Sodium 4-Chloro-2-Nitrophenoxide: A Reflection on Its Journey and Role in Science
The Roots and the Rise of Sodium 4-Chloro-2-Nitrophenoxide
Sodium 4-chloro-2-nitrophenoxide didn’t pop up out of nowhere. Its journey ties into a larger tradition of fine-tuning aromatic compounds for chemistry, stretching back a century or more. As the chemical industry grew, researchers needed sharper, more targeted ways to build and modify molecules. Scientists learned that tweaking aromatic rings—adding groups like nitro or chloro—could radically change how chemicals behave. In time, sodium 4-chloro-2-nitrophenoxide found its way into labs looking for new routes toward dyes, pharmaceuticals, and other materials. My work in organic synthesis has shown again and again how careful substitution on aromatics shapes both reactivity and the fortunes of fledgling molecules—this one sits front and center in that story.
Understanding the Compound: Tangible Properties and Identity
To an experienced chemist, sodium 4-chloro-2-nitrophenoxide stands out for its distinctive blend of electron-withdrawing groups, both nitro and chloro, which draw the eye and the reactivity. In the jar, it appears as a pale solid, usually a powder, thanks to the combined ionic and aromatic character. The bitter, sometimes sharp, odor reminds you quickly this isn’t table salt. The chemical feels robust yet reactive—soluble in water, bristling with possibilities for further reaction. Chemists often refer to it by more than one name, sometimes as sodium p-chloro-o-nitrophenoxide or just sodium nitro-chlorophenoxide, depending on the context and the quirks of local literature. Regional journals sometimes stick with their favored synonyms and it never hurts to double-check for clarity when sharing research.
Chemistry at Work: Technical Standards and Labeling
Bottles of sodium 4-chloro-2-nitrophenoxide carry sharp, strict labels for good reason. Its tech sheet rarely skips over the details—molecular weight, appearance, water solubility, purity, and specific safety warnings leap off the page. I’ve worked in labs where a slip-up with the label could mean hours of confusion or a ruined experiment, so accuracy in technical specs matters. Reliable suppliers typically guarantee high purity, traceable to spectra or chromatograms to back up the figures. This transparency becomes especially crucial in tightly regulated environments: nowhere does full disclosure feel more urgent than in active pharmaceutical ingredient (API) manufacturing or university labs training young chemists.
Getting Ready: How Sodium 4-Chloro-2-Nitrophenoxide Comes Together
Preparing this compound feels familiar to anyone who’s made substituted phenoxides before. Usually, it starts with 4-chloro-2-nitrophenol—a mouthful, but a solid starting point—reacted with a sodium base. I remember the mix of anticipation and routine the first time I tried it, watching the aromatic phenol fizz and change as the sodium salt forms. The color deepens, a sign that the electron-poor nitro group and chlorine are sculpting the electron cloud. Proper temperature control and careful stirring keep side reactions down. Over the years, process chemists have optimized these steps for purity and yield, often swapping out solvents or adjusting pH based on the scale of the preparation.
Chemical Reactions and Useful Modifications
In the world of organic chemistry, sodium 4-chloro-2-nitrophenoxide works like a gateway. Its phenoxide oxygen makes it a handy nucleophile. It’s often used in nucleophilic aromatic substitution, especially when researchers need to introduce a nitro- and chloro-substituted ring system. Reactions with alkyl halides or sulfonate esters lead to a host of useful derivatives—ethers that anchor themselves in dye chemistry, or intermediates for agrochemical production. The combination of nitro and chloro keeps the ring receptive to further tweaks, making this molecule something of a favorite for method development. In my own experience, a well-chosen starting material like this one can save a lot of time, sparing the researcher days of troubleshooting and purification headaches.
Safety and Day-to-Day Handling
No one in the lab treats sodium 4-chloro-2-nitrophenoxide lightly. The nitro group, particularly, means extra caution: careful ventilation, gloves, and dust control are non-negotiable. Regulatory agencies lay out clear guidelines, and following them protects not only individual chemists but the whole operation from unexpected accidents. Reports tie some risks to irritation of skin and respiratory tract, so working with this material in a fume hood is pretty much standard. Storage deserves attention too; the solid should live in a cool, dry spot, away from acids or reducing agents that might trigger unwanted reactions. I’ve never forgotten a close call caused by ignoring proper segregation—with this compound, respect for safety rules always earns its keep.
Practical Value: Where the Compound Makes Its Mark
Sodium 4-chloro-2-nitrophenoxide finds itself right in the heart of innovation across several industries. Dye manufacture, for instance, leans on its chemistry to introduce fastness and unique color profiles. Pharmaceutics looks toward it not as a final product but as a robust intermediate, shaping more complex molecules destined for disease treatment. In agricultural research, this molecule underpins studies searching for new pesticides or herbicides, as its structure lends itself to clever modifications that change biological activity. Having collaborated with folks in both academic and industrial settings, I’ve seen firsthand how a versatile intermediate like this one gives project teams a fighting chance to push past bottlenecks and explore novel applications.
R&D and the Push for Better Chemistry
Chemical research isn’t about standing still. Scientists aim to revisit synthesis methods, reduce waste, and make production friendlier to the environment. Green chemistry principles push us to minimize harsh reagents or energy-intensive steps, which matters for both the lab bench and the factory floor. Teams continue to test new catalysts and optimize reaction conditions—always seeking to trim costs while shrinking environmental impact. Articles in recent years showcase alternative solvents, microwave-assisted syntheses, or biocatalytic steps for aromatic substitutions connected to molecules just like sodium 4-chloro-2-nitrophenoxide. Over coffee with colleagues, I often hear about the frustration of balancing chemical efficiency against the pressure to go greener. Yet these frustrations pave the way for creative problem-solving and, occasionally, a breakthrough that changes the workflow for everyone involved.
Toxicity and Current Research Challenges
Toxicological concerns hover over any aromatic nitro compound, and sodium 4-chloro-2-nitrophenoxide is no exception. Animal studies in the literature flag risks connected to both acute and chronic exposure. The nitro group, again, usually draws the most scrutiny because it can lead to metabolites that damage cells or interfere with normal biological processes. Careful handling, regular training, and tight controls on exposure have become the norm everywhere from university labs to manufacturing plants. Some current research digs into the long-term effects of exposure at low levels, as the line between harmless and hazardous isn’t always clear-cut. I’ve found that ongoing investment in better analytical techniques has improved our understanding, but quick, easy solutions aren’t likely on the toxicity front. Toxicology data shapes regulatory limits and storage protocols, guiding workers and researchers as they design new processes for old and new chemicals alike.
The Road Ahead: Looking Toward Future Uses
What lies ahead for sodium 4-chloro-2-nitrophenoxide depends on chemistry’s evolving needs. Interest in high-throughput and automated chemistry means standardized intermediates like this one will probably play an even bigger role. As the demand for greener reactions grows, every synthetic step—from phenol to phenoxide—gets a fresh look to cut down on byproducts and boost overall atom economy. I’ve spoken with scientists mapping out plans for new materials and sensor technologies, many of whom find this molecule an appealing building block. Future applications might reach beyond the familiar grounds of dyes and medicines into completely new markets, especially as researchers mix traditional organic synthesis with fields like materials science or environmental monitoring. My own optimism springs from seeing how centuries-old compounds, given a chance and a little reimagining, can anchor tomorrow’s innovations as surely as they did yesterday’s breakthroughs.
What is Sodium 4-Chloro-2-Nitrophenoxide used for?
A Closer Look at This Niche Compound
Sodium 4-chloro-2-nitrophenoxide shows up in the kind of places most people never think to look. In my experience in laboratory work, chemicals like this serve as building blocks in bigger stories that never hit the front page. Chemists see it as one of those small but mighty pieces in the ongoing effort to create dyes, pharmaceuticals, and specialized chemical products that show up in products we all use.
Color Chemistry’s Backstage Player
Any industrial chemist recognizes that the creation of pigments depends on a whole family of modified phenols and nitroderivatives. Sodium 4-chloro-2-nitrophenoxide, with its distinctive color and reactive sites, often acts as an intermediate in producing organic dyes. Synthetic dye makers rely on it to tweak colorfastness or hue, often by making tiny changes to the molecule before moving to the next production stage. If you wear vividly colored clothes or work with printed plastics, there’s a decent chance this compound—or at least its chemical cousins—played a backstage role somewhere along the way.
Pharma Sees a Chemical Workhorse
In pharmaceutical synthesis, fine chemicals like sodium 4-chloro-2-nitrophenoxide can pop up as intermediates. The production lines for antiseptics, pain relief medicines, or even some diagnostic agents often require intermediates with nitro and halogen groups. While the molecule itself rarely makes it into the final pill or ointment, the unique combination of nitro and chloro groups helps create the reaction conditions necessary to add or remove other groups cleanly. The reliability of these reactions means less waste and more consistent quality—a big deal from both an environmental and cost perspective.
Challenges and Safety Lessons
Any hands-on chemist learns caution with nitro and halogen compounds. In the lab, strict safety steps come from real lessons learned—not just paperwork. Gloves, eye protection, and fume hoods aren't just for show. Handling sodium 4-chloro-2-nitrophenoxide demands airtight labeling, careful storage (especially to avoid mixing with incompatible chemicals), and respect for potential irritant effects. Some of the best work I’ve seen in labs came from teams who treated “minor” intermediates with as much attention as well-known toxins.
Responsible Manufacturing and the Bigger Picture
Manufacturers who use sodium 4-chloro-2-nitrophenoxide can’t just focus on yield or price. Oversight from regulatory bodies like the EPA and Europe’s REACH means anyone in the supply chain needs to provide thorough toxicity data, waste management plans, and track how every batch is handled. Purpose matters: making a life-saving drug or a permanent dye justifies the care and oversight. But too often, shortcuts in disposal or documentation can turn a routine chemical run into an environmental headache. I’ve seen small changes in waste processing or in documentation practices transform a chemical plant’s record almost overnight—from hazardous incidents to clean audits.
Practical Solutions for Moving Forward
Better training stands out as the most practical fix. Bringing in real-world case studies—accidents avoided by good labeling, environmental wins tied to efficient reuse—turns dry safety sessions into habits people actually follow. Updating storage protocols and running spot inspections keep everyone on their toes. Technological investments help too: improved ventilation, automated mixing, and digital tracking cut down on human error. These details may not grab headlines, but they make all the difference to those working with or near these substances.
What are the safety precautions when handling Sodium 4-Chloro-2-Nitrophenoxide?
Respecting the Risks
Sodium 4-chloro-2-nitrophenoxide is more than a tricky name to pronounce. With its bright yellow powder, this chemical doesn’t exactly look menacing, but don’t fall for appearances. In labs, I’ve watched colleagues drop their guard after handling routine reagents, only for unexpected burns or fumes to remind everyone that a single slip brings consequences. Any compound carrying both nitro and chloro groups, paired with sodium, delivers serious punch. Even low levels of contact can irritate skin, and inhaling dust will set off a coughing fit in seconds.
Gear Saves Skin
Talk to anyone who’s spilled this stuff without gloves, and you’ll hear some version of “never again.” Nitrile gloves stand up to most organics here. I always throw on a fresh pair before opening containers. Safety glasses are non-negotiable; fine yellow dust flies everywhere while measuring. Lab coats make cleanup way easier if powder escapes the weighing boat. It feels like overkill until you notice yellow stains on your wrists, knowing some of that seeped through the wrong glove. Closed-toe shoes keep feet out of harm’s way. Once, someone in a shared space worked in sandals—a single dropped spatula left their foot tingling for the rest of the day.
Air Quality Matters
Nobody wants to breathe chemical dust. Sodium 4-chloro-2-nitrophenoxide makes no exception. Weigh it only in a properly ventilated hood, never out in the open room. Years in the lab taught me to respect the quiet hum and sash of a well-kept fume hood. The risk grows when people get casual and prop open the sash. Keeping heads outside the hood and minimizing swirling motions during transfers keeps the powder from lifting off into the air. With proper airflow, nasty odors don’t linger and you avoid inhaling what you can’t see.
Accidents and Cleanup
Spills are common in crowded workspaces. Cleaning this powder means no sweeping or blowing about. Instead, reach for damp towels to trap and collect every trace. Dry towels multiply the mess. Proper waste bins with clearly labeled hazardous containers build good habits and prevent mixing with incompatible substances. You learn early to treat even small spills seriously. Eyes deserve extra attention—use an eye wash in case of the slightest splash. I’ve seen small splatters wake up an entire research group, as everyone drops what they’re doing to decontaminate fast.
Storage and Disposal Keep Problems Small
Storing chemicals by their hazard class does more than keep inspectors happy. Keeping sodium 4-chloro-2-nitrophenoxide away from heat and incompatible chemicals like acids or strong oxidizers prevents dangerous surprises. I keep my area organized, not just out of pride—a loose cap or improper label once cost a friend an entire Saturday scrubbing shelves. Hazardous waste collection seems tedious, but it’s the only way to protect your building and the environment. Neutralizing leftover product under supervision or working with waste management teams feels like a hassle, yet it builds trust that nobody will get hurt later.
Knowledge Means Confidence
Anyone working with these chemicals owes it to themselves and others to learn more than the basics. Reading material safety data sheets and seeking advice from seasoned staff goes further than just checking boxes. Reviewing emergency procedures with colleagues keeps everyone sharp. Real learning sticks when you see the impact of shortcuts. With steady habits and respect for risk, labs become places to solve problems—not create new ones.
How should Sodium 4-Chloro-2-Nitrophenoxide be stored?
Sodium 4-chloro-2-nitrophenoxide comes up often in labs working with organic synthesis and dye chemistry. It’s pretty common to focus on the chemistry itself and forget about the storage practices that keep everyone safe. It’s easy to underestimate how tricky chemicals like this can be if left on an open shelf or near heat. This is where experience in the lab really matters.
Chemical Stability Needs Respect
Having handled a range of aromatic salts, you can feel how moisture in the air spells trouble fast. Sodium 4-chloro-2-nitrophenoxide, like many sodium salts, absorbs water from humid air. Once clumping begins, measuring becomes tough, and more critically, decomposition speeds up. Keep containers tightly sealed, and not just for neatness. Moisture control lengthens the compound’s life and keeps the chemistry predictable. A screw-cap bottle lined with polyethylene gives extra confidence against leaks and humidity.
Temperature Matters—even on Quiet Days
Heating elements in the lab are easily ignored until something goes wrong. Even if this compound doesn’t flash into flames under a desk lamp, storing it close to heat sources isn’t a small thing. Warm spots increase the rate of chemical reactions quietly over weeks or months. Cool, dry shelving saves a lot of headaches. Most labs stick with standard room temperature, which, by all credible sources, works acceptably for this salt. Freezing isn’t necessary—just skip any spot that creeps over 25°C.
Light Can Ruin It
Some aromatic compounds break down under fluorescent lighting or sunlight. Yellowing or fading shades on a white label are warning signs. Direct sun on a windowsill may look harmless, but it slowly degrades both plastic containers and chemicals inside, sometimes before you notice. A dark drawer or a closed cupboard is plenty. It helps to get in the habit of checking where the most light hits your shelves and avoid putting sensitive materials there—even when you’re in a hurry.
Safety Isn’t Just for Daytime
Having worked late in poorly ventilated labs, it’s tempting to want easy grabs for supplies at arm’s reach. That becomes risky with compounds that release toxic fumes if spilled or exposed to acid by mistake. Laboratory safety data sheets recommend keeping sodium 4-chloro-2-nitrophenoxide away from acids, oxidizers, and reducing agents. Label storage containers clearly and keep incompatible reactants a few shelves apart. Acid and alkali cabinets exist for a reason; tossing all bottles together in the same drawer eventually bites back.
Practical Lab Reminders
Keeping an up-to-date inventory reduces the odds of old, forgotten chemicals breaking down quietly. Make it routine to check for leaks, cracks, or unusual smells around containers. If a spill happens, neutralize and clean up with care before it dries—rushing this step never ends well. Never skip gloves and eye protection, even if it’s “just a little bit.” Many accidents happen when attention slips, especially during clean-up.
Sodium 4-chloro-2-nitrophenoxide might not grab the headlines, but smart, respectful storage protects people and preserves research. Well-cared-for supplies save time, cut costs, and avoid needless risks. That’s the sort of reliability everyone in science values.
What is the chemical formula and molecular weight of Sodium 4-Chloro-2-Nitrophenoxide?
Chemical Formula Matters
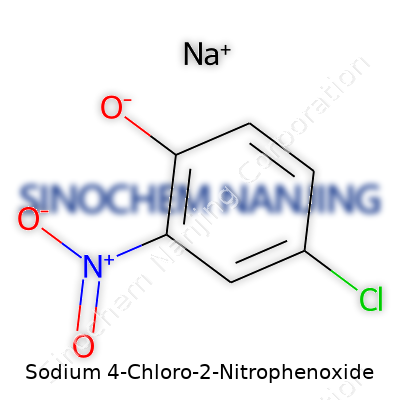
Sodium 4-chloro-2-nitrophenoxide grabs attention in the lab because its structure packs quite a punch. This compound forms when sodium reacts with 4-chloro-2-nitrophenol, stripping away a hydrogen and giving rise to a negatively charged oxygen hooked to a benzene ring. Once you break down its chemical formula, you have C6H3ClNNaO3. Each piece of this formula points to an atom crucial for its identity—carbon, hydrogen, chlorine, nitrogen, sodium, and oxygen. Seeing sodium in the mix means this isn’t an average phenol. It’s a salt, and that impacts how it dissolves, reacts, and gets used.
Molecular Weight: More Than Just a Number
The molecular weight counts for more than calculation’s sake. Anyone who has measured out powders before knows accuracy rules the roost. Sodium 4-chloro-2-nitrophenoxide comes in at 213.53 g/mol. This figure matters every time someone weighs out the compound—synthetic chemists rely on this for building blocks, environmental analysts factor it into detection, and students hunt for it in problem sets. If the amount feels off, the outcome turns unpredictable. Working in academic labs, I used to double-check calculations before and after each prep, especially with substances like this one. One slip could waste hours or botch an analysis.
Practical Importance in Research
This compound’s mix of functional groups—nitro, chloro, and phenoxide—gives it unique behavior. Researchers often use it as an intermediate when assembling more complex molecules, including pharmaceuticals and dyes. The electron-withdrawing nitro group and chloro ring bring selective reactivity. Synthetic routes in organic chemistry appreciate such versatility, and the presence of the sodium ion opens doors to water-based workups and salt metathesis. As someone who’s worked with similar molecules, the predictability of its reactions streamlines multi-step syntheses. The compound can serve as a launching pad for molecules with biological activity or color-changing properties, giving scientists reliable tools for discovery.
Quality and Safety Concerns
Every batch entering a lab deserves scrutiny. Impurities sometimes sneak in, altering results or producing side effects. In regulated sectors—think pharmaceuticals—quality assurance checks don’t get skipped. Analytical tools like HPLC, NMR, and mass spectrometry become essential, verifying both identity and purity. Dealing with sodium 4-chloro-2-nitrophenoxide, glove use and fume hoods help protect from dust inhalation and skin contact. Over the years, most accidents I’ve seen start when basic protocols get ignored. Labels fade or samples get mixed up, causing confusion and dangerous spills. Proper training prevents costly mistakes and keeps everyone safe.
Supporting Responsible Use and Future Directions
Sustainable handling always sits on the radar. Waste from phenolic compounds can harm waterways, so waste streams need careful monitoring before disposal. Treatment options include neutralization and specialized filtration. Green chemistry pushes for alternatives whenever possible, but certain syntheses still rely on specialized phenoxide salts. Researchers continue to seek safer and more sustainable synthetic routes, which sometimes means tweaking reaction conditions or switching solvents to safer ones.
Keeping Knowledge Up to Date
Accurate, up-to-date information fuels progress. Respecting the details—chemical formula, molecular weight, safety protocols—builds a solid foundation for any lab. Understanding sodium 4-chloro-2-nitrophenoxide goes beyond memorizing a formula; it means recognizing how it fits into wider scientific efforts and responsible chemical stewardship.
Is Sodium 4-Chloro-2-Nitrophenoxide soluble in water or other solvents?
Getting Practical With Lab Experience
College labs taught me most chemicals won’t play by textbook rules. Sodium 4-chloro-2-nitrophenoxide sounds intimidating, but fundamentally it’s an organic salt. I’ve watched similar salts drop straight to the bottom of a beaker, refusing to mix into water, just as often as I’ve seen them dissolve in a flash. So what’s going on? The big clue sits with the molecule’s nitro and chloro groups—they introduce some complexity.
Generally, sodium salts of phenols do show decent water solubility thanks to the sodium ion’s knack for attracting water molecules. The 4-chloro-2-nitro twists add a bit of polarity, which might help, but also can increase the stubbornness due to bulk and less hydrogen-bonding capability. Sodium 4-chloro-2-nitrophenoxide actually dissolves in water, just not always as fast as sodium chloride or sodium benzoate. Learners often get tripped up assuming anything “sodium” vanishes in water at high speed—this one takes some stirring and patience, especially if the water isn’t warm or pure.
What Happens In Other Solvents?
Switch to something less polar than water, like acetone or ethanol, and you still get some solubility, but it's noticeably lower. The nitro group does let the molecule interact with polar solvents, but you won’t see instant, complete mixing. Pouring sodium 4-chloro-2-nitrophenoxide into non-polar solvents—think hexane—will leave you with a separation and a clear, undissolved layer. The molecule prefers its company polar.
Why Does It Matter for Real Work?
Anyone making dyes, drugs, or agricultural chemicals needs to know what their raw materials do in different liquids. Poor mixing causes clogged lines, incomplete reactions, and wasted product. I’ve spent hours unclogging glassware after using the wrong solvent combination. Sodium 4-chloro-2-nitrophenoxide sits in a grey zone—soluble enough for reactions using water-based chemistry, but not the top pick for making solutions at very high concentrations.
Chemists use aqueous NaOH to improve solubility for phenolic compounds that just won’t go otherwise. This trick usually works with sodium 4-chloro-2-nitrophenoxide, turning what would be a chunky, partially dissolved situation into a clear solution. Temperature also matters—warmth speeds up the process.
Takeaways From the Literature
Research and safety sheets call out moderate to good solubility in water, especially if the solution is stirred and heated. The Merck Index, Sigma-Aldrich database, and trusted organic synthesis references put the solubility at “readily soluble” in water, but lower in alcohol and minimal in ether or hydrocarbons. If a project needs something that dumps into just about any solvent and vanishes, this is not the ideal choice. Broad solubility often gets engineered into a molecule for that reason.
Remembering the Safety Side
Working with sodium 4-chloro-2-nitrophenoxide also brings safety concerns, not just solubility. Despite going into water, it’s still an irritant, especially to eyes and skin. Gloves and goggles aren’t optional. Disposal isn’t as simple as pouring down the drain either; proper waste handling protects people and the environment.
Solutions For The Stubborn Cases
If sodium 4-chloro-2-nitrophenoxide refuses to dissolve, adding a little more heat, grinding to a finer powder, or using a co-solvent like a drop or two of ethanol in water can usually tip things in your favor. Sometimes, you have to give up and change the salt form entirely or modify the pH of your solvent.
Solubility information always matters in a real lab. The more you know about how this salt behaves, the less likely you are to end up with an unexpected mess on your hands or an incomplete reaction.
| Names | |
| Preferred IUPAC name | sodium 4-chloro-2-nitrophenolate |
| Other names |
Sodium p-chloro-o-nitrophenoxide Sodium 4-chloro-2-nitrophenolate |
| Pronunciation | /ˈsəʊdiəm fɔːr ˈklɔːrəʊ tuː ˈnaɪtrəʊˈfiːnɒksaɪd/ |
| Identifiers | |
| CAS Number | 100-00-5 |
| Beilstein Reference | 1768188 |
| ChEBI | CHEBI:87074 |
| ChEMBL | CHEMBL510892 |
| ChemSpider | 21573039 |
| DrugBank | DB14056 |
| ECHA InfoCard | 29-635-5 |
| EC Number | 215-718-6 |
| Gmelin Reference | 1263.7 |
| KEGG | C14370 |
| MeSH | D005932 |
| PubChem CID | 142098 |
| RTECS number | DB8750000 |
| UNII | Z8T5T4LIG6 |
| UN number | UN3439 |
| Properties | |
| Chemical formula | C6H3ClNNaO3 |
| Molar mass | 167.53 g/mol |
| Appearance | yellow powder |
| Odor | Odorless |
| Density | 1.61 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -0.6 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 7.1 |
| Basicity (pKb) | pKb = 10.08 |
| Magnetic susceptibility (χ) | -35.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.642 |
| Dipole moment | 5.81 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 190.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -239.2 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | Not assigned |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS05, GHS07, GHS09 |
| Signal word | Warning |
| Hazard statements | H302 + H315 + H319 + H335 |
| Precautionary statements | P261, P264, P271, P273, P280, P302+P352, P305+P351+P338, P312, P332+P313, P337+P313, P362+P364 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Explosive limits | Non-explosive |
| Lethal dose or concentration | LD₅₀ oral rat 2836 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 2830 mg/kg |
| NIOSH | SN1750000 |
| PEL (Permissible) | PEL (Permissible exposure limit) for Sodium 4-Chloro-2-Nitrophenoxide: Not established |
| REL (Recommended) | 0.5 mg/m³ |
| IDLH (Immediate danger) | Not Listed |
| Related compounds | |
| Related compounds |
Sodium 2-nitrophenoxide Sodium 4-chlorophenoxide 4-Chloro-2-nitrophenol 2-Nitrophenol 4-Chlorophenol Sodium phenoxide |

