Silver Fluoroborate: An Editorial Look at its Evolution, Properties, and Future
Roots of Silver Fluoroborate: How Science Got Here
Few chemicals spark curiosity in a lab quite like silver fluoroborate. Tracing back through journals and patents, you find traces of this compound’s use in specialized synthetic chemistry emerging in the later half of the twentieth century. A need for strong, stable non-coordinating anions drove exploration away from silver nitrate or common halides, which often brought unwanted reactivity. Once chemists realized how the tetrafluoroborate anion’s bulk could stabilize reactive cations, silver fluoroborate found recognition as a tool for precise metathesis reactions and as a helpful promoter in organic syntheses. Its significance grew from those early papers highlighting improved yields or unique selectivities, and interest trickled down from academic labs into industrial careers in pharmaceuticals, organometallics, and electronics.
Getting to Know the Compound: Appearance and Core Traits
Silver fluoroborate has characteristics that draw both respect and caution. As a solid, it usually presents as a white or off-white crystalline powder. The substance doesn’t go unnoticed in a lab, not just for its color but for its known oxidizing power and reactivity. Silver, never a cheap commodity, guarantees that every gram gets monitored and handled with intention. Its solubility in water and various polar organic solvents makes it a flexible reagent, yet every technician knows the threat posed by moisture or traces of reducing agents. Under ordinary conditions, it maintains stability, but the compound reacts if exposed to excess heat or strong bases, releasing boron-containing byproducts or forming unsuspected silver precipitates.
Packed With Chemistry: Composition, Structure, and Labeling
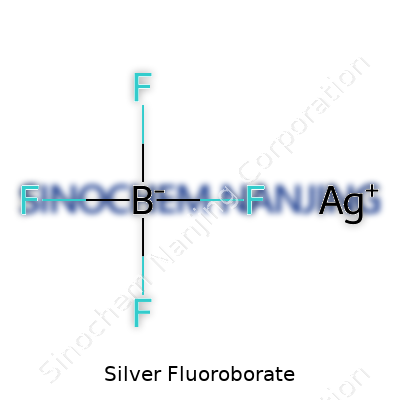
In terms of core composition, silver fluoroborate brings together a single silver ion with the tetrafluoroborate counterion. Cautious chemists see the BF4 group and think of both strength and volatility. The label should make clear its irritant potential, its need for dry storage, and the hazards tied to contact with acids or strong moisture. In the lab, you see containers with desiccant packs, clear hazard icons, and, at reputable facilities, supplemental notes describing proper ventilation and eyewash stations within arm’s reach. Chemically, it doesn’t take much to see how the slightly larger BF4- anion shifts the reactivity away from traditional halides. That shift opens doors for selective syntheses or clean-up steps in organic pipelines.
Building Silver Fluoroborate: Reactions That Matter
No one walks into a chemical supply room and finds silver fluoroborate growing on the shelves. Chemists usually prepare it from silver oxide or silver carbonate, reacting directly with fluoroboric acid or a readily-available fluoroborate salt. Some labs work up careful stoichiometric balances to limit waste and maximize yield, but the main challenge remains the isolation of a moisture-free product. Each batch demands solid purification—often crystallization from approved solvents under an inert atmosphere—and strict protocols for segregating leftover acids and metal salts. Regular glove use, careful weighing, and close attention to trace contaminants all come standard in places where this compound enters a synthetic scheme.
On the Bench: Chemical Reactions and Pathways
On the reaction front, silver fluoroborate acts both as a useful silver source and a non-coordinating anion for stabilizing sensitive organic cations. In metathesis reactions, the compound takes part in smooth silver-halide precipitation, allowing isolation of complex organic or coordination compounds free from contaminating halide species. For research teams hoping to shift the balance in favor of novel catalysts or intermediates, this reagent transforms outcomes where more reactive anions would fail. Side reactions do exist—most involve decomposition under heat, light, or prolonged standing in damp air—so labs take on required safeguards to limit waste and unwelcome surprises.
What’s In a Name: Synonyms and Aliases
Silver fluoroborate appears under a handful of names in literature and catalogs, including silver tetrafluoroborate and its formula AgBF4. Some publications treat it simply as “silver(Ⅰ) fluoroborate,” a nod to its +1 oxidation state. While the number of synonyms isn’t vast, clarity matters in safety protocols and cross-referencing research papers. The slight variations sometimes hint at different crystalline hydrates or batch-specific purities, which don’t carry the same properties when swapped in carefully controlled syntheses.
Working With Care: Safety and Operational Practices
Anyone who’s handled silver or fluorinated chemicals knows the double-edged sword these bring. Silver fluoroborate can irritate skin, eyes, and respiratory tracts; more than one researcher has learned the hard way after an accidental spill or careless glove adjustment. Contact with strong acids or bases can drive off dangerous fumes. Environmental agencies flag both silver and the fluoroborate group for concerns about aquatic systems and decomposition products, so responsible chemical stewardship guides every step from storage and labeling to disposal. In the lab, personal protective equipment feels less like an extra step and more like a requirement, and every bottle stays locked in a dry, clearly marked cabinet.
Where It Counts: Application Areas
The utility of silver fluoroborate stretches across synthetic chemistry, especially where halide-free conditions make or break a project. In organometallic chemistry, it’s used to abstract halide ligands from sensitive complexes, generating catalytically active species impossible to make with simple salts. Cyclopropanation, carbon–carbon bond construction, and transition metal catalysis—these are places where the compound earns its keep. Beyond research, process chemists have eyed applications in battery development, particularly as a component in advanced electrolyte systems. It has shown up in methods for high-purity silver plating and etching in microelectronics, underlining a relevance that moves beyond academic papers.
Pushing the Boundaries: R&D and Pursuit of New Knowledge
In research circles, silver fluoroborate dances on the cutting edge of catalyst development and new functional material synthesis. Scientists keep looking for ways to exploit its non-coordinating anion to stabilize fleeting intermediates in photochemistry and electrochemistry. Leading groups use it to push forward methods for clean halide abstraction, streamline processes in peptide synthesis, or speed up construction of complex molecular frameworks. Whenever a new electronic material or catalyst emerges, you can bet some bench worker considered this compound for its clean chemistry and unique reactivity. The value isn’t just historical—it keeps showing up in patent applications for drug discovery and specialty chemical synthesis.
Rethinking Toxicity and Health Risks
Silver’s history with human health always brings conversation; add the fluoroborate group, and questions about toxicity take on new urgency. Chronic exposure to silver salts can cause argyria, a discoloration of skin and eyes, and though the risk with this compound is low when handled properly, it’s not zero. Fluoroborate ions don’t rank among the worst environmental offenders, yet concerns grow when large-scale disposal or accidental releases enter waterways. Studies in recent decades map out thresholds for dermal or inhalation limits, but as labs push toward greener practices, chemists pay close attention to documentation and exposure records, especially in teaching environments.
Where Silver Fluoroborate Might Lead Us
Everyday experience suggests silver fluoroborate will hold a role in next-generation chemistry, even as researchers chase less toxic or more sustainable alternatives. Trends point toward smarter process design, including closed-system syntheses and novel uses in recyclable catalyst platforms. Demand may ebb and flow with the rising cost of silver, but the creative energy poured into new applications promises continued relevance. Development of recyclable or biodegradable alternative anions might trim back some applications, but for now, deep knowledge and practical skill in handling this compound shape crucial advances in pharmaceuticals, precision synthesis, and advanced materials. Looking forward, sustained funding for safety and environmental research will shape how brightly silver fluoroborate glows in the laboratory future.
What is Silver Fluoroborate used for?
The Curious Case of Silver Fluoroborate
Silver fluoroborate hardly gets a headline. That’s just the way it goes with compounds tucked away in labs or industry. But I’ll say this: Few folks realize how a specialty chemical like this one shapes real progress in fields that touch our lives. Silver fluoroborate isn’t a household name, though it takes on roles that matter in electronics, imaging, even green tech.
In Electronics, Results Depend on Chemistry
Anyone who has tried to fix an old stereo or peered into a printed circuit board knows the magic comes down to good connections. Silver fluoroborate doesn’t just make an appearance; it enables precision. Plating with silver needs reliability and consistency, especially for switches and relays meant to last. A compound like this doesn’t just gift conductivity — it gives protection against corrosion and boosts performance at surprisingly small scales.
Electronics demand more than shiny finishes. Customers want reliability, and silver fluoroborate helps deliver that. Manufacturers go for chemicals that guarantee repeat results, give steady yields, and don’t cause headaches with unpredictable side effects. The demand for advanced tech puts pressure on every tiny process, and silver fluoroborate answers the call for strong metal layering, even where only the thickness of a few atoms matters.
Photography and Imaging: Not Just Digital
Digital cameras dominate, but classic film and specialized imaging stick around. Old-school darkrooms still rely on silver-based chemistry, and silver fluoroborate fits into that lineup. What stands out is the accuracy and sensitivity it offers during development. In fields like medical imaging and scientific photography, there isn’t much room for error, so the chemical’s role in these processes can’t be shrugged off.
Having tinkered with film development myself, I’ve seen how little changes in chemistry go a long way. Professionals gravitate toward solutions that cut down on waste and keep clarity high. Silver fluoroborate supports developers and fixers that handle high loads and recover more silver from spent materials. That’s not just skillful chemistry — it’s responsible resource management.
Green Technology and Recovering Precious Metals
With the world burning through resources faster each year, questions about recycling precious metals have never been more pressing. Urban mining — recovering materials like silver and gold from discarded electronics — depends on quick, safe, and cost-effective chemistry. Silver fluoroborate serves as a tool here, helping to strip and recover silver with less environmental damage than some old-school alternatives.
There’s a quiet value in improving these recovery methods. Toxic waste has plagued traditional metal recovery, but now safer, more controllable agents like silver fluoroborate make the job less risky. From my own days helping friends at scrap yards, I’ve seen how a switch to greener chemistry can change the whole atmosphere — literally and figuratively.
Tough Rules, Trust, and Consistency
Companies must trust their chemicals. Unreliable supplies or unsafe handling lead to wasted money or worse, workplace harm. Silver fluoroborate comes with handling guidelines for a good reason. Health and safety standards mean everybody from chemists to machine operators stays protected. Responsible suppliers combine technical support with strict testing, helping users keep regulatory agencies satisfied and their own people healthy on the job.
Room for Smarter Solutions
There’s opportunity here. More research can push this chemical into cleaner, even more effective uses. Collaboration between scientists, industry, and environmental agencies will strengthen responsible consumption. People who work with advanced materials know: progress doesn’t come from shortcuts, it grows from small gains in reliability, safety, and performance.
What is the chemical formula of Silver Fluoroborate?
Understanding Silver Fluoroborate in the Real World
Silver fluoroborate carries the chemical formula AgBF4. People working in chemistry labs, battery research, or even the field of jewelry cleaning probably already know that one small formula can signal a compound with a whole set of properties and uses. With Ag representing silver and BF4 as the tetrafluoroborate ion, this compound shows up in places where reactivity and stability have to play well together.
The Value of Knowing the Formula
Getting the formula right isn’t just for those in white coats. In education, students rely on formulas to connect classroom theory with hands-on experience. Messing up something as basic as the chemical identity leads to costly errors. In industry, I’ve watched colleagues pause at procurement meetings to double-check chemical names and formulas, and for good reason. Silver salts like this one don’t come cheap, and purchasing the wrong compound can set research back by weeks.
How AgBF4 is Used—And Why That Matters
This chemical features in organic synthesis, specifically as a source for silver ions. People use it to remove halide ions from reaction mixtures, and its compatibility with non-aqueous solvents gives it a special niche. Batteries and electronics draw on its properties; for years, companies have tested silver fluoroborate in areas where electrical conductivity and stability must go hand in hand.
I remember a project where we tried to streamline a silver recovery process. Someone mixed up silver nitrate with silver fluoroborate. Instead of saving money, we chalked up a bottle of lost silver. Mistakes come from misunderstanding formulas. It’s not glamorous, but double-checking saved both budgets and reputations.
Spotting Hazards and Handling Chemistry with Care
Silver fluoroborate stands out for more than just its silver shine. Its properties mean it needs informed handling and disposal. Based on its chemistry, exposure to moisture can generate toxic fumes, and both silver and fluorine bring environmental concerns. Improper disposal contributes to pollution, and I’ve seen labs fined when paperwork slipped and waste controls failed. Treating these issues is never as simple as following one checklist. You need an up-to-date materials safety data sheet, training, and a team culture that respects accuracy as much as efficiency.
Fact-Checking and Moving Forward
Every student and professional working with silver fluoroborate should keep trusted references nearby. Over the years, I’ve relied on Sigma-Aldrich data pages and independent safety literature before I order or use a new chemical. Checking against these sources reduces confusion and ensures compliance. For students, trainers, or industry veterans, brushing up on core knowledge, such as formulas, puts everyone in a better spot.
Simple Solutions Start with the Basics
Clear labeling and thorough training don’t just help avoid mix-ups. They build the foundation for any successful lab or production floor. The formula AgBF4 is more than a piece of trivia. It unlocks doors to better practices and safer outcomes. No one gets it right all the time, but every good result starts with knowing exactly what sits on the shelf. Reliable knowledge leads to better, safer choices for people and the planet.
How should Silver Fluoroborate be stored?
Why Silver Fluoroborate Draws Caution
Silver fluoroborate isn’t something you find outside a research lab or specialized factory. The stuff packs a real punch, both for its usefulness and for the risks it introduces. If you’ve worked in labs long enough, you pick up pretty quickly how much small mistakes with the wrong acids or salts end up costing folks—damaged glassware, ruined gloves, or even chemical burns. Silver fluoroborate stands among those materials that doesn’t forgive carelessness.
Getting comfortable around chemicals leads to trouble. I’ve seen it before: someone stores a reactive salt in a regular polypropylene jar, shoves it on a shelf under a drip of condensation, and a month later, the label smudges or the contents clump. That kind of oversight turns dangerous fast with a substance like this.
Moisture Turns Things Ugly
Silver fluoroborate picks up water in the air and can react, sometimes even releasing corrosive vapors or forming acids like hydrofluoric acid, which does real harm. One of my old mentors told stories about handling dusty reagents, only to realize too late that they’d left lids loose and let in moisture. Within a couple of days, you have clumps and mystery stains, sometimes corrosion on your shelving. So, air-tightness isn’t just a technical recommendation—it keeps your workspace, your lungs, and your skin safer.
Glass and Plastic: Why the Container Matters
In practice, we go for glass bottles dotted with PTFE-lined caps. Those caps don’t degrade or let anything through. Silver fluoroborate doesn’t melt plastic instantly, but with time, the wrong kind of cap or seal lets in air or chemicals, tarnishing your compound or changing its properties so you can’t use it reliably for research. I remember one incident where a lab switched to rubber stoppers—bad cost-cutting move. The container stuck to the shelf, and they had to treat the spill as a hazmat event.
Labeling Makes a Difference
A good label saves trouble. I mark the bottle with the date received, chemical name, and a clear hazard symbol. This isn’t just bureaucracy—it’s about walking into a shared workspace and knowing right away what lurks on each shelf. Mislabeling has caused more emergencies than almost any other single error. I’ve seen acids alongside neutral salts with identical generic white labels. One mix-up sent a colleague to the eye wash station—lucky they had goggles on.
Storage Conditions: The Simple Rules That Matter
Cool, dry, and dark—it sounds boring, but these three things keep silver fluoroborate stable. I keep it away from sinks, condensation, and direct sunlight. I check cabinets for leaks and wipe away any signs of broken packaging or chemical residue. Labs that don’t enforce these rules invite accidents. In shared spaces, anybody’s mistake can become your problem.
For best practice, store the material in dedicated corrosives cabinets. I always double-check for nearby acids or bases; mixing up incompatible chemicals can lead to dangerous reactions, especially if bottles leak or break in a cramped space. You don’t want a splash of spilled solvent meeting silver fluoroborate by surprise.
Disposal and Emergency Prep
It’s not all about long-term storage. Prepare for cleanup as well: keep spill kits, absorbents, and protective gloves handy. And train everyone working around this stuff—not just the senior researchers. One small slip from someone new can put the whole team at risk.
Safe storage isn’t just a checklist for inspectors—it’s daily work for anyone who uses challenging chemistry. After years in the lab, I can say: the routines feel repetitive, until the day they save you from a problem you never saw coming.
Is Silver Fluoroborate hazardous or toxic?
No Chemistry Experiment in the Garage: Silver Fluoroborate’s Risks
Silver fluoroborate draws curiosity for plenty of people interested in chemistry. This salt, used in organic synthesis, found a home in labs long before most folks had ever heard about it. Still, it rarely shows up in household chats unless someone is asking about chemical dangers or industrial mishaps. The main question crops up fast: is it hazardous, or even toxic?
Lab Tales and Straight Talk on Safety
Folks familiar with chemistry labs have seen safety data sheets gathering dust until suddenly, a pale salt like silver fluoroborate appears on the bench. Gloves come out, and fume hoods hum for a reason. Silver fluoroborate has a reputation as a reactive and corrosive compound, not something benign. Breathing in its dust or fumes is no joke — reports tie its exposure to irritation of the eyes, skin, and lungs. Touching it without protection can trigger chemical burns. Swallow it, and you’re signing yourself up for a trip to the clinic, maybe worse.
Toxicity doesn't only hit human health. Many chemicals like this collect in water or soil, which means aquatic life, plants, and local wildlife could feel the impact if waste isn’t kept in line. Too often, stories about industrial sites that ignored proper waste disposal pop up in the news. Substances like silver fluoroborate make headlines for contamination, not because they’re common, but because their effects can last for years.
Everyday Exposure? Not Likely. Still, Caution Rules
Silver fluoroborate isn’t something you’d find under the kitchen sink or in a classroom without supervision. In the world outside chemistry labs and factories, you won’t run into it. Still, the risks don’t go away just because most people never see it in person. Lab workers and those in industries using this chemical carry responsibilities that ripple outward. Mistakes can happen — a cracked vial, spilled powder, improper disposal. Pointing out these risks can sound alarmist to outsiders, but personal experience in lab settings proves accidents move fast.
On the regulatory side, agencies highlight handling guidelines: splash goggles, chemical-resistant gloves, lab coats, and solid air ventilation. No shortcuts. In a busy setting, strict attention to protocols saves fingers, lungs, and long-term health more often than flashy safety videos suggest. Even so, older researchers swap stories about the days before strict enforcement, and their memories of damaged lungs and persistent skin issues remind us that taking things lightly leads to trouble.
Addressing the Challenge: Real-World Solutions
Handling chemicals like silver fluoroborate safely calls for more than just signs on the wall. Training must stick. Supervisors who’ve spent decades in labs know that familiar routines breed complacency. Regular safety drills, incident debriefs, and clear instructions cut real risk. Companies and universities can invest in better fume hoods and disposal systems, not because regulators demand it, but because the old way costs too much in medical bills and lost talent.
Waste disposal ranks high. Treating silver fluoroborate as just another salt lands local water and soil in trouble. Working with certified chemical waste companies makes a difference, and new technologies — think on-site neutralization stations — can reduce how much hazardous material leaves a site. Researchers and workers also benefit from ongoing education. Seminars or short workshops about both health effects and environmental risks stick better than dense manuals.
When all is said and done, silver fluoroborate proves that small, pale powders can require serious respect. Anyone entrusted with its use or disposal shapes the safety picture for themselves and everyone else down the line. Stories from labs, facts from health agencies, and lessons from environmental mishaps all agree: ignore safety, and you pay the price. Practice caution, and you keep the risks in check, for yourself and for those nearby.
What is the purity/grade of Silver Fluoroborate available?
Why Purity in Silver Fluoroborate Matters
Silver fluoroborate isn’t something the average person keeps under the kitchen sink. For folks in electroplating, electronics, and certain chemical syntheses, though, this compound steps up with a unique set of properties. The catch: purity makes or breaks its performance. Even a small trace of contaminants can skew outcomes, leading to faulty finishes, unreliable products, or safety issues. That’s not speculation; it’s what happens in practice.
Commercial Grades You'll Actually Find
Walk into a lab supplier or call a major chemical distributor, and you’re going to hear numbers: 98%, 99%, even up to 99.9%. Most customers need at least 99% purity, with the real sticklers reaching for 99.9%—what’s commonly called “high-purity” or “analytical grade.”
Some sources sell what’s called “technical grade” at 98% and above, meant for applications where a bit of iron or sodium won’t ruin the batch. That said, high-end electronics or precision plating shops almost always demand grades above 99%. A reputable example comes from Sigma-Aldrich and Alfa Aesar, both reporting assays of 99% or better. Dig into their certificates of analysis, and you’ll see the impurities broken down—iron, copper, even moisture content—because serious customers ask tough questions.
Real-World Impact
Lab work doesn’t leave room for ugly surprises. In plating, impurities translate to uneven surfaces and conductance issues. I’ve watched a colleague sweat over a faulty run only to realize that subpar silver fluoroborate was the silent culprit. The frustration lingers, especially when downstream processing gets disrupted. Money and time go out the window.
From a health and safety angle, pure chemicals keep reactions predictable. Impurities can turn a simple procedure dangerous, spiking risks unexpectedly. Worker safety organizations, like OSHA, list purity as a big deal for a reason—clean chemicals keep things under control, in both the lab and the factory.
Getting the Right Stuff: Verification and Traceability
Not all sellers play by the same rules. Documentation is essential. Responsible buyers demand certificates of analysis (CoA), batch numbers, and trusted supply chains. These details make all the difference if something fumbles—recalling a batch or tracing an issue gets a lot easier. The electronics industry has set a strong example here, with International Electrotechnical Commission (IEC) standards calling for documented purity checks and impurity limits.
Traceability also protects against fraud. Counterfeits and diluted stock have hit even established markets, especially as online trade grows. Multiple published reports in journals like Analytical Chemistry have documented these risks, and regulatory agencies have advised stricter sourcing practices.
Raising the Bar: What Actually Works
Tech companies and research labs have responded by partnering directly with reliable producers or using in-house purity testing. ICP-MS, atomic absorption, and titration give definite results, letting staff spot problems before production lines start rolling.
As demand grows, some see value in industry certifications for chemical purity, something akin to “organic” certifications for food. The European Union has already launched stricter compliance requirements for chemicals in the past decade, causing shifts even in U.S.-based supplier policies.
Moving Towards Consistency
Long term, purity issues don’t shrink unless buyers push hard for supply chain transparency, vendors add real-time reporting, and regulators enforce existing purity standards. Silver fluoroborate deserves the same attention as other building-block chemicals. Customers, chemists, and lab managers can shape a safer, more reliable system—simply by refusing to settle for less than full documentation and proven purity.
| Names | |
| Preferred IUPAC name | silver tetrafluoroborate |
| Other names |
Fluoroboric acid silver salt Silver tetrafluoroborate Silver(I) tetrafluoroborate Silver fluoroborate |
| Pronunciation | /ˈsɪl.vər ˈflʊə.rəˌbɔː.reɪt/ |
| Identifiers | |
| CAS Number | [14104-20-2] |
| Beilstein Reference | 3858657 |
| ChEBI | CHEBI:91251 |
| ChEMBL | CHEMBL3301082 |
| ChemSpider | 161438 |
| DrugBank | DB15971 |
| ECHA InfoCard | 100.031.052 |
| EC Number | 216-143-6 |
| Gmelin Reference | 47199 |
| KEGG | C18648 |
| MeSH | D015237 |
| PubChem CID | 25089454 |
| RTECS number | XT2975000 |
| UNII | B6P35FBR4A |
| UN number | UN3264 |
| CompTox Dashboard (EPA) | DTXSID6038745 |
| Properties | |
| Chemical formula | AgBF4 |
| Molar mass | 196.77 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.89 g/cm3 |
| Solubility in water | Soluble |
| log P | -3.1 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 1.5 |
| Basicity (pKb) | 10.3 |
| Magnetic susceptibility (χ) | -45.9×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.405 |
| Dipole moment | Zero Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 150.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –957 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D11AE05 |
| Hazards | |
| Main hazards | Hazardous if swallowed, inhaled, or in contact with skin; may cause burns and irritation; toxic to aquatic life. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H302, H314 |
| Precautionary statements | P210, P260, P280, P304+P340, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 1-0-2 |
| Lethal dose or concentration | Lethal dose or concentration: LD50 (oral, rat): >2000 mg/kg |
| NIOSH | Not established |
| PEL (Permissible) | Not established |
| REL (Recommended) | 2.5 mg/m³ |
| Related compounds | |
| Related compounds |
Silver tetrafluoroborate Silver nitrate Silver perchlorate Silver hexafluorophosphate Silver trifluoromethanesulfonate |

