Silver Perchlorate: Beyond the Lab Glass
Looking Back: Historical Development
Silver perchlorate entered the picture when chemists in the 1800s chased after reactive silver salts with stronger oxidizing bite and greater solubility in organic solvents. Its early use fit with the explosion of organometallic chemistry, as the need grew for silver reagents that worked in less polar liquids. Folks researching silver nitrate kept bumping against its lack of solubility in many organics. When someone realized silver could pair up with perchloric acid's robust anion, things moved forward quickly—silver perchlorate started showing up in papers and experiments, even in the pre-WWI era. Historians of chemistry will find scattered notes on its use, especially in replacing other halide salts, since perchlorate never adds halide pollution to solutions. As inorganic and coordination chemistry advanced, this salt began to play a quiet but steady role, right up to today’s synthetic challenges.
Product Overview: A Versatile Player
People don’t just grab silver perchlorate because it’s “another silver salt.” Researchers come back to it because it dissolves like few others do, especially in solvents such as acetonitrile or acetone. Some chemists even treat it as a “clean swapper”—it brings silver into play without dragging along less wanted partners like chloride or sulfate. Whether chemists aim to kick off metathesis reactions, clear out halides, or spark some delicate catalyst activation, this compound usually gets the job done fast. In the toolbox, it sits next to silver triflate and nitrate, stepping in where those salts just won’t behave.
Physical & Chemical Properties: Born Reactive
Silver perchlorate powder shines with its white, crystalline appearance, but it rarely just sits in a jar for long. Folks notice right away it pulls water from the air, a trait called hygroscopicity. Unlike some other silver compounds, it dissolves into a clear solution in both water and many polar organics. Its high solubility—easily over 500g per liter in water—sets it apart from the crowd. Chemically, it acts as a strong oxidizer and has to be handled with care near flammable solvents or reducing agents. Its oxidizing strength, paired with silver’s affinity for halides and unsaturated organics, drives its popularity among advanced lab chemists.
Technical Specifications & Labeling: Reading Beyond Labels
I’ve seen high-purity silver perchlorate get labeled not just by its minimum assay (often over 99 percent) but by its dryness, since even a cracked bottle cap means the powder starts soaking up water. Standard labeling marks the oxidation risk, storage limits, and regulatory warnings tied to perchlorates as a class. Any fine print about batch source or trace metal checks can matter, since catalysis or advanced synthesis sometimes goes awry with hidden contaminants in the millimole range. What gets missed far too often is the need for airtight storage—even for short-term lab work, a bad seal spells trouble, especially in reactions where water presence ruins selectivity.
Preparation Method: Behind the Scenes Synthesis
Making silver perchlorate in the lab follows pretty well-trodden ground. Anyone who’s done this probably remembers dissolving silver nitrate in water, then adding perchloric acid. Precipitated silver chloride usually gets filtered out if any chloride stuck around. With enough mixing, what you get is silver perchlorate in the solution, crystallizing out on cooling—sometimes filtration and lots of drying follow. The process isn’t especially tricky with good safety habits, but anyone who’s made perchlorates knows the importance of avoiding spills and of strict cleanliness to avoid accidental explosions, as perchlorate’s combination with organic matter or heat can make for nasty surprises.
Chemical Reactions & Modifications: Chemistry In Action
Silver perchlorate turns up often as a halide scavenger. Toss it into a reaction mixture where you want to drag out unwanted chloride or bromide, and it will happily precipitate those as silver halide salts, leaving behind perchlorate. This habit proves valuable when preparing organometallic cations, where the perchlorate lets the “active” bit of the molecule do its thing, mostly ignoring the counterion. In some synthetic strategies, swapping out coordinated halides from metal complexes with perchlorate unlocks reactive sites and simplifies purification. You’ll also spot this salt in cyclization reactions or alkylations that avoid nucleophilic counterions. The chemistry world still spins out new uses for this unassuming white powder.
Synonyms & Product Names: What’s In A Name?
You’ll hear “silver(Ⅰ) perchlorate,” “argentous perchlorate,” and “AgClO4” thrown around—the labels can change by supplier or journal, but all roads lead to the same salt. Whatever name lands on the bottle, the compound brings the same combination of solubility and reactivity. It might not have the brand cachet that certain catalysts or ligands enjoy, but for researchers in need of a non-nucleophilic silver source, the name recognition delivers confidence across generations of chemists.
Safety & Operational Standards: Direct Talk On Handling
Anyone who has spent time with perchlorate salts, especially in open labs, knows that safety goes way past the hazard diamond. Silver perchlorate’s strong oxidizing character shows up if you bring it near organic material—paper, grease, even bits of rubber—where heat, friction, or sparks could start a fire or worse. Storing this salt away from all fuels, using glass or plastic labware, and locking bottles shut in dry cabinets isn’t just a checklist—it’s routine survival for chemists. No one trusts silver perchlorate on their bench for long without a backup label warning newcomers about the risks. Lab tales circulate about unexpected detonations during scale-up or careless mixing, serving as sobering reminders to measure and store carefully. Regular training, posted warnings, and strict inventory audits go a long way to keeping everyone in one piece.
Application Area: Real Use in the World
Outside the theory and glassware, silver perchlorate shows up in research labs, especially in reactions that need halide abstraction or very pure organometallic species. You spot it in studies of weakly coordinating anions, where the goal is to strip away as much counterion interference as possible. Analytical chemistry sometimes draws on it for specific ion studies or precipitation reactions. Some niche electronics and imaging applications count on its precise reactivity, but you won’t find a big industrial market because safer or cheaper silver sources often edge it out. In advanced labs, where obscure synthetic routes or catalyst preparation demand clean, non-nucleophilic conditions, this salt still holds strong.
Research & Development: Pushing the Boundaries
Research on silver perchlorate covers two main tracks: safer synthesis and new uses. As regulation on perchlorates tightens—due to both environmental and explosive risks—chemists chase more controlled processes. Papers from the past decade highlight solvent tweaks, purification innovations, and a growing focus on minimizing perchlorate waste. Parallel efforts explore the salt’s role in preparing next-generation catalysts for organic and polymer chemistry, where “innocent” counterions play a key role. Advances in green chemistry, with its preference for less toxic byproducts and energy use, guide a lot of the current brainstorming, though the quest for a full alternative continues.
Toxicity Research: Facing the Risks Head-On
While silver itself has a reputation as being less toxic to humans in trace amounts compared to lead or cadmium, the perchlorate anion drags along significant concerns. Chronic exposure to perchlorates—across any silver salt or unrelated industry—links to thyroid disruption and impacts on plant and animal health. Acute chemical accidents with the salt place it firmly in the hazardous material category, not just for oxidation, but for fire and contamination risk. Modern toxicity studies now map the pathways and biopersistance of perchlorates far more closely, as environmental rules keep tightening. Good lab hygiene, personal protective equipment, and smart waste disposal are baseline, not extras, as both silver and perchlorate contamination can trip up water and soil safety far away from the lab’s doorstep.
Future Prospects: Where To Next?
Silver perchlorate’s future stands at a crossroads. Demand in advanced research and niche chemistry may sharpen, driven by cleaner synthetic methods and a push for more selective catalysis. At the same time, regulatory heat on perchlorates—in drinking water, agricultural runoff, or industrial waste—keeps pushing innovation toward less hazardous routes. Some labs already migrate to less risky silver reagents except where nothing else fits. It’s hard to give up on a tool that just works when nothing else does, but safety, cost, and sustainability will keep squeezing its field of play. Until a new, safer, equally effective compound shows up, silver perchlorate will stick around in the hands of the careful, the experienced, and those willing to weigh its risks for the right reward.
What is Silver Perchlorate used for?
The Real Science Behind Silver Perchlorate
Silver perchlorate might sound mysterious, but every chemist worth their salt knows this isn’t some random lab trick. This chemical sees real work in labs across the globe. It combines silver—famous for its shine—with perchlorate, which packs a punch in the world of chemistry. You can’t talk about analytical reagents or cutting-edge materials research without stumbling across this compound.
Standing Out in Analytical Chemistry
In my early days at the university lab, silver perchlorate showed up when the professor wanted results that didn’t leave room for mistakes. It’s well-known for helping separate out different ions in a solution. Ion-exchange reactions get a boost from its strong oxidizing power and its reliable solubility in organic solvents. Students who wrestle with techniques like gravimetric analysis rely on this stuff for clear results. In one memorable lab, our class used it to measure the chloride content in water. Watching the silver ions pull chloride out so cleanly drove home why this compound wins trust.
Organic Synthesis and Silver Perchlorate’s Role
Synthesis work can get messy. Many organic chemists turn to silver perchlorate for creating organometallic compounds. Its strong oxidizing nature means it steps up as a catalyst when building complex molecules. It also finds work in rearrangement and substitution reactions. My mentor swore by it during complex group transfer steps thanks to the clean conversions it enabled. Researchers who seek excellent yields and cleaner products often keep a flask of it nearby. Pharmaceutical labs, for example, sometimes lean on it during tricky reactions that other chemicals just can’t handle as predictably.
Helping Build Sensitive Devices and Sensors
Over the last few years, modern device work called for more reliable materials. Silver perchlorate has come in handy for making conductive films, thin wires, and nano-scale materials. I spoke with a startup technician who works on printable electronics; he mentioned using silver perchlorate in their ink formulations. They count on its ability to deliver clean, high purity silver; that makes a huge difference for building sensors or flexible circuits. There’s no patience for poor electrical performance when it comes to next-generation wearables or biosensors.
Trouble and Precaution
No talk about silver perchlorate can skip over its risks. Even experienced chemists treat this chemical with respect—it doesn’t take much to see why. Perchlorates in general can be explosive under the wrong conditions, and mixing this with reducing agents or organic material brings trouble. Safety protocols require gloves, goggles, and well-ventilated hoods. In my own experience, seeing the label on a bottle is enough to make you double-check your prep work. Labs that handle it must also keep a tight leash on disposal, since perchlorate sticks around in water and threatens ecosystems if care slips.
Cutting Waste and Making Things Safer
Labs across the world aim for greener chemistry. Teams look at waste disposal, substitute less hazardous oxidizers, and train up new chemists in responsible techniques. Some research groups now test newer compounds that try to mimic what silver perchlorate can do, hoping for less risky byproducts. Sharing experiences about accidents and near misses helps everyone tighten up routines. For real progress, more companies must invest in both safer handling and deeper studies on stability. Some of my colleagues have switched entirely to alternatives for their routine analyses, limiting silver perchlorate only to cases where nothing else delivers the same power.
The Bottom Line
Silver perchlorate isn’t something the average person will ever see, but its influence reaches further than expected. From fine-tuned lab tests to the building blocks of advanced electronics, it’s a tool that carries both opportunity and risk. For those who work with it every day, strong habits and a willingness to adapt to safer ways mean everyone walks out the lab door healthy at the end of the day.
Is Silver Perchlorate hazardous or explosive?
Putting Safety First with Silver Perchlorate
As someone who’s handled more chemicals than most people do laundry, silver perchlorate always gets my full attention. It’s a pale, crystalline compound with a reputation that could scare anybody who grew up reading stories about lab explosions. Old hands in chemistry don’t treat any perchlorate lightly, and for good reason. Right out of the gate, silver perchlorate holds both oxidizing and explosive potential. Even if you’ve been careful with more familiar compounds, it’s the unique risks tied to this material that require a sharper focus.
Hazards Lurking in the Bottle
Perchlorates by their very nature push the limits of stability. The perchlorate ion carries enough oxygen to fuel strong and sometimes unpredictable oxidation reactions. Bring in silver—well known for forming sensitive explosives like silver fulminate or azide—and it doesn’t take much imagination to see trouble brewing. Silver perchlorate itself isn’t the most sensitive explosive in its class, but lab records include incidents where static, friction, or even sunlight triggered sudden decomposition or even detonation, especially in dry conditions. In the field, most incidents came from stored samples left to dry out, or careless heating during experiments. NIOSH labels this material as hazardous, and for good reason—not just because of the usual chemical burns or corrosion, but because mistreatment can, rarely, turn it dangerous in violent ways.
Knowing the Science Keeps You Safer
I learned early on that chemicals never “look” dangerous until something goes wrong. With silver perchlorate, the main issue comes from its strong oxidizing character, which means it quickly ignites or supports burning if it contacts organic material. For instance, homemade lab benches wiped clean with acetone or alcohol become fire hazards after a single spill. Water solutions are much more stable, but letting them dry out—say, in an evaporating dish—converts them into something you can barely trust around.
Understanding the Health and Environmental Impact
The health hazards aren’t just theoretical. Silver perchlorate can burn the skin and eyes, and inhalation irritates airways. Chronic exposure to silver compounds leads to argyria, a bluish-gray skin discoloration that doesn’t wash off. The perchlorate part, meanwhile, messes with the thyroid by blocking iodide uptake, which can cause problems for sensitive groups, especially pregnant women and children. Drop a load of this into groundwater—perchlorates travel fast—and it will stick around for years, causing regulatory headaches for everyone from city water managers to local doctors.
Handling With Respect, Not Fear
At the bench, there’s a big difference between “rarely explodes” and “absolutely can’t explode.” So, it feels reckless to treat silver perchlorate as just another reagent. Full PPE (face shield, gloves, coat), careful labeling, and keeping the container cool and moist with a bit of extra water solves a lot of problems before they start. Anything that heats up or dries out deserves skepticism. Document every step and hand off your knowledge to the next person, not just because it’s polite—because it could save a hand, an eye, or even a lab.
Smarter Solutions: Handling and Cleanup
There’s no fixing a spill if you only react after the fact. Preparing for containment, using spill trays, and never letting the compound near organic solvents keeps things straightforward. In my experience, sticking to solutions and never isolating the dry solid outside of controlled, necessary experiments turns risks from “explosive” to “manageable.” Disposal means careful dilution and neutralization, following EPA and local rules. Training matters—the more you know how a chemical behaves, the more comfortable you get, and the safer the lab culture becomes. Silver perchlorate isn’t out to get you, but it doesn’t forgive shortcuts.
How should Silver Perchlorate be stored?
Silver Perchlorate: A Dangerous Beauty
Silver perchlorate stands out on lab shelves as a strong oxidizer with real potential for accidents. This chemical mixes with water easily, forms beautiful crystals, and delivers consistent results in synthesis. Still, its dangers shadow its appeal. Most folks hear about perchlorates sparking huge explosions when mixed with the wrong stuff. That’s not just a scare tactic from chemistry textbooks; it comes straight from real cases. Labs have burned, and chemists have landed themselves in the hospital. Not fun — and that's why storage isn't just a formality.
Straightforward Precautions Really Save Lives
Anyone serious about chemistry, industry, or even storage at a university needs to think of silver perchlorate as a loaded gun: respect and separation come first. Keep it away from organics—no question. Mixing it with fuels, paper, or even simple solvents like ethanol leads to disaster, sometimes through one mistaken drawer pull. A plywood cabinet won’t do. Use a steel storage cabinet, properly vented, positioned where heat sources and sunlight can’t raise the temperature. Even small leaks or vapors can kick-start trouble.
Humidity Matters More Than Most Expect
In my own lab days, humidity levels turned out to be as important as fire hazard. Silver perchlorate draws in water from the air until it gets sticky—a sticky, dangerous mess that can leach into other substances. Dessicators work for small bottles. Larger quantities mean moving the storage into climate-controlled spaces.
No Compromises on Labels and Records
Chemicals lose identities fast in a busy workplace. People forget, hand-written notes rub off, and staff change hands. I once saw a poorly labeled bottle end up on a general supply shelf. That moment made me insist on clear, full hazard labels, plus logging each transfer. Manufacturers mark lots and expiration dates for a reason, but double-checking never hurts. A tiny dose of paranoia beats cleaning up after an exposure.
Handling Spills: Training Over Guesswork
Wear nitrile gloves, goggles, and lab coats instead of relying on luck. Eye-washing stations and spill kits stay within sight, not hidden in a locked janitor’s closet. In academic labs where undergrads rotate in every term, regular drills make a difference. Students or interns don’t always know that perchlorates react to basic cleaners like ammonia. Training closes this knowledge gap.
Waste Disposal: Never Down the Drain
Pouring leftover silver perchlorate down the drain won’t just destroy plumbing. It contaminates water, seeps into soil, turns up in plant roots, and can affect city infrastructure. Hazardous waste bins with secure lids avoid those headaches. Even better, using up the entire quantity ordered means fewer disposal problems down the line. Inventory reviews—monthly, not yearly—let staff spot older, unused stocks for safe removal.
Supply Chains and Inspection: Keep Shortcuts Out
Most chemical accidents happen from laziness or taking shortcuts. Trusted vendors, up-to-date Material Safety Data Sheets, and annual audits keep things above board. Lock cabinets. Double-check key logs. Inspect containers for corrosion or powder around the seals.
Handling silver perchlorate the right way means everyone leaves the lab healthy, and accidents never get the chance to turn into headlines.
What is the chemical formula and molar mass of Silver Perchlorate?
Chemical Formula of Silver Perchlorate
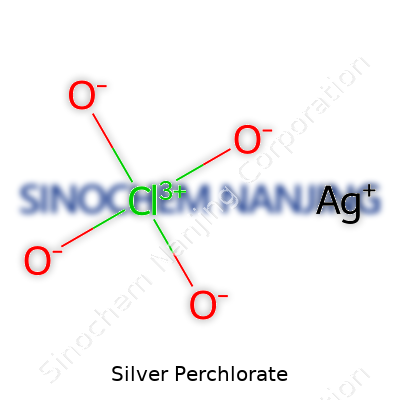
Silver perchlorate goes by the formula AgClO4. It mixes silver, known for its role in jewelry and currency, with perchlorate, a reactive group built on chlorine and oxygen. The formula reads as one silver atom joined with one perchlorate group. This structure gives silver perchlorate some unique qualities you won’t see in table salt or everyday baking soda. Not many folks realize how just swapping out the companion to the perchlorate can swing the behavior of the whole compound.
Molar Mass of Silver Perchlorate
Counting up the molar mass of AgClO4 breaks down like a solid puzzle for chemistry students and professionals alike. Silver brings 107.87 grams per mole to the mix. Chlorine adds 35.45 grams per mole. Oxygen, at four atoms per molecule here, piles on with 16.00 grams per mole each, which comes to 64.00 grams per mole from oxygen. Add it up:
- Silver (Ag): 107.87 g/mol
- Chlorine (Cl): 35.45 g/mol
- Oxygen (O4): 64.00 g/mol
So, the molar mass lands at 207.32 g/mol for AgClO4.
Why This Matters In Science and Industry
Rarely does silver perchlorate show up on a shelf at home improvement stores. Its main stage is in chemical labs, where it does the work of an oxidizer or comes in handy in analytical chemistry. Many researchers appreciate it for the way it dissolves in solvents where others won’t, making it useful for swapping out anions when other silver salts just don’t cut it.
I once watched a chemistry professor demonstrate precipitation reactions with silver salts. Silver nitrate usually soaks up the glory for these tasks, but silver perchlorate can step in when nitrate ions would throw off results. It can help chemists single out certain ions quickly, all thanks to its solubility and reactivity driven by that ClO4 group.
The role of perchlorates in environmental and industrial chemistry makes silver perchlorate important to handle properly. Both perchlorate and silver ions can have real environmental and health impacts if left unchecked. For example, perchlorates in groundwater can interfere with thyroid function in humans, while silver can be toxic to aquatic life. That makes proper storage and disposal practices essential, guided by strict regulations.
Solutions and Responsible Use
One thing I’ve found in lab work: clear procedures are essential before anyone opens a bottle labeled AgClO4. Labs maintain strict logs, provide proper protective equipment, and train researchers before use. Safe handling matters since perchlorate salts, in general, can be explosive under the right conditions.
Recycling programs for chemicals and tight controls on discharge to wastewater help limit the impact on the environment. Institutions often support third-party audits and follow national hazardous waste protocols for chemicals like silver perchlorate. Manufacturers develop safer container designs and better labeling so both rookie and veteran researchers can avoid costly mistakes.
Clear communication and updated safety training remain the best defenses for anyone working around chemicals with strong effects, including AgClO4. These steps, combined with continuous research, help keep people and the environment safe, ties into everyday chemistry work, and builds trust in scientific results.
Can Silver Perchlorate be used in analytical chemistry or synthesis?
Looking Beyond the Usual Reagents
Most people outside a lab circle have never heard of silver perchlorate. In a simple bottle, this compound blends two things: the unique reactivity of silver and the almost reckless energy of perchlorate. People like myself, who once spent hours balancing test tubes and jotting down yields, remember encounters with this reagent as brief and calculated. The buzz around silver perchlorate centers on its uses in analytical chemistry and synthesis—and for good reason.
What Silver Perchlorate Really Does in the Lab
Let’s face it, not every silver salt makes a great partner for chemical analysis. Some clog up reactions with messy byproducts or lose their punch in water. Silver perchlorate, on the other hand, brings real strengths. Its high solubility in water gives analytical chemists an edge for precipitation reactions, such as selectively pulling halides out of solution. If you’re testing a sample for chloride, bromide, or iodide, silver perchlorate shows its value by reacting without throwing a wrench in the works. This trait makes it attractive for gravimetric analysis, a method where small differences matter.
Synthesis specialists notice that silver perchlorate opens doors, too. The perchlorate ion acts as a “non-coordinating anion,” stepping back and leaving silver to interact cleanly with other molecules. In laboratories, this can mean smoother routes to complicated silver complexes or organic molecules where the byproduct’s behavior can throw an entire reaction off. There’s a sense of efficiency here, which I learned firsthand. A reaction using silver nitrate hit a dead end because the nitrate complicated the outcome; with silver perchlorate, the result came through crystal clear. Letting the perchlorate step aside can be a real win.
Why Worry? Safety and Practical Problems
Everything sounds great until we talk about safety and sustainability. Perchlorates carry a loud warning tag in chemical manuals. They react furiously with organic matter, and explosions in labs from mishandled perchlorates make news every decade or so. In my experience, knowing the real risks means reading beyond the first five pages of a lab protocol. Adding silver—expensive on its own—means the costs stack up, both in dollars and in dealing with hazardous waste. Disposal rules grow tighter for perchlorates, especially since they’re linked to environmental problems.
This kind of reagent tempts chemists into shortcuts, but anyone spending long days handling them remembers that a single slip can cost more than just supplies. With labs facing more pressure over green practices and budgets, silver perchlorate just doesn’t show up in large-scale production. Luckily, other silver salts and more benign anions get the job done for many analytical tasks, if not always with the same precision.
Searching for Solutions
People working in research and industry test newer reagents that do the same job with less drama. Green chemistry pushes researchers into finding alternatives that offer selectivity, without the environmental baggage or safety scares. Some labs shift toward ion chromatography for halide detection instead of old-school precipitation. In synthesis, careful selection of solvents and ligands sometimes cuts out the need for perchlorate entirely. Other times, improved protocols just mean less hazardous waste by smart planning on a small scale.
In the end, silver perchlorate has its place on the shelf—but only in trusted and practiced hands. Experience, training, and an honest look at the risks decide if it’s the right tool for the job. The lesson from time in the lab sticks clear: a clever approach usually wins over brute chemical force and saves everyone trouble down the road.
| Names | |
| Preferred IUPAC name | silver(I) perchlorate |
| Other names |
Perchloric acid, silver(1+) salt Perchloric acid silver(1+) salt Silver(I) perchlorate |
| Pronunciation | /ˈsɪl.vər pərˈklɔː.reɪt/ |
| Identifiers | |
| CAS Number | [10025-73-7] |
| Beilstein Reference | 353990 |
| ChEBI | CHEBI:75955 |
| ChEMBL | CHEMBL3300892 |
| ChemSpider | 8802 |
| DrugBank | DB14638 |
| ECHA InfoCard | ECHA InfoCard: 100.029.790 |
| EC Number | 231-511-9 |
| Gmelin Reference | 61300 |
| KEGG | C10915 |
| MeSH | D002561 |
| PubChem CID | 24956 |
| RTECS number | SD9800000 |
| UNII | 0E96G2DI4A |
| UN number | UN1512 |
| Properties | |
| Chemical formula | AgClO4 |
| Molar mass | 207.32 g/mol |
| Appearance | White crystals |
| Odor | Odorless |
| Density | Density: 2.49 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.1 |
| Acidity (pKa) | -3.29 |
| Basicity (pKb) | -10 |
| Magnetic susceptibility (χ) | −27.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.744 |
| Viscosity | Viscous liquid |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 212.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -235.7 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB38 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H271, H302, H312, H332 |
| Precautionary statements | P210, P220, P221, P222, P280, P370+P378, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Explosive limits | Non-explosive |
| Lethal dose or concentration | LD50 oral rat 117 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 117 mg/kg |
| NIOSH | SN1220000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Silver Perchlorate: 0.01 mg/m3 (as Ag) |
| REL (Recommended) | 50 g/L |
| IDLH (Immediate danger) | IDLH: 15 mg/m3 |
| Related compounds | |
| Related compounds |
Perchloric acid Sodium perchlorate Potassium perchlorate Ammonium perchlorate Silver nitrate |

