Sodium 4-Aminophenylarsonate: A Hard Look at Its Journey and Its Future
Historical Development
Long before modern pharmaceuticals started rolling out synthetic molecules by the millions, curiosity drove chemists and doctors to experiment with different combinations of organic and inorganic compounds. Sodium 4-aminophenylarsonate shows up as a chapter in that bigger book—born from the age when arsenic, for all its infamous toxicity, was seen through a different lens. Early work with arsonic acids and related derivatives stretched back into the late 19th and early 20th centuries, as researchers like Paul Ehrlich explored their use in treating infectious diseases. At one point, arsenic-containing drugs offered hope against diseases that otherwise left communities helpless. The legacy of this compound carries the shadows of both progress and ethical dilemma, since even the most promising molecules called attention to safety and human risk. Anyone walking through a lab today might forget how research used to skirt the edge between promise and peril—yet Sodium 4-aminophenylarsonate’s lineage roots it in those choices.
Product Overview and Applications
Sodium 4-aminophenylarsonate stands out in organoarsenic chemistry—marked by a unique structure that combines an aromatic amine group with an arsonate functional group, tethered together by a sodium counterion. It never exploded into commercial industry in the way some synthetic drugs did, yet its contributions to both chemical research and biological investigation shouldn’t be overlooked. Investigators trying to parse complex biochemical pathways in the last few decades found it useful as a starting point, intermediate, or reagent. Some approaches leverage this molecule for introducing arsonate functionality into larger frameworks or modifying the behavior of organic compounds, especially in controlled lab settings. Others look at its role as a prototype—almost a checkpoint in the story of organoarsenic and pharmaceutical chemistry, serving as a cautionary tale and inspiration in equal measure.
Physical and Chemical Properties
The combination of an aromatic ring with amine and arsonate groups makes this compound a laboratory workhorse for certain specialized synthetic routes. At room temperature it takes the form of a white to off-white powder, soluble in water—thanks to the sodium ion facilitating dissolution. Molecular stability often hinges on pH and storage conditions, and exposure to light and moisture can spark concerns over hydrolysis or reduction, with dangerous byproducts always lurking. The structural features—namely free amine and arsonate groups—invite a rich variety of reaction schemes, but also demand strict handling. Unlike many of today’s commercial chemicals, even simple contact or inhalation poses important health risks, and any work with this material quickly brings to mind the broader balance between utility and hazard.
Technical Specifications & Labeling Practices
A lot rides on clear, accurate information when dealing with hazardous reagents. But labels aren’t just about regulatory compliance or plain good housekeeping—they actually set the tone for safety and collaboration in the lab. Sodium 4-aminophenylarsonate, with its no-nonsense chemical profile, requires unmistakable labeling. Specific warnings about toxicity, possible carcinogenicity, and storage instructions take priority over marketing claims or purity percentages. For researchers, what matters goes beyond a chemical formula: everyone on the team should respect the power and risks behind the name. More often than not, the best labels spell out routes of exposure and handling guidelines in large, readable type. Removing ambiguity directly saves lives.
Preparation Method
The path to Sodium 4-aminophenylarsonate starts with recognizable precursors—usually an aromatic amine like aniline, pushed through an arsonation reaction with arsonic acid or similar agents under precisely controlled heating and stirring. Success or failure often hangs on minor changes in temperature or pH. Any slip-up can turn a straightforward synthesis into a cauldron of unplanned byproducts or toxic fumes. Batch-to-batch purity depends not only on chemical math but on the vigilance, experience, and institutional memory of the people running the process. Looking back over years spent in research facilities, the lesson stands out: textbooks might provide the reaction, but only disciplined protocol really delivers clean material, without avoidable risk.
Chemical Reactions and Possible Modifications
Once inside the lab, researchers tap into the molecule's potential for coupling, reduction, or further substitution—using its amine group as a handle for downstream chemistry, or tweaking the arsonate functionality in controlled conditions. Sometimes, I’ve seen teams attempt complex coupling reactions to introduce new moieties or design analogues that mimic the parent structure but with changed reactivity or biological profiles. The dense electron cloud around the aromatic system creates both opportunity and challenge—invitation to attack by electrophiles but also the risk of generating unpredictable intermediates. In practice, every hand in the glove box or fume hood knows to respect both the complexity and the danger. Heavy regulatory scrutiny comes with the territory; nobody wants a repeat of the bad old days when unchecked laboratory improvisation led to hospital visits or long-term contamination.
Synonyms & Product Names
People in the field recognize this compound under several names—some dry and technical, others relics of older nomenclature conventions. Alongside Sodium 4-aminophenylarsonate, references will surface to para-aminophenylarsonic acid sodium salt, or sodium p-aminophenylarsonate, depending on the lab or the context. Chemists often keep a running list of synonyms to avoid confusion, especially when literature searches span decades and international borders. Standardization helps, but it’s on each user to double-check the material in front of them and cross-reference alternate labels. Anyone who’s ever mixed up reagents because of name confusion feels the sting: a strong culture of verification safeguards against expensive mistakes.
Safety and Operational Standards
Toxicity commands the spotlight. No matter how valuable the compound might be on paper, every ounce must be treated as a potential health threat. Protocols mandate sealed gloves, goggles, fume hoods, and spill kits—minimum standards, not luxuries. Training sessions emphasize that arsenic compounds leave little room for error, so new staff learn both procedures and the underlying reasons behind them. I recall more than one instance where a minor breach in PPE led to a near-miss; those events stick with you as much as any published guideline. Disposal practices require careful containment, neutralization, and documentation before leaving the facility. Regulatory bodies, from OSHA to international equivalents, weigh in with strict rules, but the real margin of protection comes from culture—a sense of shared responsibility and institutional memory among staff who’ve seen what happens if you drop your guard.
Application Area
This isn’t a compound you’ll find in mass-market production. Most of its impact comes from focused laboratory and academic use. Some researchers draw on it as a bridge to understanding how arsonic groups behave in larger bioactive molecules, or as a model system for testing new detection methods for arsenic and similar contaminants. In certain studies, variants of Sodium 4-aminophenylarsonate serve as benchmarks for environmental toxicology—providing a controlled source for tracing how organoarsenic substances move through soil, water, or biological systems. Modern synthetic chemistry sometimes calls for precisely this sort of rare, targeted reagent, but always with a deep respect for what’s at stake if protocols go wrong.
Research & Development
A molecule like this fuels a particular kind of curiosity—the slow, painstaking work of building out the chemistry toolbox. Development today revolves less around large-scale deployment and more around fine-tuning reactivity, understanding structure-activity relationships, and inventing safer analogues. Newer research efforts look for ways to harness the unique properties of the arsonate group without dragging along so much toxicity. In my own work, teams often debate which synthetic intermediates can deliver similar results without the health and environmental baggage. In this crowded research landscape, the compound acts as both a springboard and a warning, reminding us that innovation means balancing promise against risk, and that the past shouldn't dictate the future of chemistry.
Toxicity Research
No discussion about Sodium 4-aminophenylarsonate could ignore the cold facts of toxicity. Arsenic’s dark pedigree runs through epidemiological data, clinical case reports, and a long legacy of poisoning cases from both accidental and intentional exposure. Research teams devote significant energy to understanding how the compound enters cells, interferes with enzymes, or disrupts natural chemical cycles in the body. Traditional animal studies, paired with cutting-edge cellular assays and computational models, hunt for weak points or unexpected side effects. Institutional review boards rarely green-light work with this compound unless benefits clearly outweigh the risks; the paperwork alone reads like a sober, cautionary tale. For me and many others, the hope is that continued research lays a stronger foundation for decontamination, remediation, and better predictive models—tools that can then step in before accidents turn tragic.
Future Prospects
So what’s next for this compound and others like it? The answer will always run through hard scrutiny, constant learning, and above all, humility in the face of chemical power. Advances in green chemistry and reagent design push the field to find new ways to run old reactions without relying on hazardous intermediates. Some laboratories pivot to theoretical and computational modeling, exploring the utility of arsonate groups without handling the stuff firsthand. Others dig into remediation technologies—turning the challenge of arsenic contamination into an opportunity for sustainable clean-up or safer waste management. The road ahead will likely see less bench-top use of Sodium 4-aminophenylarsonate and more application as a lesson. If anything, this compound reminds today’s generation that progress promises nothing without an honest reckoning with risk, and that chemistry—at its best—uses history as a tool, not a chain.
What is Sodium 4-Aminophenylarsonate used for?
Digging Into Its Roots
Sodium 4-aminophenylarsonate isn’t a chemical you come across in daily news, but its story weaves through decades of medicine and science. Scientists first studied it for its potential as an arsenic-based drug, seeing if it could tackle parasitic infections that plague livestock. The backbone of this compound connects chemical innovation with agricultural and early human health applications.
Its Big Moment: Veterinary Medicine
Doctors and farmers once relied on sodium 4-aminophenylarsonate to fight trypanosomiasis—a disease that devastates cattle, horses, and donkeys across Africa and parts of Asia. Arsenic-based treatments stood out in the early 1900s, before safer drugs arrived. Old research papers describe how this compound didn’t just prop up a sick herd. It helped entire communities, improving food security and livelihoods where animal-borne illness threatened survival.
While the drug has faded from mainstream veterinary medicine in regions with modern alternatives, for years, people viewed it as a lifeline. No one wants arsenic near dinner, but with careful dosing, this medicine offered hope in places with few choices.
Human Medicine and the Shadow of Risk
Researchers tried sodium 4-aminophenylarsonate for treating certain types of human sleeping sickness, a disease that can be deadly without treatment. The challenge? It’s tough to balance killing parasites with not harming the patient. Arsenic compounds often walk a fine line between therapy and toxicity. Reports point to serious side effects—anything from stomach pain to kidney problems. Drug safety standards grew stricter, pushing old treatments like this out of regular use.
Safer options for trypanosomiasis exist now, but those developing medicines today owe plenty to the chemical pioneers who experimented with compounds like this. Learning from past mistakes, we developed drugs with fewer risks.
Environmental Echoes and Modern Lessons
The environmental footprint of arsenic-based drugs, including sodium 4-aminophenylarsonate, can’t be ignored. Discarded animal waste and untreated chemical runoff seeped into soil and water. Long-term, these traces end up in crops and groundwater, putting whole communities at risk. I grew up on a family farm and noticed the way agricultural chemicals, helpful as they may be, have a knack for sticking around long after the job’s done. The responsibility falls on scientists, regulators, and everyone in the supply chain to manage and clean up these effects.
Building Smarter Solutions
With global regulations getting sharper around hazardous chemicals, most countries now restrict or outright ban these old arsenic drugs in agriculture and medicine. Progress means not just finding quick cures, but making sure the solution doesn’t spark fresh problems. Innovators now target parasites with medicines that break down easily, leave no dangerous residue, and truly heal without causing new problems downstream.
If we want safer treatments and cleaner soil, researchers have to team up with policymakers and communities. The story of sodium 4-aminophenylarsonate reminds us: every “miracle drug” comes with a cost. Nothing develops in a vacuum—health, food supply, and the environment intersect every day. The future hinges on putting that full picture into practice, never settling for quick fixes.
What is the chemical formula of Sodium 4-Aminophenylarsonate?
Breaking Down the Name
Chemistry sometimes feels like a foreign language, but every name tells a story. Sodium 4-aminophenylarsonate falls into this category. This compound carries an interesting mix of elements. The core pieces are sodium (Na), a phenyl ring with an amino group at the fourth position (4-aminophenyl), and an arsonate group, which features arsenic bonded to oxygen atoms. Many might recognize the “amine” group as -NH2, which often signals high reactivity and unique binding properties in pharmaceuticals and dyes.
To spell it out: the sodium cation stabilizes the negatively charged arsonate group, while the phenyl ring and amino group organize the molecule’s unique identity. The formula comes together as NaC6H7AsNO3. Getting this right matters. Chemists and pharmacists count on clear, reproducible formulas—an error can mean surprises in synthesis, safety, or downstream effects.
Why Proper Formulas Matter for Real People
During an undergraduate stint in organic and medicinal chemistry labs, a single misplaced element could derail a week’s work. Textbooks stress accuracy for a good reason. Sodium 4-aminophenylarsonate has cropped up in research exploring new medicines, especially when arsenic-based drugs still saw routine use before safer options arrived. With sodium sitting at the edge and the arsonate group ready for interaction, researchers could turn their attention to fighting parasites like those that cause sleeping sickness—a key part of some drug development in the early-to-mid 20th century.
Improper formulas don’t just trip up scientists. Downstream, they can affect labeling, transport, and storage requirements. For substances involving arsenic, mistakes become particularly serious—arsenate compounds need thoughtful handling due to their toxicity. Chemical workers, doctors, and regulators need to see the correct molecular formula on everything from shipment labels to clinical documentation. If one digit or atom is missing, outcomes can shift from simple confusion to serious accidents.
Trusted Sources and Fact-Checking
Everyone from new students to seasoned researchers should double-check their facts. Peer-reviewed chemistry journals or reference works like the Merck Index often confirm formulas like sodium 4-aminophenylarsonate’s NaC6H7AsNO3. In practical settings, online databases—when curated by recognized scientific groups—keep compound information up to date. The reliability doesn’t stem from convenience; rather, it grows from the track record of institutions dedicated to accuracy, transparency, and user safety.
Addressing Knowledge Gaps and Moving Forward
Even with all the right information, people occasionally let errors slip through. Labs and educational programs could do more by encouraging open verification—sharing peer cross-checks and test runs. Uncertainty multiplies when someone assumes information is obvious or settles for incomplete references. Encouraging rigorous habits, such as consulting two or three sources for sensitive substances, arms everyone with the best chance to avoid dangerous confusion. For compounds involving elements like arsenic, these extra checks save more than just face—they can protect health, studies, and future progress.
Clear, correct chemical formulas form the backbone of scientific progress. Sodium 4-aminophenylarsonate’s formula—NaC6H7AsNO3—stands as a reminder that chemistry depends on small details, collaboration, and a commitment to checking our work every step of the way.
Is Sodium 4-Aminophenylarsonate hazardous or toxic?
What We Know About Sodium 4-Aminophenylarsonate
Sodium 4-aminophenylarsonate, a compound that blends the chemistry of arsenic and aniline, doesn't turn up often in popular conversation. Whenever a chemical contains arsenic in its structure, I start paying attention. Arsenic is notorious: it's linked to a raft of health problems, including cancer, organ damage, and developmental issues. Many folks know stories about contaminated well water or dangerous pesticides. So, questions about its safety aren’t just smart—they’re necessary.
The Science Points to Risk
Chemists and toxicologists flag aromatic arsenicals like this as risky for plenty of reasons. Even at low exposures, arsenic compounds cause trouble. Researchers connect chronic exposure to skin, bladder, and lung cancer. They also link arsenic to heart disease, diabetes, and immune system breakdown. Some arsenic chemicals pass into the body through inhalation, skin contact, or swallowing. Workers handling powders or liquids get the worst of it, but the wider community faces exposure through contaminated water, air, or soil.
Animal studies use sodium 4-aminophenylarsonate as a model for arsenic poisoning because it replicates the symptoms doctors see in arsenic-exposed humans: fatigue, gastrointestinal issues, and sometimes neurological symptoms. A single spill or improper disposal may taint groundwater. Once there, compounds break down slowly, lingering for decades. Outdated pesticides and industrial chemicals haunt abandoned sites and, without cleanup, turn up in crops or downstream water taps.
Protecting People and Places
The good news is, we know quite a bit about handling dangerous chemicals. Strict rules limit allowable worker exposure levels. Safety gear—masks, gloves, full suits—blocks skin and air contact. Workers also train to manage spills, track chemical inventories, and respond fast if something leaks or spills. In labs where I’ve worked, teams run regular air sampling and test workspaces for invisible contamination.
Disposal gets equal attention. Hazardous waste firms incinerate or neutralize arsenic-bearing material, keeping it out of landfills and water systems. Engineers design treatment plants with separate arsenic removal stages. Water utilities in high-risk regions test arsenic levels weekly. The push for digital records and barcoding means public health teams catch problems faster than ever.
Solutions and Safer Choices
Better options exist for most tasks. Agriculture now relies on arsenic-free pesticides. Modern dyes and pharmaceuticals rely on safer synthetics. Most companies phase out arsenic-based reagents for alternatives that don’t carry human health baggage. If I look around the lab, shelf labels stand out in bold red if a bottle holds an arsenic compound—an immediate visual cue for extra care.
Communities near chemical plants or legacy industrial sites demand transparency. Public health advocates push for full chemical reporting, prompt spill notifications, and regular groundwater monitoring—all backed by law and local organizing. Parents get school water tested, local governments secure grants for cleanup, and whistleblowers call out unsafe practices.
Why It Matters
Living in a world with heavy metals and industrial byproducts means every person deserves straightforward information. Sodium 4-aminophenylarsonate cannot be dismissed as a harmless ingredient. No matter how exotic the name sounds, the risks are concrete, and the science backs up the need for robust safety measures, strong community action, and real investment in alternatives.
How should Sodium 4-Aminophenylarsonate be stored?
Understanding the Risks
Sodium 4-Aminophenylarsonate doesn’t show up on most shopping lists, but for folks who handle chemicals for a living, awareness about its storage could make a real difference. This substance includes an aromatic amine and an arsenic group. Anyone around the world who’s spent time working in a lab or a research facility knows that certain chemicals never sit loosely on a dusty shelf, and this is one of them. The arsenic component demands respect due to its hazardous nature—fine dust or residues aren’t something you want in your eyes, lungs, or on your skin.
Choosing the Right Location
I’ve seen labs where chemicals were stashed in any spot with open space. That always felt like waiting for trouble. Sodium 4-Aminophenylarsonate should stay locked in a dedicated chemical storage cabinet that’s clearly labeled, separate from incompatible substances like strong oxidizers or heavy acids and bases. A cluttered shelf or an unlocked door isn’t good enough—not only for compliance, but also for peace of mind.
Temperature and Environment
People sometimes overlook the influence of temperature on chemical stability. Keeping this compound at controlled room temperature, away from heat or direct sunlight, helps prevent slow degradation or moisture absorption. Arsenic-based materials can release dangerous fumes under the wrong conditions, so I’ve always favored well-ventilated storage rooms. Humidity and chemical vapors never mix well.
Physical Security and Access Control
In any research lab I’ve worked in, giving open access to toxic substances just isn’t smart. Only trained personnel should handle or even approach containers of Sodium 4-Aminophenylarsonate. Labs with sign-in logs for restricted areas protect both staff and anyone who might mistakenly wander in. Setting up access barriers, proper signage, and written procedures for handling spills sets a higher standard for everyone’s safety.
Container Choice and Labeling
Original, tight-sealing containers serve a purpose beyond convenience. They cut down on accidental contact and slow the spread if a spill happens. Using non-reactive plastics or glass helps avoid corrosion or accidental chemical reactions. A visible, durable label showing the compound’s full name, hazard class, and the date received always helps in a hurry. I remember an incident when an unlabeled bottle led to confusion, which nearly escalated to a full evacuation. Labels stop accidents before they start.
Emergency Preparedness
Early in my career, someone spilled a few grams of an organo-arsenic compound. Nobody panicked because safety showers, eyewash stations, and spill kits lined the corridor. Having the right gear within easy reach, coupled with regular safety drills, could mean the difference between a minor hiccup and a disaster. Staff also need clear contact information for emergency responders and poison control. It strikes me that simple things—knowing where to wash off a splash or where the nearest phone is—offer the quickest protection amid chaos.
Waste Handling and Accountability
Disposal of leftover chemical or contaminated items must stick to regulations. Nobody wants these compounds headed for a landfill where groundwater could be threatened. Logging every use and keeping an inventory helps spot theft, leaks, or unauthorized disposal. Authorities require this, but it’s just as much about keeping co-workers healthy and the environment intact. I’ve watched organizations build cultures of responsibility, and the payoff stands out in cleaner, safer workplaces for all.
What precautions should be taken when handling Sodium 4-Aminophenylarsonate?
Putting Safety Before The Work
Sodium 4-Aminophenylarsonate just sounds like trouble because, frankly, it can be. Anyone who spent time in a chemistry lab knows some substances ask for real respect. This one checks that box, thanks to the arsenic in the mix. Long sleeves, solid gloves, protective goggles—those become the uniform. I always looked at substances like this and remembered how skin contact and accidental inhalation stay with you far longer than the moment you open a bottle.
It’s not just about following a rulebook. Stats from the CDC lay out how arsenic compounds, even in small doses, mess with nerves and organs. Symptoms don’t always show up right away, so anyone handling the material should treat spills or leaks as serious. I always kept a proper fume hood running for anything like this—indoor ventilation buys you peace of mind in ways nothing else does.
Why Training Never Ends
Calling experts obsessive only tells half the story. Training sticks because every small misstep with toxic chemicals like this throws away months of caution. Eyewash stations and emergency showers should always stay a few steps away from the workspace. At one company, we ran drills because the worst time to figure out what happens in an accident is during the real thing. If you work around this kind of compound often, you already know regular training leads to quick reactions and fewer regrets.
Double-checking chemical storage also becomes habit once you see a poorly sealed container leak through a shelf. Sodium 4-Aminophenylarsonate calls for clear labels and compatible containers. Out of sight means out of mind, so regular checks on seals, expiration dates, and chemical logs pay off.
Waste, Spills, and Environmental Stakes
Some still underestimate the cost of a spill. This isn’t sugar—trace amounts of arsenic impact water and soil in a big way. For disposal, the old sink is not the answer. I’ve seen entire shipments halted because someone forgot the hazardous waste protocols. Sealed hazard containers, professional pickup, and paperwork stand between responsible handling and a pollution slip-up with state regulators at your door. Following EPA guidelines is more than a “good practice”—it keeps your community safe. The research is plain: arsenic doesn’t just disappear, and small labs create big problems without clear disposal plans.
Room for Better Tools
Labs keep improving their safety gear. Sensors can flag gas leaks, and new glove materials block almost anything you throw at them. I remember moving from latex to nitrile gloves and being surprised how big a difference it made for strong chemicals. Scaled-down sensors now fit into most budgets, so waiting for the smell isn’t an excuse.
Wrapping up Responsibility
Making Sodium 4-Aminophenylarsonate safer to handle isn’t about ticking boxes. It’s about real routines, practical training, and using the best gear. Neighbors, lab workers, and anyone downstream from your waste count on you doing the job right. That commitment shows, not just in spotless records but in peace of mind at the end of every shift.
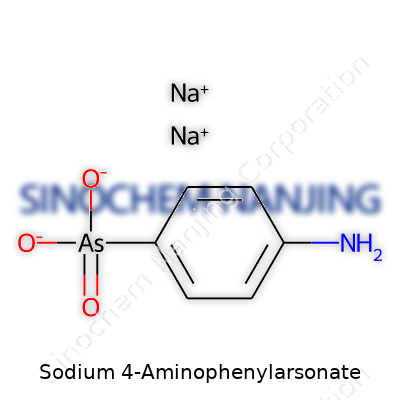
| Names | |
| Preferred IUPAC name | sodium 4-aminophenylarsonate |
| Other names |
Aminylarsenate sodium Atoxyl Sodium aminophenylarsonate 4-Aminophenylarsonic acid sodium salt Sodium p-aminophenylarsonate |
| Pronunciation | /ˈsəʊdiəm ˈfɔːr əˌmɪnoʊˈfiːnɪl ˈɑːrsəˌneɪt/ |
| Identifiers | |
| CAS Number | 13064-82-9 |
| 3D model (JSmol) | `3DModel:JSmol=C1=CC(=CC=C1N)As(=O)(O)ONa` |
| Beilstein Reference | 4098752 |
| ChEBI | CHEBI:52445 |
| ChEMBL | CHEMBL1233672 |
| ChemSpider | 21569739 |
| DrugBank | DB01394 |
| ECHA InfoCard | 100.007.778 |
| EC Number | 217-595-7 |
| Gmelin Reference | 81654 |
| KEGG | C06797 |
| MeSH | D000325 |
| PubChem CID | 67698 |
| RTECS number | CN6950000 |
| UNII | IQ542B47T8 |
| UN number | UN2014 |
| CompTox Dashboard (EPA) | DTXSID5050853 |
| Properties | |
| Chemical formula | C6H7AsNNaO3 |
| Molar mass | 263.07 g/mol |
| Appearance | White to yellowish crystalline powder |
| Odor | Odorless |
| Density | 1.77 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -2.6 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 6.0 |
| Basicity (pKb) | 6.52 |
| Magnetic susceptibility (χ) | -49.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.630 |
| Dipole moment | 6.14 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -889.1 kJ/mol |
| Pharmacology | |
| ATC code | QH51AB04 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. May cause damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS05, GHS07, GHS08 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H312, H332, H351, H372, H410 |
| Precautionary statements | P264, P270, P273, P301+P312, P305+P351+P338, P330, P337+P313, P501 |
| NFPA 704 (fire diamond) | 1-2-0-NA |
| Flash point | 138°C |
| Lethal dose or concentration | LD50 oral rat 900 mg/kg |
| LD50 (median dose) | LD50 (median dose): 600 mg/kg (oral, rat) |
| NIOSH | AS8575000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Sodium 4-Aminophenylarsonate: "0.01 mg(As)/m3 |
| REL (Recommended) | 0.5 mg As/m3 |
| IDLH (Immediate danger) | IDLH: 5 mg As/m3 |
| Related compounds | |
| Related compounds |
4-Aminophenylarsonic acid Arsanilic acid Roxarsone |

