Sodium 2,4-Dinitrophenoxide: A Close Look at its Journey and Real-World Impact
Historical Development
Sodium 2,4-dinitrophenoxide ties back to an era when chemical craftsmanship demanded a balance between curiosity and necessity. Chemists first produced dinitro derivatives of phenol over a century ago, driven by the hunger to manipulate aromatic compounds for dyes, explosives, and agricultural chemistry. In those early days, the milk-glass flasks and copper kettles of European labs filled with new possibilities, but the knowledge passed down was hard-won, cut from experience rather than digital simulation. My own chemical roots begin in a dusty university lab where handling reactive nitro-aromatics taught me caution and deep respect for the unpredictable. Whether students or seasoned inventors, chemists quickly learned that compounds like sodium 2,4-dinitrophenoxide packed both utility and risk, promising breakthroughs if you learned their language. Its adoption in the industrial age mirrored the growth of synthetic intermediates—vital yet rarely acknowledged by those outside the trade. This compound carved its niche through the tireless, sometimes hazardous, experiments that mirrored the ambitions and dilemmas of 20th-century chemistry.
Product Overview
This isn’t a compound that shows up under a spotlight, and yet it underpins a surprising set of modern processes. Sodium 2,4-dinitrophenoxide, the sodium salt of 2,4-dinitrophenol, serves as a stepping stone for synthesizing more complex molecules. Organic chemists cherish it as a reliable intermediate in nitroaromatic chemistry, especially where the phenoxide functional group improves solubility and reactivity. The formula, C6H3N2O5Na, only partly tells the story. Its use stretches from chemical synthesis to process optimization in specialty materials, sometimes even popping up in exploratory agricultural work. My years in specialty synthesis revealed that chemists rely on such intermediates to achieve selectivities and purities that would otherwise take months to engineer. Many innovative processes get their start from unassuming salts like this one—each batch, in essence, holds the potential for new discoveries.
Physical & Chemical Properties
Taking sodium 2,4-dinitrophenoxide into your hands (with gloves, always), you see a yellow-orange powder that dissolves quickly in water, thanks to the phenoxide ion. This color signals the strong conjugation in the aromatic system and hints at the molecule’s photochemical behavior. In solution, its ionic form encourages a swift transport of electrons, making it reactive to both oxidants and reductants. From my own benchwork, I remember the unmistakable smell of nitroaromatics filling the fume hood, pushing home the reality that what makes a compound useful often makes it hazardous. Storage brings its own lessons—heat, moisture, and contamination can tip the scales from stable to hazardous without warning. This is no waxy, stable block—exposure and even mild acidic conditions can trigger decomposition, creating challenges for transport and storage. Sodium 2,4-dinitrophenoxide, like many nitroaromatic salts, resists casual handling and demands focus from anyone in proximity.
Technical Specifications & Labeling
Technical details aren’t just bureaucratic hurdles—they’re everyday shields. Labeling often states purity levels, typically above 97 percent, but what matters in the lab is confirming the absence of by-products, particularly unreacted dinitrophenol or sodium contaminants that might steer the chemistry off course. Chemical suppliers mark hazardous properties clearly: toxic if swallowed, contact hazard, and environmental contaminant. Overconfidence on the bench can lead to costly mistakes, which I learned the hard way after a minor but memorable spill that reinforced the need to triple-check seals and compatibility with other reagents. Regulations differ by country, but informed chemists track SDS sheets, GHS pictograms, and local transport codes—omitting any of these can cost time, money, or safety. Calibration with analytical protocols such as UV-Vis absorbance or HPLC often ensures product profiling, as standardization underpins both trust and traceability in the supply chain.
Preparation Method
Building this compound on a practical scale usually starts with 2,4-dinitrophenol and a stoichiometric excess of sodium hydroxide in water or alcohol as a solvent. The reaction’s simplicity masks a wealth of nuance. Stir too quickly and you create foam that overflows glassware. Go too slow and you risk incomplete reaction and wasted precursor. I’ve watched students wrestle with the fine line separating efficiency from runaway exotherms. Dialing in the pH controls salt formation, while too much base generates side products. The purification process can force you to choose between economy and performance—use cold precipitation for cleaner product, or settle for a simple filtration and risk impurities landing in your next synthesis. Large-scale production often calls for automation, but even then, real-world production lines need constant maintenance to prevent fouling and off-spec batches. These lessons, learned across a career, underline the need for methodical thinking, practical patience, and the flexibility to adapt recipes as starting materials or regulations evolve.
Chemical Reactions & Modifications
The fun begins once you have this salty yellow powder in hand. In organic synthesis, sodium 2,4-dinitrophenoxide reacts as a nucleophile, displacing halides or activating positions on other aromatic rings. In my time synthesizing complex heterocycles, I’ve drawn on its utility to swap out leaving groups and assemble more intricate scaffolds. Its tendency to participate in nucleophilic aromatic substitution reactions makes it a winner for creative molecular construction. Some researchers modify it with electrophiles, chasing specialty dyes or pharmaceuticals, while others focus on reduction reactions that tease out new functional groups. The same electron-rich phenoxide that encourages reaction also spawns side chemistry if you don’t monitor conditions closely: over-reduction, unwanted coupling, hydrolysis of sensitive groups. Careful selection of solvents, temperature, and stoichiometry minimizes detours and maximizes the return on both effort and investment.
Synonyms & Product Names
Multiple names circulate in research circles, and each brings a slightly different flavor of history or intended use. Besides “sodium 2,4-dinitrophenoxide,” you might see references to “sodium dinitro-o-phenol” or simply “dinitrophenoxide sodium salt.” Identifiers like CAS numbers are more than regulatory arcana; from personal experience, searching by these precise numbers shaves hours off literature dives and avoids cross-contamination with similar but subtly different compounds. Academic and commercial suppliers may tweak these names to flag specific grades or purity levels. The key isn’t memorizing every synonym, but staying alert to context—nomenclature drifts between subfields, and context determines whether a name points to the substance you want or to something dangerously off-mark.
Safety & Operational Standards
Safety around nitroaromatics, especially phenoxides, boils down to deep-rooted habits. Handling requires gloves and real eye protection, not the decorative kind that sits in a drawer. Airflow is non-negotiable: vents, fume hoods, and scrubbers protect against inhalation and accidental release. In my working years, I’ve witnessed too many seasoned chemists get complacent, underestimating the persistent toxicity of nitrophenol derivatives. Ingestion, skin contact, and inhalation all threaten health—rapid onset of symptoms like headache, nausea, and even serious systemic effects become real dangers. The environmental hazard grows even greater, as run-off contaminates water and soil. Waste management must follow local hazardous waste disposal requirements; turning a blind eye here only pushes the risk downstream. Ongoing education and open discussion about near-misses build a culture where safety matters as much as technical prowess.
Application Area
This compound doesn’t parade around as an everyday household chemical, but its reach travels far. Synthetic chemistry claims it as a prized intermediate, especially for constructing more complex nitro derivatives. The most visible uses slip into specialty dyes, pesticides, and sometimes as a building block for pharmaceutical research. Some applications explore controlled energy-release systems, where the stability of the nitro group becomes both asset and liability. Having worked on colorant chemistry, I’ve seen researchers lean on the unique chromophore provided by the conjugated dinitrophenyl group to craft new visible indicators and responsive coatings. These innovations start in small flasks but sometimes find their way into smart materials or agricultural improvements. Its role as a starting material keeps it relevant, with new uses emerging as research evolves.
Research & Development
One of the eye-opening things about this field today lies in how research now spans bench to bioinformatics. Advances in analytical chemistry, crystallography, and in silico modeling help teams predict and tune the reactivity of 2,4-dinitrophenoxide well before any flask fizzes. While much of the early work felt like trial and error, today’s methods rapidly screen for environmental persistence, reactivity hotspots, and biological activity. I learned that collaboration between organic chemists, toxicologists, and environmental scientists now shapes the direction of research, not just for output, but for responsibility. Outside big-budget labs, creative use of this salt is growing in sensor technology, coordination chemistry, and even green chemistry experiments designed to replace heavier environmental burdens. Funding bodies now push for dual goals: performance and sustainability, with the hope that tomorrow’s products can harness the same power with a lighter environmental footprint.
Toxicity Research
Toxicity shadows every use of sodium 2,4-dinitrophenoxide. Its parent, 2,4-dinitrophenol, is infamous for uncoupling cellular respiration, making it acutely toxic across mammalian and aquatic systems. Early studies showed how minimal doses disrupt metabolic pathways, a fact that marks every protocol and justifies rigorous exposure limits. Watching the development of toxicity assessment, I noticed how modern in vitro assays and computational toxicology now predict impacts long before animal testing. Regulatory agencies monitor not just occupational exposure, but downstream effects through soil and water contamination. Chronic exposure links to respiratory symptoms and possible carcinogenicity. Disposal remains thorny—accidental release can linger in soil and water, outlasting the careers of the people who handled it. This drives ongoing calls for green chemistry solutions, safer alternatives, and improved remediation techniques. Advances in protective equipment and process automation mean that compared to 40 years ago, scientists can operate with a greater degree of security—but no one working with these materials ever truly relaxes.
Future Prospects
The story for sodium 2,4-dinitrophenoxide doesn’t end with its historic uses. As lab technology advances, demand for tailored building blocks grows in electronics, sensors, and medical diagnostics. Innovators now look to aromatic nitro compounds for roles in responsive materials or as triggers in controlled-release systems. The real progress will come from chemists who reimagine how to control reactivity and reduce environmental impact, maybe through smarter catalyst systems, greener manufacturing protocols, or more robust waste management. Peer networks, improved regulatory frameworks, and open-access toxicology databases offer hope that tomorrow’s research will prevent the pitfalls of yesterday. In my own experience, the most meaningful changes happen when regulatory insight, research funding, and on-the-ground know-how meet. If history serves, sodium 2,4-dinitrophenoxide will keep showing up wherever chemical complexity meets creative problem-solving, its risks kept in check by diligence and the collective memory of those who learned its lessons firsthand.
What is Sodium 2,4-Dinitrophenoxide used for?
Sparking Reactions in the Lab
Sodium 2,4-dinitrophenoxide carries a name that might sound unfamiliar outside a chemistry classroom, but its uses reach further than a dusty shelf in a school lab. Chemists often work with this compound to kick-start reactions that need a solid nudge. The nitro groups on the molecule power up its reactivity. With those nitro groups in place, this compound steps into the role of a strong nucleophile in organic synthesis. That means it helps build new molecules, including dyes, pharmaceuticals, and even agricultural chemicals.
In research, it isn’t just about mixing and waiting. A chemist uses sodium 2,4-dinitrophenoxide to break bonds and forge new ones, opening up different routes to produce valuable compounds. This approach saves time, money, and sometimes stays away from nastier or more toxic chemicals. The safety profile and ease of handling matter here—no one wants to trade one problem for another.
Link to Making Dyes and Colors
Dye manufacturers know this chemical well. In the world of colored fabrics and printed images, sodium 2,4-dinitrophenoxide takes part in reactions that create intense yellows, oranges, and reds. The textile industry needs these bright, lasting dyes. Without stable molecules like this one, colors fade or wash out. Investing in the right kind of building block means your new shirt keeps its color longer, and producers avoid the waste from botched dye batches.
Pharmaceutical Pathways
Antibacterial drugs and other medicines sometimes start with sodium 2,4-dinitrophenoxide. It doesn’t end up in your stomach, but it lends a hand during manufacturing. Its special shape and properties help form bonds in molecules that end up fighting infection or inflammation. Chemists aim for efficiency, steering away from routes that produce lots of waste. Research suggests that this compound keeps the process cleaner and more controlled. For anyone interested in affordable medicine, that’s a win.
Sparking Change, But at a Cost
With benefits come concerns. Safety is a question that never goes away. Sodium 2,4-dinitrophenoxide comes from dinitrophenol, a name that raises eyebrows among toxicologists. Dinitrophenol has a reputation for dangerous side effects if ingested, thanks to some tragic misuse in diet pills nearly a century ago. Anyone handling sodium 2,4-dinitrophenoxide needs to respect gloves, goggles, and fume hoods. Waste disposal matters, too. Dumping any nitro compound into waterways spells disaster for wildlife.
In practice, industry standards push for tight controls. Modern labs and manufacturers typically track every gram. Accidents still happen; the safer route is always backing up chemical supplies with training. Regulators in many countries demand thorough record-keeping for chemicals like this one, and audits catch surprises before they turn into bigger problems.
Looking for Smarter Substitutes
Green chemistry keeps nudging industries to rethink old habits. For years, sodium 2,4-dinitrophenoxide filled a niche. Newer research brings safer, less toxic reagents onto the market. Some companies now try to swap in these alternatives whenever possible, especially in teaching labs or small batch processes. For large-scale work, finding a balance between reactivity and impact on health and environment will stay important.
Innovation often comes from unexpected problems. The push to lower risk, cut costs, and increase safety drives the hunt for better chemicals. Sodium 2,4-dinitrophenoxide still plays a supporting role in plenty of industries, but every year brings a new round of ideas on how to do the job with less fallout for people and the environment.
What are the safety precautions when handling Sodium 2,4-Dinitrophenoxide?
Recognizing the Risks
Sodium 2,4-dinitrophenoxide doesn't show mercy. Anyone inside a lab who has dealt with aromatic nitro compounds learns this fast. The bright yellow color is easy to spot, but it hides a nasty secret: this stuff is toxic, reactive, and can burn your skin or your airways. I remember my first real safety drill in a university chemistry lab. There, we don't just read paperwork. We role play scenarios with chemical spills. You learn from mistakes you’d rather not make with the real thing. People sweat under their goggles for good reason.
Why Personal Protection Means Everything
No one wants to end their day at the hospital because they skimped on gloves or goggles. Good gloves—nitrile, not those flimsy latex ones—matter. Chemical splash goggles keep your eyes protected from tiny splatters. Some old-timers in labs wear lab coats that look worn, but a torn sleeve can give sodium 2,4-dinitrophenoxide the perfect entry point to your skin. Fast action helps, too. Wash off spills right away using the emergency shower or eyewash, even if you think it’s just a drop. Medical data and anecdotal stories both show that quick rinsing prevents deep tissue injuries and scary health risks down the line, like kidney damage.
Taming the Fumes and Dust
Airborne dust from this compound creates invisible danger. More than a few people have discovered an irritating cough after working with dry powder in poorly ventilated rooms. Fume hoods aren't there for looks—they catch dangerous particles and draw them away from your lungs. I spent a summer in a research group analyzing toxic residues. The only reason no one developed health issues: our PI enforced a strict “fume hood or no work” rule. Inhaling even low levels puts long-term health on the line. Reports from occupational safety surveys tie improper ventilation to higher rates of respiratory illnesses among lab techs who handle chemicals like these.
Labeling and Storage—No Shortcuts
Poor storage turns an average lab into a powder keg. Sodium 2,4-dinitrophenoxide demands a cool, dry, well-marked space—never next to acids or combustible materials. A spark, spilled liquid, or even a knocked-over beaker changes everything in seconds. Getting lazy about labeling doesn’t just put your lab in danger. Unlabeled containers kill. Just read any chemical incident report database. Those tragic stories almost always start with a mystery bottle and someone in a hurry.
Emergency Plans That Actually Work
Everyone jokes about fire drills until a real accident happens. I’ve watched teams freeze when alarms sound because they never practiced more than the basics. Real emergency plans get tested in drills. The people who pay attention to exit routes, spill protocols, and phone trees stay safe while confusion reigns. Direct access to material safety data sheets, printed and in plain view, saves valuable minutes. Data from agencies like OSHA shows that properly drilled staff respond faster and with fewer mistakes, which translates to fewer injuries and deaths.
Training the Next Generation
Good habits stick when senior staff actually teach, not just supervise. I learned more from a mentor’s five-minute walk-through than from an hour buried in a safety manual. Visits from environmental health officers or safety trainers make things real, too. Labs that invest in hands-on training for their students and technicians end up on the right side of accident statistics. Safe handling doesn’t just protect individuals—it keeps whole teams alive and well enough to finish the project and make it to next semester.
How should Sodium 2,4-Dinitrophenoxide be stored?
Why Storing Chemicals Demands Attention
Anyone who’s worked around a chemical storeroom knows that good storage makes all the difference. Sodium 2,4-Dinitrophenoxide isn’t your average shelf dweller. This yellowish powder comes with a set of risks that people sometimes overlook. In my early lab days, someone stashed a reactive compound near a rusty sink. It ended with an emergency call and a lesson none of us forgot.
Recognizing What’s at Stake
This substance reacts with acids, metals, and oxidizers faster than most, so mistakes with storage could lead to more than cleanup duty. OSHA files warn of fire and explosion if the chemical comes in contact with incompatible substances. National Institute for Occupational Safety and Health recommends keeping it away from moisture—water contact releases toxic gases. These guidelines have teeth for a reason. Chemical burns and lung issues aren’t just textbook warnings; they show up in ERs.
Choosing a Place: Out of Sun, Out of Harm’s Way
Store sodium 2,4-dinitrophenoxide in a dedicated corrosive cabinet. Preferably, use a steel-lined cabinet with self-closing doors. It should live far from open flames, direct sunlight, and any spot with high humidity. Most seasoned lab folk use temperature controls and humidity indicators to avoid surprises. Don’t even think of putting it near acids, bases, or oxidizers. Over my years in university labs, the best storage spots have limited foot traffic and strong security measures—a lock beats a label any day.
Container Matters: Beyond the Original Packaging
The original container makes sense if there’s no sign of damage. If the cap cracks or the seal fails, get a chemical-resistant bottle with a solid, airtight closure. Polyethylene or glass with plastic-lined caps keeps out moisture. A mistake I saw—someone used a metal lid. Corrosion led to a slow leak before anyone noticed. Replace damaged containers right away. Don’t transfer between bottles unless absolutely necessary; every transfer adds risk.
Labeling and Documentation: Accountability Counts
Every jar or bottle needs a bold, legible label. Include the chemical name, date received, and hazard symbols. When a rotating group uses the chemical, everyone should sign a usage log. Tracking usage is more than bureaucracy; it stops dangerous guesswork. In my time working with shared spaces, clear logs caught a case where the compound’s expiration was missed by months.
Ventilation and Emergency Equipment
Good air flow makes accidents much less likely. Never keep sodium 2,4-dinitrophenoxide in a sealed, stuffy space. Most chemistry stockrooms use mechanical ventilation or a vented cabinet. Eyewash stations, safety showers, and fire extinguishers need to be within easy reach. A class ABC or class D extinguisher deals with the type of fire this chemical could start.
Training Makes a Difference
Seasoned workers run drills. New students and techs need to practice spotting spills, leaks, and unsafe conditions. Training isn’t a bureaucratic step—it’s the best way to keep people safe. One overlooked protocol or missing glove can mean a trip to the hospital. I’ve seen experienced staff intervene just in time because they remembered dry chemical spills don’t mix with wet cleanup. These skills prevent headlines, not just accidents.
Solutions: Building Better Habits
Fixing storage issues starts with honest audits. Bring in a fresh set of eyes every six months. Swap out weak containers. Update logs. Don’t let familiarity lead to shortcuts. Sometimes management skips these steps to save a few bucks. Later, the costs stack up in ways no one wanted. By building routines around airtight containers, honest labeling, smart cabinet placement, and straightforward training, everyone walks away safer. That’s not just chemical hygiene—it’s common sense built by hard lessons.
What is the chemical formula of Sodium 2,4-Dinitrophenoxide?
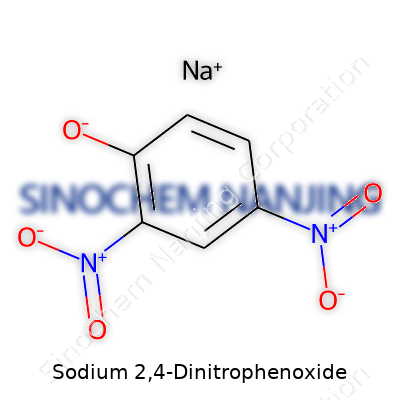
Chemical Formula: NaC6H3N2O5
Sodium 2,4-dinitrophenoxide comes up a lot in labs and classrooms. The formula, NaC6H3N2O5, packs more history than one might think at first glance. At its core, this substance is just a phenol ring loaded up with two nitro groups, one in the second spot and the other in the fourth, all sitting together with a sodium ion tagging along. It’s simple, yet it represents a class of organic compounds that play a part in real-world chemistry and industry.
In my years working alongside other chemists, a compound like this can become the hinge for a whole range of reactions. Pull off its sodium and you get 2,4-dinitrophenol, which brings its own danger and usefulness. Sodium 2,4-dinitrophenoxide has been used as an intermediate in the creation of dyes, explosives, and even some pharmaceuticals—not just as a curiosity for the keen-eyed student.
Back in college, we handled related compounds in synthesis labs. Some of us thought these reactions were routine, but the bright yellow color left on the glassware told us otherwise. The reason for the color? Those nitro groups. They don't just look interesting, they give the molecule power, both in reactivity and in what it can do for industry.
Importance Outside the Textbook
This formula represents more than numbers and letters on a whiteboard. Take the case of its cousin, dinitrophenol, which turned up in early diet pills and even chemical pesticides. That specific chemical formula—NaC6H3N2O5—reminds everybody that small structural changes can lead to massive changes in safety and application. As a sodium salt, its reactivity shifts, often making it easier to dissolve and manipulate in water-based settings.
Over the years, accidents involving these kinds of nitro compounds have made safety training stick in my head. Dust, fumes, and accidental spills have all been issues in academic and industrial labs. One friend had to evacuate a lab at midnight thanks to a small mix-up with a dinitro compound. That memory hangs around longer than any quiz score. Whenever I see NaC6H3N2O5 on a bottle, I remember the warnings—gloves on, eye protection up, keep it contained, respect the volatility.
Deeper Challenges and a Way Forward
Safety remains the key issue with substances like sodium 2,4-dinitrophenoxide. I’ve seen colleagues push for digital tracking in inventory and automated spill detection as practical steps. Teaching new chemists exactly what’s in the bottle and why it matters can go a long way. The industry can invest in better labeling and faster disposal techniques to keep both people and environments away from harm. Strong habits around PPE, chemical storage, and proper label reading become second nature after a few close calls.
This chemical formula, simple on paper, doesn’t exist in a bubble. It connects to human safety, environmental risk, and practical innovation. Every professional who works with it holds a bit of responsibility, and the details behind a formula can shape how we work, teach, and plan for the future.
Is Sodium 2,4-Dinitrophenoxide hazardous to health or the environment?
How Exposure Affects Health
Many people don’t recognize Sodium 2,4-Dinitrophenoxide by name, but it’s a chemical that deserves real scrutiny. I came across it in old lab safety notes, flagged for both its toxicity and unpredictable effects. Handling even a small amount can put someone at risk. It reaches into the body with alarming ease, irritating skin and eyes. Getting some on your hands or breathing in its dust can set off symptoms. The bigger issue—once inside, it's no friend to your organs. Studies have linked compounds in its family to liver and kidney problems. I remember one case where a colleague suffered headaches and nausea after spilling a related compound, only realizing the source hours later. That’s the kind of real harm this chemical can cause.
Environmental Impact
Talking with environmental chemists, I’ve learned that chemicals like Sodium 2,4-Dinitrophenoxide stick around in water and soil longer than anyone would want. It doesn’t break down easily. Instead, it soaks into groundwater or runs into streams, where it lingers, potentially poisoning aquatic life. Fish are particularly sensitive; even low concentrations can stress their systems and disturb their ability to reproduce. One study from the late 2010s showed clear evidence of dinitrophenol derivatives affecting the hatching rates of freshwater fish eggs. It’s not just fish—plants have a tough time, too. Roots can absorb these residues, which then move up into leaves and stems. I’ve seen farmers struggle when fields next to contaminated sites yield stunted, damaged crops.
Why It’s a Growing Concern
Sodium 2,4-Dinitrophenoxide often slips through the cracks. It’s used in some industrial processes, especially older ones where waste handling didn’t get much attention. Towns near these industries sometimes find traces in their local water. The problem grows quietly with each season. You don’t smell it or see it, but over the years, communities notice increases in unexplained health issues. As someone who has visited areas with legacy pollution, I’ve seen the frustration firsthand—the sense that something invisible is undermining safety.
Reducing Harm Starts With Awareness
Addressing the dangers means starting with tighter control. Factories should switch to safer alternatives wherever possible. If a process can’t run without Sodium 2,4-Dinitrophenoxide, using strict containment with regular checks keeps leaks out of the environment. Workers deserve routine health screenings and proper gear. I’ve also seen cities adopt better water monitoring, using sensors to track pollution right from the source. Early warnings save wildlife and keep families safe.
Education counts, too. People living near production sites need honest updates about risks and responses. In schools, teachers can help by discussing these chemicals in science lessons, turning abstract dangers into real-world challenges students can help solve. Regulators and community leaders play their part by enforcing existing rules and supporting efforts for cleanup.
Moving Toward Safer Practices
Progress means learning from the past. Replacing hazardous chemicals like Sodium 2,4-Dinitrophenoxide with less toxic choices should always be the goal. Technologies to clean up polluted soil and water do exist—they just need more funding and political will. People have a right to know about threats to their health and environment, and that starts with facts, clear conversations, and shared action.
| Names | |
| Preferred IUPAC name | Sodium 2,4-dinitrophenolate |
| Other names |
Sodium dinitrophenolate Sodium DNPO Sodium 2,4-dinitrophenolate |
| Pronunciation | /ˈsoʊdiəm tuː fɔːr daɪˌnaɪtroʊˈfiːnˌɒksaɪd/ |
| Identifiers | |
| CAS Number | 119-75-5 |
| 3D model (JSmol) | `3DModel:JSmol/{"mol":"\nNa+.O=[N+]([O-])c1cc([N+](=O)[O-])ccc1[O-]\n"}` |
| Beilstein Reference | 120924 |
| ChEBI | CHEBI:38032 |
| ChEMBL | CHEMBL136527 |
| ChemSpider | 19738 |
| DrugBank | DB13732 |
| ECHA InfoCard | 03-2119944272-47-0000 |
| EC Number | 210-569-8 |
| Gmelin Reference | 85889 |
| KEGG | C14453 |
| MeSH | D017376 |
| PubChem CID | 11442 |
| RTECS number | TT2975000 |
| UNII | B7YKP6IK56 |
| UN number | UN3318 |
| Properties | |
| Chemical formula | C6H3N2NaO5 |
| Molar mass | 198.08 g/mol |
| Appearance | yellow crystalline powder |
| Odor | Odorless |
| Density | 1.69 g/cm3 |
| Solubility in water | Soluble in water |
| log P | -0.63 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 7.08 |
| Basicity (pKb) | 10.9 |
| Magnetic susceptibility (χ) | -38.6 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.688 |
| Viscosity | Viscous liquid |
| Dipole moment | 6.56 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 203.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -214.0 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1158 kJ mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation, toxic to aquatic life with long lasting effects |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 3-2-1 |
| Lethal dose or concentration | LD50 oral rat 340 mg/kg |
| LD50 (median dose) | LD50 (median dose): 202 mg/kg (oral, rat) |
| NIOSH | SN 15750 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Sodium 2,4-Dinitrophenoxide: "PEL: 0.15 mg/m³ (as dinitrophenol) |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | IDLH: 50 mg/m3 |
| Related compounds | |
| Related compounds |
2,4-Dinitrophenol 2,4-Dinitrophenylhydrazine Sodium phenoxide 4-Nitrophenol 2,4,6-Trinitrophenol (picric acid) |

