Sodium 4,6-Dinitro-2-Aminophenoxide: The Curious Case of a Niche Chemical
Historical Development
Chemistry often reminds us how a small tweak in structure can open doors to whole new possibilities or even unearth concerns. Sodium 4,6-dinitro-2-aminophenoxide has traveled an interesting path since researchers first studied dinitrophenolic compounds over a century ago. The early 1900s saw chemists, driven by the search for better dyes and pharmaceuticals, poking around with nitroaromatic compounds. Later, as the nitro group’s electron-withdrawing power caught attention, scientists kept exploring substituted aminophenols for their analytical and industrial uses. With nitro groups in the 4 and 6 positions on the phenol ring, and a sodium salt to boot, this molecule didn’t fade into obscurity. Instead, it found a few corners of the lab and manufacturing sectors where its curious blend of properties mattered.
Product Overview
Sodium 4,6-dinitro-2-aminophenoxide doesn’t show up on store shelves or in typical household goods, but its mix of aromatic rings, nitro groups, and an amino group mean it’s far from trivial. As a sodium salt, it dissolves in water better than its neutral form. Chemists appreciate compounds like this, which combine reactivity and water solubility, for their role in synthesis and as reference standards. Years in the lab teach that only certain molecules check all the right boxes for niche applications, and this one often gets pulled out when specific reactivities are needed or when its vivid yellow-orange color can be put to work.
Physical & Chemical Properties
With two nitro groups and an amino group tipping the balance on the ring, sodium 4,6-dinitro-2-aminophenoxide stands out for more than just its color. Subtle changes in the electron distribution change everything from how it dissolves to what it reacts with. Salts like this usually come as crystalline solids, sometimes hygroscopic, sporting strong absorption in the visible spectrum. In the right light, the solid pulses in yellow to orange hues, hinting at how the structure grabs and releases photons. The molecule’s polarity—a result of both the nitro and amino functional groups—pushes it toward water solubility, tracking with what most sodium salts do. In a regular lab, compounds with this structure spark discussions about hydrogen bonding, resonance stabilization, and how substitutions dictate both physical handling and reactivity.
Technical Specifications & Labeling
No two labs label specialty chemicals the same way, but sodium 4,6-dinitro-2-aminophenoxide gets handled with particular care. The need for clear hazard statements stems from its nitro content, which suggests explosives risk or at least sensitivity to strong shock or heat. Chemists who’ve been on the wrong end of a nitro compound know why regulatory warning labels run longer than the compound’s name. It’s rarely enough to slap a “toxic” sticker and move on. Anyone moving this molecule across a campus or factory respects the protocols for tracking, packaging, and secondary containment. Inventory management—down to the gram—becomes second nature when you’re dealing with compounds that need this amount of respect.
Preparation Method
Lab synthesis usually traces a clear path: start with a phenol backbone, bring in nitro groups, and control the temperature to avoid runaway reactions. For sodium 4,6-dinitro-2-aminophenoxide, precision matters. Dinitration requires careful handling of nitric and sulfuric acids. Too much heat or acid leads to a mess; too little, and the yield plummets. Introducing an amino group—often by reduction or direct amination—demands just as much patience. It’s not a reaction suited for those looking for quick results. The sodium salt forms in a final step, usually by neutralizing with sodium hydroxide. This one-two punch—careful substitution followed by neutralization—comes up often in organic synthesis courses because it drives home the importance of stepwise monitoring, pH control, and purification.
Chemical Reactions & Modifications
With two strong nitro groups and an amino group, the structure acts almost like a chemical playground. The electron-withdrawing and -donating effects interplay in ways only encountered with careful experiment planning. In a synthetic sense, the product offers a starting point for nucleophilic and electrophilic substitutions. Its aromatic nature stands up to certain oxidative and reductive conditions, but the nitro groups impose limits. Experienced chemists watch for signs of decomposition or side reactions, especially under harsh conditions. Substituents can be replaced or further functionalized, but not without running into issues of reactivity and selectivity, which drive even experts to double-check their reaction monitoring. In practice, these challenges highlight why precise lab technique matters and why understanding aromatic substitution keeps paying dividends throughout a chemist's career.
Synonyms & Product Names
Anyone who’s ever juggled a dozen names for one chemical knows how confusing the field gets. Sodium 4,6-dinitro-2-aminophenoxide often pops up under alternate spellings or synonyms in old journals or in research catalogs. Older texts toss around names like sodium dinitroaminophenolate, DNP-aminophenol sodium salt, or even more arcane variations depending on translation or naming conventions of the day. This can trip up researchers hunting for previous studies or ordering reference samples. Anyone diving into literature reviews, especially across languages or decades, learns fast to cross-check molecular formulas and not trust that one name suffices. As always, CAS numbers or InChI codes help cut through the confusion, underscoring how chemists anchor their work in confusing seas of nomenclature.
Safety & Operational Standards
The nitro groups on this molecule bring more than color—they bring risk. I’ve seen what happens when someone gets too casual around energetic compounds, and it’s never pretty. Chemists and lab techs faced with sodium 4,6-dinitro-2-aminophenoxide check for proper ventilation, grounding, and secondary containment. The need for chemical-resistant gloves and splash-proof goggles can’t get overstated. The compound’s water solubility can fool newcomers into underestimating its risks, but the ingestion and inhalation hazards remain serious. Emergency protocols hang within sight. Disposal steps require neutralization and walk-through from environmental health staff, preventing any wishful thinking about pouring leftovers down the drain. Insurance audits and worksite reviews hammer this point home—chemicals with energetic potential demand more than just a nod to compliance. They demand proven methods, tested by time and, sadly, sometimes hard lessons.
Application Area
This isn’t a workhorse chemical found in every industry, but where it finds a niche, its influence stands out. Analytical chemists sometimes use derivatives like this to detect or quantify transition metals, leveraging the color change that occurs upon binding. Its vivid pigment properties also tempt researchers aiming to blend new dyes, particularly in specialty coatings or high-contrast stains for microscopy. As an intermediate, it pops up in syntheses of more complicated organic molecules. Some routes leading toward pharmaceuticals or advanced materials pass through molecules like this one, either for their reactivity or their signal properties. In my own research, aromatic nitro derivatives often show up as key reagents when pushing the boundaries on redox chemistry or in cases where a precise electron distribution on a ring sets the pace for downstream steps. Still, widespread industrial adoption always bumps up against the cost and safety hazards, reminding developers to weigh utility against risk.
Research & Development
Research teams drawn to sodium 4,6-dinitro-2-aminophenoxide usually come from either analytical chemistry or the fringes of materials science. I’ve seen projects where its electron push-pull character enabled new insights into charge-transfer complexes or molecular recognition. The challenge? Taming the reactivity without inviting unwanted side-products or accidents. Detailed kinetic studies stack up, seeking not just better yields but also safer reaction conditions. Industrial chemistry labs sometimes dig into structure-activity relationships, hoping to eke out better performance in niche applications—think detection assays, UV-Vis dye complexes, or intermediary steps for more valuable end products. As always, funding agencies want both results and reassurances that safety standards won’t slip, prompting constant dialogue among chemists, engineers, and environmental health officers.
Toxicity Research
It’s tough to overstate the importance of solid toxicity data for compounds like this. Nitroaromatic compounds have a checkered history—some cause acute toxicity, others linger in the environment. Animal studies and in-vitro tests typically drive the generation of LD50 data, mutagenicity assays, and bioaccumulation profiles. I’ve worked with labs that spend months chasing down metabolic breakdown pathways, looking for any sign of unexpected toxicity in derivative or downstream products. Those working in regulatory or safety roles often study solvents, reaction byproducts, and waste streams for traces, recognizing that even small releases can create headaches. The bottom line from the data: respect for these molecules keeps people safe. Detailed risk assessments run alongside any discussion of process scale-up, and it’s not uncommon for proposed uses to get shelved due to toxicity concerns, particularly in consumer-facing applications.
Future Prospects
Advances in synthetic chemistry and green process engineering have revitalized old molecules in surprising ways, and sodium 4,6-dinitro-2-aminophenoxide stands on the edge of that trend. As computational chemistry tools get sharper, researchers model how the subtle interactions of nitro and amino groups influence reactivity and functionality, searching for safer or more efficient analogs that keep the benefits without the baggage. Environmental scientists, for their part, look for routes to degrade or neutralize waste, bringing bioremediation and advanced oxidation processes into the conversation. The chemical industry knows it can’t rest on legacy products and needs to demonstrate sustainability alongside performance. There’s cautious optimism that, with the right investment in safer manufacturing and disposal, compounds like this don’t have to become relics—if they can find a path that balances value with lower risk. As someone who’s watched once-obscure chemicals earn a new reputation through smart, responsible innovation, I bet the story for sodium 4,6-dinitro-2-aminophenoxide has room for a few more chapters.
What is Sodium 4,6-Dinitro-2-Aminophenoxide used for?
What Is This Chemical?
Sodium 4,6-dinitro-2-aminophenoxide sounds like something plucked straight from a chemist’s wildest dreams. Dig past the long name and you’ll land on a specialized compound, made by tweaking the familiar aminophenol molecule and tacking on nitro groups. That means it comes out bright yellow, and it’s loaded with energy and reactivity. It doesn’t show up in corner stores or the household cleaning aisle for a good reason—its knock-on effects are a double-edged sword, both in the good and bad sense.
Who Really Uses It?
Most folks don’t bump into this chemical in everyday life. Scientists in labs, engineers in very specific industries, and folks working in explosives research see it pop up. The structure sets it apart. Those nitro groups make it behave differently than its cousin molecules.
One major area: research focused on energetic materials and dye synthesis. In simple terms, that’s anything from new types of explosives for mining to specialty dyes for high-tech applications. Some pharmaceutical researchers also watch it closely because the nitro and amine groups can link onto other compounds—think of it like extra “hands” in the world of molecules, latching onto stuff for building or testing new medicines.
Why Does It Matter?
The backbone of this compound is its ability to donate electrons and break apart under controlled conditions. This characteristic plays straight into the demands of industries wanting rapid, powerful reactions. Certain types of explosive or pyrotechnic compositions lean on such chemicals because they deliver a hefty punch in a controlled chain reaction.
From my own work in a chemical lab, synthesis and handling of such energetic compounds always demands respect. Even a minor mistake can mean more than a burnt hand—sometimes it’s a full emergency shutdown. The safety considerations go hand in hand with the reason industries still work with this compound: it often does what nothing else can. It comes down to a simple fact—if a reaction needs strong donor groups or special coloration, there aren’t many better choices.
The dye industry also gets a say. That intense color—almost a warning flag—makes it valuable in developing sensitive colorimetric sensors. A project I followed at a university lab used it to build a new sensor for detecting toxic metals in water. The positive results fueled interest, but the process raised constant questions about toxicity, waste handling, and long-term safety.
What About Safety?
Every year, stories pop up of labs mishandling reactive intermediates like sodium 4,6-dinitro-2-aminophenoxide. Its energetic properties mean storage and disposal guidelines aren’t simply good hygiene—they’re mandatory. People sometimes cut corners, thinking a splash here or there won’t do harm. I’ve learned, sometimes the hard way, that cutting corners never pays when dealing with such reactive stuff.
Regulations catch up slow, but the answer lies in vigilance and transparency. Clear protocols, dedicated training, and willingness to seek help—not just relying on printed MSDS sheets—lower risk. The world may never see this chemical in a household product, but it plays a quiet, serious role in the background. It reminds us that cutting-edge science almost always pairs promise with real risks, and both deserve equal attention.
What are the safety precautions when handling Sodium 4,6-Dinitro-2-Aminophenoxide?
Respecting a Potent Chemical
Sodium 4,6-dinitro-2-aminophenoxide ranks among chemicals that demand real care from anyone handling them. You remember the first time a teacher cracked open a bottle of something like it in my college lab: the smell hit sharply and the professor’s voice jumped half an octave as she laid down the ground rules. One mistake with these kinds of substances can leave a person with burns, harmful dust in the lungs, or worse. The bright yellow crystals might not look too sinister, but don’t let their appearance mislead you.
Powdered aromatic nitro compounds like this one break down quickly under the wrong conditions, and they can be toxic through skin contact, inhalation, or accidental ingestion. They also risk dust explosions in the right mix of air and powder. Good practices keep damage at bay, not just for the person behind the goggles, but for everyone sharing the space where it’s being used.
Personal Safety Starts With Gear
For most people, goggles, a decent lab coat, and gloves seem standard—almost obvious. Plenty of people forget that splash-proof eye protection beats basic safety glasses. Polycarbonate goggles form a shield that cuts down on vapor and particle exposure if something splashes or clouds up. I pick heavy-duty nitrile gloves for this kind of material. Latex lets compounds soak through; nitrile holds up a lot longer in the face of chemical assault. Closed shoes, not sandals, protect your feet from what falls.
People sometimes skip wearing a mask with powdery compounds. That got me into trouble during a rushed experiment. Dust inhalation can lay a person out flat, so a certified particulate respirator with an N95 or better rating keeps harmful particles out of the lungs.
Physical Control and Ventilation
Fume hoods earn their reputation for a reason. Sodium 4,6-dinitro-2-aminophenoxide lets off tiny particles and fumes that shouldn’t linger. Work right inside the sash, never outside it. I spent a summer in an under-ventilated shop and can say headaches and strange taste sensations taught most people to double-check the airflow before any chemical moved out of storage. Keep containers closed unless you’re measuring or using them, since exposure to air encourages the release of dust and breakdown products.
Clean Habits, Safe Results
Nobody likes cleaning, but with chemicals this hazardous, thorough decontamination isn’t an option—it’s survival. Wipe down benches with appropriate solvents or neutralizers after every session. Don’t let crumbs of powder linger, as dry residues can go airborne with a gust of wind or an elbow bump. Dedicated disposal bins lined with chemical-resistant plastic grab all wipes, gloves, and used paper, sealing them away from the open air. Forget about tossing this waste in regular trash. Even trace amounts must meet guidelines for toxic material disposal, set by your city or region’s hazardous waste service.
People, Procedures, and Training
No one masters chemical safety by intuition. Many labs run safety briefings before touching something new. Fact sheets with Safe Work Procedures hang in easy view, not just so regulators see compliance, but so no one needs to gamble. If you’re unsure about anything, find a more experienced voice. I once saw a colleague take a shortcut “because he’d done it a hundred times.” That shortcut nearly caused a fire. The most skilled chemists treat every transfer and measurement as an opportunity for disaster if precautions drop.
Solutions for Safer Handling
Routine checklists, strong mentorship, and a worn-in respect for risk keep labs safe. Automated dispensing systems help reduce direct contact with powders. Proper emergency protocols like waterfall decontamination and fresh eyewash stations give everyone a safety net. No level of familiarity ever erases the dangers here; only vigilance and preparation hold serious accidents at bay.
How should Sodium 4,6-Dinitro-2-Aminophenoxide be stored?
Experience at the Bench and Storage Room
Messing around with chemicals in a research lab taught me a few lessons about respect and caution, especially with energetic compounds like Sodium 4,6-Dinitro-2-Aminophenoxide. Once, a poorly labeled jar caused a real scare, sending everyone scrambling for the safety shower. Nobody ever forgot the smell. Looking back, proper storage could have saved time, nerves, and the janitor’s mop.
Chemical Stability Isn’t a Joke
This sodium salt pulls from a family of compounds known for instability. Dinitro-substituted aromatics react fast, and an exposed bench in a humid, warm room can trigger surprises. Leaving such a substance open ages it quickly. Away from moisture and heat—that’s essential. Sticking it inside robust, screw-top containers with a gasket beats cheap plastic bags. I learned to invest in glass bottles lined with Teflon. Pop the jar in a chemical-resistant secondary tray for an extra hedge against leaks.
Temperature and Light Play a Role
The hazard grows with heat. In one lab, we kept it in a basic flammable-chemicals cabinet, out of direct sunlight, at temperatures closer to the cool end of room temperature. Heartier labs push for refrigeration—dedicated chemical fridges, not the same one holding people’s lunches. Light breaks down energetic compounds faster. So, packaging in amber glass helps, and storing the bottle deep inside storage reduces the chance of a careless sunbeam doing damage.
Keep It Dry
Any chemist who cracked open a bottle in a humid lab knows the crusty mess that follows. Water in the air reacts with sodium salts, leading to degradation, color changes, and hard-to-clean puddles. I’ve seen silica gel packets or small canisters work to reduce humidity in storage cabinets. No one shrugs off moisture where safety is on the line.
Safety Above All
This is not a typical household chemical. Contact risks include inhalation, skin absorption, and possible explosion on mixing with the wrong reagents. Personal protective gear matters. Gloves, goggles, and long sleeves belong on the list. After seeing a buddy pick up a rash—and a visit to occupational health—no one in the group ever skipped PPE. Label every container in plain language: compound name, date of opening, and hazard info. Up-to-date Safety Data Sheets (SDS) need to stick near where stocks sit, not lost in some binder no one checks.
Segregation and Security
Accidents happen in crowded storage rooms. Nitroaromatics stay well away from acids, bases, oxidizers, and anything that can spark a fire. Dedicated shelves, separated by physical dividers, work much better than open racks. Once, a hasty across-the-room shakeup dumped incompatible powders within inches—luckily, nothing mixed that time.
Disposal and Real Costs
Holding onto old, unused reagent turns into a risk. Disposal isn’t a ‘deal with it later’ problem. Trained staff, in coordination with waste handling firms, provide the final safeguard. A moment’s shortcut can end up costing lives, health, or tens of thousands in clean-up. No shortcut ever pays off.
Keeping Track Means Everything
Inventory lists do more than satisfy an inspector; they save lives. A clear log prevents mystery jars and forgotten hazards from lingering and shifting hands. Reviewing these lists, I once found an expired jar nobody remembered—avoided a disaster nobody would ever want to explain to their boss or their family.
Is Sodium 4,6-Dinitro-2-Aminophenoxide hazardous to health or the environment?
Looking Closer at the Chemicals in Our World
I remember the first time I heard of sodium 4,6-dinitro-2-aminophenoxide. The name alone sounded like something best left inside a chemistry book. Yet, it isn’t just jargon for scientists. This compound shows up in research, manufacturing, and possibly in products that touch daily life.
Why It’s Worth Paying Attention
This chemical comes from a group called nitrophenols, which are no strangers to hazard warnings. Nitrophenols aren’t just buzzwords in toxicology—they’ve made headlines over the years for their links to water pollution and industrial accidents. They pack a punch when it comes to toxicity. Their track record in history serves as a red flag, reminding folks not to brush off new relatives without a second look.
What the Science Says
So what’s the deal with sodium 4,6-dinitro-2-aminophenoxide? It doesn’t show up as often in research as some of its chemical cousins, but we know enough to proceed with caution. Take nitro groups—those tend to make a molecule more toxic. They help chemicals slip through cell walls and interfere with life’s most basic functions.
Lab tests show that dinitrophenol compounds can mess with cellular respiration, the process every living thing depends on for energy. It’s not hard to see why people working with these substances get proper training and lab coats—a little exposure has been linked to headaches, breathing troubles, and even organ failure. Stories of industrial mishaps tell us what can go wrong when these warnings get ignored.
Environmental Fallout: A Bigger Picture
People living downstream from factories might not always recognize the name sodium 4,6-dinitro-2-aminophenoxide, but they feel its impact if it escapes into local waterways. Nitro-derivatives, in general, last a long time in the environment. They stick around in soil and water, building up over time. Fish and other wildlife pick them up, and the chemistry doesn’t just disappear—it climbs the food chain.
I’ve seen local river cleanup projects spend years reversing damage from related chemicals, only to learn it’s nearly impossible to get these residues out completely. Data from the Environmental Protection Agency warns that even trace contamination may put local ecosystems at risk. Amphibians and insects show higher death rates after exposure. Once these compounds reach groundwater, cleanup costs skyrocket, hitting towns and taxpayers right in the wallet.
How to Turn Things Around
Solutions start with information. Companies have to be honest about what they handle. Communities deserve transparency. There’s real progress to be made in switching to less toxic replacements in manufacturing and research. Government rules, like the Toxic Substances Control Act, give a strong legal framework for inspection, but enforcement hasn’t always met the mark.
Organizations can invest in smart waste handling, keeping hazardous compounds out of regular trash and sewers. Regular monitoring and emergency response play a big role in stopping small spills from turning into local disasters. Lessons from chemical safety classes and industry training don’t just stay in the classroom—they protect lives. Building a habit of respect toward hazardous chemicals saves futures. Every policy update, every better-designed lab process, brings a safer workplace and a healthier environment closer to reality.
What This Means Moving Forward
Sodium 4,6-dinitro-2-aminophenoxide belongs among chemicals deserving our respect. It’s possible to handle it safely, but shortcuts and ignorance lead to real harm. The best path keeps people, wildlife, and water out of harm’s way. Staying informed, following good practices, and holding industries accountable all matter. Whether you work in a lab, live near an industrial site, or just care about the planet, this isn’t just a science lesson—it’s a call for watchfulness in the everyday decisions around us.
What is the recommended method of disposal for Sodium 4,6-Dinitro-2-Aminophenoxide?
Why Proper Disposal Matters
The way we get rid of chemicals like Sodium 4,6-Dinitro-2-Aminophenoxide shapes more than just workplace safety. It affects soil health, public water sources, and the lives of people who work with waste. This compound isn’t one of those mild, biodegradable substances a person could just rinse down the drain. Dinitro structures, like those on this chemical, are known for both toxicity and persisting in the environment. Mishandling can lead to pollution that sticks around.
During my years helping university labs close out research projects, I saw how improper disposal led to extra costs, facility shutdowns, or emergency calls. One forgotten container of dinitro compound prompted a visit from hazardous materials response, and the whole building shut down for hours. Experiences like that drove home how a clear process benefits everyone.
Risks and Facts
Sodium 4,6-Dinitro-2-Aminophenoxide’s toxicity can harm people and wildlife, especially if it leaches into water systems. Health studies connect dinitro compounds to skin irritation, long-term organ damage, and even genetic risks if exposure goes unchecked. According to the U.S. EPA, nitroaromatics such as this do not break down easily, and simple landfill disposal or dilution undoes years of regulation aimed at keeping these poisons out of our food and water.
Waste treatment plants cannot neutralize or filter out these molecules with their standard procedures. Pouring leftovers in the trash or drain sends those dangers downstream, out of sight but never really gone.
Recommended Disposal Method
It takes more than closing a lid and writing “hazardous” on the label. Strict rules call for professional chemical waste handling. The right method begins with segregating Sodium 4,6-Dinitro-2-Aminophenoxide waste inside clearly labeled, leak-resistant containers. Use compatible materials, like high-density polyethylene, since certain metals react with strong bases or nitrated organics. Each container gets logged in a chemical inventory, noting who produced it and in what quantity.
After collection, contact a licensed hazardous waste disposal contractor familiar with energetic and toxic organics. They send the material to a permitted treatment, storage, and disposal facility (TSDF), where real chemical engineers oversee the process. Common solutions include chemical degradation—using reagents like sodium sulfite or hydrosulfite under controlled, ventilated conditions—or incineration in a rotary kiln built to break nitroaromatics into less worrisome substances. This step eliminates persistent molecules instead of shuffling them from one landfill to the next. Shredding, burial, and DIY neutralization do not reach the temperatures or controls required for safety here.
Improving Safety Through Awareness
I’ve watched trainers turn safety drills into box-checking exercises that miss practical details. Labels fade or spill logs go unfilled. Teams that connect the dots—linking proper disposal to real-world protection—show fewer accidents and regulatory penalties. Posting clear signage, conducting regular refresher training, and drilling on emergency cleanup techniques create a culture where no one tosses unknown powders into the regular trash bin.
Some labs, especially at smaller institutions, lack resources for full-time safety staff. Grouping hazardous waste pickups across departments or sharing contact lists for approved vendors can lower costs and reduce confusion. Digital waste tracking tools reduce errors from messy paperwork, letting everyone follow the journey of dangerous chemicals from benchtop to final destruction.
Looking Ahead
Governments and local agencies set rules, but it falls to the people handling chemicals to respect both the letter and the spirit of those laws. Substituting less hazardous reagents in new research, minimizing leftover stock, and robust record-keeping all shrink the chance of future mishaps. Experience shows that the best disposal method for anything as toxic as Sodium 4,6-Dinitro-2-Aminophenoxide stays rooted in knowledge, diligence, and a team that understands exactly why each step matters.
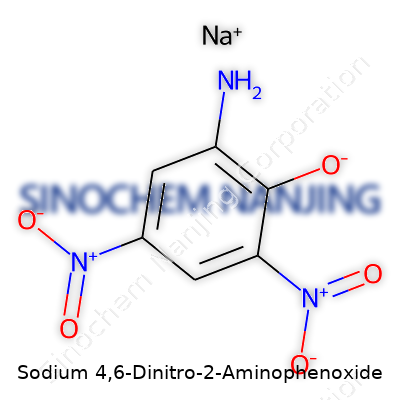
| Names | |
| Preferred IUPAC name | sodium 2-amino-4,6-dinitrophenolate |
| Other names |
Dinitram Dinitramin Sodium dinitramide 4,6-Dinitro-o-aminophenol sodium salt Sodium 4,6-dinitro-2-aminophenolate |
| Pronunciation | /ˈsəʊdiəm ˌdaɪˈnaɪtrəʊ ˈtuː əˈmiːnəˌfiːnɒksaɪd/ |
| Identifiers | |
| CAS Number | 36634-90-1 |
| Beilstein Reference | 136315 |
| ChEBI | CHEBI:51455 |
| ChEMBL | CHEMBL3341224 |
| ChemSpider | 21569728 |
| DrugBank | DB13305 |
| ECHA InfoCard | 100.017.641 |
| EC Number | 208-734-8 |
| Gmelin Reference | 107490 |
| KEGG | C14315 |
| MeSH | Dinitrophenols |
| PubChem CID | 12383 |
| RTECS number | ZE5425000 |
| UNII | CS16H7T7VU |
| UN number | UN1320 |
| Properties | |
| Chemical formula | C6H4N3NaO5 |
| Molar mass | 213.08 g/mol |
| Appearance | Yellow solid |
| Odor | Odorless |
| Density | 1.73 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -1.2 |
| Acidity (pKa) | 4.0 |
| Basicity (pKb) | 9.1 |
| Magnetic susceptibility (χ) | -39.0e-6 cm³/mol |
| Dipole moment | 6.56 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 261.6 J K⁻¹ mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -172.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1670 kJ/mol |
| Pharmacology | |
| ATC code | N03AX11 |
| Hazards | |
| Main hazards | Explosive; Harmful if swallowed; Causes skin and eye irritation; May cause respiratory irritation |
| GHS labelling | GHS05, GHS07, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H302 + H315 + H319 + H335 |
| Precautionary statements | P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Lethal dose or concentration | LD50 oral rat 300 mg/kg |
| LD50 (median dose) | LD50 (median dose): 175 mg/kg (oral, rat) |
| NIOSH | # 61788 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Sodium 4,6-Dinitro-2-Aminophenoxide: Not established |
| REL (Recommended) | 36 Months |
| Related compounds | |
| Related compounds |
4,6-Dinitro-2-aminophenol 2-Amino-4,6-dinitrophenol Sodium 2-aminophenoxide Nitrophenol derivatives Phenoxide salts |

