Silver Selenite: Substance with a Storied Background and Growing Potential
Historical Development
Silver selenite hasn’t gotten half the press of other silver compounds, but it’s played a surprisingly quiet role in inorganic chemistry for over a century. The earliest notes on selenites came from nineteenth-century mineralogists chasing odd earths, and silver salts drew curious experimenters thanks to both their shine and unusual colors. Practical uses trailed far behind noticing, though old lab records show this compound getting studied while chemists puzzled over selenium’s quirky properties and silver’s long-known medical applications. Researchers learned that while silver nitrate claimed more fame, silver selenite belonged to the side roster of compounds crucial for understanding selenium’s structures and for fine-tuning analytical chemistry.
Product Overview
Silver selenite appears as white or off-white powder or crystalline fragments, standing out from the darker hues of elemental selenium or the metallic sheen of pure silver. Its molecular formula, Ag2SeO3, tells a lot about reactivity, with selenium in the +4 oxidation state. Chemists and lab suppliers also call it disilver selenite or simply silver(II) selenite. For anyone stocking a chemical shelf, reliable suppliers deliver this product in tightly sealed amber bottles. Each lot includes thorough labeling, showing batch number, purity, and sometimes trace contaminants, reflecting the careful handling needed to avoid unwanted reactions.
Physical and Chemical Properties
Silver selenite looks pretty unremarkable at first glance. It doesn’t jump out in the way metallic silver does. The powder barely dissolves in water, which frustrates anyone hoping for easy solution handling. It stays more stable in air than some selenium compounds, resisting quick shifts from moisture or sunlight. One critical trait centers on light sensitivity: silver compounds often change color under strong light, and selenite is no different, especially when exposed over time. It melts at high temperatures, above 300°C, and decomposes rather than turning liquid, so melting isn’t a practical way to use or modify the substantive powder. It reacts mildly with acids, forming silver salts and releasing toxic gases, which means proper fume hoods stay a must in any real lab.
Technical Specifications & Labeling
Chemistry demands clarity, both in the flask and on paper. Packaging for silver selenite comes with clear hazard warnings: toxic if swallowed, eye and skin irritant, dangerous for aquatic environments. Besides product name and chemical formula, reputable sellers include CAS number (which ensures correct identification among countless similar compounds), purity percentage, manufacturer information, and chemical structure. Most offer a minimum purity, often 99% or higher for analytical uses. Each label carries hazard pictograms and up-to-date safety instructions, nudging purchasers toward compliance with strict laboratory protections.
Preparation Method
Silver selenite rarely shows up in nature, so it gets produced in controlled lab setups. Typical preparation uses a metathesis reaction. A measured solution of silver nitrate reacts with sodium selenite in distilled water. After stirring at room temperature, silver selenite precipitates as a fine white solid. The reaction goes like this: AgNO3 + Na2SeO3 → Ag2SeO3↓ + 2 NaNO3. Filtration and repeated washing with cold water remove lingering nitrates—otherwise, the impurities could alter reactivity or purity. Careful drying under vacuum or with gentle heat preserves quality, preventing decomposition or unwanted side-products. This batch method stands dependable for small-to-mid scale applications.
Chemical Reactions & Modifications
In the lab, silver selenite takes on many faces. Its key reaction uses lie in reduction or transformation to elemental silver and selenium compounds. Hydrogen or organic reducing agents break bonds, splitting selenite into silver and selenium oxides. Strong acids, such as hydrochloric, swap out selenite’s anion to yield silver chloride and free selenous acid—one reason to keep acids and selenite apart unless you’re aiming for a deliberate synthesis. With heat or certain metals present, selenite also helps form more complex silver-selenium alloys, a route explored in semiconductor research. The compound’s chemistry tells a lot about redox processes in selenium and how silver interacts with less-common chalcogenides.
Synonyms & Product Names
Disilver selenite, silver(II) selenite, or simply Ag2SeO3 all refer to the same material. In commercial and industrial catalogs, names shift with region or supplier. Academic texts still prefer the systematic name, but you can spot “silver selenious acid salt” or older references that tie the compound to related minerals, even when those minerals don’t match laboratory purity or structure. On global trade lists, translations vary, so checking for CAS number prevents confusion when ordering internationally.
Safety & Operational Standards
Despite its lack of notoriety, silver selenite poses real risks seldom discussed outside technical circles. Both silver and selenium compounds rate as toxic, and selenite’s low solubility doesn’t mean low danger. Gloves, lab coats, and splash goggles act as baseline protections. Dust easily irritates lungs and eyes, and accidental ingestion can damage internal organs—cases exist of mild poisoning from careless handling. In my own lab, a loose cap and knocked-over bottle set off the fume hood alarms, making clear that old hands and newcomers alike need frequent safety reminders. Storage takes place away from acids and organic solvents. Lab waste gets labeled for controlled disposal, shipped to chemical waste handlers who know how to neutralize selenium residues safely. Regulations in the EU and North America treat all selenium-containing waste as hazardous, underscoring the attention required even for disposal.
Application Area
Silver selenite has found real traction only in specialty corners of science and technology. Analysts use it for trace selenium detection, as the compound’s defined structure aids in calibration and spot-checks for contamination. It also appears in semiconductor research as a stepping stone toward more complex chalcogenide materials. A handful of studies test its value as a precursor for specialized glass or ceramic coatings—fields where chemical durability and photoactivity matter. Sometimes, its photo-reactivity drives creative experiments in alternative light-sensitive materials. The small but dedicated user base blends academic research and commercial curiosity, frequently crossing over into solid-state physics.
Research & Development
Over the last two decades, the interest in selenite chemistry has regained speed. Researchers examine how subtle tweaks in crystal structure influence its usefulness in photoelectronic devices. There’s ongoing work in modifying its surface for higher selectivity in sensor applications, especially those detecting rare gases or biological toxins. Physical chemists keep turning up unexpected magneto-optical effects, which could redefine silver selenite’s footprint in emerging optical technologies. A handful of patents already cover modified selenites mixed with rare earth elements to improve light-harvesting or energy-transfer features.
Toxicity Research
Toxicological studies address silver selenite in both acute and chronic exposure. Short-term effects range from mild irritation on skin contact to severe gastrointestinal upset when swallowed. Selenium’s push into cells makes chronic exposure especially harmful; symptoms move from fatigue to organ malfunction at higher doses. Animal studies prove severe toxicity at doses far below those of pure silver or less-reactive selenium salts. Wastewater and lab-acquired contamination threaten aquatic environments, given the bioaccumulation of selenium species. Research continues into safer handling, and emerging treatments for potential exposure include chelation therapy, similar to that for other heavy-metal toxins.
Future Prospects
Looking ahead, silver selenite’s main growth will probably come from its adaptability in niche technologies. The push for cleaner sensors, more efficient solar cells, and targeted chemical detection keeps selenite on the radar, especially as teams test greener synthesis routes and smarter recycling. If industry or academic breakthroughs boost the value of hybrid chalcogenides, the doors could open wider for this compound beyond the lab niche. Training new chemists and engineers in its handling and potential will remain vital, since mishaps with hazardous substances hold progress back as much as any technical hurdle. Silver selenite may never rival silver nitrate or selenides for volume, but its unique spot between classic analysis and future-facing technology provides a solid reason to keep watching its development.
What are the main benefits of using Silver Selenite?
Cutting Through the Chemistry
Silver selenite hasn’t grabbed big headlines, but anyone digging into advanced materials knows its name. Most people associate it with high-tech labs or niche industrial processes, and for good reason. Produced by combining silver and selenium oxides, silver selenite carries some real punch—not just another compound in a catalog, but a material with genuine impact.
Antimicrobial Edge
Bacteria don’t play favorites, but silver selenite sure has a way of making their lives difficult. Hospitals face enough trouble trying to keep surfaces clean. Resistant bugs keep pushing back, evolving past standard disinfectants. Silver, already famous for its germ-killing properties, finds a strong partner in selenite. Together, they short-circuit bacteria’s survival strategy. Research from respected journals shows silver selenite disrupts key cellular processes, making it valuable in coatings for medical equipment. With more devices turning into breeding grounds for infection, the need for smarter, more reliable antimicrobial materials only grows.
Boosting Solar Cell Performance
Clean energy isn’t just a slogan—it becomes reality with each leap in technology. Silver selenite comes up often in research on solar cells, especially the thin-film variety. It offers solid photoconductivity, meaning it handles light and turns it into electricity with real efficiency. The element combination lets manufacturers break past some traditional limits. Labs working with this compound have recorded stronger charge collection and better resilience against environmental wear-and-tear. Where sunlight is abundant but resources are limited, materials like this could help communities leapfrog old, expensive installations and put simple, robust power into homes and clinics.
Electrical and Optical Flexibility
I spent years fiddling with electronics for fun, digging through piles of components and custom projects. Silver selenite piques my interest because it manages both electrical conductivity and optical clarity. In advanced circuits—especially sensors and optoelectronic devices—a material that moves current efficiently and handles light signals cleanly is worth its weight. Smart windows, light-sensitive switches, and compact cameras could all benefit from silver selenite layers. Research articles point out its stability under heat and exposure, reducing failure rates and maintenance costs for manufacturers and users alike.
Challenges and Paths Forward
No one should ignore the flip side. Silver and selenium don’t grow on trees, and mining or processing them comes with environmental baggage. I’ve seen supply chain hiccups slow down promising products before. Slowdowns drive home the point: if adoption of silver selenite really picks up, recycling and responsible sourcing become non-negotiable. Markets that focus on lifecycle—cradle to grave—find ways to reuse and recycle components, keeping both costs and waste down. Teams developing silver selenite applications can work with suppliers on tracking sources, find creative ways to reclaim used material, and openly report their practices. When companies and scientists put real-world impact over market splash, all these technical advances mean more than just a brighter lab report—they stick around for the long haul.
Is Silver Selenite safe for everyday use?
Looking Beyond the Hype
Silver selenite shows up in scientific circles and niche applications, thanks to its unique blend of silver and selenium. From time to time, new compounds like this make a splash, billed as innovative solutions for everything from medicine to electronics. The real story, though, isn’t just about what a substance can do—it’s about its safety for the people who handle it and the environment that deals with its leftovers.
Chemistry Brings Both Benefits and Risks
Silver selenite doesn’t appear at the dinner table or the local pharmacy. Both silver and selenium have well-known roles: silver pops up in antimicrobial agents and electrical circuits, and selenium is an essential trace element, but can easily become toxic if misused. Put them together, and you don’t get a magical substance that’s free from concern.
Lab manuals highlight the risks around silver compounds: even small doses can build up in the body, leading to tough conditions like argyria, which stains skin blue-grey. Selenite compounds, meanwhile, can disrupt cellular functions when they land in living systems at higher doses. I’ve worked with chemical inventories and cleanup crews—protocols for substances like silver selenite usually come with thick binders full of hazard data instead of “safe for daily use” stickers.
Real Risks in Everyday Exposure
You won’t see silver selenite listed as a dietary supplement or beauty product ingredient. Regulatory agencies, such as the EPA and OSHA in the United States, consider both silver and selenium compounds as materials needing careful control. Chronic exposure carries cumulative risk, because body systems can’t quickly clear either one without stress. Reports from factories and labs sometimes show people developing strange symptoms even after brief, low-level contact. Symptoms run the gamut from skin and eye irritation to more serious systemic effects—sometimes neurological, sometimes metabolic.
Let’s talk environmental impact. Waste streams from industrial labs or battery plants can release selenium and silver into water and soil, where they don’t stay put. Selenium leaching from old electronics, for example, has caused mass fish deaths when runoff reaches rivers. Silver isn’t much kinder; it can disrupt aquatic life and throw local ecosystems off balance after repeated disposal. Some may argue the risk is overblown, but anyone who has tried growing veggies on contaminated soil doesn’t need a debate to know what’s at stake.
Solutions for Safer Handling
Strict labeling and storage help reduce accidents. In my work with hazardous materials, teams insisted on gloves, masks, and clearly labeled containment. This helped prevent careless mistakes—one unlabeled bottle becomes one emergency room visit too many. Schools and small companies could use more training and user-friendly hazard communication, not just legal warnings in fine print.
Industry can step up with greener processes, tightening the controls on emissions and taking responsibility for recycling or safely neutralizing byproducts. Governments can encourage research into safer substitutes that deliver the same benefits without as many long-term risks. People have a right to know what they’re handling, even if it comes in a shiny package, and to push manufacturers for transparency about every new compound introduced into daily life.
Trust isn’t built on promises—people protect themselves best when information is open, clear, and comes from experience on the ground, not just the front page of a marketing brochure.
How should Silver Selenite be stored?
Why Silver Selenite Calls for Respect
Silver selenite isn’t something most people bump into at the hardware store, but in labs and certain industries, it turns up with a list of straight-forward but essential rules. The first thing that really sticks out about silver selenite is its instability, especially when heat and light start playing around. Even simple room light can start to set off changes in its chemical structure, which leads to problems with both shelf life and safety. I’ve spent enough time handling compounds like this to know that a little planning on the front end can save a lot of headache later.
Smart Storage is About Prevention, not Repair
No one wants to clean up a spill or explain a surprise chemical reaction. I always reach for sealed glass containers, not plastic, because silver selenite likes to interact with some materials. Airtight containers help keep out moisture and airborne contamination. Moisture sneaks up on you faster than you’d think, and with this compound, water encourages slow breakdown. When a worker once left a cap loose on a reagent bottle, we caught a crystallization mess and a ruined sample. The lesson stuck with me.
Cool, dry, and dark spaces keep silver selenite in good shape. Lab storage isn’t about fancy vaults — a basic chemical storage cabinet does the job, but it has to be away from sources of heat. Heating vents, sunny windows, and even shared shelves with chemical oxidizers or acids can encourage trouble. The rules may sound simple, but I’ve seen even seasoned folks overlook them and pay the price with ruined material or minor exposure scares.
Human Health Remains a Top Priority
Silver selenite is more toxic than its “silver” label suggests. The real danger comes from inhaling or ingesting dust, but even a minor spill calls for gloves and eye protection. A memory that sticks out to me comes from a summer intern who wiped up a spill with a paper towel and forgot gloves. Luckily, quick showers and reporting meant no lasting harm, but it drove home the value of consistent personal protective habits. Every container should stay labeled — not a sticky note, but a real chemical sticker so that no one plays guessing games about safety.
Looking Ahead: Handling and Disposal
Once silver selenite breaks down or reaches the end of its usefulness, tossing it in the trash is off the table. The heavy metals and selenium content raise environmental flags. I keep used or unwanted samples in labeled waste containers, only handing them off to certified hazardous waste handlers. Local and federal regulations demand strict compliance with disposal, and in every workplace I’ve known, the long-term safety concerns keep people extra cautious.
Many problems start with inattention, not malice. Silver selenite needs respect, not fear. Setting up a routine — check the labels, keep the gloves stocked, don’t skip PPE — builds habits where safety becomes second nature. In my experience, these habits don’t just protect against one-off incidents. They add up to years of safe, steady work with some of the oddest materials on the shelf.
What are the common applications of Silver Selenite?
Industrial and Technological Benefits
Manufacturers and engineers often turn to Silver Selenite for its unique properties. People working with glassmaking have probably come across this grayish-white compound, since it serves a special purpose in coloring glass. The slightly red or purple tones delivered by Silver Selenite let glassmakers produce shades that stand apart from those created by more common metal oxides. Stained glass artists and companies making specialty lighting benefit from this reliable coloring agent.
Beyond color, the compound serves as a starting material for synthesizing other selenium-based chemicals and alloys. Chemical labs in industry might use Silver Selenite to generate selenium dioxide or sodium selenite. These chemicals later end up in everything from pharmaceuticals to animal feed supplements, offering a thread that quietly ties Silver Selenite to global supply chains.
Electronics and Sensors
Silver Selenite doesn’t often get the spotlight compared to materials like silicon or gold, but it helps drive advances in electronics. Its photoelectric properties lend it to roles in photocells and light sensors. Certain research efforts, looking to improve solar energy capture or boost sensor sensitivity, have experimented with thin films and powders made from Silver Selenite. The results so far suggest that it responds to light in useful ways, which means it plays a small part in the shift toward smarter energy systems.
Some circuit designers also value Silver Selenite for its conductive and optical characteristics. It helps researchers push past the limits of traditional semiconductor materials, searching for that practical edge in efficiency or response time. This is especially true in academic settings and R&D labs, where folks have the time and resources to try out new compounds in search of a breakthrough.
Pharmaceuticals and Medicine
People who follow developments in health sciences might hear about selenium compounds in the context of nutrition and disease prevention. Silver Selenite has no direct, widespread use as a drug, but chemists use it as a reagent for preparing other selenium compounds. Selenium itself is essential to human health in tiny doses, playing a part in immune function and antioxidant activity. While Silver Selenite isn’t given straight to patients, it still helps support the broader pharmaceutical industry by making other useful chemicals possible.
Any laboratory considering Silver Selenite faces safety questions, since selenium compounds can become toxic at high concentrations. Toxicology research continues to define safe practices for using and disposing of Silver Selenite, ensuring that industrial and scientific workers handle it carefully.
Challenges and Future Directions
Silver Selenite offers specialized answers, but groups using it must think about resource management and environmental safety. Producers pay close attention to how much of the compound enters waste streams, since heavy metals and selenium derivatives pose risks to water supplies and animal health. Proper containment and recycling of used materials go a long way toward reducing negative impacts.
New applications might appear as scientists learn more about its chemical structure. Modern material science often builds on yesterday’s expertise, so compounds like Silver Selenite could stand at the root of next-generation sensors or purification technologies. Keeping eyes open for safe, cost-effective uses promises steady progress, both in new gadgets and old industries.
Does Silver Selenite have any side effects or health risks?
Understanding Silver Selenite
Silver selenite doesn’t pop up in everyday conversation, yet it holds a spot among chemical compounds catching attention in science and alternative health circles. These days, people search for new supplements or remedies, sometimes trusting what they find online without checking what science has to say.
The Problem With Silver Selenite
One big question carries a lot of weight: is silver selenite safe? Based on what we know right now, there’s no clear sign that humans should tinker with this compound, either as a supplement or additive. Selenium, on its own, plays an important role in health—think thyroid function and defense against oxidative stress. The body doesn’t need much, but enough to avoid deficiency.
Silver, on the other hand, tells a thistly story in medicine. Silver wound dressings have their place. Swallowing or applying it without medical reason and supervision leads to side effects—skin turning blue-gray being just the most visible sign. Most regulators warn against using silver in dietary supplements, and for good reason.
Silver selenite combines both silver and selenium. This means it brings health risks from both elements, and possibly more. The compound hasn’t been studied in people, at least not directly. Its toxicity has cropped up in animal studies. Rats exposed to selenite compounds developed liver and neurological problems. Silver salts have hit kidneys and the nervous system hard in lab tests.
What Real-World Experience Shows
I’ve spoken with colleagues in both pharmacy and toxicology who won’t touch silver selenite outside controlled labs. They point to stories of people experimenting with silver supplements out of curiosity, only to wind up with permanent pigment changes in their skin or unwanted kidney trouble. Anyone with kidney disease faces extra danger, since the body can’t flush out these metals well.
The environmental side deserves a look, too. Both selenium and silver contaminate water in regions with mining or industrial runoff, where fish show deformities and local residents develop chronic health issues. Silver selenite isn’t designed for medical use; local poison control centers and government agencies like the EPA and CDC would classify it as hazardous.
Finding a Better Way Forward
People reach for chemical supplements for many reasons—sometimes out of frustration with standard treatments, sometimes out of hope. As a healthcare professional, I recommend starting with known facts and science-backed advice. Selenium requirements get met with a handful of Brazil nuts or seafood. Most people don’t lack this mineral. Using silver selenite to chase vague wellness benefits introduces risks with zero proven rewards.
Both the FDA and World Health Organization routinely review new compounds and update lists of what’s considered safe. Anyone tempted to buy something not approved for human use should pause and talk with their doctor or pharmacist first. Think of compounds like silver selenite as best left for research, not recipes or medicine cabinets.
Long story short, the real prescription is clear information, not silver bullets. No one gains lasting health by gambling on chemicals without legitimate evidence or oversight.
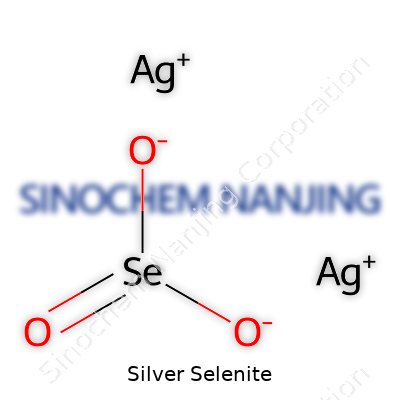
| Names | |
| Preferred IUPAC name | Silver(I) selenite |
| Other names |
Disilver selenite Silver(I) selenite Argentous selenite |
| Pronunciation | /ˈsɪl.vər səˈliː.naɪt/ |
| Identifiers | |
| CAS Number | [10102-43-9] |
| Beilstein Reference | 4045546 |
| ChEBI | CHEBI:86452 |
| ChEMBL | CHEMBL3300955 |
| ChemSpider | 23313985 |
| DrugBank | DB11092 |
| ECHA InfoCard | EU-InfoCard-100.018.089 |
| EC Number | 240-832-2 |
| Gmelin Reference | 84573 |
| KEGG | C18799 |
| MeSH | D014017 |
| PubChem CID | 166864 |
| RTECS number | VV7525000 |
| UNII | 8X6I2GWG6K |
| UN number | UN3283 |
| Properties | |
| Chemical formula | Ag2SeO3 |
| Molar mass | 246.84 g/mol |
| Appearance | Colorless or white, with a silky or pearly luster. |
| Odor | Odorless |
| Density | 6.96 g/cm3 |
| Solubility in water | Insoluble |
| log P | 0.35 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 11.25 |
| Magnetic susceptibility (χ) | +94.0e-6 cm³/mol |
| Refractive index (nD) | 1.894 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 120.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -175.0 kJ/mol |
| Pharmacology | |
| ATC code | V03AX |
| Hazards | |
| Main hazards | Toxic if swallowed; contact with acids liberates toxic gas; may cause burns; environmental hazard. |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS08 |
| Signal word | Danger |
| Hazard statements | H301+H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P210, P220, P261, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P313, P314, P330, P337+P313, P362, P501 |
| NFPA 704 (fire diamond) | 2-0-2-OX |
| Autoignition temperature | 350 °C (662 °F) |
| Lethal dose or concentration | LD50 (oral, rat): 3.6 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 4 mg/kg |
| NIOSH | Not Listed |
| PEL (Permissible) | 0.01 mg/m³ |
| REL (Recommended) | 286.86 |
| Related compounds | |
| Related compounds |
Silver(I) oxide Silver sulfide Silver selenide Selenium dioxide |

