Sodium 2,4,6-Trinitrobenzenesulfonate: A Down-to-Earth Look at a Storied Chemical
Historical Development
Long before most modern labs opened their doors, researchers searched for versatile compounds that could serve both as analytical tools and as stepping stones to bigger discoveries. Over a century ago, chemists started isolating and tinkering with nitroaromatic sulfonates, drawn by their striking colors and interesting reactions. Sodium 2,4,6-trinitrobenzenesulfonate sprang up in this context—first as an academic curiosity, later as a valued reagent in analytical and synthetic work. The bumps and turns in its story mirror the growing pains of chemistry itself, as safety standards caught up to the energetic punch of nitroaromatics, and applications multiplied from textbook examples to real-world utility in diagnostics and biochemistry.
Product Overview
Sodium 2,4,6-trinitrobenzenesulfonate brings more to the bench than a mouthful of a name. The compound stands out for its ability to act as a sensitive chromogenic reagent, especially for detecting primary amines and amino acids. Its solubility in water and intense yellow hue make it instantly recognizable. In any well-stocked organic or analytical chemistry lab, this sulfonated trinitrobenzene salt earns its shelf space both as a dependable workhorse and a specialist’s reagent.
Physical & Chemical Properties
Sodium 2,4,6-trinitrobenzenesulfonate presents as a yellow solid, owing to its high nitro group density, which leads to intense color absorption. The salt dissolves in water, a blessing for those doing aqueous chemistry or bio-relevant work, and the sulfonate group keeps it ionic and easily handled in buffered solutions. Stability depends on conditions—dry and cool storage helps keep it from slowly breaking down. Reactivity centers around those nitro and sulfonate groups, with the molecule acting as an electrophilic partner in various substitutions. Carefully measuring what happens when it meets nucleophiles, like simple amines, reveals much about both the target and the compound itself.
Technical Specifications & Labeling
Regulators and suppliers tend to focus on clear labeling, especially given the safety concerns tied to nitro compounds. This salt usually ships in tightly sealed drums or smaller laboratory bottles, marked with key identification like CAS number and hazard references. Handling advice emphasizes keeping away from heat, flame, and incompatible reducers, as well as minimizing dust. Chemists have learned to give respect to these requirements: even if the trinitrobenzenesulfonate is less dangerous than some of its explosive relatives, it deserves caution in handling, storage, and disposal.
Preparation Method
The synthesis starts with benzene sulfonation followed by thorough nitration under controlled acidity and temperature. The route favors careful temperature ramps because the nitro groups don’t play nice with sloppy heat management. Once achieved, neutralization with sodium hydroxide gives the sodium salt. For many years, chemists carried out variations of this process at ever-larger scales, learning lessons about purification, washing, and yield optimization. Efficient crystallization separates the product from side products. Large-scale manufacturing requires process controls that catch runaway reactions early—lessons learned the hard way during the early history of aromatic nitrations.
Chemical Reactions & Modifications
This trinitrobenzenesulfonate hardly sits in the bottle for long. Chemists prize it as an agent for identifying free amines, due to the specific and rapid reaction it offers. The result—a vividly colored complex—makes for quick, visible tests in both academic labs and industrial settings. More recently, researchers working in protein chemistry have used it to investigate lysine residues and probe reactivity on macromolecules. These reactions offer a glimpse into how fine control and selectivity unfold in practice. Modifications have also explored partial reduction and other functional taps, but the main action stays close to its original use: a flashy diagnostic tool in chemical analysis.
Synonyms & Product Names
Names tend to be a mouthful in this field: sodium trinitrobenzenesulfonate goes by alternatives like TNBS, sodium picrylsulfonate, or sodium 2,4,6-trinitrobenzene-1-sulfonate. Generic catalog codes come and go, but the core set of synonyms persists in academic literature and supplier lists. Finding the same compound under these different names can sometimes trip up early-career chemists, but clear cross-referencing in publications helps prevent confusion.
Safety & Operational Standards
Hard experience teaches that handling nitroaromatics comes with risk. TNBS doesn’t explode as readily as picric acid or dinitrobenzene, but it still commands respect for being both an irritant and a potential sensitizer. Good laboratory practice calls for gloves, eye protection, and a fume hood—not just because of TNBS’s reputation, but because skip one precaution and you quickly learn why safety gets so much emphasis. Waste disposal, too, gets attention: nitro groups resist biological breakdown, so responsible disposal means chemical reduction followed by neutralization, according to published environmental guidelines. No one wants to fumble disposal or have to justify a preventable accident to a safety committee.
Application Area
TNBS plays a central role in labeling and quantifying primary amines, whether in protein sequencing or in monitoring amino acid modifications. Biochemists reach for it to map out lysine positions or follow subtle changes in protein structure. In environmental chemistry, its strong reactivity underpins sensitive field tests for pollutants bearing reactive groups. Analytical chemists often tell stories about devising new tests based on the quick visual feedback TNBS delivers. In some manufacturing contexts, the compound helps monitor raw materials and spot low-level contamination by amines. Whether for academic insight or quality control, its presence keeps experiments on track.
Research & Development
It’s easy to underestimate how much research revolves around reliable reagents, but the track record of TNBS in protein chemistry speaks for itself. The field advanced considerably with the introduction of colorimetric assays leveraging its sulfonate activity. These protocols underpin modern day standards for evaluating protein hydrolysis or screening enzyme activity. Newer research explores derivatization routes aimed at improving detection limits or creating even more selective assays. Many of us remember the first time a TNBS reaction made a protein band pop out bright yellow in a crowded gel, a little eureka moment in a sea of routine lab work.
Toxicity Research
Despite its utility, the dark side of TNBS demands attention. Early studies found strong sensitization potential, especially for skin and respiratory systems, and more recent animal trials have illuminated its potential for causing inflammation or allergic responses. The compound’s reactivity helps explain its ability to trigger immune responses, making it a go-to compound in immunology for inducing models of colitis, but raising red flags in handling at scale. Toxicology lessons learned over decades now shape every risk assessment: thorough ventilation, rigorous spill protocols, and mitigating exposure risks for those in routine contact.
Future Prospects
Chemistry doesn’t sit still. Environmental concerns drive ongoing research into greener reagents, and some hope to design analogs of TNBS that keep the color but shed the hazard. At the same time, the specificity and utility that TNBS delivers remain hard to beat. Advances in assay sensitivity, waste treatment, and even automation all link back to reliable reactions like the ones it powers. As long as chemists value dependable, vivid results for detecting amines, sodium 2,4,6-trinitrobenzenesulfonate looks set to keep its place on lab shelves. Sustainable synthesis and safer alternatives may shift the landscape in coming years, but for today’s science, TNBS continues to offer a powerful reminder of what a well-chosen chemical reagent adds to both research and routine practice.
What is Sodium 2,4,6-Trinitrobenzenesulfonate used for?
Understanding Its Purpose
Sodium 2,4,6-Trinitrobenzenesulfonate, often called TNBS, does not show up in the headlines, but its impact reaches labs all over the world. To me, this yellow, powdery chemical always meant the start of a day working through rows of tubes, handling reactions that can’t be taken lightly. TNBS gets attention in science for one main reason: it reacts strongly and predictably with amino groups in proteins.
Researchers rely on it for something called the TNBS assay. The test helps measure how many free amino groups are present in proteins and peptides. Knowing this, scientists can figure out changes in protein structure and trace reactions during the making of modified foods, medicines, or biodegradable plastics. I remember a colleague, deep in food science, mixing TNBS into batches of hydrolyzed protein to gauge just how much processing altered the original structure. Without TNBS, tracking subtle protein changes would take a lot more guesswork and time.
A Tool in Immunology and Disease Research
Beyond the lab bench for food or plastics, immunologists turn to TNBS for something that hits closer to home: understanding disease. This compound gets used to create models of chronic inflammation, including colitis in lab animals. With a single injection, TNBS can trigger a strong immune response, and this mimics human inflammatory bowel disease. Scientists then watch, learn, and search for new drugs or strategies to treat the condition. I have seen how insights from these models lead to real-world treatments for human health problems.
Because TNBS helps mimic disease, it allows controlled conditions when testing therapies or unraveling the steps in disease development. Tracking these reactions without TNBS would come with less certainty and more error. It has sped up research and helps provide more ethical options by refining animal models and sometimes reducing the number of animals needed, matching ongoing efforts for humane laboratory practice.
Safety and Environmental Impact
Handling TNBS comes with challenges. Its chemical structure packs several nitro groups, which means researchers must take safety seriously—explosions or toxic exposure remain real risks. Proper ventilation, sealed storage, and respectful handling are non-negotiable. Years ago, a careless moment in a workshop next door led to a tiny spill, and the lesson still gets passed down: always read up, and never skip gloves or face protection. Labs need to keep staff trained and ready for accidents.
Disposal raises more issues. TNBS doesn’t just break down safely in the environment. Improper disposal can cause problems for water supplies and aquatic life, so regulations guide labs on neutralizing it before sending waste outside. Efforts to design safer or greener alternatives have cropped up, and researchers weigh cost, reliability, and safety with every choice. Exploring less hazardous reagents for protein analysis or immune models may one day replace TNBS, but for now, it remains a tool with real weight.
Room for Better Practices
The story of TNBS highlights a basic truth: science leans on practical, sometimes risky tools to make progress. Training, awareness, and good habits matter as much as the compound itself. Open discussion about chemical hazards and broader efforts to reduce harm can shift not only safety practices, but the future of research. More investment into green chemistry could lead to safer, just-as-reliable substitutes down the line—something both experienced scientists and students welcome.
What are the safety precautions when handling Sodium 2,4,6-Trinitrobenzenesulfonate?
Why Extra Caution Matters
Handling sodium 2,4,6-trinitrobenzenesulfonate, better known as TNBS, isn’t something for the casual lab worker. This chemical plays a role in research—often in immunology—but packs a risky punch due to its strong reactivity and potential for causing harm. I’ve seen experienced researchers get caught off guard when hurrying through tasks with TNBS, thinking gloves and goggles do all the work. Getting too comfortable can lead to serious skin irritation, eye issues, and even more severe reactions if the powder hits the air or water the wrong way.
Understanding the Hazards
TNBS doesn’t just stain fingers. It carries a reputation for causing allergic responses on skin contact and can damage eyes if splashed. Lab workers can become sensitized to it after repeated exposure, and that often sneaks up over time. Inhaling tiny airborne particles can trigger respiratory issues, and spilled powder, if not handled fast and carefully, can ignite or even explode under certain conditions. Better to stay steps ahead than to scramble after an accident.
Gearing Up the Right Way
Forget flimsy gloves. Nitrile gloves hold up best against this chemical. I like wearing two pairs for that extra buffer—removing and tossing the top set if anything gets on them prevents spreading the substance. Eye protection isn’t optional: wrap-around safety goggles stop dust and liquid splashes. Toss in a well-fitted lab coat, preferably a flame-resistant one, and closed shoes. Labs that use TNBS need good airflow, so I always stick to working in a certified chemical fume hood—no open room work, no shortcuts.
Solid Working Habits
Attention to detail makes all the difference. I steer clear of distractions: no phones, no open drinks, and no loose papers on the bench. Measuring and transferring TNBS with spill trays or on disposable pads keeps the workspace clean and makes it easier to grab all residue once the experiment wraps up. I clean my hands right after removing gloves, scrubbing under nails and up past the wrists. If there’s a spill, I never use water right away—water can make things worse. I’ve found that gentle dry sweeping or vacuuming with specialized equipment, followed by a thorough chemical neutralization, works better. Spill kits built for reactive chemicals save time and prevent panic.
Disposal Demands Respect
Piling contaminated gloves, pipettes, or paper towels in the regular trash isn’t just lazy—it’s dangerous. Used material goes in dedicated, clearly marked hazardous-waste containers. Liquids and slurries with TNBS follow strict waste procedures, never mixed with incompatible waste streams. Reliable chemical waste companies know how to handle pick-up and disposal, removing the risk from the building. Labs that try to shortcut or DIY disposal end up with leaks, angry safety inspectors, or worse.
Training, Communication, Preparedness
Every new team member needs a detailed walkthrough of the lab’s protocol, not just a stack of papers with warnings. Practicing emergency procedures together beats reading instructions during a crisis. Eye wash stations and safety showers must work, and the access paths can’t be blocked with boxes or stools—a trip across the lab with burning hands or splashed eyes isn’t just unpleasant, it’s dangerous. Keeping updated material safety data sheets nearby means no one’s caught guessing during a shaky moment.
Looking Ahead
Sodium 2,4,6-trinitrobenzenesulfonate’s usefulness doesn’t give it a free pass on safety. Following clear processes, suiting up right, and taking clean-up seriously prevents painful health scares and keeps labs open for work. I’d rather spend a few extra minutes cleaning than a week off work recovering from a preventable mistake.
How should Sodium 2,4,6-Trinitrobenzenesulfonate be stored?
Trust Built on Respect for Hazards
Lab safety takes a real turn when you work with compounds like Sodium 2,4,6-Trinitrobenzenesulfonate. This material doesn’t leave much room for casual handling. The reason sounds straightforward—its structure belongs to the trinitrobenzene family, linking it closely to known explosives and strong oxidizers. Years in research settings taught many of us to never assume stability purely from a chemical’s day-to-day appearance. You let your guard down, you get burned—some lessons strike hard.
Real-World Practices Beat Textbook Storage Rules
It makes more sense to use a dry, dedicated chemical cabinet, one built to stand up against heat and stray sparks. Cardboard or open shelving doesn’t cut it. Fireproof storage works better because it keeps the material away from flames and limits damage during emergencies. Some teams add extra layers by storing small quantities in sealed glass bottles, then sealing those inside durable plastic containers. This approach keeps moisture and reactive vapors out. A single moisture leak can lead to clumping or unpredictable chemistry.
Strong labeling helps too. Labels should show the full chemical name, date of receipt, and risk signals. It’s not just about obeying regulations. Tired hands can grab the wrong bottle when storage systems turn sloppy. Old habits from years in the lab still carry weight—every bottle wears its risks right on the front, and no one’s embarrassed to double-check.
Reality Check: Room Conditions Matter
Temperature in your storage makes a make-or-break difference. Temperatures above room range increase volatility. I’ve seen what happens in the summer; a couple of degrees too high and containers start sweating. Placing bottles away from sunlight, heaters, or hot equipment keeps runaway reactions at bay. Keep away from acids, bases, reducing agents, and anything flammable. Chemical neighbors can team up for nasty surprises, turning normal days toxic or explosive.
A dry space protects from sudden moisture surges. Silica gel packs or other moisture-absorbing materials serve as cheap insurance. If you open the container and see any crusty buildup or odd coloring, treat everything with suspicion. Dispose of compromised material in line with hazardous chemical guidelines. Never dump or flush—call on a certified disposal service. Past mistakes showed how local spills rival fires for damage. Fast action comes from good habits, not regulations glued to the wall.
Training Protects More than Property
Every new chemist or technician must receive real training on handling and storage. Stories sink in better than warning posters. One older colleague shared how a rushed move of chemicals to an unprotected shelf led to a close call; nothing replaces hearing about what can go wrong from someone who's lived through it. Protective equipment always stays nearby: gloves, goggles, thick lab coats—even for quick samples or transfers. Emergency shower and eyewash stations get checked often, because fast seconds mean everything when trouble hits.
Accountability and Continuous Improvement
Routine inspection means catching risk before it grows legs. Each chemical inventory brings an opportunity to spot leaks, outdated stock, or misfiled containers. Years of audits and walkthroughs highlighted how teams grow complacent. A system that pairs written logs with regular reviews prevents surprises. Handling dangerous compounds demands both respect for protocols and willingness to question habits that grow stale over time.
Every safe day comes from understanding the risks tied to every bottle, not just trusting the labels or store-bought cabinets. Safety grows through action—one safe practice at a time.
What is the chemical structure of Sodium 2,4,6-Trinitrobenzenesulfonate?
Understanding the Chemistry
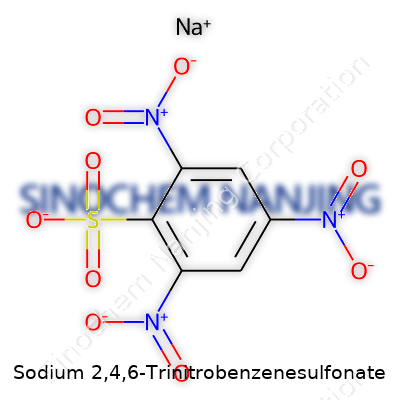
Sodium 2,4,6-trinitrobenzenesulfonate may sound intimidating, but the name tells you exactly what's in it. The compound features a benzene ring, a stable plate-shaped arrangement common in organic chemistry. This ring carries three nitro groups—one each at positions two, four, and six. That pattern puts hefty electron-withdrawing groups across the molecule, making it reactive in interesting ways. Attached to the ring, there's a sulfonate group, bonded at a remaining position, and this group forms a salt with sodium. Chemically, you’ll see the structure as C6H2(NO2)3SO3Na. The sodium ion serves to balance out the charge from the negatively charged sulfonate group.
Why Structural Features Matter
A structure might seem like a puzzle for chemists, but practical life brings out its importance. The three nitro groups make the compound highly electron-deficient. That property stands out during reactions; molecules with electron-rich partners will be drawn to this compound. In the context of analytical chemistry, I’ve used similar trinitro-substituted compounds to identify functional groups in proteins. The sodium sulfonate part means the chemical dissolves in water, an essential trait for lab work or industrial applications. If this compound wasn’t water-soluble, handling and measurement would become a challenge.
Safety and Environmental Impact
The arrangement of those nitro groups and the sulfonate makes sodium 2,4,6-trinitrobenzenesulfonate similar in reactivity to some explosives, although not as unstable. Still, its use needs careful management. Nitrated aromatics can have toxic effects. Long-term handling or improper disposal could add contaminants to water or soil. From my own experience, even lab-scale handling demands good ventilation, gloves, and solid waste management steps. Data show that trinitro compounds can affect aquatic life through chronic exposure, even if acute toxicity stays moderate. Many companies and research labs follow strict protocols, including dilution and neutralization, to make sure this chemical doesn't end up untreated in waterways.
Applications and Ethical Considerations
The unique interplay of electron-poor and water-soluble features opens doors in research. Most often, sodium 2,4,6-trinitrobenzenesulfonate acts as a diagnostic tool in protein chemistry. The compound reacts with sulfhydryl groups, helping to measure the extent of sulfhydryl content in a sample. That’s crucial for biochemists working on enzymes or protein folding. Yet with research freedom comes responsibility. Any chemical with nitro groups requires attention in procurement and tracking, not only for safety but for regulatory compliance.
Potential Solutions and Best Practices
If anyone works with this compound, reducing environmental impact means a combination of secondary containment, chemical inventory tracking, and staff education. My own lab invested in specialized waste containers just for nitro-aromatics. Periodic audits keep protocols sharp. Labs use chemical neutralization and choose water-soluble derivatives, like this sodium salt, to simplify waste processing. Product data sheets and material safety sheets play an essential role, guiding safe handling from start to finish. As more universities stress green chemistry principles, the next generation of researchers grows up with these habits built in.
Is Sodium 2,4,6-Trinitrobenzenesulfonate hazardous or explosive?
Chemical in Focus
Mention “trinitro” to a chemist, and most think about explosives—TNT especially. That sets off alarms for good reason. Sodium 2,4,6-trinitrobenzenesulfonate stands out because it carries the same trinitro aromatic core as TNT, yet adds a bulky sulfonate group with a sodium salt twist. Structurally, it’s a cousin, and that resemblance prompts some to worry about hazards related to explosiveness or instability. Once, I worked on a project where even mentioning a trinitro compound got the safety officers buzzing and setting up special protocols.
Hazards: Perception Versus Reality
TNT shatters concrete for a reason—it’s both volatile and packs a chemical punch. Some research chemists expect the same from anything with a trinitrobenzene ring. But facts spin a different story here. Sodium 2,4,6-trinitrobenzenesulfonate swaps some explosive traits for increased water solubility and charge separation, thanks to its sulfonate and sodium. Studies, including MSDS entries and the scientific literature, suggest that the substance doesn’t act like TNT or picric acid in a lab or industrial space. Instead, it behaves more like a somewhat irritant, stable crystalline powder.
During my own years handling specialty reagents and teaching new lab members how to look up substance risks, I found this product got flagged less for fire risk and more for causing skin or eye irritation. I never read about it detonating or posing a fire hazard under routine conditions. Most safety datasheets say you should use gloves, eye protection, and common sense, just like with any chemical. Accident records don’t show spontaneous explosions by this material, even when mistakes occur.
Potential Health and Environmental Issues
Most concerns come from its irritant properties, not its bang. Get it in your eyes or on your skin and you feel it. Breathe it in as dust and you cough. These are not small annoyances, but they don’t bring the risk of a surprise lab explosion. Like many aromatic sulfonates, it can linger if dumped, and since its salts are water-soluble, it can travel through water systems. Responsible labs and factories limit environmental release; regulatory controls exist in some places for disposal.
Someone curious about chemical safety will notice that it lacks the tell-tale indicators for explosive classification under GHS or OSHA rules. You won’t find it sold with explosives or listed as a restricted substance for fireworks unless you misuse it or mix it with much stronger reducers or oxidizers. Emergency first responders train on chemical databases, and none list this as a high-risk explosive problem.
Safer Handling and Broader Lessons
Given what I’ve seen in the lab and confirmed in safety databases, simple personal protective equipment and chemical hygiene practices keep workers safe from both spills and splashes. The bigger obstacle appears when new users see the name and freeze up thanks to the “trinitro” tag. It proves that chemical safety depends more on solid, up-to-date information and ongoing training than on fear or urban legend.
For teachers, R&D chemists, or industry safety officers, this is a lesson—look past a scary-sounding name, check modern safety data, and stick to good lab habits. The sulfonate cousin of TNT might raise eyebrows, but it doesn’t raise much dust or danger in daily use.
| Names | |
| Preferred IUPAC name | Sodium 2,4,6-trinitrobenzenesulfonate |
| Other names |
TNBS Picrylsulfonic acid, sodium salt Trinitrobenzenesulfonic acid sodium salt |
| Pronunciation | /ˈsəʊdiəm ˌtraɪˌnaɪtrəʊˌbɛnˌziːnˈsʌl.fəˌneɪt/ |
| Identifiers | |
| CAS Number | 2508-10-7 |
| Beilstein Reference | 358726 |
| ChEBI | CHEBI:8687 |
| ChEMBL | CHEMBL3230983 |
| ChemSpider | 119213 |
| DrugBank | DB14045 |
| ECHA InfoCard | 03ffbdb6-7ec9-48a5-893f-ab64114cd5a0 |
| EC Number | 208-734-8 |
| Gmelin Reference | 1221 |
| KEGG | C14304 |
| MeSH | D013074 |
| PubChem CID | 9549046 |
| RTECS number | WZ9825000 |
| UNII | W73BZZ7A4Z |
| UN number | UN1325 |
| CompTox Dashboard (EPA) | DTXSID1060454 |
| Properties | |
| Chemical formula | C6H2N3NaO9S |
| Molar mass | 367.14 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.89 g/cm³ |
| Solubility in water | soluble |
| log P | -2.2 |
| Vapor pressure | Negligible |
| Acidity (pKa) | -2.81 |
| Basicity (pKb) | pKb: 6.48 |
| Magnetic susceptibility (χ) | -62.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.630 |
| Viscosity | 200 cP (25 °C, 20% in H2O) |
| Dipole moment | 6.68 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 384.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −610.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1210 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V04CL04 |
| Hazards | |
| Main hazards | Explosive; harmful if swallowed; causes skin and eye irritation. |
| GHS labelling | GHS02, GHS05, GHS07, GHS09 |
| Pictograms | GHS07, GHS09 |
| Signal word | Warning |
| Hazard statements | H302 + H315 + H319 + H335 |
| Precautionary statements | P210, P220, P221, P280, P305+P351+P338, P370+P378, P402+P404, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 1, Instability: 1, Special: - |
| Lethal dose or concentration | LD50 oral rat 324 mg/kg |
| LD50 (median dose) | Mouse oral LD50: 324 mg/kg |
| NIOSH | TTQ9438000 |
| PEL (Permissible) | PEL (Permissible): Not established |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | IDLH: Not Listed |
| Related compounds | |
| Related compounds |
Trinitrobenzenesulfonic acid Picric acid Sodium picrate Dinitrobenzenesulfonic acid 2,4,6-Trinitrotoluene (TNT) |

