Silver Nitrate: A Deep Dive into Its Story, Uses, and Impact
Historical Development of Silver Nitrate
Long before white lab coats filled with chemists working under fluorescent lights, humans discovered the sharp and unusual reactions of silver salts, especially silver nitrate. Early records dating back to the Middle Ages describe artisans and alchemists experimenting with this striking compound, drawn by its ability to blacken upon contact with light and organic matter. Later, famous pioneers like Albertus Magnus wrote about methods to create silver salts, recognizing their transformative potential. Everyone from medieval physicians treating wounds to 19th-century photographers mixing solutions in studio darkrooms valued silver nitrate for reasons that ranged from health care to the birth of modern photographic imaging. During the late 1700s, German chemist Carl Wilhelm Scheele detailed the synthesis and properties, marking a turning point that opened its path for industrial and scientific adoption. Today’s industries still lean on a legacy built up through centuries of discovery, trial, and curiosity-driven research.
Product Overview
Silver nitrate appears as colorless, nearly transparent, crystalline granules or powder. Unlike other silver compounds, it actually dissolves in water quite well, granting it versatility in application. This material shows up in small bottles on hospital trays, as reagent kits in classrooms, and in manufacturing sites preparing advanced coatings or electronics. Its antiseptic punch doesn’t take much labeling: the mere touch sets dark stains on skin, a hint about its reactive power. With a molecular formula of AgNO3, the simplicity of structure belies the broad reach it holds across scientific, medical, and industrial fields. In commerce, silver nitrate is sold in varied grades, from analytical pure to industrial grades, clearly indicating the role purity plays depending on use—this fine differentiation influences outcome in both the lab and in final product quality.
Physical and Chemical Properties
Many chemicals boast a particular look or behavior, but silver nitrate’s personality stands out. It forms clear, rhombic crystals melting at 212°C, and once it finds water, those crystals disappear without much coaxing. You won’t pick up any smell, but the sharp, metallic taste can linger (though direct tasting should never occur given toxicity). The substance oxidizes everything from skin proteins to printing emulsions. Exposed to light, silver nitrate decomposes, giving off fine particles of pure silver—this property paved the way for photography. In nitric acid, it stays stable, resisting further breakdown, but throw in organic matter, acids, or bases outside the mild-to-neutral range, and reactions change rapidly. At room temperature, silver nitrate dissolves in various solvents: water, alcohol, and even acetone to a lesser degree.
Technical Specifications and Labeling
Every bottle or packet carries specification labels detailing composition, concentration, purity, hazards, and recommendations for storage. Analytical reagent grades reach purity levels above 99.9%, while technical and industrial forms may allow for traces of contaminants like chlorides, sulfates, or heavy metals within strict boundaries. Labels often highlight the hazard symbol for oxidizers, instructions for avoiding exposure to light, and storage temperature limits. In my own experience working with chemical supply chains, the paperwork and regulatory certifications shoulder as much importance as the chemical itself—mislabeling can affect entire production runs or prompt laboratory audits. Batch numbers, test results, and manufacturer certifications increase traceability, a critical factor across pharmaceuticals and electronics manufacturing.
Preparation Method
Silver nitrate comes from the reaction of pure silver with concentrated nitric acid. Add silver shavings or metal to the acid in a glass vessel; a fizzing reaction releases nitric oxide and gives a blue-green solution of silver nitrate. Slow evaporation concentrates the solution, precipitating crystals as it cools. Filtration, multiple washings, and slow drying ensure a product fit for high-purity needs. The efficiency relies on quality starting materials and careful control over temperature and evaporation speed—the entire setup can falter if handled impatiently or with impure silver, revealing the intersection of science and artisanal care in the chemical industry.
Chemical Reactions and Modifications
Silver nitrate works as both chemical tool and building block. In the laboratory, chemists use it to test for the presence of halides (chlorides, bromides, iodides) by noting visible precipitates—an immediate, unmistakable reaction. In medical practice, silver nitrate sticks and solutions cauterize wounds and treat infections because of direct protein precipitation and antimicrobial action. For organic synthesis, it oxidizes and reshapes molecules efficiently. Industry moves further, converting silver nitrate into photographic emulsions and cutting-edge nanomaterials—both areas where precision and predictability shape outcomes. Change the base, add reducing agents, or blend with other metal salts, and modifications yield products like silver oxide, silver chloride, or even colloidal silvers for countless technical purposes.
Synonyms and Product Names
Alongside the name “silver nitrate,” chemical catalogs and patents call this compound by older or alternative names: lunar caustic, nitric acid silver salt, AgNO3, or simply nitrate of silver. Sometimes, in medical texts and older pharmacopoeias, you’ll see “argenti nitras”—the Latin touch reflecting ages of scholarly use. These synonyms exist both because of historical usage and the diverse array of industries drawing upon silver nitrate, each carving out its terminology tied to tradition, trade, or regulatory compliance.
Safety and Operational Standards
Few substances demand as much respect in handling as silver nitrate. Direct skin or eye contact burns with dark stains and real tissue damage, never mind the risk of accidental ingestion. Strongly oxidizing, it heightens fire risk in the presence of organic materials—storage away from paper, cloth, combustibles is non-negotiable. Laboratories and industrial spaces install local exhaust ventilation, train personnel on the use of PPE (gloves, goggles, coats), and set up spill and wash stations. Documented in both OSHA and international guidelines, silver nitrate storage and disposal require sealed, light-resistant bottles and neutralizing solutions at hand. Speaking from personal lab hours, following these standards is not about regulatory boxes—it is about avoiding hard-earned mistakes, poverty of attention, and the cost of accidents.
Application Area
Silver nitrate fills more niches than one would expect. Medical practice leans heavily on its antibacterial and caustic properties. Doctors treat warts and ulcers, prevent newborn eye infections, and manage wound care, especially before antibiotics went mainstream. In laboratories, silver nitrate acts as an analytical staple, its halide tests a basic step in water quality surveys or forensic work. Photography’s ancestry ties to silver nitrate—the magic behind early film and print development. Electroplating, mirror-making, and high-tech electronics benefit from its precise release of silver. Water purification systems harness its biocidal edge to kill contaminants. Even artwork and restoration turn to silver nitrate’s reactions to produce authentic, time-worn finishes.
Research and Development
Innovation rarely skips over a material as versatile as silver nitrate. Researchers have probed its role in nanotechnology, creating silver nanoparticles fine enough to disrupt bacterial and viral membranes, an area showing direct impact for surgery, wound dressings, and new drug delivery systems. Scientists studied advanced coatings embedding silver nitrate to resist microbial growth in medical devices, showing positive results in reducing hospital-acquired infections. In electronics, precise deposition from silver nitrate solutions allows for microcircuitry manufacture well beyond older techniques. Recent journals highlight the use of silver nitrate in catalysis, especially for green chemistry push, transforming reactions for less waste and greater selectivity. In my time at interdisciplinary conferences, silver nitrate always surfaced when people talked about translating foundational chemical principles into near-futuristic devices.
Toxicity Research
Toxicologists have dug deeply into the effects of silver nitrate on humans and the environment. Ingestion or repeated skin exposure leads to argyria, a gray-blue discoloration of tissues that does not fade easily. Though acute toxicity in small laboratory settings can be managed through fast decontamination, chronic exposure builds up silver particles in the body, creating lasting effects. Airborne dust or droplets bring risk to eyes and lungs, prompting strict airborne exposure limits. Environmentally, silver nitrate runoff can harm aquatic life—tiny concentrations disrupt cell membranes and lead to widespread toxicity across food chains. Regulators track movement and handling as closely as pharmaceuticals in many regions. In our own review for a university environmental safety board, the biggest risks came during improper disposal or accidental leakage during transport, making training and accountability more important than ever.
Future Prospects
The future of silver nitrate draws on its old strengths while pushing toward new frontiers. Nanotechnology and medicine will likely turn to silver nitrate as starting point for smarter antimicrobial surfaces and advanced diagnostics, especially as societies confront antibiotic resistance and hospital-acquired infections. Electronics manufacturing keeps drawing on its reliability to lay down fine silver lines for flexible circuits and solar panels. Environmental science looks toward improved ways to detoxify and recover silver from waste, both for economy and sustainability. Regulatory tightening makes safe handling just as valuable as anything discovered in the lab, but innovation does not pause for paperwork. Global demand for high-purity silver nitrate, paired with evolving standards and green chemistry techniques, points to a future where this old compound still writes fresh chapters.
What is Silver Nitrate used for?
More Than Meets the Eye
Silver nitrate doesn’t get much attention at family dinners or backyard barbecues, yet it finds its way into both history books and medicine cabinets. I remember the first time I used it—it was in a biology lab, watching how a few drops made fingerprints jump out under gentle light. Its action wasn’t some trick; it hooked me on chemistry’s usefulness.
Medicine’s Old-Timer Still Going Strong
Doctors and nurses trust silver nitrate for a reason. Its power to stop bleeding and zap bacteria doesn’t fade with new tech. People might see small brown sticks in emergency rooms—they use those to treat nosebleeds that won’t quit. A few dabs seal off the trouble spots by causing blood vessels to close up. It can even take care of stubborn granulation tissue that grows where it shouldn’t, especially around wounds or after surgeries.
Some might think of it as a throwback, but silver nitrate remains on hand for newborn care too. Until the late 20th century, nurses dropped a dilute solution into babies’ eyes to protect against infections from birth—an approach that saved sight back when antibiotics hadn’t joined the scene. The practice faded as new medicine arrived, but in places where money or supplies run short, those tiny vials still protect freshmen lives.
Stains, Science, and Surprises
Silver nitrate isn’t limited to clinics. In chemistry labs around the globe, this compound marks its place in classes studying reactions or analyzing minerals. Drop silver nitrate into a solution—if chloride is lurking there, it shows up as a cloudy white or gray. That easy color test helps water treatment plants check purity, and guides environmental scientists tracking down heavy metals near factories. I’ve seen science students use it for quick checks on tap water, learning in minutes what could take hours by other means.
Traditionally, it also served as the first ingredient for black-and-white photography. Silver nitrate reacts with light and lays down an image on film, capturing memories before the smartphone came along. This chemistry powered the photos pasted in generations’ family albums and makes me appreciate the patience old photographers had for timing and technique.
Pitfalls and Solutions
Trouble can follow this potent powder. Silver nitrate burns skin, stains clothes forever, and stings if it touches eyes in the wrong way. I’ve seen the blackish marks it leaves on fingers linger for days. Places that rely on it—labs and clinics—handle it with careful training and tight rules. Gloves and goggles aren’t optional, and every spill gets a full cleanup. Manufactures print warning labels bold and clear, pushing safety to the front so that a valuable tool doesn’t turn into a hazard.
The world steadily searches for greener, safer options. Some innovators use silver nanoparticles in bandages, aiming for the same bacteria-zapping effect with fewer side kicks. Researchers also experiment with lower doses and alternatives, measuring what gets the job done without adding risks. Solutions grow from shared experience—trade knowledge, honest error, and smart regulation.
Everyday Impact, Quietly Used
Silver nitrate shapes health, science, and even art, mostly without fanfare. Its reach stretches further than people see, and its story isn’t finished yet. Even as new materials appear, silver nitrate holds a steady role because it gets results—fast, clear, and trusted by those who know what careful chemistry can do.
Is Silver Nitrate safe to handle?
A Closer Look at a Common Chemical
Silver nitrate shows up in classrooms, clinics, and even in darkrooms where photography lives on. In my chemistry classes, we used it for tests and experiments, watching it turn skin dark brown and spotting it on glassware days later. Silver nitrate grabs attention—both for its reactions and for questions about its safety.
What Happens on Contact
Touching silver nitrate leaves a mark. The compound reacts with skin proteins, turning fingertips or hands dark or black. In my own school lab, nobody forgot which students skipped gloves after silver nitrate made its way onto their hands. The discoloration sticks around for days, sometimes over a week, even when you scrub hard.
A dark stain may look odd, but the bigger risk comes from silver nitrate's chemical nature. It's caustic. On skin, it can burn or blister in higher concentrations or after long exposure. Eyes take a heavy hit if the chemical gets in—damage happens fast, and it won’t wash away without lingering pain or possibly permanent effects.
Not All Risks Are Easy to Spot
Even after stains fade, the chemical keeps working behind the scenes. Breathing in dust from silver nitrate starts headaches and irritates the respiratory tract. Eating or drinking any by mistake—dangerous. Silver nitrate in the bloodstream can trigger nausea, stomach cramps, and in the worst cases, organ damage. This isn’t a common accident, but poor labeling or spilled powder on shared surfaces raises the odds.
Silver nitrate’s reactivity can surprise, especially mixed with reducing agents or other metals. Living through an unexpected classroom fizz or pop makes you respect even small amounts of this crystal.
Regulations and Real-World Use
Silver nitrate appears in medical offices for a reason. It stops bleeding and removes warts with care from a trained professional. The FDA still lists it among essential drugs, but everything hinges on careful measurement and secure packaging. In photography and industry, chemical fume hoods and goggles block the worst from reaching people. Labs that take shortcuts see more accidents, and stories spread quickly about mishandled chemicals causing evacuations or trips to campus clinics.
Better Handling Means Fewer Problems
Wearing gloves, eye protection, and lab coats might sound like overkill, but after even one burn or stain, few students or workers want to chance it. Rinsing instantly after contact reduces harm, but thorough instruction—and repeated reminders—make a bigger difference. Labeling containers, securing lids, and storing silver nitrate away from heat or acids drops risk even further. Some places have moved to pre-mixed solutions or single-use ampules to limit exposure in schools or clinics.
The stakes grow larger in places without proper training. Young students and untrained staff take more risks, and sometimes people treat silver nitrate like regular salt, not realizing the impact. Sharing real stories and facts keeps safety at the front of the conversation.
Learning From Each Other
Most injuries and mistakes come from skipping safety steps or misunderstanding what silver nitrate can do. Routine drills, visible safety data sheets, and upfront conversation go further than quiet warnings buried in paperwork. In classes, we made sure to stop and talk before bottles opened or pipettes dipped in. People remember what they see and experience, not just what they read.
Respect for silver nitrate starts with recognizing its double-sided nature—useful in the right place, but always worth handling with extra care. It’s more than a science lesson. It’s a tool that shapes habits for every other chemical down the road.
How should Silver Nitrate be stored?
Respecting the Risks
Anyone who has worked in a lab knows silver nitrate by sight: those crystalline white chunks pack a punch well beyond their delicate look. Over time, I’ve seen careless storage practices create headaches ranging from ruined glassware to damaged skin. It’s easy to underestimate how quickly moisture, warmth, and a loose cap can cause problems with this chemical. Silver nitrate reacts with organic material, light, and even greasy fingertips; in the wrong setting, that means resources wasted, safety threatened, and expensive clean-ups that no one wants.
The Trouble with Light and Air
Science textbooks show silver nitrate turning brown or black. The culprit is light. If you leave a container out under the lights, even for a few days, you start to see that ugly gray hue. At best, you lose purity. At worst, you end up with a mess tougher to handle the next time someone opens the jar. Air also plays a role. Humid environments transform clumps into a sticky situation, which makes accurate weighing a guessing game. Glass containers with tight-fitting lids provide real value here—simple, but proven. Polyethylene or amber glass helps cut down on light exposure.
Labeling Isn’t Optional
Labeling may sound boring, but you can spot sloppy labeling anywhere, and it always catches up with people. In shared spaces, I’ve dug through shelves of mystery bottles—no date, no hazard warning, no hint of who brought in what. All it takes is one person reaching for what they think is salt and ending up with a burned hand, or a spill nobody wants to own. Proper hazard labels save a world of trouble. The best labs put the purchase date on container labels, too, so no one uses expired or decomposed material.
Choosing the Right Storage Spot
Locking up silver nitrate isn’t about paranoia, it’s about responsibility. Even small children at home could suffer severe harm from exposure. Facilities that handle silver nitrate owe an ethical duty to keep these chemicals away from unauthorized hands—locked cabinets, restricted key cards, whatever it takes. You want it dry, cool, and well away from sunlight or heat sources. I’ve seen some places keep these jars in the same cabinets as acids—never a good call, since chemical fumes accelerate deterioration and create dangerous interactions if leaks occur.
Disposal and Clean-Up Habits Matter
Silver nitrate solutions stain skin for days, so even short contact leaves a mark. More importantly, any leftover spills must get proper treatment—not just water and a rag, because that sends silver down the drain and risks environmental damage. Most municipalities classify silver nitrate as hazardous waste, and ignoring that comes with heavy fines, not to mention an ethical hit if silver leaches into the water supply. Dedicated spill kits, with clear instructions and personal protective equipment, should always sit within easy reach wherever this material finds use.
Solutions That Work in Practice
The best storage keeps these risks front and center. That means routine checks for leaky containers, regular retraining of staff, and a culture where nobody feels shy about reporting a problem. Simple systems—like color-coded shelves or checklists—help prevent complacency. In a world where regulations grow tighter and raw materials more expensive, respect for safety and the environment isn’t an extra, it’s part of handling chemistry with care.
What precautions should be taken when using Silver Nitrate?
Why Silver Nitrate Deserves Respect
Silver nitrate has made its name as a handy chemical in medicine, photography, and research labs. I remember seeing those small, dark bottles stashed away in my university’s chemistry storeroom—always treated with extra care. It looks harmless, just another crystalline powder, but its punch reveals itself fast once the seal breaks. Silver nitrate can burn, stain, and cause tissue damage before anybody realizes what’s happened. A tool with this much power always requires forethought and a good deal of caution.
The Risks Everyone Should Know
Even a small drop can put a dark, persistent stain on skin. The real concern lies deeper: direct contact can cause serious irritation or even burns. When splashed in the eyes, silver nitrate quickly damages delicate tissue, sometimes leading to vision loss if not rinsed away at once. Inhalation brings its own risks. The dust lingers in the air and, if breathed in, can inflame lungs and airways. Swallowing silver nitrate pushes the body into distress—nausea, vomiting, and damage to internal organs often follow. Knowing these risks means taking precautions isn’t optional; it’s a necessity.
Putting Safety Plan Into Everyday Practice
Every time I’ve handled silver nitrate, proper barriers made all the difference. Gloves stop it from reaching exposed skin, and lab coats prevent accidental spills from soaking into clothing. Standard prescription glasses won’t cut it for eye protection, so proper safety goggles need to be worn whenever there’s a risk of splashes. Sometimes I saw colleagues try to skip this step, only to regret it once their hands picked up those tell-tale black stains for a week or longer. If a spill does happen, water is the first line of defense. Immediately rinsing the area with plenty of water can wash away most of the danger before it sets in.
Respecting Storage and Disposal
Silver nitrate sits best in a dry, dark spot. Light sparks off a chemical reaction, so tightly sealed amber bottles become essential. Keeping it away from organic materials and strong reducing agents avoids unwanted fires or explosions, a risk not always obvious until something goes wrong. Out-of-date or spilled silver nitrate doesn’t just go down the drain. Over the years, I learned that specialty waste bins, set aside for heavy metals and hazardous chemicals, are the only place these leftovers belong. Wastewater plants can’t easily remove silver, and even small amounts of it find their way into rivers and soil. Those with environmental stewardship in mind should make sure their labs follow local hazardous waste disposal regulations.
Training, Teamwork, and Clear Labels
Safety talks in the lab can feel repetitive, but with chemicals like silver nitrate, no one should work alone if they aren’t fully trained. I still remember a classmate panicking during their first spill, unsure what to do. With regular drills and a clearly posted protocol, confusion stays at bay, and the team moves into action. Every bottle and container must carry a clear label—hazard icons, concentration, date received. No room for mystery when mistakes could cost someone an eye or their skin’s health.
Simple Changes Bring Safer Outcomes
The right mindset goes a long way. Using silver nitrate without shortcuts, keeping workspaces uncluttered, and sharing firsthand stories about mishaps all help keep people aware. Learning from accidents, not just in textbooks but from real people’s experiences, sticks far longer. By looking out for one another and refusing to cut corners, everyone can keep silver nitrate’s power working where it belongs—never at the expense of safety.
What are the potential side effects of Silver Nitrate exposure?
More Than Stained Fingers
After spending time in a chemistry lab and seeing silver nitrate in action, it’s easy to forget how risky some of these chemicals can be. Silver nitrate is famous for leaving black marks wherever it touches skin—something anyone who’s picked up a pipette remembers. Beneath those dark stains sits a whole range of other health problems, most of which rarely get talked about outside safety training or long pharmaceutical brochures.
Direct Contact Isn’t Just a Nuisance
Silver nitrate makes its mark by reacting with skin proteins, turning them a deep brown or black. This reaction isn’t just cosmetic. Even small amounts of exposure can lead to irritation, burns, and sometimes painful ulcers if the solution stays on too long. Growing up, a family friend who worked in photography—an industry where silver nitrate used to play a major part—described regular cracked, itchy skin after accidental splashes. Gloves matter for a reason.
Getting the Eyes Involved
Contact with the eyes takes the conversation to an entirely different level. Silver nitrate causes immediate pain, redness, and a risk of permanent eye damage if it gets past an eyelid. Even a splash of diluted solution means a trip to an emergency room. The American Conference of Governmental Industrial Hygienists highlights how quickly this compound can eat away at sensitive tissues.
Breathing It In? Not Good News
Sometimes, working with powdered silver nitrate means there’s a risk of airborne particles. Breathing these in can lead to everything from coughs and sore throats to burning sensations through the nose and lungs. If exposure keeps up, there’s a chance for chronic respiratory issues. I remember being told stories by former glassworkers who joked about ‘silver dust cough’—it stopped being funny when co-workers started migrating toward jobs with better ventilation.
Swallowing Is an Old Problem
Ingesting silver nitrate isn’t likely during most jobs now, but stories of accidental or experimental consumption point to serious health risks: stomach pain, vomiting, and even tissue necrosis. Historically, medics used silver nitrate drops for newborns’ eyes to kill bacteria, yet high concentrations caused burns. Medicine keeps tweaking protocols for just this reason: overdoing it or sloppy handling can do more harm than good.
Long-Term Build-Up Plays Rough
Regular silver nitrate contact can cause a condition called argyria where skin and tissues turn a bluish-gray. Beyond the change in appearance, argyria becomes permanent and sometimes affects internal organs too. Reports from medical literature make it clear that this isn't just an urban legend. The National Institute for Occupational Safety and Health documents workers developing this condition after years of exposure. Silver builds up with each contact, so even tiny amounts over a long period leave their mark.
Prevention Starts With Basics
Practical fixes beat panic every time. Ventilation—both on the workbench and overhead—carries away dangerous dust. Nitrile gloves, goggles, and lab coats cut down the risk of direct contact. Training people to recognize the dangers involves more than just theory; regular, hands-on drills help everyone know what to do when a splash does happen. Keeping wash stations and eye rinses close makes accidents less harmful in a hurry.
Thinking Beyond the Lab
Silver nitrate shows up outside science labs, too. Tattoo artists sometimes use it to treat overactive granulation tissue. In some places, silver-based medicines show up in unregulated online products. Anyone tempted by miracle cures needs to double-check health warnings. Side effects aren’t theoretical—they’re well-documented and can follow people for years.
Knowing the hazards, keeping up good practices, and speaking up when something doesn’t feel safe make all the difference. At the end of a day in any workshop or lab, preventing a single accident outweighs cleaning up after it.
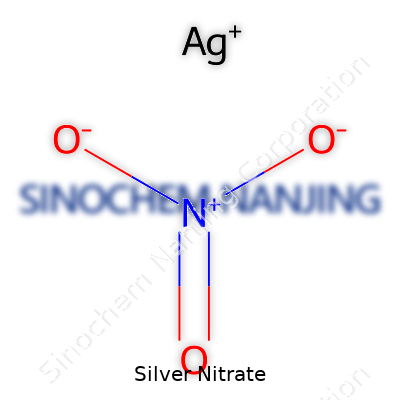
| Names | |
| Preferred IUPAC name | Silver nitrate |
| Other names |
Lunar caustic Argentum nitricum Nitric acid silver(1+) salt Silver(I) nitrate |
| Pronunciation | /ˈsɪl.vər ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 7761-88-8 |
| Beilstein Reference | 12768 |
| ChEBI | CHEBI:31344 |
| ChEMBL | CHEMBL1402012 |
| ChemSpider | 20652 |
| DrugBank | DB02794 |
| ECHA InfoCard | 03b0dfb8-0498-4f27-a9ee-62e61fbfa3c6 |
| EC Number | 231-853-9 |
| Gmelin Reference | 82242 |
| KEGG | C01086 |
| MeSH | D012704 |
| PubChem CID | 24470 |
| RTECS number | VW4725000 |
| UNII | 202U1VXT6B |
| UN number | UN1493 |
| Properties | |
| Chemical formula | AgNO3 |
| Molar mass | 169.87 g/mol |
| Appearance | Colorless or white crystalline solid |
| Odor | Odorless |
| Density | 4.35 g/cm³ |
| Solubility in water | 122 g/100 mL (20 °C) |
| log P | -2.42 |
| Vapor pressure | Negligible |
| Acidity (pKa) | -1.98 |
| Magnetic susceptibility (χ) | '-21.0 × 10⁻⁶ cm³/mol' |
| Refractive index (nD) | 1.50 |
| Viscosity | 1.58 cP (20°C) |
| Dipole moment | 7.23 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -123.0 kJ/mol |
| Pharmacology | |
| ATC code | S01AX04 |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation, very toxic to aquatic life. |
| GHS labelling | GHS02, GHS05, GHS07, GHS09 |
| Pictograms | GHS03,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | Hazard statements: "H272, H314, H410 |
| Precautionary statements | P210, P220, P221, P260, P264, P273, P280, P302+P352, P305+P351+P338, P308+P313, P332+P313, P337+P313, P362+P364, P370+P378, P391, P501 |
| NFPA 704 (fire diamond) | 2-3-2-OX |
| Lethal dose or concentration | LD50 (oral, rat): 117 mg/kg |
| LD50 (median dose) | LD50 (median dose): 50 mg/kg (oral, rat) |
| NIOSH | 0166 |
| PEL (Permissible) | 0.01 mg/m³ |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Silver carbonate Silver chloride Silver sulfate Silver iodide Silver fluoride |

