Silver Chlorate: Past, Present, and What’s Next
Tracing Back the Story
Silver chlorate stands out as one of those compounds that science textbooks often mention in passing, but the actual story goes much deeper. Chemists first encountered it as far back as the nineteenth century, often during experiments focused on silver’s broad reactivity and the search for new oxidizing agents. In those early years, silver nitrate dominated attention, but silver chlorate quickly attracted interest, especially in the wake of electrification and advances in analytical chemistry. While it never became as famous as its nitrate or chloride cousins, it carved out a unique space on lab benches, showing itself to be much more than a curious blend of silver and chloric acid. From my own work with legacy textbooks, it’s clear generations of chemists have circled back to silver chlorate every time a need for a strong oxidizer or a sensitive analytical reagent popped up.
What Silver Chlorate Brings to the Table
Looking at a jar marked ‘AgClO3’, you get a fine, white, crystalline powder. Unlike many other silver salts, silver chlorate dissolves easily in water, and you don’t see much fuss over precipitation or insolubility, which makes it more manageable in solution-based reactions. Industrial or pure research labs keep it handy because it drops out silver ions and the chlorate group at once, both ready to drive different chemical actions. Technically, it’s a salt, but its behavior differs from more routine choices like sodium chlorate. Because silver’s ions carry weight—figuratively and literally—it can do things lighter metals just can’t match.
The Hard Science: Properties and Identity
Silver chlorate melts around 230°C before breaking down, setting it apart from both the stubbornly stable halides and the delicate, explosive nitrates. Water soaks it up easily, and it doesn’t mess around with low solubility thresholds. In my own bench time, I've seen it act swiftly—whether spurring oxidations or splitting apart with the right kind of heat. Its blend of high density and immediate reactivity makes it easier to handle and trickier to ignore. If you ever see it given a title like ‘silver(I) chlorate’, ‘chloric acid silver(I) salt’, or even just the chemical shorthand AgClO3, you’re in the right ballpark. These synonyms pop up across older textbooks and current chemical catalogs.
Making Silver Chlorate: Tried and Tested Methods
For decades, the go-to method to make silver chlorate has involved a metathesis reaction—silver nitrate solution meets sodium chlorate solution. That’s textbook chemistry, right down to the glassware stains. Mix them, filter off the byproduct sodium nitrate, then concentrate and crystallize your silver chlorate. Any high school with halfway decent lab supplies could pull this off, though strict supervision remains a must given the toxicity and oxidizing nature. I remember old university labs where grad students would run these syntheses just to stock up on oxidizers for organic tests, always under a vent and with PPE, because the risk of unwanted reactions never vanished. There’s rarely a shortcut: purity demands careful washing, and experienced chemists watch for stray contaminants that could destabilize the final batch.
What Silver Chlorate Does in Reactions
Silver chlorate loves to donate oxygen. That knack makes it a bulletproof oxidizer, especially in organic labs. Mix it with combustible compounds and it’ll push reactions forward with speed, sometimes with more heat and vigor than expected. I’ve seen it used to kick-start chlorination, fuel pyrotechnic formulas, and help with titrations where precise endpoints matter. Add alkali chlorates, and you get the silver version right away due to silver’s tendency to precipitate out unwanted ions. Heat it, and decomposition kicks in—releasing oxygen and sometimes all but exploding if conditions aren’t tightly controlled. Researchers keep an eye on these wild-card dynamics, making sure any chemical modifications respect the material’s inherent volatility.
Keeping People and Places Safe
Safety always shapes how we handle any reactive salt, and silver chlorate has earned extra caution. Even tiny amounts pose real risks, not just from the irritant angle but from explosion hazards, especially as it can act as both oxidizer and fuel in the wrong setting. Most chemical safety standards approach it in the same breath as other strong oxidizers: keep it away from paper, oil, and the random bits of experimental detritus that inevitably accumulate during long bench sessions. Standard procedure: seal it tight, store it in the dark, and never handle it with sweaty or greasy gloves. Toxicity studies show silver ions persist in biological systems, giving rise to concerns around bioaccumulation—something I’ve seen drive R&D teams to look into safer disposal techniques and more robust labeling requirements.
Application: Silver Chlorate’s Role Today
Despite not being a household name, silver chlorate still finds dedicated use. Analytical chemists value it for precision redox titrations, while pyrotechnics experts sometimes reach for it to enable burn rates and brilliant flashes that less reactive salts can’t match. Now and then it turns up in small-scale syntheses of other silver compounds, exploiting its solubility and reaction speed. Over the years, I’ve seen its name appear on procurement lists whenever a specific oxidizing task needed that extra push—not as an everyday reagent, but as a specialist’s tool. Environmental controls and regulatory scrutiny have tightened, but demand for reliable oxidants with unusual profiles keeps silver chlorate relevant in a way few others can claim.
Keeping up with Research and Future Directions
Current research mainly focuses on two tracks: understanding the health effects of silver compounds broadly, and seeking greener ways to use or neutralize potent oxidizers like silver chlorate. Toxicologists track how silver ions shift through ecosystems and bioaccumulate, sometimes toggling between fascination and alarm at just how persistent silver can be. On the production and use side, R&D teams chase after new synthetic routes that cut down on waste and lower risks—often swapping legacy glassware for microreactor setups and digital monitoring to prevent runaway reactions. With all the buzz in sustainable chemistry, there’s even talk of repurposing legacy oxidizers for advanced energy systems, though these projects stay mostly in the realm of speculation so far. What stands out above all is the ongoing, relentless push to pin down every risk, so the benefits of this old-school compound can be tapped without opening up new environmental headaches.
What’s Next for Silver Chlorate?
Calls for safer chemistry and stricter regulations around heavy metals shape the path forward for silver chlorate. There’s every reason to believe research labs and specialty applications will keep it around, especially when nothing else delivers the same punch as a selective oxidizer. Those in the field often eye replacement compounds with skepticism—hard-earned experience shows that real-world behavior rarely matches brochures. Green chemistry researchers keep poking and prodding, trying to develop alternatives or at least neutralization methods that truly close the environmental loop. For now, silver chlorate keeps hanging on, a relic from chemistry’s formative years still pulling its weight beneath the surface of modern science.
What is Silver Chlorate used for?
Understanding Its Place in the Lab
Silver chlorate isn’t something that pops up during a supermarket run or a casual cleaning spree. This compound lands in the world of chemistry labs and specialized industrial processes. I remember the first time I saw it in a teaching lab—it looked like an unassuming white powder, but the professor made it clear that a little bit could go a long way in setting off some fast, energetic reactions.
Uses That Stand Out
Silver chlorate shows up mostly when folks need a strong oxidizing agent. Chemists reach for it when nothing else will push a reaction far enough or fast enough. Its power makes it valuable for certain synthesis reactions, especially those dealing with the challenge of adding oxygen to a molecule. This isn’t the stuff for simple rust removal or scrubbing silverware; it carries more serious punch.
Another well-known use involves explosives research. I’ll be honest, nothing about working with explosive compounds is routine or comfortable. Lab coats and goggles stop feeling like overkill when risking a violent reaction. Silver chlorate can act as a primary explosive or a component in matches, primers, and other pyrotechnic mixes. Safety isn’t just a concern—it defines the whole approach to handling it.
Why Safety Gets Top Billing
I’ve seen safety officers stop by the chemistry department just to double-check how people store oxidizers like silver chlorate. Combine it with almost any organic material, and you’re asking for unpredictable heat and possibly an explosion. The dangers aren’t just hypothetical; accidents have closed buildings. The US Occupational Safety and Health Administration (OSHA) and the Environmental Protection Agency (EPA) both set limits and rules to keep people out of harm's way.
Handling silver chlorate outside strict supervision doesn’t make sense. It can irritate skin, damage mucous membranes, or harm organs if someone inhales it or lets it linger on their skin. The best labs use solid ventilation, real containment, and experienced hands. Lab workers learn pretty quickly that respect for a chemical like this isn’t optional. Skip the rules and you risk lives, not just grades.
Good Science Relies on Responsible Use
We’re living in a time with information at everyone’s fingertips, but not all details found online give a real picture of what it takes to use substances like silver chlorate. Regulations and expert oversight keep the benefits flowing to industries without spilling over into the hands of the unprepared or reckless. What works in a chemistry classroom or a professional R&D lab won’t always translate to a hobbyist’s garage.
By relying on strong rules and transparent practices, scientists put safety first, even while exploring new materials and reactions. Tracking inventory, labeling everything clearly, and sticking to disposal protocols keeps accidents rare and knowledge growing. I’ve seen these steps save more than just paperwork headaches—they make sure nobody gets hurt chasing innovation.
Looking Ahead: Shaping the Future Minimizes Risk
Finding alternatives with fewer risks remains a goal for chemists. There's research into using different oxidants or designing processes that cut down on hazardous waste. Until better options emerge on a larger scale, silver chlorate stays in use in narrow, well-controlled circles. Its role remains important, but it won’t show up in friendly household products—and for good reason.
Is Silver Chlorate hazardous or explosive?
Understanding Silver Chlorate
Silver chlorate might sound like something you’d only encounter in a chemistry class, but it turns up in real-world labs and, sometimes, industrial settings. An important thing to know right away: this chemical doesn’t belong in homes, schools, or anywhere folks aren’t taking safety seriously. Silver chlorate packs an explosive punch, both in how it reacts and in how little provocation it needs to go off.
A Closer Look at the Risks
Silver chlorate never earned a reputation for being friendly. Years ago, learning about it meant hearing cautionary tales: “Don’t store it near organic material. Don’t bump it. Don’t even scratch your head too close to it.” That’s not just teacher paranoia. The compound is extremely sensitive—shock, friction, heat, or accidental mixing with combustible substances create perfect conditions for an explosion. It doesn’t wait for a fire to break out; a stray spark or rough movement could set off a violent reaction.
I remember a lab safety seminar where an instructor shared stories about those who ignored warnings about reactive chemicals. Lessons stuck because the dangers weren’t hypothetical. Data backs up what experience already taught: silver chlorate is a strong oxidizer with a history of setting off accidental blasts. Common reference works (such as the CDC NIOSH Pocket Guide and peer-reviewed chemical safety databases) all flag it for explosive risk, even in very small quantities. Explosions have been recorded during handling, purification, and even simple transfers from one bottle to another.
What Does This Mean for Lab Safety?
Working near silver chlorate means taking strict safety measures. Personal protective equipment isn’t just for show; goggles and gloves are the minimum. Good ventilation, spark-proof tools, and strict storage protocols count for everything. A clean lab—free of dust, paper, or wood shavings—can make all the difference. Spills need immediate attention, using methods designed for oxidizers. Disposal requires patience and respect for chemical rules, with regulatory guidelines giving clear steps for safe neutralization and removal.
Safe storage separates silver chlorate from anything organic or flammable. In my experience, strict labeling and tight inventory control kept close tabs on every gram. No gray areas—nobody took shortcuts, because the smallest mistake led to the greatest risks. Those who worked around these chemicals trained hard, reviewed MSDS sheets, and stuck with written procedures, even for the most routine tasks.
Fact-Based Caution, Not Fear
People sometimes ask whether silver chlorate belongs on the list of chemicals to avoid altogether. There’s good reason for caution, but it serves specialized purposes for analytical work and high-end technical processes. Handling it safely isn’t just about avoiding personal injury; improper use creates risks that can extend beyond lab doors.
Strict laws govern transportation, storage, and disposal of all chlorates, especially the silver salt. Regulators require incident reporting for good reason. Reporting helps spot trends and fine-tune safety advice. These steps, along with rigorous training and safety audits, reduce accident rates and help keep dangerous situations in check.
Looking For Solutions
Switching to less hazardous alternatives solves many headaches. Some labs seek substitutes, using chemicals with lower reactivity to avoid the risk of accidental explosions. Improved manufacturing controls and real-time monitoring keep storage safer and help prevent contamination. Teaching lab staff about chemical reactivity—not just during orientation, but with regular refreshers—builds a culture of vigilance.
Experts recommend ongoing research into safer handling techniques and reviewing historical incident reports. Knowledge-sharing between labs and industries can lead to smarter practices. Safety isn’t a one-time fix with silver chlorate; it’s a daily discipline that relies on facts, mutual respect, and an understanding that even tiny amounts command serious attention.
What is the chemical formula of Silver Chlorate?
Understanding the Basics
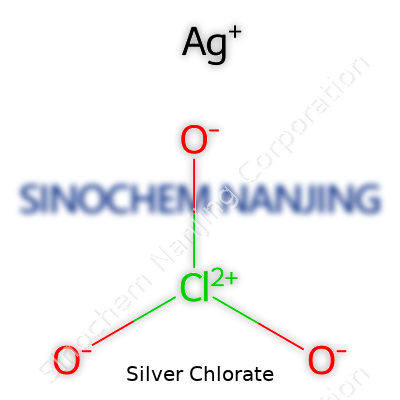
Silver chlorate might not show up in everyday conversation, but it plays a clear role for anyone who works with chemicals in labs or teaches high school chemistry. The chemical formula for silver chlorate is AgClO3. One silver atom teams up with one chlorate ion, which itself is made of one chlorine atom and three oxygen atoms. Mixing silver and chlorate gives us something with very distinct properties, and knowing its makeup helps people stay safe around it and use it properly.
Why People Care About Silver Chlorate
This compound does not just sit unused on a shelf. Scientists pay attention to silver chlorate because it breaks down easily, releasing oxygen gas in the process. That makes it a strong oxidizing agent. Some folks in the field of chemistry push the limits of their experiments using strong oxidizers like this one. Ignoring its properties can lead to dangerous surprises. I remember mixing strong oxidizers in a school lab years ago, and safety rules kept us out of trouble. Without a clear understanding of the formula, it would have been a risk we did not need to take.
Safety First: The Hidden Risks
Anyone who picks up a bottle of silver chlorate in a supply closet should see it as more than just a white powder. Its reactivity demands respect. A few accidental sparks or the wrong combination with other chemicals can cause violent reactions or even explosions. Several years ago, I read about a group of university students caught off-guard by the violent nature of this compound during an experiment. Incidents like that push home the lesson: knowing exactly what you're dealing with isn’t an academic exercise, it’s a safety issue.
Silver Chlorate in Education
Teachers bring out silver chlorate to show chemical decomposition or to discuss oxidation reactions. The chemical formula isn’t just a combination of symbols; it signals exactly how much of each element sits in the lab sample. That becomes important in calculations, making sure no one uses the wrong dose or creates unnecessary hazards. Experience in classrooms proves that most accidents come from underestimating the power within a seemingly simple formula.
Better Labeling and Access
Silver chlorate’s reputation for strong reactivity means it deserves clear labeling and responsible storage. All chemicals deserve respect, but the strong oxidizers sit at the top. Proper labeling must include the chemical formula and potential risks, not just a common name. By taking extra care with storage and signs, people working with these compounds cut down on the potential for surprise reactions. Many labs now use sturdy containers and color-coded labels to make these materials stand out. Good habits protect lives, not just test results.
Solutions for Safer Use
Changing the way we teach chemical formulas could help. Interactive lessons that link formulas like AgClO3 directly to their risks stick much better than rote memorization. Digital tools and videos, even simple experiments with close supervision, help new users see why specific combinations of elements create different dangers. Some universities have started updating their training videos and lab manuals to show how fast strong oxidizers react, keeping students alert and prepared.
Moving Toward Smart Chemistry
Keen attention to the details pays off. Silver chlorate highlights the divide between seeing a chemical formula as just another homework problem, and seeing it as a real-world safety sign. By building a culture of informed, careful use, labs protect people and equipment, and create a safer place to learn and discover. That starts with treating the chemical formula as more than scribbles in a textbook.
How should Silver Chlorate be stored safely?
Understanding the Risks Behind the White Crystals
Ask anyone who's worked in a chemistry lab. Silver chlorate isn't the stuff you leave out on a cluttered shelf and forget about till next semester. This compound has a reputation for turning minor oversights into major emergencies. Its toxic nature pairs with an uncomfortable habit of exploding on contact with the wrong materials. The world saw what chlorate-blended fireworks could do in the wrong hands — and that’s not a risk worth taking in a laboratory, warehouse, or classroom.
Prevention Starts with Dry, Cool Spaces
Silver chlorate reacts badly to humidity, heat, and light. Keeping it dry and cool isn't just common sense, it’s non-negotiable. I’ve seen eager scientists leave bottles near windows, only to come back to caked up crystals or worse. Don’t jam it next to the Bunsen burners or toss it among all-purpose reagents. Stash it in a dedicated cabinet, far from sunlight, away from drafty vents that can swing the temperature.
Choose the Right Containers
Glass jars with sturdy lids strap down the threat of contamination. Forget about using metal, as silver chlorate reacts with a surprising number of metals to create unstable substances. Some labs keep a small batch in amber glass bottles — blocking out light that can degrade the chemical and lower the risk of accidental ignition through static or sparks.
Tightly sealed caps keep away stray fingers and, more importantly, moisture from the air. If a screw cap gets stuck, never force it open in a hurry. Take it slow. Stress and glass aren't friends, and one sharp twist can send crystals into the air. Contain any mess and let a supervisor know. Safety showers and eyewash stations won't help much if the air fills with toxic dust.
Keep Distance from Fuels, Papers, and Acids
A big mistake is storing silver chlorate next to common supplies. It may just be sitting there in powder form, but exposure to organic material spells disaster. Paper towels, alcohol, acetone, or acid bottles nearby stretch the odds for an accident. It’s not paranoia to keep it in its own coded bin, apart from oxidizers, reducers, or combustibles. That simple step beats cleaning flecks of white crystal off everything after an “unplanned reaction.”
Ligatures, Labels, and Logs
Don’t lay blame on the label-maker, but too many labs use mismatched bottles with torn stickers. Always mark containers clearly, listing chemical name and date received. Hazards like “Oxidizer—explosive with organics” deserve to stand out. Use color-coded tape or labels others recognize at a distance. Digital logs back up physical records. Trace who opened what, when, and how much left the supply. This isn’t just bureaucracy — if something goes missing or spoils, you want answers fast.
Training: Fact, Not Formality
Booklets warn that untrained hands can trigger a mishap with a simple transfer or cleanup. I remember fresh students treating silver chlorate like sodium chloride, scooping loosely without a fume hood, inhaling dust, or brushing spilled crystals under a bench. Refresher courses keep these mistakes rare. Share a few disaster stories, show old news clippings. Remind every user—silver chlorate’s danger is real, and so is the responsibility that comes with storing it.
Where can I buy Silver Chlorate?
The Real Deal Behind Silver Chlorate
Silver chlorate isn’t something you see every day on a hardware store shelf. It’s not sitting next to baking soda or vinegar in a supermarket. This chemical carries a level of risk that goes far beyond cleaning up the kitchen or polishing metal. Having spent some years working in both lab supply distribution and as a high school science instructor, I learned that asking about substances like silver chlorate puts you squarely in the crosshairs of regulatory authorities. Silver chlorate’s strong oxidizing properties mean safety concerns climb high on the list for anyone looking to get their hands on it.
Access Heavily Regulated for Good Reason
If you’re part of a university lab or working in industrial manufacturing, you likely follow strict protocols before you ever touch reagents like this. I remember spending hours filling out forms and attending safety briefings just for far less reactive chemicals. Companies like Sigma-Aldrich, Fisher Scientific, or Alfa Aesar do offer silver chlorate, but only with the proper documentation—business licenses, end-use statements, and often a detailed background check.
Private individuals rarely succeed in placing an order for this compound without clearance. It’s labeled under the Department of Homeland Security’s Chemical Facility Anti-Terrorism Standards for its potential misuse. Stories run through the science community about random visits from authorities after an unusual chemical order triggers automated red flags. Trust me, no one wants to end up on that list by accident.
Risks Outweigh Curiosity
The temptation to find a shortcut and grab things off obscure sites always pops up online. Vendors lurk on shady marketplaces, offering chemicals with little oversight. Here’s an honest fact: going around legal channels brings severe risks. Health hazards aside, criminal charges stack up quickly if you possess regulated chemicals without the right paperwork. Accidental mixing or improper storage can lead to fire and toxic releases, outcomes I saw personally in university labs before stricter reforms came in around 2010.
Building Safety Through Education
Chemicals like silver chlorate underscore the need for public education around hazardous substances. Too often, young students or curious hobbyists stumble blindly into dangerous territory. Watching bright, talented teens blow up a beaker through simple ignorance proved heartbreaking every year I taught. Responsible science means more than following rules. It involves understanding why those rules exist. Teaching proper handling, sharing horror stories without censoring the consequences, and making chemical safety part of science culture helps prevent disaster.
Looking for Safer Alternatives
If your project truly needs an oxidizer, alternatives with fewer hazards exist. Potassium chlorate or sodium chlorate sometimes fill the role without quite the same explosive reputation. Even then, sourcing happens through approved vendors after proper vetting. The legal path may feel tedious, but it saves lives and reputations.
So, buying silver chlorate isn’t a simple matter of a click and some cash. It’s a gateway to bigger conversations about responsibility, risk, and respect for the power hiding in a bottle of white crystals. Anyone trying to purchase it should take time to ask whether their goal justifies the dangers ahead.
| Names | |
| Preferred IUPAC name | Silver(I) chlorate |
| Other names |
Chloric acid silver salt Silver(I) chlorate Argentous chlorate |
| Pronunciation | /ˈsɪl.vər ˈklɔː.reɪt/ |
| Identifiers | |
| CAS Number | [7783-92-8] |
| Beilstein Reference | 392029 |
| ChEBI | CHEBI:75955 |
| ChEMBL | CHEMBL1231871 |
| ChemSpider | 21567739 |
| DrugBank | DB15815 |
| ECHA InfoCard | 100.033.963 |
| EC Number | 232-043-7 |
| Gmelin Reference | 1277 |
| KEGG | C18637 |
| MeSH | D013035 |
| PubChem CID | 24657 |
| RTECS number | VW3500000 |
| UNII | 9V1R49109U |
| UN number | UN3363 |
| CompTox Dashboard (EPA) | `'DTXSID6050917'` |
| Properties | |
| Chemical formula | AgClO3 |
| Molar mass | 191.32 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 4.24 g/cm³ |
| Solubility in water | High |
| log P | -2.17 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 6.38 |
| Magnetic susceptibility (χ) | +3.1×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.924 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 151.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -6.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -155.6 kJ/mol |
| Pharmacology | |
| ATC code | V09AX05 |
| Hazards | |
| Main hazards | Oxidizes, fire and explosion risk, toxic if swallowed, skin and eye irritant. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS03,GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H302, H314, H410 |
| Precautionary statements | P210, P220, P221, P280, P370+P378, P371+P380+P375, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 0, Instability: 3, Special: OX |
| Autoignition temperature | 250 °C |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 24 mg/kg |
| NIOSH | SN38500 |
| PEL (Permissible) | PEL (Permissible exposure limit) for Silver Chlorate: 0.01 mg/m3 (as Ag) |
| REL (Recommended) | 0.05 mg/m³ |
| Related compounds | |
| Related compounds |
Potassium chlorate Sodium chlorate Silver nitrate Silver perchlorate Silver chloride |

