arsenate falls into the group of inorganic arsenic salts. In the lab, chemists often grab it as colorless crystals, easy to dissolve in water. Its formula, Na3AsO4, tells you the ratio: three sodium ions, one arsenate. You’ll find it sold as either the anhydrous variety or with 12 water molecules attached in the dodecahydrate form, which changes how you store it. Its main selling point is how easily it dissolves, releasing arsenate ions ready to react. Companies must issue Material Safety Data Sheets, warning users about its acute toxicity and high hazard level, and regulators flag containers for dangerous goods transport. Every shipment needs correct hazard statements, proper storage guidance, and disposal plans to limit harm to people and the environment.
Physical & Chemical Properties
Clear, odorless crystals of sodium arsenate may look harmless to the untrained eye, but experience around chemicals teaches caution. The compound melts below 900°C and easily dissolves in water, producing a basic solution (pH usually lands above 9). It doesn’t burn, but it can break down under strong heat or reaction, letting out toxic fumes. Like most ionic salts, it stands up to average air and room temperatures. The chemical’s strong oxidizing behavior causes reactions with reducing agents, and its ions like to bind with certain metals, making waste disposal a technical and legal headache. For all its simplicity on paper, sodium arsenate packs a complex risk to those working with it.
Technical Specifications & Labeling
Regulations force suppliers to declare the purity of sodium arsenate on product labels—most research-grade stock promises at least 99% purity, but industrial forms sometimes contain minor impurities such as sulfate, iron, or chloride. Labels highlight danger symbols: skull and crossbones for acute toxicity, environmental hazard icons, and signal words like “Danger” in bold print. Modern supply chains demand containers with tamper-evident seals, hazard labels that comply with GHS (Globally Harmonized System) guidelines, and clear batch numbers for tracking. Storage recommendations focus on keeping the chemical dry, cool, and away from acids or organics, as accidental mixing may trigger dangerous reactions.
Preparation Method
Simple but hazardous protocols shape the manufacture of sodium arsenate. Industry standard involves neutralizing arsenic acid with sodium carbonate or sodium hydroxide in water. The reaction produces sodium arsenate that crystallizes as the solution cools and water evaporates. Past decades saw bulk production in large reactors with open access; tightening safety standards now create demands for closed systems, improved ventilation, and strict monitoring. Most chemists agree that handling reacts with basic respect and full PPE: gloves, goggles, and face coverings at a minimum, since spills pose big risks. Scaling up for industrial quantities forces companies to account for arsenic-contaminated wastewater, which mandates on-site treatment to keep arsenic out of rivers and groundwater.
Chemical Reactions & Modifications
People working in chemistry labs use sodium arsenate as a base to build or break down more complex arsenic compounds. Its anions substitute in many double-replacement reactions. If you mix sodium arsenate with calcium salts in water, the reaction produces insoluble calcium arsenate, which made its way for years into pesticides and fungicides. Chemical modifications sometimes turn sodium arsenate into mixed salts or derivatives by reacting it with metals, acids, or reducing agents. Most real-world incidents connecting sodium arsenate to accidents or contamination stem from these secondary reactions, as the resulting arsenic species often slip into water or soil with serious biological effects. Exactly because of these reaction risks, chemical engineers and researchers promote safer alternatives and reaction protocols.
Synonyms & Product Names
Over the years, sodium arsenate picked up a basket of names: trisodium arsenate, sodium ortharsenate, EINECS 231-138-1. In commerce, its hydrate form gets listed as sodium arsenate dodecahydrate, while some technical circles just use “Na3AsO4.” Old pesticide records may call it “white arsenic” or “arsenate of soda,” but modern inventory systems need exact identifiers to avoid confusion. In every case, the CAS number (7778-43-0) sets it apart, key for import, export, and regulatory tracking across borders.
Safety & Operational Standards
Chemical safety officers look at sodium arsenate with respect and a little dread. Its acute toxicity claims the spotlight, as it can cause fatal poisoning by inhalation, ingestion, or even skin contact through wounds. Chronic exposure unrolls a catalogue of health troubles: skin lesions, neuropathy, cancer, and kidney damage lead the list, backed by hundreds of studies. Protective gear forms the frontline: gloves, face shields, chemical-resistant clothing, and tightly sealed workspaces. Air handling and filtration systems stop invisible dust or droplets from spreading. Emergency procedures include eye washes and showers close at hand, spill kits prepped for neutralization, and strict disposal rules to keep waste arsenic from recycling into the environment. OSHA in the United States caps exposure, and many plants run real-time monitoring for airborne arsenic to catch trouble before it harms staff or leaks beyond the fence line.
Application Area
Sodium arsenate’s past stretches wide—agriculture, pigment making, glass, electronics, tanning, and even in some cases as a reagent in analytical chemistry. Most of these doors have closed because of environmental and human health impacts. Medical labs or university research sometimes include sodium arsenate as a model compound to study toxicity, enzyme inhibition, or metabolic pathways in plants and animals. Environmental engineers sometimes use it to simulate arsenic contamination or test filtration systems. Some specialty manufacturing, such as glass or ceramics, makes selective use because of the unique optical or chemical properties arsenic grants. Despite these uses, most modern businesses move away from arsenic chemistry except for essential or highly controlled projects.
Research & Development
Academic journals keep turning out studies that deal with arsenate’s effect on living things or methods to clean up contamination. Environmental scientists design new absorbent materials or bacteria-based solutions aimed at soaking up arsenic from water and soil, using sodium arsenate as a benchmark. Biomedical researchers use it to probe enzyme function, since arsenate disrupts ATP synthesis and blocks many cell processes. Materials science teams explore arsenate’s role in battery or glass chemistry, even though regulations steer industry away. My own time assisting in environmental labs showed just how few researchers want to deal with arsenate, but every study adds pieces to our understanding of how to limit damage and recover from accidental release.
Toxicity Research
Sodium arsenate emerges as a textbook example in toxicology handbooks. Animal and cell studies document its impact on the nervous system, cardiovascular function, and DNA repair. Large population studies in Bangladesh and West Bengal, where arsenic-laced water remains a tragedy, link even trace exposures to skin disease and cancer. Laboratory safety manuals feature sodium arsenate’s ability to pass through membranes and cross cell boundaries, making it a hazard at nearly every point of the process. Public health guidelines restrict total arsenic to barely-detectable levels in food and water. Toxicity drives research for new filtration, remediation, and biomarker tools, since no one wants a repeat of old arsenic tragedies. First aid guides for scientists warn that ingestion or inhalation needs immediate, aggressive emergency care. No simple antidotes—the main treatment focuses on getting the poison out fast and preventing damage while monitoring for long-term effects.
Future Prospects
Across industries and countries, policy pushes for minimization and replacement of sodium arsenate. As green chemistry expands, industry looks to non-arsenic alternatives for everything from wood preservation to herbicides, and research funds tend to follow this direction. The only growing field for sodium arsenate rests in scientific research and environmental remediation, where understanding how arsenic behaves in soil, water, and living tissues drives new cleanup technology. In places with historic arsenic contamination, researchers and site managers depend on controlled use for monitoring and treatment design. As analytical techniques get more sensitive, regulators ratchet limits ever lower, and most manufacturers see less commercial value for sodium arsenate beyond the research bench. Scientists and companies that care about sustainability choose to avoid it whenever possible, as future generations benefit more from removing old toxins than from adding new risks.
What is Sodium Arsenate used for?
What People Do With Sodium Arsenate
Sodium arsenate has played a part in science and industry for decades, popping up in places many folks don’t realize. In the lab, researchers use this compound to break down proteins, run chemical reactions, and study cell signals. It’s sometimes chosen for its unique reactivity—especially when scientists look for specific chemical changes that other substances won’t produce. This might sound a bit abstract, but getting a new treatment for an illness or figuring out a disease at the molecular level can depend on compounds like this.
Away from the microscope, farmers and wood processors have reached for sodium arsenate as a pesticide and preservative. For a long time, it showed up in formulations meant to protect crops from pests or keep wood free from rot and bugs. There’s a reason industries used arsenic-based blends: they pack a toxic punch that other bugs and fungi can’t shrug off. I remember visiting a rural plant long ago and seeing workers shape lumber treated with solutions that included sodium arsenate. It kept fence posts in the ground far longer than untreated wood.
Growing Concerns Over Safety
The same toxicity that makes sodium arsenate useful also turns it into a public health problem. Exposure links to skin lesions, nerve issues, and increased cancer risk—not just in workers, but in communities nearby. Environmental runoff has left its footprint in some groundwater sources in farming towns. Reports from the EPA and World Health Organization pull no punches: arsenic compounds stick around, and even small quantities can pose real dangers to people and wildlife.
Over time, regulations have tightened. Today, many uses of sodium arsenate in pesticides and wood preservation have been phased out or heavily restricted in countries like the United States, Canada, and those within the European Union. Research institutions have standards set by OSHA and similar agencies to keep scientists safe, from glove protocols to specialized fume hoods.
Lessons Learned and Safer Replacements
Watching the fallout from sodium arsenate in agriculture and industry has taught us a few things. We don’t always see the risks until a lot of folks are exposed. Once those risks show up in medical records, it can take years to undo the harm and even longer to clean up a contaminated site. In my own experience working with chemical safety teams, nothing beats a culture of vigilance. Teams spend hours on training, labeling, and double-checking procedures to make sure nobody breathes in or touches a material they shouldn’t.
Researchers now look for safer chemicals with similar functions. Borates and copper-based formulas replace arsenic in a lot of wood preservatives. Synthetic pesticides get designed to break down more easily and cause less trouble for the environment. It’s never a perfect process, but keeping people safe, keeping food safe, and keeping groundwater clean all push science forward.
Looking Ahead
The history of sodium arsenate pushes us to ask tougher questions before greenlighting industrial chemicals. I’ve seen the change happen firsthand, from lost trust to community activism driving better standards. Today, agencies expect visible proof of safety long before new products hit the shelves. We owe it to people who live near farms, work in labs, and raise families on properties where these chemicals once lingered.
Is Sodium Arsenate toxic or hazardous?
Understanding Sodium Arsenate's Risk
Sodium arsenate lands on the list of substances that raise alarms among chemists and industry workers alike. This compound comes packed with toxic potential, leading researchers, regulators, and health workers to tread carefully around it. Mixing sodium and arsenic, scientists created something with a long, checkered past in everything from pesticides to labs, but the hazards it brings overshadow any benefit.
Personal Encounters in Scientific Settings
During a stint in an old college chemistry lab, I ran across a shelf labeled "Arsenates: Poison." If anything shouts "do not touch," it’s that word. Stories get handed down about how even a careless touch, a puff of dust, a forgotten glove can cause damage. The protocol was clear: double gloves, fume hoods, every accident logged. That kind of respect comes from real incidents where people faced headaches, nausea, nerve damage, even organ failure after exposure. Years later, the same fear persists in academic and industrial spaces. People react strongly to sodium arsenate for a reason—science and stories both warn against taking this powder lightly.
Why Public Health Officials Worry
The World Health Organization lists arsenic compounds among the most toxic elements. Sodium arsenate is water-soluble, creeping into groundwater if handled poorly. Many small towns in the American West still feel the impact of arsenic pollution from mining days. Residents deal with higher cancer rates, kidney issues, and skin disorders. Each case became another lesson about why strict rules and safety measures mean more than red tape. The Environmental Protection Agency keeps sodium arsenate on its hazardous substance list and limits the amount allowed in drinking water to barely a trace. Overexposure isn’t hypothetical—it’s a repeated health crisis across continents.
Hazardous by All Measures
Sodium arsenate carries hazard symbols for a reason. Its dust settles easily on clothes, tools, or skin. Ingesting as little as a few milligrams can poison a person. Inhaling or absorbing it can cause cumulative harm, with arsenic building up in the system, damaging nerves and organs. There’s no antidote for arsenic poisoning, just time and medical intervention. Hospital rooms have seen workers and accident victims fighting for recovery after just one careless incident. The only way to use it safely is under tight controls, in spaces where every spill gets treated as a potential disaster.
Solutions and Safer Practices
Communities and industries are stepping away from sodium arsenate wherever possible. Farmers now choose pesticides and herbicides without arsenic compounds, keeping food and soil safer. Labs limit access to sodium arsenate, training staff in accident response and protective methods. Mining and wastewater cleanup projects now put millions towards removing arsenic from water and soil, repairing decades of damage. These measures come from a shared understanding: the risk isn’t an abstraction—it’s a real threat to anyone exposed.
Practical Advice for Everyone
Everyday folks may never see a jar labeled sodium arsenate, but the risk can still show up in contaminated well water or old industrial lands. Get water tested by a certified lab, especially in areas with a mining legacy. Follow public warnings about ground contamination—those signs don’t come from guesswork. As for industry and science, staying up to date on regulations and using best practices saves lives every year. Everyone benefits when the most hazardous chemicals stay locked away, handled by those trained for the job.
What are the storage conditions for Sodium Arsenate?
Understanding Sodium Arsenate and Why It Demands Careful Storage
If you’ve ever spent time in a laboratory or worked near industrial chemicals, you know most substances don’t play around. Sodium arsenate ranks high on that list, bringing serious toxicity and environmental risk. People rely on careful handling because exposure to this white, water-soluble powder does more than just sting your nose. It binds to human tissues, disrupts cell function, and even increases the risk of cancers. There’s a good reason chemists and technicians treat its storage with respect.
Why Modern Facilities Choose Isolation and Precautions
My years handling toxic reagents taught me that mistakes creep in fastest when someone stores hazardous substances like sodium arsenate as if they’re just another bag of salt. It doesn’t take much: a leaky cap or a shelf too close to heat, and pretty quickly you’re looking at a potential health disaster. That’s why smart setups ensure sodium arsenate sits in tightly sealed, corrosion-resistant containers — glass, HDPE, or steel with secure closures — and always labeled with clear warnings. No exceptions.
Safe Storage: The Details That Matter
Lab managers I’ve worked with insist sodium arsenate stays locked away in a well-ventilated, cool, dry area. Humidity and heat trigger chemical changes and can even promote the release of toxic dust or vapors. Forgetting to control these factors could easily create an unsafe environment for workers and nearby communities. Fire-resistant cabinets make a huge difference. Unlike less hazardous compounds, storing it next to acids, reducing agents, or organic materials could mean violent reactions. Segregated storage prevents accidents.
Accidental Exposure: Keeping the Team Informed
Some of the worst stories in chemical safety result from people not knowing what’s actually in a container. I once saw a well-meaning janitor mishandle a poorly labeled bottle, leading to a scary, all-hands-on-deck cleanup. Everyone in the lab or facility should understand the risks and know how to respond if a spill occurs. Consistent training and easy access to safety data sheets make a difference. Nobody wants to fumble for answers after a dangerous exposure.
Protection Beyond Physical Storage
Sodium arsenate doesn’t stay put if a facility has poor engineering controls. Good airflow, HEPA filtration, and well-maintained fume hoods add a serious line of defense. I’ve watched a single equipment failure leave a storage room hot and stagnant — a recipe for vapor and dust spreading far beyond the confined space. Building managers who check their systems regularly prove that preparation beats luck any day.
Disposal and Community Responsibility
Getting rid of sodium arsenate brings up another important factor: environmental safety. Dumping it down the drain or with ordinary trash poisons water, soil, and can creep up the food chain. Certified hazardous waste handlers follow strict protocols. My community saw an uptick in arsenic levels in groundwater once, traced back to improper chemical disposal a few miles up the road. Those lessons stick with you. Responsible storage goes hand-in-hand with responsible disposal.
Routine Inspections: Nipping Problems Early
Spot checks, logs, and regular inspection schedules keep small mistakes from snowballing. In my experience, the most reliable teams use checklists, keep storage spaces uncluttered, and track inventory by the book. No mystery containers, no expired chemicals forgotten in the back. That discipline helps everyone go home safe, every day, with peace of mind about what’s staying behind in the storage room.
How should Sodium Arsenate be handled safely?
Understanding the Real Risks
Sodium arsenate creates big safety concerns. If you spend any time in a laboratory or deal with chemicals in an industrial setting, you know some materials force you to slow down, take stock, and double-check every step. This compound sits right up there with the ones that shouldn’t be underestimated.
It’s a known carcinogen, with a very bad reputation for causing both acute and chronic health problems. The stuff doesn’t just irritate—it can damage organs and cause cancer with repeated exposure. Even brief contact burns skin, stings the eyes, and can cause breathing trouble if inhaled or ingested. Stories from old workplaces—where gloves were skipped and spills cleaned without thought—linger as somber reminders of how much harm comes from cutting corners.
The Importance of Prevention Over Cure
Nothing replaces strong prevention in handling this chemical. Half-measures do not cut it. Relying on an extra layer of PPE isn’t enough without strong training culture. Take, for instance, loading sodium arsenate for dissolution; wearing gloves and goggles seems obvious, yet I’ve seen folks skip proper face shields or forget to check for leaks in fume hoods. This is where trouble starts. One slip, one unexpected splash, and contamination spreads. Eating or drinking near your workspace gives arsenic compounds a one-way ticket into your system. The habits you set matter as much as the technical steps.
Ventilation and Containment
A fume hood always makes the difference. Open bench work turns any contamination into a group problem. Hoods suck away hazardous particles, protecting anyone in the area—no hoping the draft carries away a stray dust, no relying on luck. Closed systems matter even more during transfers. I’ve watched seasoned chemists pause, refuse to move forward until the flow is confirmed strong, because they know what’s at stake.
Physical containment also helps—secure, properly labeled containers stop accidents before they start. The labels should scream hazard, not whisper it. Shared spaces demand clear warnings and dated records, so anyone who picks up a container knows exactly what it holds and how old it is.
Personal Gear: More than Just Gloves
No one likes wearing stifling protective gear, especially in the summer or during long shifts. Still, that full-coverage face shield, thick nitrile gloves, chemical-resistant apron, and closed shoes must come out. Laboratory coats need regular cleaning or disposal, since arsenic residues linger on fibers. It isn’t paranoia—it’s accepting the way skin absorbs and lungs inhale.
Clear Emergency Response Helps Everyone
Spills demand immediate action. Anyone working with sodium arsenate should know how to grab a spill kit and which steps to take. Cover the spill with an appropriate absorbent; sweep gently without stirring up dust. Bag waste double and label every bag. This isn’t overkill—it’s respect for a poison that hides in the smallest traces.
Healthcare professionals must have current information sheets available. Exposures must get reported straight away, without fear of blame. Cultivating a speak-up culture in the lab keeps everyone safer.
Disposal—Don’t Pass It Down the Line
Some cut corners by dumping down the drain. That should never happen. Waste contractors, licensed for hazardous materials, know how to neutralize and destroy arsenic effectively. Diluting and dispersing only shifts the burden to water supplies, hurting people and wildlife. Holding yourself, your team, and your workplace to this standard protects both your immediate circle and the wider community.
Staying safe with sodium arsenate starts with knowledge and discipline—earned through experience and enforced by an unrelenting commitment to do things right, every single time.
What is the chemical formula and molar mass of Sodium Arsenate?
Not Just a Name: Behind Sodium Arsenate
In the long list of industrial and laboratory chemicals, sodium arsenate grabs attention for reasons that stretch far beyond its name. This compound walks a fine line between utility and caution. The chemical formula, Na3AsO4, tells a story about its structure: three sodium atoms team up with one arsenic atom and four oxygen atoms. The simplicity of the formula doesn’t reflect the complexity and care needed in real use.
Breaking Down the Numbers
Anyone familiar with chemistry knows that molar mass sits at the crossroads where math meets matter. Sodium arsenate draws its weight from each of its elements:
- Sodium (Na): 22.99 g/mol × 3 = 68.97 g/mol
- Arsenic (As): 74.92 g/mol
- Oxygen (O): 16.00 g/mol × 4 = 64.00 g/mol
Altogether, sodium arsenate has a molar mass of 207.89 g/mol. To an analytical chemist, that number means precision and accuracy in weighing up reagents. To someone less familiar with lab work, it’s the kind of detail that makes formulas feel personal and grounded in reality. These molar values shape every calculation in research, manufacturing, and medical testing involving sodium arsenate.
Deep Roots in Industry and Environment
Sodium arsenate has played roles as a wood preservative, pesticide ingredient, and laboratory reagent. Its strong oxidizing properties come into play during chemical synthesis, helping to shape molecules that appear in countless products. The consistency of its molar mass allows for predictable outcomes in those processes. But there’s a dark side to this convenience. Arsenic does not forgive carelessness. The same property that makes sodium arsenate valuable in chemical reactions can threaten ecosystems and health. Arsenic compounds remain highly toxic, especially when mishandled. I have seen firsthand how even a small miscalculation in handling arsenic-based materials can spiral. Regulations grew tighter for a reason; years of environmental and health effects forced change and vigilance.
Facing the Challenges
No one should minimize the risks found in sodium arsenate. Molar mass calculations sound numerical, though each set of numbers points to impact in people’s lives. Water contamination with arsenic—whether from old wood treatments or chemical plant wastes—still hits communities worldwide. Testing and remediation get fueled by chemists who rely on these solid calculations. Understanding its chemical formula gives a snapshot of the risk. Knowing the exact molar mass makes it possible to control dosage, exposure, and disposal. Working in labs taught me that safety doesn’t mean fear; it means respecting details.
Toward Safer, Smarter Uses
Everyday reliance on sodium arsenate calls for better protocols and transparent communication. Industries that still use this compound must commit to upgrading handling practices. Community-level education about arsenic sources helps bring awareness to families at risk. Researchers have mapped out several ways to neutralize or sequester arsenic from water and soil. It’s up to decision-makers to invest in those solutions.
In the last decade, I’ve watched as improved labeling, better storage, and clear molar mass documentation cut down on workplace accidents. This tells me respect for the science behind the formula—Na3AsO4 and its carefully measured 207.89 g/mol—pays off. Sodium arsenate’s story is about more than chemistry; it reflects how well we care for each other in the details.
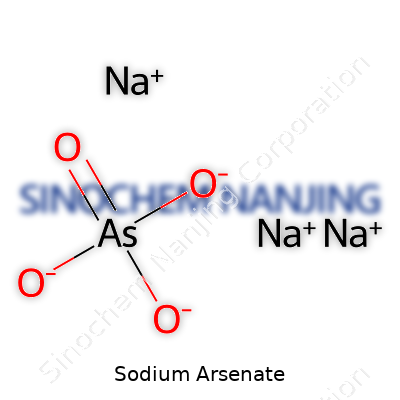
| Names | |
| Preferred IUPAC name | sodium arsenate |
| Other names |
Arsenic sodium salt Disodium arsenate Sodium arsenate dibasic Sodium orthoarsenate |
| Pronunciation | /ˌsəʊdiəm ɑːˈseniːt/ |
| Identifiers | |
| CAS Number | 7631-89-2 |
| Beilstein Reference | 358990 |
| ChEBI | CHEBI:75263 |
| ChEMBL | CHEMBL1201111 |
| ChemSpider | 21171395 |
| DrugBank | DB01378 |
| ECHA InfoCard | 035bf65a-c31d-4410-ac87-b65f387c1ee1 |
| EC Number | 231-993-5 |
| Gmelin Reference | 778 |
| KEGG | C01356 |
| MeSH | D012512 |
| PubChem CID | 24243 |
| RTECS number | CG3325000 |
| UNII | 7U4F883D3T |
| UN number | UN #1685 |
| Properties | |
| Chemical formula | Na3AsO4 |
| Molar mass | 311.888 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 2.87 g/cm³ |
| Solubility in water | Highly soluble |
| log P | -4.26 |
| Vapor pressure | Negligible |
| Acidity (pKa) | ~11.5 |
| Basicity (pKb) | 11.54 |
| Magnetic susceptibility (χ) | +40.0e-6 cm³/mol |
| Refractive index (nD) | 1.333 |
| Viscosity | 60 cP (20°C) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 207.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1278.4 kJ/mol |
| Pharmacology | |
| ATC code | V03AB05 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; may cause cancer; causes damage to organs through prolonged or repeated exposure; environmental hazard. |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H300 + H330: Fatal if swallowed or if inhaled. H350: May cause cancer. H400: Very toxic to aquatic life. |
| Precautionary statements | P264, P270, P273, P280, P301+P310, P302+P352, P305+P351+P338, P308+P313, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Lethal dose or concentration | LD50 oral rat 41 mg/kg |
| LD50 (median dose) | LD50 (median dose): 40 mg/kg (oral, rat) |
| NIOSH | TT2325000 |
| PEL (Permissible) | 0.01 mg/m3 |
| REL (Recommended) | 34 mg/kg bw |
| IDLH (Immediate danger) | 5 mg/m3 |
| Related compounds | |
| Related compounds |
Sodium arsenite Disodium hydrogen arsenate Potassium arsenate Arsenic acid |

