Silver Permanganate: Unpacking the Science, Promise, and Peril
Historical Development
Chemistry keeps surprising us with compounds that turn out to be far more than laboratory curiosities. Silver permanganate is one of those fascinating substances. Chemists started paying attention to permanganates over a century ago for their powerful oxidizing abilities. Silver compounds, on the other hand, won early favor for uses in medicine and photography. Put the two together—silver and permanganate—and you get a compound that stands out for its unusual color, instability, and reactivity. Over time, this material picked up a scattered trail of research papers, especially as scientists looked for stronger oxidizers for analytical chemistry and as accelerants in specialized applications.
Product Overview
This salt, made from silver and permanganate ions, shows off a deep violet to black shade, a visual hint of its intense chemical potential. While most permanganates catch attention for their brilliant purple color, the silver salt dials up the drama, both in looks and behavior. It’s not an everyday material; instead, lab stockrooms and chemistry benches see it mainly in research or in experiments where a powerful oxidizer makes a difference. Its rarity and instability make it something pursued more for what it can teach, rather than as a staple raw material.
Physical & Chemical Properties
Silver permanganate typically forms a fine, dark crystalline powder. It’s not something you want to shake up—the material has a reputation for being both light-sensitive and subject to explosive decomposition. Even the moisture in the air poses a risk to its stability, and dry conditions often can’t prevent its natural tendency to fall apart or react with organic contaminants. With reactivity that makes it a stronger oxidizer than potassium permanganate, it’s hard to ignore both the promise and the danger packed into each grain.
Technical Specifications & Labeling
Handling silver permanganate takes real care. Not just gloves and goggles, but real respect for the warnings printed on every label or material safety data sheet. Even a tiny bit of contamination, extra heat, or rough handling can lead to runaway reactions. Lab storage must mean sealed containers, out of the light, at cool, steady temps. Labels highlight not just oxidizer warnings, but the need for containment and spill controls. Every technical document comes with bold hazard pictograms, and rightfully so.
Preparation Method
Most lab prep methods for silver permanganate rely on reacting a soluble silver salt, like silver nitrate, with a permanganate salt in water. If done right, a dark precipitate falls out, filtered, washed, and dried cautiously—a step where many accidents can occur. The chemistry stays straightforward; the real challenge comes with isolation and purification. Each grain becomes a risk—the drier and purer the compound, the more unstable and reactive it becomes.
Chemical Reactions & Modifications
Silver permanganate isn’t one to sit still. Add heat or even mild reducing agents and it breaks down fast, often with enough energy to trigger explosions if not contained. Its oxidizing reaction attacks organics fiercely, sometimes with no warning, making it a strong tool for chemists but a hazard for everyone else. Modifications mean swapping ions for slightly less reactive versions or suspending the solid in silicone oil just to slow down its appetite for decomposition. Chemists learned early on that using smaller quantities in diluted forms could harness its power without the same level of risk.
Synonyms & Product Names
Silver permanganate sometimes goes by names like silver manganate(VII) or simply AgMnO4. Chemists prefer shorthand in the lab, but those in the know never forget what lurks beneath the formula. Batch labels and supply catalogs rarely stray from these scientific monikers, which also act as reminders of its reactivity.
Safety & Operational Standards
Recklessness around silver permanganate courts disaster, plain and simple. Safety standards keep evolving as case reports of burns, fires, and even lab explosions still roll in. Anyone using it tapes checklists above their workbench—double gloves, face protection, chemical splash gear, and explosion shields, even when handling tiny amounts. Waste needs special buckets, never mixed with organics or any reducing agent. Labs with strict operational codes have far fewer accidents, and training happens every year—not just once per scientist. The compound never leaves controlled storage, and only a handful of workers ever get near it without direct supervision.
Application Area
While most industries steer clear, a handful of specialty labs dig into silver permanganate’s oxidizing strength for analytical chemistry purposes, trace metal analyses, and the occasional synthesis of unusual coordination compounds. Some research explores its use in rocket propellants, but only under carefully controlled test conditions. Regular industrial use remains off the table because safer alternatives—like potassium permanganate—do much of the same job without nearly as much risk.
Research & Development
Researchers keep coming back to silver permanganate, not for high-volume production, but for what it can reveal about the relationship between oxidizer strength and chemical stability. Its shelf-life issues become textbook examples for students learning to respect hazardous compounds. Analytical chemists sometimes study the reaction pathways, hoping a tweak or additive will make it more useful and less treacherous. Funding bodies tend to avoid projects using it outside strict academic settings, and peer-reviewed journals keep pushing for risk mitigation in any new experiments.
Toxicity Research
Scientists learned early that any permanganate salt holds health risks, from the metal content to the ability to act as an irritant or worse. Silver permanganate amplifies those risks due to its volatility. Inhaled dust or accidental skin contact can provoke serious responses—irritation, burns, or longer-term tissue damage in high enough concentration. Chronic exposure, while rare, raises worries over both silver accumulation and permanganate toxicity. Toxicologists press for strict exposure controls, since current data show no safe level for this compound outside a controlled laboratory. Training includes drills for accidental spills, and there’s no expectation of a safe “everyday” dose; simple avoidance stays the rule.
Future Prospects
The future for silver permanganate doesn’t point toward new commercial markets. Its explosive behavior and the ease with which safer compounds do the same job mean most researchers study it for targeted discoveries or for teaching tough lessons about chemical hazards. There’s curiosity about whether new stabilizers or nano-carriers could tame it for specialized fuel or battery uses, but nothing yet suggests a breakthrough on the horizon. The compound remains a warning as much as it’s a tool, showing the limits of what strength in chemistry can offer if safety and practicality can’t keep up. For now, silver permanganate stands as a story more about chemical understanding than broad adoption, and maybe that’s its value: it reminds chemists not all power is meant to be tamed.
What is Silver Permanganate used for?
Looking Beyond the Textbook
Silver permanganate doesn’t show up in most household cleaning aisles, but people who spend hours in chemistry labs know its value. This deep-purple crystal belongs in the family of permanganates, a group known for strong oxidizing abilities. Whenever someone asks about its real-world use, my mind goes back to stories from former colleagues experimenting with small glass vials and the unusual spark of excitement that came when something new reacted fast and powerfully. Silver permanganate holds that reputation: potent, sensitive, and, for specific jobs, irreplaceable.
The Chemical Industry and Power of Oxidation
Those working in specialty chemistry recognize silver permanganate as a serious oxidizing agent. During organic synthesis, this compound helps break down tough molecular bonds or facilitate reactions that stall without a forceful push. Trying to design a new organic compound, you often hit a dead end: either a reaction won’t proceed, or you need a cleaner split of molecules. People in R&D labs sometimes rely on silver permanganate to break through these blocks, especially in situations where weaker oxidizers let impurities slip through.
Silver’s role makes it stand out. Other permanganates feature sodium or potassium, but adding silver brings greater sensitivity and reactivity. In the hands of a seasoned chemist, that means more control over outcomes. But with extra power comes risk. Accidental friction or a stray spark can set off a violent reaction, enough to raise concerns about safe handling, proper storage, and even legal restrictions in several countries.
Field Uses: Analytical Chemistry, Detectors, and Specialty Applications
Silver permanganate finds use in analytical chemistry. Labs may use it to test for oxidizable substances or detect traces of organic matter by watching color changes or measuring reactions. Urban water treatment rarely relies on such a sensitive compound because of cost and safety. But research facilities value the accuracy that comes with a sharp, visible reaction.
I remember seeing researchers use silver permanganate in early-stage sensor development, especially those looking for ultra-sensitive detectors for pollution or rare contaminants. It’s impressive how the right chemical reveals a hidden toxin at parts per billion. In those cases, speed matters; a material able to trigger a bold, unmistakable response can help catch a problem before it spreads.
Why Safety and Strict Handling Matter
Few chemicals draw as much caution as silver permanganate. Strong oxidizing agents can react violently with organic materials and reducing agents. Just a little contamination or friction can produce heat, smoke, or even explosions. It surprised me at first to learn how many shipping companies avoid it entirely, and how storage requirements include temperature controls, non-combustible shelving, strict logs, and constant staff education.
One solution involves better training: anyone handling reactive materials needs to understand both the science and the risk. Clear protocols save lives—there’s no shortcut around this. More labs now invest in digital monitoring of storage conditions and automated dispensing systems, limiting direct human contact. Regulatory oversight continues to evolve, but grassroots improvements by chemists themselves drive the biggest changes.
The Takeaway: Value Balanced With Vigilance
People outside of research circles may never see a gram of silver permanganate, but those aiming for the next discovery or a breakthrough sensor lean on its reliability. Powerful materials like this keep the world pushing forward. The key rests in using them wisely, backed by respect for their strengths and a commitment to safety that never lets up.
What are the safety precautions when handling Silver Permanganate?
Why Silver Permanganate Demands Respect
Silver permanganate sits high on the list of chemicals that can turn a routine task into a disaster without warning. It doesn’t just tarnish your tools and stain your skin; it reacts with common stuff in ways most folks don’t expect. You won’t hear it fizzing or popping, but rubbing two grains together or dropping a speck on dusty cloth can spark more than just curiosity—sometimes literally.
Personal Experiences with Unpredictable Reactions
A few years ago, our lab brought in a new shipment of silver permanganate. The safety advisor’s eyes widened as she unpacked it. Her first step: reinforce the rule that nobody measures this compound out alone, and gloves aren’t optional. We’d all heard stories: mixing with organics or a splash of alcohol can set off instant combustion, and even light friction—like what you’d barely notice with sugar or salt—can launch purple sparks. After that day, nobody looked at this powder the same way.
Safety Steps Worth Taking Seriously
Storage: People who handle this chemical keep it isolated—locked, even—well away from organics, acids, and anything likely to shed dust or fibers. Shelves stay cool and dry. Everyone’s checklist starts with tight lids, no glass-on-glass contact, and labels that scream, “Don’t grab casually.”
Protective Gear: Forget thin lab mitts. Thick nitrile or neoprene gloves, goggles, and flame-resistant coats run standard. Before opening the bottle, sleeves get rolled down and beards get tied off. In my experience, anyone skipping goggles even once usually ends up regretting it—maybe not with burns, but with panic in their eyes after a close call.
Workspace: Fume hoods make a big difference. No open flames, no static, and strictly no food or drink nearby. The counter holds only what you’ll use. If you can’t keep the workspace immaculate, find another job, because even a stray paper towel can turn a spill into a firestorm.
Measuring and Transferring: Slow movements matter. No scraping spoon edges. Scoop or spatula stays non-metallic and perfectly clean. Double-check all utensils. At my last job, someone hurriedly used a wooden stick—never again. The heat and smoke cleared the room and forced a full review of every step we’d gotten lazy about. We switched to special glass, washed thoroughly and dried every time, with not even a hint of leftover chemical.
Troubleshooting Accidents and the Right Way to Respond
Spills call for immediate action, not panic. Smother with sand or vermiculite—not paper towels, never rags. Water makes it worse; it’s not a mop-up job. Every year, hospitals see burns from folks thinking they can rinse off permanganate spills. The stained fingers are the least of their problem when skin starts to blister. So we keep buckets of absorbent around, sweep carefully into hazard bins, and watch for airborne dust. Exhaust fans run as backup.
Training and Ongoing Caution
What really keeps people safe is stubborn, routine training. If someone’s new, I show them how close a call can get, then shadow their first dozen tries. Refresher sessions, not just one-and-done checklists, keep the lessons sharp in everyone’s minds. This isn’t one of those chemicals that gets easier with time. You keep holding respect for it, or you learn the hard way—maybe from a headline, or worse, from an ambulance ride. Strict habits and honest wariness save hands, eyes, and careers.
How should Silver Permanganate be stored?
Understanding the Need for Caution
Silver permanganate isn’t your garden-variety chemical. This stuff ranks high on the list of powerful oxidizers. Even after working in different labs, the respect for this compound stays the same: treat it right, or face real trouble. Over time, cases of lab fires and dramatic reactions have traced back to mishandled oxidizers like this one. The lessons stick—always handle and store it properly.
Safe Storage: Not Just a Guideline, but a Necessity
A cool, dry, and well-ventilated space stands out as the first line of defense. Temperatures should stay steady, always below room temperature. A lot of people overlook humidity, but that’s where moisture seeps in and trouble starts. Water causes silver permanganate to decompose and release oxygen, raising the explosion risk—even from a minor spark. That’s not just textbook advice. I’ve seen small drips wreak havoc on chemical shelves during a hurried cleanup.
Flammable organic materials and reducing agents can’t sit nearby. Permanganate mixes badly with those—one accidental spill equals a recipe for instant combustion. In my early days, I watched someone store oxidizers beside solvents. A small leak turned into a real emergency. Lesson learned: always use separate, dedicated cabinets, preferably metal or corrosion-resistant polymer, with clear labeling.
Sealing and Containment Practices
Containers should seal tightly and resist corrosion. Only glass or tough plastic fits the bill. Metal, given its reactivity, can’t guarantee safety—especially with an oxidizer this reactive. No shortcuts here. I’ve had batch inventories grow old or go unnoticed, so regular checks with fresh labeling stave off accidental misuse or decay.
Locked storage isn’t about paranoia. It keeps unauthorized hands away and reduces the chance of routine mishandling. The best labs I’ve walked through used secondary containment trays. In case of a spill or breakage, the compound stays confined. This isn’t a hassle; it’s an investment in accident prevention and downtime avoidance.
Limiting Quantity and Secure Disposal
Don’t stockpile for convenience. Only keep what’s essential for your immediate work. Lesser quantities translate into a smaller target for disaster. I remember cleaning out an old storage room—finding chemical bottles past their prime always brings home the point. If you haven’t used it in a while, arrange safe disposal with specialists.
Clear Policies, Shared Responsibility
A single sloppy habit doesn’t just risk property, but lives. Training never stops after onboarding. Periodic refreshers and labeled storage spaces set the right routines. My best professional environments made secure chemical management a group effort, not just a formality for compliance. If everyone pitches in, fewer mishaps happen.
Treating silver permanganate with the respect it deserves keeps people, workplaces, and communities safer. The unglamorous side of chemical science means being careful every day, especially with substances that don’t forgive mistakes. Attention and diligence never go out of style.
Is Silver Permanganate hazardous to health or the environment?
Understanding Silver Permanganate
Silver permanganate isn’t something most folks find lying around their kitchen or garage. This salt, formed from silver and permanganate ion, packs a real chemical punch. Commercial labs, researchers, and some niche industries keep it for certain oxidation reactions. The dark hue and crystalline form might give it an air of mystery, but the real story lies in how it behaves and the risks that come with it.
Real Dangers in Everyday Use
Some chemicals fade into the background, barely noticed outside textbooks. Silver permanganate refuses to go unnoticed. This compound combines the explosive potential of permanganates with the sensitivity of silver. Friction, heat, or the wrong mixing can set off violent reactions. A simple spark or knock on the table can become a trip to the emergency room.
I remember working in a university lab that prohibited silver permanganate, even for demonstration. Too many stories made their way through the chemistry departments—an explosion here, a nasty fire there. It sticks as a hazard in safety handbooks.
Touching, Breathing, Swallowing: Health Hazards
Skin burns readily under permanganates’ oxidizing power. Even a speck can cause trouble if it lands on bare skin. Inhaling dust causes problems in lungs, with doctors reporting coughing, shortness of breath, and possible tissue damage if exposure lasts. Ingesting even small amounts poisons the nervous system and kidneys.
A 2015 toxicology report found that manganese, a breakdown product, accumulates in the body when permanganates go unchecked. High levels damage the brain and nervous system—a risk that shows up in workers exposed for long periods. Silver, on its own, also accumulates in tissue and can turn skin a bluish tone, called argyria.
Environmental Fallout
Silver and permanganate ions both threaten the environment. Permanganates kill aquatic life, especially fish and amphibians, disrupting delicate streams and ponds. Silver doesn’t break down, instead collecting in soil and sediment, where it harms earthworms and plants.
Leaching from spills, improper disposal, or even small lab mishaps sends these ions into groundwater. Many municipal water systems install special filters for these ions, yet not every region has the budget or know-how.
Remediation comes at a heavy cost, not just in cleanup but in lost biodiversity. I recall a local river where fish populations failed to recover for years after an oxidizing chemical spill—permanganates among the suspects. The fear stretches beyond humans, reaching every organism in the food chain.
What Makes a Safer Path?
Control starts with education and strict handling rules. Manufacturers and labs who use silver permanganate set detailed safety plans, proper containers, and strict guidelines. Fume hoods, gloves, face shields, and spill kits become mandatory. Skylarking or shortcuts bring real consequences.
Some researchers hunt for less risky substitutes in industrial and academic work. Switching to milder oxidants or non-toxic catalysts cuts risk not just for chemists but for downstream users and the environment.
For those dealing with silver permanganate, knowledge and respect keep harm at bay. Regulators and scientists owe it to users and the community to demand best practices—before disaster writes the next cautionary tale.
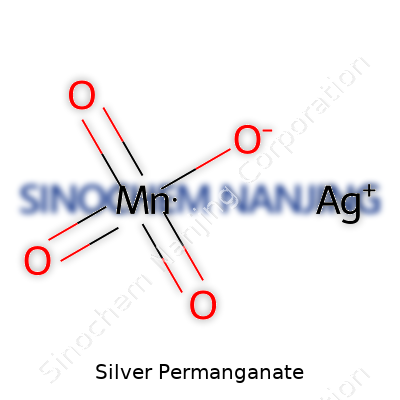
What is the chemical formula and appearance of Silver Permanganate?
Digging into Silver Permanganate
Silver permanganate comes with the formula AgMnO4. Over countless lab hours, the formula never gets old—one part silver, one part permanganate, four oxygens. That’s the simple side; the moment this stuff lands on a watch glass, you notice its deep, almost haunting, purple hue. It isn’t the bright flash of potassium permanganate you find in every high school chemistry set. Instead, it reels in a darker, more somber shade, suggesting right off the bat it’s not a compound you want to handle casually.
Appearance Up Close
Any chemist who has spilled this crystalline powder remembers the mess. Silver permanganate piles up in clusters, giving off a metallic sheen under good light, thanks to the silver. Touching or brushing it creates an unmistakable smear on everything—most lab bench stains tell a permanent story. That blackish-purple color doesn’t wash off easily either. For me, the striking color difference compared to other permanganates forges a direct sense of caution. Nature rarely gives warning labels with such intensity, but this compound manages it loud and clear.
Why Understanding Silver Permanganate Matters
It might come across as one more chemical tucked at the back of a shelf, but its reactivity gives it importance far beyond a color pigment. Silver’s reputation for being useful in electronics and antimicrobial coatings doesn’t soften the hazards when combined with a strong oxidizer like permanganate. Stories of spectacular, unintended decompositions still run through the halls of many research buildings. Mixing silver and permanganate creates a tandem that heats up fast—even slight physical shocks can start decomposition. In one case, a professor illustrated how mixing a trace of organic dust was enough to spark combustion. There’s real risk behind the visual beauty.
Weighing Hazards and Handling Solutions
Research data tracks the instability of silver permanganate. Sudden light, friction, or a tiny bit of moisture can start a chain reaction. Lab safety guidelines don’t treat it as an everyday salt. Working with it meant handling only microscale batches, often inside a fume hood, with full PPE—a must for chemists who value their eyebrows. Years spent watching new technicians, it stands out how much emphasis mentors put on handling oxidizers: never store with flammables, protect from sunlight, and always double-check dating since age can prompt unpredictable breakdowns.
Some research groups search for stabilizing agents—common choices like silica gel or keeping the compound damp under carefully controlled conditions. But the margin for error never widens enough to let your guard down. Even properly labeled containers can be a problem after months on the shelf. Regular inspections and drills in spill response help keep surprises to a minimum. These steps look tedious, but in my experience, strict discipline around storage and use prevents emergencies most folks never have to witness.
Learning from the Chemistry
Silver permanganate’s look and chemical makeup send clear signals: treat it with respect, know your material, and never gloss over the risks. People see the stark color and may think it’s all for show. Instead, it’s a warning—one that routine, awareness, and the right protocols help to answer. Better understanding and better practices are the best tools for any researcher working with substances that carry power far beyond their visual impression.
| Names | |
| Preferred IUPAC name | silver;oxo(dioxo(oxomanganio)oxy)manganese |
| Other names |
Permanganic acid silver salt Silver(I) permanganate |
| Pronunciation | /ˈsɪl.vər pərˈmæŋ.ɡə.neɪt/ |
| Identifiers | |
| CAS Number | [7783-98-4] |
| Beilstein Reference | 3589903 |
| ChEBI | CHEBI:86364 |
| ChEMBL | CHEMBL1201577 |
| ChemSpider | 20811 |
| DrugBank | DB15775 |
| ECHA InfoCard | 03bdfa41-08eb-4ae2-a403-27e09adb5c5d |
| EC Number | 232-019-5 |
| Gmelin Reference | Gm. 1418 |
| KEGG | C18687 |
| MeSH | D012710 |
| PubChem CID | 24554 |
| RTECS number | SJ6475000 |
| UNII | 92G0M2HH36 |
| UN number | UN1481 |
| Properties | |
| Chemical formula | AgMnO4 |
| Molar mass | 262.81 g/mol |
| Appearance | dark gray odorless crystals |
| Odor | Odorless |
| Density | 3.96 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.69 |
| Vapor pressure | Negligible |
| Basicity (pKb) | -6.5 |
| Magnetic susceptibility (χ) | +57.0·10⁻⁶ cm³/mol |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 238.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -80.9 kJ/mol |
| Pharmacology | |
| ATC code | V09AX02 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07, GHS09 |
| Pictograms | GHS03,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H271: May cause fire or explosion; strong oxidizer. H301: Toxic if swallowed. H331: Toxic if inhaled. H314: Causes severe skin burns and eye damage. |
| Precautionary statements | Precautionary statements of Silver Permanganate: "P210, P220, P221, P280, P305+P351+P338, P370+P378, P371+P380+P375, P501 |
| NFPA 704 (fire diamond) | 2 3 1 OX |
| Lethal dose or concentration | LDLo oral rat 25 mg/kg |
| NIOSH | SN122 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.01 mg/m³ |
| Related compounds | |
| Related compounds |
Potassium permanganate Sodium permanganate Calcium permanganate Silver nitrate |

