Sodium 3-Hydroxybutyrate: More Than Just a Compound
How History Shaped Sodium 3-Hydroxybutyrate
Looking back, researchers didn’t stumble upon sodium 3-hydroxybutyrate overnight. It arrived on the scientific scene as doctors and biochemists dug deeper into metabolic states like fasting and ketosis, tracing how the body keeps going when sugar gets scarce. The mid-20th century brought the real traction, when scientists began to realize the compound’s role in energy transport, especially in ketoacidosis and infant metabolic disorders. Sodium 3-hydroxybutyrate moved from a clinical curiosity to an important subject in labs and hospitals alike, as kids with energy metabolism issues found new hope through its supplementation. The real change came as researchers cracked the code on how our bodies make and use ketone bodies, and this compound became a bit of a household name for anyone digging into metabolic health, neurological diseases, or even endurance sports science.
Getting to Know the Product
Sodium 3-hydroxybutyrate isn’t some random white powder destined to sit on a shelf. In practical terms, it’s an organic sodium salt, derived from 3-hydroxybutyric acid. When it hits water, it dissolves well, making it fairly easy to administer orally or even in hospital settings through infusion. You can spot it in labs as a crystalline or slightly powdery substance with a slight salty taste. Its role as a key energy carrier in the body gives it a place in nutritional supplements and medical therapies alike. What’s unusual about this compound is that it doesn’t just serve as a dietary ingredient or a chemical curiosity; it’s already woven into the fabric of human metabolism, showing up whenever fats are broken down for fuel.
Physical and Chemical Properties Worth Attention
Talking chemistry, sodium 3-hydroxybutyrate stands out for its stability and solubility. In most labs, it presents as a colorless crystalline solid, fairly stable in air and showing no tendency toward rapid decomposition. Mix it with water or saline, and it dissolves without fuss—a property critical for both bench scientists and hospital pharmacists. The compound has a sweetish smell, faint enough not to offend, and doesn’t bring any nasty surprises under standard storage conditions. Thermally, it holds up until temperatures far beyond typical laboratory environments. Chemists will point out how its sodium salt form matters, since free 3-hydroxybutyric acid would present more handling headaches and wouldn’t suit ingestion or infusion nearly as well. Its predictability under routine lab use matters—a trait sometimes missing from more reactive molecules.
Technical Details and Labeling Practices
Anyone buying or working with sodium 3-hydroxybutyrate will want to check the label for accurate content percentage, trace impurities, and batch purity. The material typically comes standardized for sodium content, since the amount can impact medical outcomes or research reproducibility. Current industry standards often require a certificate of analysis showing at least 98% purity, with residual moisture levels and sodium ion content declared. Avoiding cross-contamination from other related ketones matters, especially if being used in clinical trials or for patient care. Safety labeling, transportation information, and clear hazard classifications—these aren’t just bureaucratic hoops but front-line defenses for anyone who handles the compound. Even with a strong safety profile compared to other chemicals, clarity and transparency on the label make life easier for researchers and caregivers.
How Preparation Shapes Its Use
The actual production process for sodium 3-hydroxybutyrate leans on classic organic synthesis. Start by sourcing either fermentation-derived or synthetic 3-hydroxybutyric acid. Neutralize it with a stoichiometric dose of sodium hydroxide, usually in aqueous solution, and impurities drop away with crystallization and washing. Some manufacturers prefer enantiomerically pure feedstock to keep the final product consistent—a nod to both regulatory pressure and the realities of biological specificity. Scale-up steps face their challenges, but the main hurdle remains cost and purity rather than feasibility. Keeping the process safe means closely monitoring pH, controlling water content, and filtering out any by-products. These basic steps decide whether what ends up in a pharmacy is suitable for a clinical setting or if it languishes on a chemical supply shelf.
Chemical Reactions and Room for Tweaks
Sodium 3-hydroxybutyrate itself doesn’t show wild reactivity under standard conditions, but it can undergo mild oxidation to acetoacetate or reduction under the right conditions. Chemists interested in making derivatives won’t find much challenge in esterification or conversion to related ketone bodies, opening up research possibilities. This adaptability supports development of analogs that might cross the blood-brain barrier more efficiently or demonstrate improved pharmacokinetics. Basic manipulation in the lab remains straightforward, benefiting from the compound’s clean baseline. For those in the medical field, what matters is its reliable behavior in biologic contexts; there, the molecule mostly serves as an energy shuttle, moving between tissues and helping bridge the gap for patients with metabolic shortfalls.
Common Names Pop Up Everywhere
Trying to keep track of sodium 3-hydroxybutyrate in literature or product catalogs means watching for several names: sodium beta-hydroxybutyrate, sodium 3-hydroxybutanoate, or simply “NaBHB.” Scientific articles occasionally stick to the IUPAC handle, but most researchers shorten it for convenience. Awareness of synonyms helps avoid confusion, especially during literature reviews or sourcing checks. This flexibility in naming reflects its centrality in both industry and academic contexts. For those jumping in from a clinical background, “beta-hydroxybutyrate” often gets used interchangeably, though the sodium version specifically matters for stability and dosing.
Staying Safe and Following the Rules
Working with sodium 3-hydroxybutyrate doesn’t raise the same level of alarm as more potent chemicals, but sticking to good laboratory practice and storage guidelines keeps incidents rare. Eye and skin protection remain a default, especially in environments where powders or concentrated solutions get handled. Keeping the compound dry avoids clumping and maintains flow characteristics, while a tightly sealed container preserves shelf life. Waste management stays simple; standard aqueous waste disposal usually fits. Regulatory bodies place sodium 3-hydroxybutyrate in a low to moderate risk bracket, but medical-grade material draws extra scrutiny for manufacturing controls, sterility, and traceability. Reporting requirements and training ensure that the substance gets used wisely without cutting corners on staff safety.
How Real Use Cases Drive Demand
Doctors and researchers turn to sodium 3-hydroxybutyrate not out of theoretical interest, but because it addresses real clinical needs. Kids with inborn errors of metabolism or certain mitochondrial disorders rely on it as a lifeline, converting chemical supplementation into real metabolic relief. Hospitals use it during severe hypoglycemic episodes, knowing it can backstop critical energy needs when glucose alone won’t do the job. Neurology researchers run studies on its neuroprotective effects—a topic that keeps drawing attention, especially with rising cases of dementia or traumatic brain injuries. Athletic trainers and biohackers dabble in its performance applications, betting that a supplemental ketone could push human boundaries. Doctors prescribing it keep a careful eye on dosing, bloodwork, and response, because while the molecule isn’t new, the contexts in which it gets used keep changing.
Research Opens Up New Possibilities
Research into sodium 3-hydroxybutyrate doesn’t rest easy on the basics. Scientists dig into neuroprotection, examining pre-clinical models for evidence that the compound shields brain cells against oxidative damage. Metabolism labs measure how it shifts energy usage, swapping carbohydrates for fats, hoping to nudge outcomes in epilepsy, Alzheimer’s, or exercise performance. Clinicians see anecdotal reports and preliminary trials suggesting benefits in heart failure patients, or those recovering from acute illness. The field isn’t just lip service—well-controlled trials and mechanistic studies collect hard data, parsing differences between oral supplements and intravenous infusions. As new delivery forms and dosing strategies emerge, the push for more rigorous safety data follows, bringing improved protocols and careful patient selection.
Toxicity Gets Close Scrutiny
Few stories are more sobering in clinical chemistry than unexpected side effects. Sodium 3-hydroxybutyrate, for all its metabolic compatibility, still demands a careful look at dose and duration. Reports show that, when used as directed, adverse events remain rare—think mild gastrointestinal issues, a bump in serum sodium, a transient effect on pH. Animal studies and human case reports haven’t flagged severe risks at customary dosing, but long-term effects in special populations (such as kids with metabolic disorders or older adults with kidney trouble) need more attention. Responsible scientists test for off-target effects, measuring everything from blood electrolyte levels to organ function. Establishing robust guidelines for chronic use and careful screening for interactions means toxicity doesn’t sneak up on users or care teams.
What’s Coming Next?
The future for sodium 3-hydroxybutyrate looks set for growth, not just in its old roles but in new arenas still being mapped out. The explosion of interest in ketogenic diets and exogenous ketone supplements pushes both consumer and clinical demand. Researchers track how the compound could support aging populations, those with rare neurological conditions, or people recovering from trauma. New formulations and delivery modes—slow release tablets, injectable forms, or combination therapies—aim to improve convenience and broaden the compound’s reach. Responsible stewardship means keeping real-world evidence ahead of marketing claims. Doctors, scientists, and everyday users look forward to data-driven answers and carefully monitored protocols. In a world searching for better energy management, sharper cognition, and novel approaches to chronic disease, sodium 3-hydroxybutyrate stands out not just for its history, but for its chance to meet today’s and tomorrow’s needs with clarity and scientific rigor.
What is Sodium 3-Hydroxybutyrate used for?
What is Sodium 3-Hydroxybutyrate?
Sodium 3-hydroxybutyrate has a mouthful of a name, but its story is about as real as science gets. This compound comes from the body’s process of breaking down fat for energy. The body naturally produces it when glucose runs low, such as during fasting or low-carb eating. Here, “ketones” often pop up in conversation, and sodium 3-hydroxybutyrate is part of that discussion. It’s the sodium salt form of beta-hydroxybutyrate, one of the main ketone bodies behind the scenes in metabolism.
Why Doctors Use It
Doctors turn to sodium 3-hydroxybutyrate mostly for some of the rarest of medical conditions. Take certain inherited metabolic disorders. In folks with issues using energy from regular carbs or fats, sodium 3-hydroxybutyrate gives them a backup plan, feeding their cells when the main power lines have trouble. Conditions like multiple acyl-CoA dehydrogenase deficiency (MADD) or certain mitochondrial disorders show up in real life, not just in textbooks. For families facing these diseases, every treatment option matters.
Researchers have published studies, such as the one in Brain in 2016, that showed sodium 3-hydroxybutyrate made a difference for children with specific metabolic problems. Patients who received the supplement showed better cognitive and motor function, enough to convince more clinicians to offer it in severe cases. This isn’t a miracle cure; it’s a tool that makes life a little easier for people who feel forgotten by most of medicine.
Interest from Athletes and the Keto Community
Supplement companies have caught wind of sodium 3-hydroxybutyrate, especially among athletes and those experimenting with ketogenic diets. The idea is to give the body a shortcut to ketosis: take in the ketone salt, get the energy, skip the uncomfortable beginnings of a strict low-carb diet. Some users swear by better mental focus or improved endurance. Studies in the journal Cell Metabolism explored ketone salts during cycling, noting mild improvements in some performance markers. Still, not everyone gets the same results, and research in healthy populations stays mixed. The price tag also runs high for a supplement that doesn’t always stack up against a well-structured diet plan.
On the other hand, dumping lots of extra ketone salts into the system may come with digestive trouble. Too much sodium also raises a red flag, especially for folks with blood pressure or kidney concerns.
Research and the Future
There’s curiosity growing around sodium 3-hydroxybutyrate for brain health. Scientists want to know if it can help with conditions like Alzheimer’s or epilepsy, where the brain’s regular energy sources run short. The hope is that ketone supplements could offer another way to keep those neurons firing. Early animal studies offer hints, but hard proof in people hasn’t arrived.
Access remains an issue. Unlike many medications, sodium 3-hydroxybutyrate often falls outside insurance coverage, with imports filling the gap. The European Medicines Agency and FDA both watch closely, but most access relies on special permission or hospital programs for rare diseases. Broader use calls for more research, honest safety data, and responsible advice from professionals who put patient needs above quick trends.
What Comes Next
Sodium 3-hydroxybutyrate doesn’t grab headlines like new cancer drugs or blockbuster pain relievers. For a small group, it offers a shot at a more regular life. For athletes and biohackers, it’s another tool to test, though results often depend on the discipline of the person using it. Every step forward shows how old molecules can find new importance in medicine. Real-world trials and honest conversations between doctors and patients keep its story going.
Is Sodium 3-Hydroxybutyrate safe to take?
Looking at What Sodium 3-Hydroxybutyrate Actually Is
Sodium 3-hydroxybutyrate, often called a ketone salt, gets a lot of attention in the world of diet and supplements. Some people use it to try to boost energy or sharpen mental focus. It comes from the same family as other ketones that our bodies make during fasting or when cutting carbs. Instead of relying on natural ketosis, folks turn to these supplements hoping to get quicker effects.
Potential Benefits—And Where the Evidence Falls Short
Some early research suggests sodium 3-hydroxybutyrate may provide an alternative energy source for the brain. It makes sense for people in certain medical situations, like those with specific metabolic disorders or struggling with epileptic seizures, and in those cases under close doctor supervision.
Many folks outside those groups try ketone supplements for athletic performance, mental clarity, or quick weight loss. Here, scientific support looks much thinner. Most published studies either focus on different ketone compounds or involve small groups of people, so firm claims stay shaky.
Risks and Side Effects—What We Know So Far
Too much sodium in the body can strain the kidneys and hike blood pressure. Sodium-based ketone products naturally add to your salt load. Research flags the risks to those with heart, kidney, or blood pressure problems. Even people without health problems report stomach upset, headaches, or muscle cramps after taking these.
Long-term safety is still a big unknown. Most trials run for a few weeks at most. Nobody can guarantee harmless results after months of regular use. Supplements aren’t held to the same safety standards as medicines, so purity and dose can shift from one manufacturer to another. People get a false sense of security just because a product is for sale on a health food shelf.
Medical Use, Self-Experimentation, and What Doctors Say
Hospitals may use sodium 3-hydroxybutyrate for rare metabolic conditions under skilled supervision. This isn’t the same as picking up a supplement bottle at the shop. Doctors look at blood levels, organ function, and other medications before using it.
The FDA does not approve over-the-counter ketone salts for any health condition. European regulators treat them with caution. The scientific community isn’t ready to sign off on these products as safe or effective for general consumers.
Better Solutions and Safer Paths to Health Goals
People chasing more energy or mental sharpness often try many shortcuts over time. A well-balanced diet, good sleep habits, and regular physical movement still offer more lasting, proven benefits, without extra strain on the heart, brain, or kidneys. For anyone thinking about trying sodium 3-hydroxybutyrate, checking with a healthcare provider comes first. Honest conversations with a registered dietitian or doctor can help sort hype from reality before putting anything new in your body.
Looking for short-term gains without understanding long-term effects often leads to disappointment—or even setbacks. Health usually rewards patience, real food, and a bit of common sense.
What are the possible side effects of Sodium 3-Hydroxybutyrate?
The Risks that Come with Medical Ketones
Sodium 3-hydroxybutyrate has made its way into emergency rooms and doctor’s offices as a prescription therapy for rare metabolic conditions. Usually prescribed for children with certain epileptic disorders or metabolic diseases, this salt mimics the effects of ketosis—the state usually produced by fasting or very low-carb diets. It's a medical innovation that often brings hope to families dealing with tough diagnoses. Alongside these potential benefits, people need to know about the possible downsides. As someone who’s seen families work through long medication guides and complicated treatment schedules, the side effect list can feel overwhelming, especially with newer treatments like this one.
Understanding What Can Happen
The body handles sodium 3-hydroxybutyrate differently than other nutrients. One of the bigger concerns centers on salt load. This compound brings a hefty dose of sodium. If a patient already deals with high blood pressure or heart problems, extra sodium can push their numbers in the wrong direction. That kind of complication isn’t minor, especially for kids with fragile health or adults with a long health history. Research has shown that too much sodium relates directly to heart disease risk in the general population.
Stomach troubles pop up often with this compound. Nausea, vomiting, and cramping hit some users right after dosing. This is especially true when higher amounts are given or when someone’s not used to ketone supplements. I spoke to a parent whose child started therapy and ended up skipping school more because of stomach aches and loose stools, not the original metabolic disorder. That’s a tradeoff no family wants to have to make.
Shifts in Chemistry and Safety Checks
Another issue comes from shifting the body’s internal chemistry. Sodium 3-hydroxybutyrate, as a ketone salt, impacts blood pH. That means the body can swing toward being too basic, a condition called metabolic alkalosis. Mild forms of this can cause confusion, muscle twitching, or tingling. More severe cases can land someone in the hospital for IV fluids and electrolyte correction. These swings show up in lab tests—something doctors check for, but patients don’t always notice until symptoms appear.
On top of that, shifting the body’s electrolyte balance can throw off potassium and calcium levels. Both of those minerals matter for heart rhythm and muscle function. I remember one case where a teenager had heart palpitations, only to find out potassium had dropped from frequent dosing. The fix meant monitoring labs closely and adjusting other meds, which built extra steps into everyday care.
Room for Improvement
Doctors and pharmacists want to help patients, but predictable side effects from high sodium or GI distress frustrate both sides. One practical solution could be new formulations with less sodium, or slow-release forms that ease absorption. Dieticians sometimes step in to help families manage salt intake elsewhere in the diet. Tech could help too—apps that track side effects or home testing kits for electrolytes make quick action more likely.
Every medicine comes with tradeoffs. With sodium 3-hydroxybutyrate, the risks linked to sodium, gut upset, and chemistry shifts demand real attention. Families living with rare disease often don’t get a break, but simple changes in monitoring and drug design could tip the balance toward better and safer use for everyone involved.
How should Sodium 3-Hydroxybutyrate be dosed?
Understanding Sodium 3-Hydroxybutyrate and Its Importance
Sodium 3-hydroxybutyrate, known to some as a supplemental ketone salt, catches plenty of attention among researchers and clinicians. People talk about its use for managing rare metabolic disorders, like certain inborn errors affecting energy production. Others explore it as a way to support brain energy in tough situations—think pediatric epilepsy or complicated mitochondrial disease. There’s even some buzz in athletic circles, where folks want to boost workout recovery or mental clarity. Yet every benefit circles back to one simple question: how much should someone take?
Moving Away from Guesswork
I remember asking a pediatric metabolic doctor what stood out when figuring out the right dose for kids missing enzymes needed for regular ketone production. She didn’t give a magic number. The only honest answer she could give was: start low, monitor closely, go by response and labs. She’d seen children respond to as little as 400 mg per kilogram of body weight a day, divided over several doses. Some needed a bit more. Too much often led to stomach upset, loose stools, or occasional shifts in blood salt levels. So, moderation and frequent checks matter—a standard “one size fits all” mindset falls short.
In adults, metabolic physicians seem to lean toward similar calculations but adjust for weight and general tolerance. Research from European clinics points to splitting the total daily amount into two or three portions, especially in patients who have trouble making their own ketones under stress. It’s common to check blood and urine ketones, sodium, and acid-base balance at least twice a week when the regimen starts, and then scale back to monthly as people settle into a routine. Families at home, as I’ve seen, often keep a journal, noting physical energy, mental clarity, and any gut complaints—data that guides every tweak and adjustment.
The Risks and Looking Out for Trouble
Sodium isn’t a harmless bystander in this formula. Every dose brings more sodium into the bloodstream, which can add strain to the heart, especially for those with kidney concerns or underlying high blood pressure. A 2022 review in the Journal of Inherited Metabolic Disease drives home the risk of overloading the body’s salt-handling systems. Kids with mitochondrial diseases may already face heart or kidney stress, so doctors double-check labs and blood pressure before raising the dose at all.
Some people expect a miracle and take too much, hoping for a cognitive or exercise lift, only to battle nausea or headaches. No over-the-counter supplement can guarantee safety or purity, so each change should run through a licensed prescriber, ideally with experience in rare metabolic illnesses. Trust in quality of compounds matters almost as much as the amount—something that has led most metabolic clinics to source medical-grade material directly from hospital pharmacies, not commercial supplement vendors.
Paving a Safer Path Forward
Doctors and scientists would do well to keep expanding published case reports documenting each dosing journey and the symptoms patients face—both benefits and side effects. More everyday people now have access to commercial sodium 3-hydroxybutyrate, so restrictions and labeling regulations deserve real scrutiny from regulators. Getting dosing right means more than numbers; it builds on relationships, honest feedback, and medical supervision that listens to lived experience above all else. New research will shape tomorrow’s protocols, but until then, a careful and personal approach stays essential.
Can Sodium 3-Hydroxybutyrate be taken with other medications?
Understanding Sodium 3-Hydroxybutyrate
Sodium 3-hydroxybutyrate, used for some rare metabolic conditions and thought of by some as a possible way to mimic ketosis, has become a talking point for both doctors and curious patients. Unlike the everyday supplements picked up at the pharmacy, this compound brings real pharmacological action. That means anyone interested in using it needs to think hard about how it plays with other medicines.
The Chemistry in the Mix
A quick look at chemistry helps here. Sodium 3-hydroxybutyrate acts as a ketone body, which the body can use for energy when glucose is low. While its main use lands in rare genetic disorders, people have asked if it fits next to other prescriptions, like anti-epileptic drugs or meds for metabolic control.
Drug interactions aren't just guesswork. Each medicine can change how another works—sometimes speeding things up, sometimes slowing down enzymes in the liver, or shifting electrolyte levels like potassium and sodium. With sodium 3-hydroxybutyrate, the sodium load matters, especially for those already taking blood pressure pills, diuretics, or medications that tip the fluid balance. Someone who’s had heart problems or chronic kidney disease knows too much sodium can nudge the body in the wrong direction, raising blood pressure or putting stress on the kidneys.
The Human Side of Polypharmacy
No science course matches the lessons from watching people stack prescriptions. I remember watching a family member juggle meds after a stroke—the neurologist carefully cross-checking each new prescription against the old ones because one wrong move could mean dizziness, confusion, or worse side effects. The same care applies here.
Some anti-seizure medications (like valproic acid or carbamazepine) already alter liver function and certain metabolic pathways. Add sodium 3-hydroxybutyrate to the mix, and the picture changes. Research is still thin in this space, but small case studies have surfaced warning about possible changes in sodium, shifts in acid-base balance, and altered seizure control.
Why Honest Conversations with Doctors Matter
Clinicians—the ones in the trenches—spot these drug interactions faster than any online database. Rushed appointments get in the way, but finding time to ask questions can save a lot of pain. Patients who bring a full list of everything they take, including vitamins and “natural” pills, help their doctor spot the sharp edges in advance. Pharmacies also offer medication reviews, and that extra set of eyes often catches problems missed during the hurried moments in the clinic.
Finding Safe Ground
Solutions start with transparency. Patients should always tell their healthcare provider about plans to add in sodium 3-hydroxybutyrate. Doctors have access to resources that compare possible interactions, but no resource replaces practical judgment. For anyone on heart medications, blood pressure drugs, or treatments affecting liver or kidney function, the risks go up. Monitoring blood sodium, watching for odd symptoms, and scheduling regular follow-ups cut down on surprises. Ultimately, mixing this compound with other prescriptions rarely fits a “one size fits all” formula, so clear communication lays the best groundwork for safety.
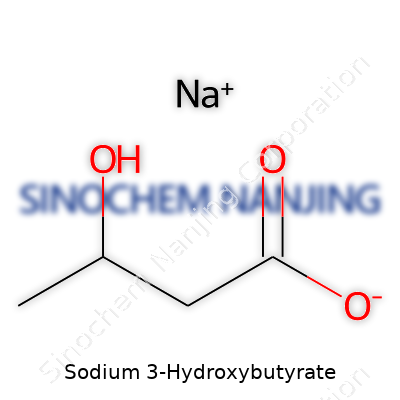
| Names | |
| Preferred IUPAC name | Sodium 3-hydroxybutanoate |
| Other names |
Sodium beta-hydroxybutyrate Sodium 3-hydroxybutyrate Sodium β-hydroxybutyrate Sodium 3-hydroxy-butyrate |
| Pronunciation | /ˈsəʊdiəm θriː haɪˌdrɒksɪˈbjuːtɪreɪt/ |
| Identifiers | |
| CAS Number | 15746-33-5 |
| Beilstein Reference | 2038650 |
| ChEBI | CHEBI:133349 |
| ChEMBL | CHEMBL1201110 |
| ChemSpider | 21541254 |
| DrugBank | DB14555 |
| ECHA InfoCard | echa-ec-253-703-0 |
| EC Number | 206-536-7 |
| Gmelin Reference | 62635 |
| KEGG | C01089 |
| MeSH | D017711 |
| PubChem CID | 23665745 |
| RTECS number | EL8450000 |
| UNII | F2J6X60A5X |
| UN number | 2811 |
| Properties | |
| Chemical formula | C4H7NaO3 |
| Molar mass | 126.09 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.268 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -3.2 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 9.7 |
| Basicity (pKb) | 9.3 |
| Magnetic susceptibility (χ) | -57.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.422 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.81 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 228.4 J∙mol⁻¹∙K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -669.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | ΔcH⦵298 = -2242 kJ/mol |
| Pharmacology | |
| ATC code | A16AX30 |
| Hazards | |
| Main hazards | Not hazardous according to GHS classification |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | No signal word |
| Hazard statements | Hazard statements: Causes serious eye irritation. |
| Precautionary statements | Precautionary statements: P264, P270, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | NFPA 704: 1-1-0 |
| Flash point | > 100 °C |
| Lethal dose or concentration | LD50 (rat, oral): 16,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 16,600 mg/kg |
| NIOSH | RN:914-927-0 |
| PEL (Permissible) | Not Established |
| REL (Recommended) | 500 mg/kg bw |
| Related compounds | |
| Related compounds |
β-Hydroxybutyric acid Sodium acetoacetate Acetoacetic acid Sodium butyrate 3-Oxobutanoic acid |

