Silver Dichromate: A Closer Look At Its Place In Science And Industry
Historical Development
Scientists didn’t always have sleek digital sensors or cleanroom-friendly reagents. They built chemistry from the ground up, driven by big questions and practical problems. Silver dichromate entered the lab scene in the late 19th century, carved out of curiosity and a hunger for vivid reactions. Chemists first churned out this burnt-orange compound by reacting silver nitrate with a source of dichromate ions, most often potassium dichromate. Through the decades, researchers fine-tuned the isolation process, tweaking temperature and concentration to coax out cleaner, purer crystals. It stood beside other vibrant chromates that once filled teaching labs and early photochemistry experiments, back when color changes told the whole story. Silver dichromate's legacy traces back to those days, when the color orange signaled reactivity, risk, and promising lines of inquiry.
Product Overview
Silver dichromate, with the molecular formula Ag2Cr2O7, catches the eye long before anyone dives into the chemical books. Its deep orange hue signals chromate’s power—strong oxidation, high reactivity, and unmistakable presence, even in small amounts. Used in analytical chemistry, organic synthesis, and occasional industrial processes, people count on its ability to oxidize alcohols and other organics, but always with a wary eye on toxicity. The compound ships as a crystalline powder, fabricators pushing for purity above 98% to avoid surprises in the lab. Packaging typically lands in glass or high-density plastic, reflecting a deep respect for the material’s stability needs and hazard profile.
Physical & Chemical Properties
Silver dichromate crystals stack orange with a metallic sheen, their density hitting roughly 5.8 g/cm3. Don’t expect them to melt easily, either. Breakdown happens before a smooth transition to liquid; the compound decomposes above 250°C, liberating noxious chromium oxides and silver particles. Solubility lies near zero in cold water and only small in hot—this matters when handling waste, diluting, or isolating reaction by-products. The real star quality comes from its oxidizing strength. Silver dichromate packs both transition metal reactivity and the signature punch of the dichromate ion. In the right hands, it transforms organic molecules completely, cleaving alcohols down to aldehydes and carboxylic acids, hinting at its role in classic organic protocols and complicated synthetic recipes.
Technical Specifications & Labeling
Industrial suppliers issue silver dichromate under strict regulatory rules. Labels flag it as both toxic and environmentally hazardous. The typical batch rolls out with identification like CAS 7784-41-0, EINECS 232-043-3, and the unmistakable orange color. Shipments ride under UN 3288 for toxic solids and CMR (carcinogenic, mutagenic, reprotoxic) warnings direct from REACH or EPA guidance. Certificates must back up purity claims, outlining a breakdown of trace metals and cation contamination. Sometimes, trace chloride or nitrate triggers a red flag about synthesis shortcuts or improper washing.
Preparation Method
To make silver dichromate, labs generally start with soluble silver nitrate and potassium dichromate solutions. Everything hinges on careful addition—dump too quickly and side products muddy the mix. Stirring brings the ions together, and orange precipitate begins to form. Isolation calls for filtration, then thorough rinsing with cold water to strip away leftover potassium and nitrate. Drying at low heat protects the product from premature breakdown. Mistakes often lurk in the washing and drying steps, where contaminated water or excess heat can degrade purity and cause partial reduction to silver chromate or metallic silver. This stepwise, controlled production holds big implications for safety, lab efficiency, and final yield.
Chemical Reactions & Modifications
Chemists reach for silver dichromate when standard chromates lack punch or selectivity. In organic synthesis, it stands as an efficient oxidizer, transforming alcohols to carboxylic acids and aldehydes. It shows a neat trick of favoring harsher oxidations, a property that makes it valuable for specific, challenging transformations. Heat or strong acid can prompt it to shed oxygen, reducing Cr(VI) down to greenish Cr(III) and freeing silver ions along the way. In the presence of reducing agents, it can join redox cascades, making it useful for stepwise transformations or even as a colorimetric indicator. People in research labs sometimes tinker with modifications to tune the oxidizing power, pairing it with various ligands to shape selectivity, even though risks always tail the rewards.
Synonyms & Product Names
Apart from the main name, silver dichromate shows up under synonyms like disilver dichromate, disilver chromium oxide, and just plain Ag2Cr2O7. Chemists sometimes shorten it to SDC or run across it as part of specialty reagent blends, particularly in catalogues selling to advanced academic or industrial customers. Trade names don’t often stick; in this field people trust the formula much more than a brand. For inventory, proper labeling rests on IUPAC standards, not creative marketing.
Safety & Operational Standards
Handling silver dichromate wakes up memories of chemistry safety drills. Chromium(VI) compounds earn their toxic reputation, and this material fits squarely in that group. Direct contact burns skin, while inhalation or ingestion poses even greater risks—damage reaches kidneys, liver, and acts as a carcinogen over prolonged exposure. Any use calls for gloves, goggles, working under a fume hood, and thorough disposal protocols. Mixing or heating without exhaust quickly clouds a room with toxic dust. Emergency eyewash stations, proper training, and chemical-resistant containers push back against the dangers, though nothing ever erases the risks fully. Yearly updates from OSHA and international toxicology reviews keep staff on alert and ensure ongoing vigilance.
Application Area
People working in organic synthesis lean on silver dichromate most often. It offers selectivity that alternatives sometimes miss, cutting through complicated multi-step projects. In analytical chemistry, its unique color change under reduction helps in redox titrations and detection work. Some electrochemical researchers try it as a component in specialty electrodes, leveraging its mixed conductivity and robust catalytic activity. A few niche pigment producers eyed its vivid color, but regulatory headwinds turned most of them away. Environmental agencies track its use tightly—few enterprises outside scientific R&D get access, and even then, paperwork and containment protocols reign.
Research & Development
Every year, research journals publish new methods for controlling the reactivity of chromium compounds, silver dichromate included. Researchers test it as a model system for oxidation experiments, probe its detailed crystal structure with X-ray diffraction, and occasionally expand its reach in materials science. Ongoing work tries to curb its toxicity by anchoring dichromate in polymer matrices or using nanostructuring to keep it from leaching into waste. Some research groups try substituting in less toxic chromates or tuning the redox profile by adding ligands, searching for pathways that match (or beat) silver dichromate without bringing the same risks. Funding rarely flows to big expansion, though—the hazards keep it niche. Still, those pushing the boundaries sometimes find new uses for the robust oxidizing power in drug synthesis or environmental remediation.
Toxicity Research
Toxicologists spare no effort tracking the impact of chromium(VI) compounds. Silver dichromate, like its cousins, triggers cell damage and increases cancer risk, confirmed through animal studies and workplace exposure data. Modern guidelines draw a hard line: chronic exposure links to respiratory diseases, skin ulcers, and acute organ toxicity. Researchers test filtration and chemical reduction, aiming to limit the damage downstream, since legacy waste still pops up near abandoned labs or old pigment factories. Reviews call for alternatives whenever possible. Some recent studies focus on biological markers and rapid detection, hoping for early warnings and real-time exposure tracking. Despite improvements in containment, nobody underestimates the need for vigilance at every stage—handling, storage, use, and disposal all stand as flashpoints for risk.
Future Prospects
Regulatory focus tracks toward limiting all chromium(VI) use, especially in open processes and student labs. For fields hooked on silver dichromate’s unique reactivity, the search for replacements continues. Catalyst designers and green chemists race to build similar oxidizing systems out of less toxic metals or organics. Some see a future in specialized reactors using microencapsulation, where operator exposure nearly vanishes. Other groups try adapting old reactions to less hazardous salts, even if yields drop. Demand drops as labs modernize, but the need for robust oxidizers never fades completely—especially where nothing else does the job as precisely. As safety standards climb and synthetic routes diversify, silver dichromate shifts further into the world of trained hands, tightly controlled spaces, and careful risk/reward calculations. Watching developments in this space sheds light on science’s ability to find balance between powerful chemistry and the practical, pressing need to protect people and the planet.
What is Silver Dichromate used for?
Understanding What Silver Dichromate Brings to the Table
If you’ve ever found yourself deep into an old chemistry book or poking around a laboratory supply catalog, you’ve likely run across the term Silver Dichromate. It’s not something most folks hear about in everyday life, mainly because it doesn’t claim a seat at ordinary dinner-table discussions. Silver Dichromate’s bright red color might catch the eye on a research shelf, but its main role comes down to chemistry experiments, industrial processes, and some wider scientific corners.
Why Chemists Value Silver Dichromate
I remember working in a college lab and asking why anyone would use something like Silver Dichromate instead of more common silver salts. The answer was clear: chemists use this compound to oxidize organic substances. This means it grabs onto electrons from other molecules, pretty much forcing a chemical change. For anyone working with pharmaceuticals or organic synthesis, a strong, reliable oxidizer is worth its weight in gold—or, in this case, silver dichromate.
Let’s talk about reliability and results. Silver Dichromate’s ability to react predictably with alcohols, turning them into aldehydes and ketones, shapes lab experiments and guides production lines. Certain research projects focus on discovering or improving medical drugs. Making one small change to a molecule can create a big difference in how a medicine acts in the body, so having predictable tools matters.
Where Science Meets Industry
It’s one thing to admire its use in research, but another to realize Silver Dichromate supports actual industry work. Electronics companies, for example, sometimes use chromium-based compounds to treat metal surfaces or alter conductivity. They don’t toss Silver Dichromate around by the bucket, but when a very particular effect is needed—changing a metal’s surface so it doesn’t rust or prepping delicate components—Silver Dichromate plays its role.
There’s a catch, though, that experience in the lab teaches pretty quickly: Silver Dichromate isn’t something you handle lightly. The chromium in it exists in what chemists call the “+6 oxidation state”. That’s just a fancy way of saying the stuff is even more reactive, and—let’s be real—more toxic than most everyday chemicals. Even as it helps build new molecules, the waste it leaves demands respect and proper disposal methods, because those same properties that help with reactions can cause long-term health and environmental harm.
Considering Safety and Responsibility
Safety stays at the front of the conversation. It doesn’t only affect the person holding the test tube but grows into something bigger: environmental health, community responsibility, and long-term ecological impact. No lab or factory should treat these responsibilities lightly. Good training, strict handling rules, thorough disposal—these protect workers and the world outside their doors.
Thinking About Alternatives and Solutions
While Silver Dichromate deserves its reputation as a useful tool, chemists and engineers spend years searching for safer or less toxic alternatives. Some set their sights on new catalysts or procedures that don’t rely on chromium. They also explore recycling programs and “green chemistry” initiatives that lower both cost and danger. These projects ask all of us—scientists, companies, regulators—to keep questioning practices and push for safer tools and smarter solutions so that progress never comes at the cost of safety.
Is Silver Dichromate hazardous or toxic?
What Makes Silver Dichromate a Concern?
Everyday chemistry can be deceiving. Some compounds carry less risk, but others, like silver dichromate, ask for extra care. Silver dichromate lands on the list of substances people should approach with caution. The eyes and skin react quickly and harshly to exposure, and breathing in its dust causes trouble for the lungs. The conversation doesn't stop with discomfort. The reputation of hexavalent chromium, a main player in silver dichromate, causes scientists and health authorities around the globe to pay close attention.
Getting Real with Health Risks
Silver dichromate, with its bright reddish-orange color, doesn't just stain surfaces; it leaves a mark on human health. People working in labs or industrial settings recognize the risks just from looking at the safety gear stashed nearby. The form of chromium inside the crystal offers almost no mercy. Hexavalent chromium has a long record as a carcinogen, and silver brings its own set of challenges for people who handle it regularly. Skin contact often leads to itching or burning, but some people develop wounds that take weeks to heal. A bit of dust on a cut or in the eyes makes for severe pain, sometimes even long-term vision problems.
Breathing In, Toxins Out
I spent a summer in a materials lab during college, learning how quickly an innocent mistake could lead to a coughing fit or, worse, an emergency shower. Inhaling even a tiny bit of silver dichromate dust can cause headaches and dizziness, and inhaling larger amounts leads to lasting lung damage. Hexavalent chromium, combined with silver, puts extra pressure on the body's detox system. Some people find out they're allergic to compounds like these after just one exposure.
Chromium's Dark Side
Not all chemicals deliver the same punch. Hexavalent chromium stands out. The American Cancer Society and OSHA both point to this compound as a cause of lung cancer, kidney trouble, and reproductive damage. Some countries have slapped strict controls on hexavalent chromium. The European Chemicals Agency even lists it as a substance of very high concern, meaning jobsites and laboratories need special licenses and controls just to keep it around. Indirect exposure counts too — air, water, and soil around industrial plants sometimes test positive for chromium, causing local health alerts.
Silver Brings Its Own Risks
Anyone who has handled enough silver salts knows that skin stains can last weeks, and in extreme cases, deposits of silver can collect in organs or skin. Medical literature describes argyria and other conditions linked to too much silver, and mixing this with toxic chromium makes for a dangerous cocktail. Waste management systems have to treat every bit of residue, down to wash water, as hazardous. Most labs treat spills with special absorbents and remove every trace using dedicated gear.
Moving Toward Solutions
Getting careless with silver dichromate doesn't just put employees in danger — it puts entire communities at risk. Progress depends on tight controls, modern ventilation, and replacing this chemical with safer alternatives when possible. Real improvements show up in student labs, where safety training starts on day one, and in industries that swapped out toxic chemicals for greener methods. Regulators suggest strict tracking, constant employee education, and quick response plans for spills. Facing the risks head-on lets science workers keep advancing without risking lives.
What is the chemical formula of Silver Dichromate?
Digging Into the Formula
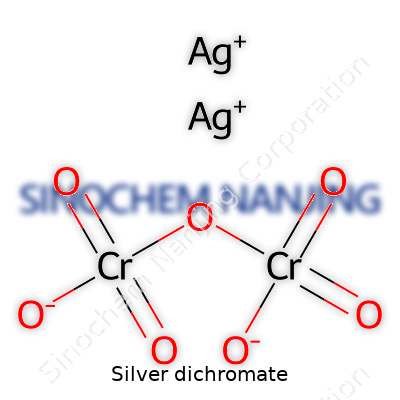
Silver dichromate lands among those compounds in chemistry that spark both interest and a healthy respect, especially in labs and classrooms. Its chemical formula is written as Ag2Cr2O7. Think of two silver atoms joining up with a dichromate ion, which itself carries two chromium atoms and seven oxygen atoms. That dichromate part gives the compound its signature deep orange-red color and a certain caution flag for anyone who's handled it before.
Safety Shaped by Science
Some chemicals only cross your desk in textbooks, but silver dichromate deserves more than a footnote. Its structure isn’t just some trivia—it shapes the way it behaves and how folks are supposed to treat it. That dichromate ion? It contains chromium in the +6 oxidation state, which stacks the risks higher. Hexavalent chromium compounds have picked up attention for their toxicity and impact on health and the environment. The U.S. Occupational Safety and Health Administration (OSHA) and World Health Organization both warn about hexavalent chromium exposure, linking it to respiratory problems and increased cancer risk. I’ve worked with these compounds in academic labs, always behind a fume hood and suited up with gloves, because a little carelessness means real trouble.
Why Knowledge of Formulas Matters
Some might say it is just a matter of memorizing symbols, but the formula Ag2Cr2O7 carries weight. Take water treatment, for example. Chromium compounds show up in industrial wastewater. Misreading or mixing up formulas could mean the difference between safe disposal and a regulatory headache. Educators hammer this home: getting these formulas straight forms the backbone of solid, safe lab practices. It echoes in my own chemistry classes; nobody gets a pass on this sort of detail.
Lab Use, Disposal, and Respect
Silver dichromate usually enters the picture as an oxidizing agent. In research, it helps turn up reactions that need strong oxidizers. In practice, handling and disposal demand attention to detail. EPA guidelines set strict limits on chromium levels in waste due to long-term environmental risks. Experiences in chemical stockrooms spell it out—the storage area always separates chromium compounds from organics, and disposal never heads down the regular drain. Instead, it requires special neutralization and containment to prevent contamination.
Moving Toward Safer Solutions
Scientists and industry professionals recognize the risks connected to chromium(VI) chemistry. Many labs push for less hazardous oxidizing agents or seek ways to trap and reduce hexavalent chromium before disposal. Some waste handlers use conversion processes: turning toxic Cr(VI) into the safer Cr(III) form before releasing water back into the environment. Colleges now teach both the chemistry and the responsibility, encouraging budding scientists to look past the test tube and consider the world outside the lab.
Education as Prevention
Formulas like Ag2Cr2O7 do more than fill exam papers. They reinforce habits that safeguard health and keep public water systems clean. By learning the right chemistry, folks everywhere can steer clear of common pitfalls, keep labs safer, and help drive smarter environmental decisions.
How should Silver Dichromate be stored?
Why Proper Storage Matters
Silver dichromate doesn’t pop up in the average household. Its deep orange color signals danger for good reason. With chromium(VI) in the mix, this compound carries real risk for health and the environment. Chromium(VI) compounds rank among some of the stronger oxidizers and show toxic effects on humans, including skin irritation and respiratory problems if inhaled.
Years back in the lab, I learned quickly how careless storage practices can ruin more than just an experiment. A coworker once used a damaged cap on an oxidizer bottle. Humidity crept in, the powder clumped, and a strong odor filled the air—luckily no harm done that time. Handling materials like silver dichromate safely means you’re looking out for everyone, not only yourself.
Key Storage Principles
Silver dichromate asks for respect and attention. The best practice starts with a dedicated, well-ventilated chemical storage cabinet. Steer clear of wood shelving because a spill or leak could start a fire. Stick to cabinets lined with inert, corrosion-proof material.
Moisture causes headaches with many dichromate salts. A tight-sealing container stands between safety and a crime scene-level mess. Glass and high-density polyethylene serve well because they don’t react. Avoid metal lids—silver dichromate can corrode many household metals. Label containers with chemical name, date received, and appropriate hazard symbols in tough-to-miss lettering.
Avoiding Unwanted Reactions
Oxidizers and combustible materials mix about as nicely as gasoline and matches. Never keep silver dichromate next to solvents like alcohol, paper, cardboard, or any material that could act as a fuel. Spilled dichromate on a wooden shelf or paper towel turns small mistakes into big emergencies.
Keep it separate from acids, too. Mixing dichromate salts with acids releases toxic chromium(VI) fumes and heat. A divided storage area, especially one that physically separates acids, oxidizers, and bases, cuts the risk of accidents.
Protecting People and the Planet
Good ventilation helps control vapors and fine dust that can escape during handling. Even with the best containers, chemical dust turns up if you handle powders often. Use a fume hood for weighing or transferring the compound, never out on the open bench. Gloves made of nitrile or neoprene protect skin better than disposable latex.
When disposal rolls around, treat silver dichromate as hazardous waste. Don’t wash it down the drain or toss it in general trash. Work with your institution’s environmental safety office to arrange safe removal. Chromium(VI) in the groundwater creates problems that linger for generations.
Don’t Guess, Stay Informed
Consult the Safety Data Sheet (SDS) every time, even if you think you remember the rules. Guidelines evolve, so regular checks with trusted sources make a difference. The US Occupational Safety and Health Administration (OSHA) and National Institute for Occupational Safety and Health (NIOSH) both offer up-to-date handling practices for toxic and oxidizing chemicals.
If you don’t keep materials like silver dichromate, make sure you know who does, and review storage practices once a year. Mistakes, even small ones, change lives when powerful chemicals enter the picture. Responsible storage forms the first defense against health risks, environmental impact, and accidents nobody wants to face.
What safety precautions should be taken when handling Silver Dichromate?
Recognizing the Hazards Early
Silver dichromate isn’t the average chemical tucked away on lab shelves. Its bright orange color might look interesting, but this compound lands squarely in the category labeled “toxic and dangerous.” One glance at the safety data, and you see the red flags: both silver and chromium compounds bring trouble for skin, lungs, and the environment. Cr(VI) compounds carry well-documented cancer risks, and silver salts can mess with tissues, especially after repeated exposure.
Engineering Controls and Safe Work Practice
A fume hood becomes more than a helpful accessory—it’s non-negotiable. Roads paved with experience tell me that trying to cut corners with adequate ventilation makes more problems than it solves. Even tiny amounts stirred up during weighing or transfer can linger and cause harm. The fume hood pulls those stubborn particles away from breathing space. Forget about open benches or slapdash setups; if the fume hood isn’t cleared for use, it’s time to wait it out.
PPE isn’t just a checklist item. I set myself up with layered nitrile gloves, safety goggles, and a lab coat. Regular cotton or latex gloves invite trouble, and a simple splash could change a day for the worse. Experienced chemists keep their eyes open for accidental spills every step of the way. Closed-toed shoes and long pants are no-brainers. My hands get washed after handling or even being near the bottle.
Strict Storage, Thoughtful Disposal
Silver dichromate keeps best in a cool, locked chemical cabinet, away from fuel sources and incompatible materials like organics and strong reducers. Labels stand out bold and clear—this isn’t a time for faded handwriting. Moisture, heat, or sunlight don’t mix with this chemical. Once the stuff comes out, it doesn’t go back in the way common solvents might. Waste needs prompt labeling and ends up in an isolated hazardous waste container, set aside for chemical waste pick-up. In no situation does it go down the drain.
Decontamination and Spill Response
Accidents do not wait for slow decision-makers. Spill kits must sit nearby, with absorbent pads, neutralizing powders, and a solid plan for containment. Sweeping up dry, toxic powder means risking inhalation, so dampening with controlled agents under the hood helps avoid airborne particles. I make sure only people trained for hazmat cleanup step in—guesswork gets sidelined when silver dichromate is involved. If skin touches the compound, the area gets flushed with water immediately, then soaped and rinsed again. Clothes splashed with it never return to the daily laundry—straight to chemical isolation.
Training and Culture
No lab worker should walk in blind. Training on handling this compound goes beyond printed handouts; it’s about hands-on demonstrations and questions answered before mistakes happen. Supervisors who model correct behavior encourage everyone to keep safety habits sharp. The trust built among team members—willing to speak up if procedures slip—guards against overconfidence. This unspoken agreement protects not just one person, but the entire group.
Looking Beyond the Lab
Some see chemical rules as paperwork or red tape, but these written protocols come from people who learned lessons at real cost. Fact is, exposure to substances like silver dichromate leaves a mark that lingers for years, both on health and the environment. Adopting rigorous safety safeguards signals a respect for those who’ve gone before and for those who share the same workspace. Keeping chemical hazards in check doesn’t only protect careers—it saves lives.
| Names | |
| Preferred IUPAC name | disilver dichromate |
| Other names |
Disilver dichromate Silver(VI) dichromate |
| Pronunciation | /ˈsɪl.vər daɪˈkrəʊ.meɪt/ |
| Identifiers | |
| CAS Number | 14307-33-6 |
| Beilstein Reference | 3944682 |
| ChEBI | CHEBI:86361 |
| ChEMBL | CHEMBL3312949 |
| ChemSpider | 22223 |
| DrugBank | DB15885 |
| ECHA InfoCard | 100.954.572 |
| EC Number | 232-043-7 |
| Gmelin Reference | 78478 |
| KEGG | C16613 |
| MeSH | D015547 |
| PubChem CID | 24556 |
| RTECS number | LC5425000 |
| UNII | U2AEJ2D00D |
| UN number | UN1479 |
| CompTox Dashboard (EPA) | DTXSID7089607 |
| Properties | |
| Chemical formula | Ag2Cr2O7 |
| Molar mass | 411.87 g/mol |
| Appearance | Orange-red crystalline powder |
| Odor | Odorless |
| Density | 5.68 g/cm3 |
| Solubility in water | Insoluble |
| log P | 0.02 |
| Vapor pressure | Negligible |
| Basicity (pKb) | No data |
| Magnetic susceptibility (χ) | +79.0e-6 cm³/mol |
| Refractive index (nD) | 1.92 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 238.1 Jmol⁻¹K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -578.4 kJ/mol |
| Pharmacology | |
| ATC code | V09AX05 |
| Hazards | |
| Main hazards | Strong oxidizer, toxic, harmful if swallowed or inhaled, may cause cancer, causes severe skin burns and eye damage, environmentally hazardous. |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS07,GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H301, H314, H317, H334, H341, H350, H360, H372, H410 |
| Precautionary statements | P273, P280, P301+P312, P302+P352, P305+P351+P338, P308+P313, P333+P313, P337+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 2-0-3-OX |
| Explosive limits | Not explosive |
| NIOSH | RA3850000 |
| PEL (Permissible) | PEL: 0.01 mg/m3 (as Cr(VI)) |
| REL (Recommended) | 0.004 mg/m³ |
| IDLH (Immediate danger) | IDLH: 15 mg Cr(VI)/m³ |
| Related compounds | |
| Related compounds |
Potassium dichromate Sodium dichromate Ammonium dichromate Silver chromate Silver nitrate |

