Silver Arsenite: A Comprehensive View
Historical Development
The history of silver arsenite weaves through the early days of chemical discovery, tracing back to 19th-century laboratories where researchers tested the properties of new compounds to understand both their potential and their risks. Over time, chemists began to note silver arsenite’s reactivity and striking appearance, leading to scattered reports in scientific journals. The path to commercial recognition proved slow. Toxicity and limited natural occurrence kept it in the shadows of more common silver and arsenic compounds. Still, academic curiosity and advancements in analytical chemistry brought it to the attention of a few specialists interested in exploring its diverse reactivity. Early syntheses took place under controlled conditions, with small yields that challenged even the most skilled practitioners. As a result, silver arsenite never entered mainstream industrial use, but it kept a presence in research institutions devoted to developing sensitive analytical reagents and conducting fundamental studies on the behavior of metal-arsenite complexes.
Product Overview
Silver arsenite appears as a yellow to reddish-yellow powder, often noted for its limited solubility and sensitivity to light. Its shelf presence has stayed mainly in laboratories and specialized educational settings. Suppliers usually package the compound in airtight, light-resistant containers to preserve its chemical integrity and mitigate accidental decomposition. The product targets the niche segment of chemical research, with specialized demands rather than mass-market applications. Whenever scientists seek a reagent to catalyze unusual reactions or to serve as a test object in toxicological modeling, silver arsenite sometimes enters the conversation. Its commercial journey, unlike that of sodium arsenite or silver nitrate, builds on word of mouth, careful review of chemical catalogues, and a keen understanding of precise technical needs.
Physical & Chemical Properties
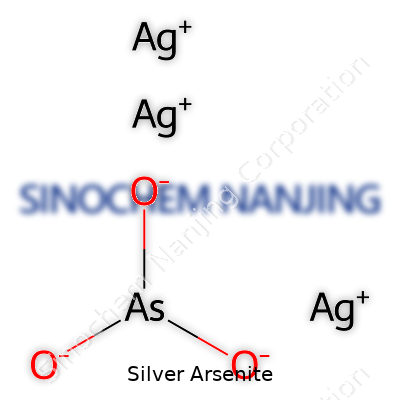
Silver arsenite, with the formula Ag3AsO3, carries a molar mass of about 501.6 grams per mole. The powder generally resists easy dissolution in water, showing only marginal solubility in strong acids. This chemical sits on the delicate line between two toxic elements: silver and arsenic. Its color—anywhere from pale yellow to orange, depending on purity—immediately signals a warning to those who recognize the hazards of arsenic-based compounds. Sensitivity to light can prompt slow degradation, releasing free silver or changing the color, which helps seasoned chemists judge quality by sight. In the lab, handling always takes place under good ventilation and proper containment, given the high risk of poisoning if inhaled or ingested.
Technical Specifications & Labeling
Every container of silver arsenite carries meticulously detailed labeling, including the percentage purity, batch lot number, recommended storage conditions, and precautionary pictograms for acute toxicity and environmental hazard. Manufacturers typically publish rigorous specifications for form (fine powder or crystalline), levels of trace contaminants (such as chloride or nitrate), and key performance indicators like reactivity in comparison tests. Technical data sheets also provide melting points, storage temperature guidelines, and disposal protocols in accordance with global chemical safety regulations. The labeling always includes supply chain traceability and technical contact details, so that professionals can directly engage with suppliers regarding any ambiguity or emergency.
Preparation Method
Lab-scale preparation of silver arsenite starts with a controlled reaction between a soluble arsenite, such as sodium arsenite, and a solution of silver nitrate. Chemists introduce silver nitrate slowly, stirring constantly to keep the system homogeneous, and monitor precipitation closely. The signature yellowish precipitate appears quickly upon mixing, and its purity depends on stoichiometric balance and rigorous exclusion of contaminants. To isolate the compound, filtration separates the solid from the mother liquor, and repeated washing with cold, distilled water helps minimize soluble impurities. The product then dries away from light and atmospheric moisture. Practitioners quickly learn to take small batch sizes seriously—large-scale synthesis without proper engineering controls elevates the risk profile due to the acute toxicity of arsenic species. Every repetition of this process underscores the importance of PPE and strict adherence to laboratory best practices.
Chemical Reactions & Modifications
Silver arsenite belongs to a reactive family. In the presence of strong acids, the arsenite ion breaks apart, sometimes leading to the formation of arsenous acid and silver salts. Reductive conditions risk converting the arsenite component into elemental arsenic while liberating metallic silver, which can stain glassware and complicate clean-up. Alkaline solutions may offer enough strength to partially dissolve the solid, creating new pathways for synthesis or measurement. Researchers occasionally use gentle heating to speed up a chemical transformation, but thermal decomposition releases hazardous arsenic vapors and silver dust, so precise thermal control remains crucial. Modifications of the compound usually stem from attempts to increase solubility, tune light stability, or anchor the arsenite moiety onto an inert matrix for analytical use. Scientists consistently push to eke out a few more insights or create a new derivative compound, each time balancing safety with scientific ambition.
Synonyms & Product Names
Across chemical registries, silver arsenite shows up under several names: argentous arsenite, silver(I) arsenite, and trisilver arsenite. Each designation gives a glimpse into its composition and oxidation state, yet the name silver arsenite persists in lab culture. Product catalogues sometimes list the compound as Ag3AsO3, in keeping with its molecular formula, or as silver meta-arsenite in older texts. Safety data sheets reference both the chemical name and these synonyms, ensuring clarity across jurisdictions and supply chains. In chemical literature, all these terms point to the same core compound, encouraging researchers and regulatory agencies to treat the substance with identical caution.
Safety & Operational Standards
Handling silver arsenite demands unwavering commitment to health and safety standards. Laboratory workers suit up with gloves, eye protection, and—if powder must be weighed or transferred—wear fit-tested respirators to prevent accidental inhalation. Material Safety Data Sheets lay out clear warnings about systemic toxicity, environmental hazards, and signs of arsenic exposure, including skin lesions and neurological symptoms. Every step in a lab protocol aligns with procedures found in arsenic risk management manuals: use of fume hoods, locked storage far from food or drink, and rigorous spill containment plans. Emergency response measures—activated carbon for containment or rapid evacuation if a significant quantity spills—join regular training drills. Disposal laws in major economies classify waste silver arsenite as hazardous, often mandating incineration with scrubbing technology or encapsulation in stable matrices before landfill placement. In my own experience, few substances command as much respect in a laboratory. Direct skin contact or accidental ingestion would trigger immediate medical intervention and environmental monitoring, underscoring the gravity of safe handling.
Application Area
Silver arsenite never became a widely used industrial chemical, likely due to its toxicity and lack of scalable advantages over other silver or arsenic compounds. Its role stays mostly technical, supporting research in analytical chemistry and environmental science, where trace analysis of arsenic or silver benefits from tailored reagents. In academic environments, the compound pops up in investigations of precipitation reactions, solubility equilibria, or electron microscopy studies interested in fine-grained crystalline structures. Certain projects in water quality assessment or heavy metal detection reach for silver arsenite as a reference standard, but these remain rare and well-regulated. I’ve found its main value arises in niche projects where traditional silver salts fail to deliver a desired precipitation pattern or where advanced researchers need to model arsenic’s behavior in unusual matrices.
Research & Development
Silver arsenite attracts continued attention from academic scientists interested in the overlap of transition metal chemistry and toxicology. Development work investigates its electronic structure, explores possibilities for labeling in advanced imaging, or studies interactions with organic ligands. Collaboration between materials scientists and toxicologists sometimes drives work toward safer handling protocols or breakthroughs in remediation techniques—should silver arsenite accidentally contaminate an ecosystem. The drive to contain arsenic’s threat to human health has also inspired attempts to embed silver arsenite particles inside stable polymers, aiming to immobilize toxicity while still offering a useful laboratory standard. My reading of recent literature shows persistent debate over improved stabilization and trace-level detection methods, with new ideas surfacing in metal recovery and advanced waste treatment.
Toxicity Research
Toxicologists list silver arsenite among compounds deserving the strictest control. Exposure routes include inhalation, ingestion, or accidental skin absorption. Studies in live models and cell cultures reveal disruption of cellular respiration and severe interferance with enzyme function—effects connected directly to the arsenic atom’s behavior as an enzyme poison. Acute poisoning can trigger headaches, abdominal pain, cardiovascular distress, and potentially fatal organ failure. The compound joins a broader class of arsenic minerals responsible for legacy environmental hazards in abandoned ore processing sites, spurring ongoing epidemiological research to track long-term risks. Environmental scientists frequently test soil and water samples near former industrial zones for evidence of silver and arsenic contamination, using sensitive analytical tools capable of detecting parts-per-billion levels. Recognizing these risks, researchers consistently warn about taking short-cuts in lab protocol and stress the need for routine biomonitoring of those who handle the compound. No safe threshold has emerged for chronic exposure, so all handling takes place under strict occupational exposure limits, generally enforced by national agencies like OSHA or their equivalents.
Future Prospects
Looking ahead, silver arsenite remains a chemical for specialized tasks. Its future most plausibly lies in analytical contexts, where its unique properties allow development of new reference standards for ultra-trace metal detection. I think the only real chance for broader application, possibly as a component in new remediation techniques or advanced sensing devices, comes with innovations that encapsulate its toxic components more effectively and reduce exposure risk down to zero. Researchers eye improved chemical stabilization for safe laboratory education, more robust analytical protocols for heavy metal monitoring, and greener disposal strategies under circular economy principles. With regulatory pressures tightening and greater attention on lab worker safety, demand won’t likely jump in general use. Still, within science’s need for highly specialized tools, silver arsenite’s future involves careful use, strong technical oversight, and persistent research into making high-risk compounds safer to manage and study.
What is Silver Arsenite used for?
Looking at Silver Arsenite Beyond a Simple Formula
Silver arsenite doesn’t make headlines often, yet it carries weight in a few specialist circles. In chemistry class, it stood out for its striking yellow hue and swift reaction with acids. There’s a mix of curiosity and caution that follows it—with good reason. Anyone who works with heavy metals or arsenic compounds knows the risks. It’s not the kind of ingredient people choose on a whim, yet its uses reflect the fine balance between discovery and responsibility.
Main Role: Analytical Chemistry
Walk into a quality control lab, and you’ll spot shelves lined with colorful compounds earmarked for testing. Silver arsenite shows up here, helping scientists and students identify and measure arsenite ions in samples. A droplet of silver nitrate mixed with an arsenite solution forms a yellow precipitate—silver arsenite. The procedure isn’t just textbook theory; companies and universities rely on reactions like this to monitor arsenic content in water, soil, or factory waste.
Monitoring arsenic matters. Contaminated groundwater in some rural areas of Southeast Asia taught the world a hard lesson: arsenic poisoning can ruin lives. The color change from this reaction means labs can detect even low concentrations, raising a flag before serious health threats multiply. That’s a small but real step in the bigger fight against contaminated water supplies.
Spotlight on Safety
Every few years, a new story breaks about chemical spills or improper handling of lab chemicals. Silver arsenite deserves respect. Both silver and arsenite ions sound familiar, but their combination makes a compound far from friendly. Arsenic exposure links to hundreds of thousands of deaths worldwide. The EPA reports that chronic low-level arsenic exposure causes skin lesions, cardiovascular problems, and cancers. Anyone handling silver arsenite suits up in gloves, goggles, and works with hoods humming overhead. Mishandling, even at the scale of milligrams, risks serious harm.
I remember once seeing a misplaced container in a university lab. A sharp-eyed technician called it out and walked us through the right way to store, track, and dispose of substances like silver arsenite. It stuck with me: Practices and protocols matter as much as the science itself.
Why the Trade-off?
Industries stick with silver arsenite in testing partly because alternatives don’t always match its precision. Spectrophotometric methods use machines, but in stretches of the world where labs still lack cutting-edge equipment, classic reagents won’t go away soon. Many labs still train students using the old tests. There’s an argument for moving toward safer techniques—ion-selective electrodes or advanced chromatographs. Yet costs and local realities shape choices.
Looking Forward: Reducing Risks
Stronger regulations and clear labeling cut down on everyday risks. Training that sticks, not just a bored slide show, keeps labs safer. Disposal needs more than pouring down the drain—formal programs for hazardous waste management bring peace of mind, not just compliance.
Innovation comes slowly, but it comes. Researchers now chase greener chemistry, developing fewer hazardous alternatives and digital sensors that take people away from messy benches. Advocacy—or just sharing stories about botched safety—nudges institutions toward better choices.
Silver arsenite, for now, keeps its narrow place in chemistry. Its legacy doesn’t just rest on its uses, but on how well we understand and respect its power.
Is Silver Arsenite toxic or hazardous?
Getting Real About Chemical Hazards
The name itself gives a good hint. Silver arsenite isn’t a chemical that belongs in a home or school. Someone who spends time in a lab or works in mining probably recognizes the “arsenite” part right away. Any compound with arsenic brings up red flags for toxicologists, clinicians, and anyone who knows the infamous history of arsenic in both industry and crime.
Known Hazards Behind the Chemistry
Silver arsenite forms through a reaction between silver nitrate and sodium arsenite. The resulting yellowish powder may seem harmless at first glance, but beneath the surface, things look uglier. Arsenic compounds are notorious for causing health problems after either short or long-term exposure. Breathing arsenic dust leads to everything from coughing to lung injury. Swallowing it takes things to a whole new level—think organ damage and a real risk of death. Skin can absorb arsenic, especially if protective gear goes ignored. Disorders like arsenicosis, linked to contaminated water in places like Bangladesh and India, show just how devastating chronic arsenic exposure can be.
Bringing silver into the picture doesn’t soften the blow. On its own, silver doesn’t pose much of a problem at low doses—after all, it’s found in bandages and medicines from ancient to modern times. Still, nobody needs silver building up in their tissues. Argyria, a strange blue-gray skin discoloration, won’t kill you but can be a social nuisance if you stare down years of it. Combining silver with arsenite shifts the risk profile back to the arsenic side of the story. No regulatory body puts these compounds in the “safe” category, and for good reason.
Why Silver Arsenite Stays on Watch Lists
OSHA, NIOSH, and the EPA take exposures like arsenic seriously. Permissible exposure limits drop to near zero. The EPA regulates arsenic in water down to parts per billion, not because it likes alarmist headlines, but because chronic exposure rolls out a list of health hazards—cancer, cardiovascular disease, and neurological issues. Those standards guide actions in labs handling chemicals like silver arsenite. Spill something in a research lab, and immediate cleanup with proper respirators, gloves, and even evacuation isn’t overkill. Regulations make sense only when the compound itself carries significant risks, and it’s hard to find many substances with more red flags than arsenic.
Protecting Health at the Source
For chemists and manufacturers, the real answer is simple: Avoid exposure if possible. The right PPE, spot ventilation, and chemical fume hoods save lives. Regular health checks for employees in environments where arsenic compounds show up shouldn’t feel like an optional extra. Waste disposal matters, too. Dumping silver arsenite into regular trash lines up as reckless, contaminating soil and groundwater with toxic elements—something no community wants in its backyard. Safe disposal through hazardous waste facilities should be non-negotiable.
Personal Takeaway: Respect Over Routine
I’ve worked alongside chemists who treat arsenic compounds with the kind of respect they’d give a rattlesnake. Gloves, masks, and a sharp eye for spills turn into second nature. A former colleague once shared a story about an old lab where arsenic dust tainted the bench surfaces from decades of carelessness. Years later, people cleaning up that space wore hazmat suits and took away entire countertops for hazardous waste disposal. That’s the legacy of ignoring the risks. Education, vigilance, and a healthy dose of caution matter with silver arsenite—and with anything carrying arsenic. Safety starts long before any exposure happens.
What is the chemical formula of Silver Arsenite?
Understanding Silver Arsenite: More Than a Name
Silver arsenite isn’t something you hear about in daily conversation, but in every chemistry classroom and lab, knowing what you’re dealing with can go a long way. The formula for silver arsenite rings out as Ag₃AsO₃. You get three silver atoms, one arsenic atom, and three oxygen atoms coming together in a single compound. That’s where things start to get interesting, because every one of those elements brings its own story.
Why the Formula Matters
Back when I first learned about these kinds of compounds, memorizing the chemical formulas felt like a chore. But seeing how these numbers and letters translate into real-world impacts is a whole different ballgame. Silver, for example, is more than just a shiny metal. It’s got antimicrobial properties, which explains its long use in medicine and water purification. Arsenite — the ion formed from arsenic and oxygen — has a notorious reputation. Arsenic in its trivalent state, as seen in arsenites, brings toxicity concerns people shouldn’t ignore, whether in chemistry labs or industry settings.
Health and Environmental Implications
Discussing silver arsenite without touching toxicity would be missing the big picture. Arsenic compounds represent some of the most toxic things found in earth’s crust. Chronic exposure even at low levels can lead to health problems, including skin disorders, cancer risks, and issues with internal organs. Regulatory agencies, such as the World Health Organization and the US EPA, flag arsenic compounds as serious threats when present in water or soil. Silver isn’t exactly harmless, but the real danger in silver arsenite lies mostly with the arsenite ion.
The Real-World Challenges with Arsenite Compounds
In the lab, you can make silver arsenite by reacting silver nitrate with sodium arsenite. The light yellow precipitate tells you the reaction went through. That’s cool on a technical level, but the bigger worry is waste handling. Disposing of arsenic-containing compounds like this brings heavy regulation. The days of dumping lab waste down the drain are long gone — environmental laws clamp down hard for good reason. There’s growing evidence showing environmental arsenic leaches into groundwater, ending up in the food chain and affecting millions globally.
Potential Solutions and Safer Practices
Some communities face groundwater arsenic contamination, often without the resources to fix it. The knowledge about compounds like silver arsenite drives innovation in safer handling and water purification. Adsorption materials, better filters, and improved monitoring show promise. In labs and industry, containment and neutralization get prioritized, with agencies doing regular audits and demanding transparency on waste.
Learning the formula for silver arsenite looks like a minor achievement on paper, but it sets the stage for bigger conversations. Chemists, environmental scientists, and public health workers collaborate to limit arsenic’s harm. Education plays a huge role — knowing what you’re handling, why it matters, and what can go wrong leads to smarter, safer choices. Every element and number in Ag₃AsO₃ forms a part of a larger story about responsibility in science.
How should Silver Arsenite be stored safely?
Understanding the Risk
Silver arsenite is bad news if someone handles it carelessly. Many people don’t realize how much damage this compound can do, both for human health and the environment. Even a small mistake during storage can turn into a big headache. It carries toxicity because of the arsenic content, so a lapse in judgment could lead to exposure that brings on headaches, dizziness or long-term illness. Knowing the risks right from the start pushes people to take storage seriously—not just for themselves, but for colleagues and anyone nearby.
Choosing the Right Location
Every time I’ve set up storage for hazardous materials, nothing beats a room with solid ventilation. Air movement in and out matters. Imagine a spill or slow release—those fumes don’t just disappear. They build up, and that’s where the trouble starts. A storage area sits best away from direct sunlight, which can cause heat buildup or subtle degradation of some chemicals, including silver arsenite. No one wants to risk accidental reactions or advisories about compromised inventory. High-quality air extractors aren't just a luxury; they should be the norm wherever people use or store chemicals like this one.
The Right Containers Matter
Glass or high-quality plastic airtight containers make a real difference. Metal containers run a risk of reacting with the compound itself, leading to contamination or even corrosion that weakens the container. Tightly sealed jars stop powders or dust from escaping into the air, where people can breathe it in. I’ve seen labs cut corners and use whatever’s at hand—one mistake and cleanup becomes a nightmare. Proper labeling using clear, chemical-resistant stickers with hazard symbols backs up safe storage. A wipe with a wet rag isn’t enough. Proper record-keeping saves time in an emergency, too—there’s no squinting at smeared pen on a jar when urgency strikes.
Protecting People and the Environment
Handling arsenic compounds means always thinking two steps ahead. Emergency gear—spill containment kits, gloves, and respirators—should be easy to grab, not locked away in another building. By keeping materials in secondary containment trays, spills are less likely to spread. If someone asks why, I remind them it’s easier to clean a plastic tray than to replace a floor. Only trained personnel belong near silver arsenite. One of my old supervisors kept strict sign-in sheets for every chemical access, and enforced training updates—no credential, no entry. It meant fewer accidents and more confidence among team members.
Legal and Ethical Responsibility
Regulations from agencies like OSHA don’t exist just to annoy managers. They guide facilities on labeling, reporting, and safe handling. I’ve watched regulators conduct surprise checks, and they look for signs that people care about these rules—not just following them, but understanding the point. Each incident of improper storage risks heavy fines, reputational harm, and most important, puts people in harm’s way. Simple actions like regular inspections and staff refresher courses build a culture where safety is part of daily work, not an afterthought tacked onto the end of a shift. People in charge carry the weight—not only for compliance, but for every person who depends on their commitment.
What precautions are needed when handling Silver Arsenite?
Understanding the Stakes
Silver arsenite carries a nasty sting if handled without proper care. As a compound that contains both silver and arsenic, two elements with reputations for risk, it demands respect in any lab or workplace. I remember walking into my first chemistry lab and seeing the big red hazard diamonds marked all over a container not too different from what stores this material. Safety protocols weren’t empty rules—they protected everyone breathing the air in that space.
Health Risks that Hit Hard
This compound produces toxic effects on touch, inhalation, or ingestion. It isn’t just a theoretical danger—long-term exposure to arsenic compounds can cause skin lesions, nervous system issues, and in some cases, cancer. Silver, while less notorious, can still build up in body tissues. I’ve seen lab mates scramble after realizing a simple dust cloud can turn personal risk into a reality. Sweat-soaked gloves and careful breathing become second nature after a scare like that.
Protective Gear: Non-Negotiable
Good gear makes all the difference. Proper lab coats, impervious gloves (think nitrile, not latex), and certified safety goggles belong on anyone handling this material. Resisting the urge to grab bare-handed or skip coverage saves skin later. Disposable gloves need to come off before touching anything else, especially phones or doorknobs. I’ve seen someone absentmindedly brush dust from their face and get a stern talking-to about how quickly accidents can spiral.
Safe Storage that Stops Trouble Before It Starts
Keep this chemical locked away, clearly labeled, in a cool and dry cabinet—real separation from food, drink, and untrained hands keeps mistakes rare. Containers need strong seals—no cracks, no leaks. Extra caution goes into making sure it doesn’t mix with acids since that can release poisonous arsine gas. Any spill, large or small, needs immediate action with finely tuned mop-up steps using dedicated tools, never the regular cleaning kit.
Air and Surface Precautions
Ventilation matters a lot. Breathing airborne particles ends badly, so working under a certified fume hood becomes a must. No shortcuts. I remember getting halfway through weighing a sample and seeing the telltale shimmer in the air—someone had left the hood half-open. One lesson learned fast, thanks to a mentor who took safety as seriously as results. Surfaces need regular checks and dedicated cleaning. Any tiny residue, especially the fine powdery dust, sticks to shoes, sleeves, even notebook pages.
Preparedness Beats Panic
Emergency procedures should stay fresh in memory. Eye washes and showers need clear access—never blocked by boxes or extra chairs. Spill kits tailored for toxic metals must always be ready, and everyone shares responsibility to remind each other about where the material lives and how to neutralize spills. Once, our team ran a drill where seconds counted; speed and clarity made all the difference.
Disposal Done Right
No dumping down the drain, and no tossing with regular trash. This material falls under hazardous waste regulations. Each container gets hand-delivered to licensed professionals for safe destruction or storage, following strict chain of custody rules. The risk doesn’t stop the moment a bottle empties—treat every last scrap like it could cause real harm.
Knowledge and Respect: The Everyday Safeguards
Handling silver arsenite safely doesn’t happen by accident. Grit, respect for hidden dangers, and a community focused on watching out for each other matter more than any checklist. I’ve witnessed mishaps nearly avoided by someone speaking up or just lending a spare pair of gloves. The habits built over days and years—the careful double-check, the simple question, "Have we missed anything?"—end up keeping disasters away and letting real science happen.
| Names | |
| Preferred IUPAC name | Silver(I) arsenite |
| Other names |
Disilver arsenite |
| Pronunciation | /ˈsɪl.vər ˈɑː.sɪ.naɪt/ |
| Identifiers | |
| CAS Number | 7784-41-0 |
| Beilstein Reference | 3561348 |
| ChEBI | CHEBI:86456 |
| ChEMBL | CHEMBL509054 |
| ChemSpider | 22293 |
| DrugBank | DB15765 |
| ECHA InfoCard | 100.013.980 |
| EC Number | 215-204-2 |
| Gmelin Reference | Gmelin Reference: "Gmelin 37 |
| KEGG | C16630 |
| MeSH | D014023 |
| PubChem CID | 166844 |
| RTECS number | CM9650000 |
| UNII | W3A096729B |
| UN number | UN1512 |
| CompTox Dashboard (EPA) | DJ8942T58Y |
| Properties | |
| Chemical formula | Ag₃AsO₃ |
| Molar mass | AgNO₂: 214.87 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 5.6 g/cm3 |
| Solubility in water | Insoluble |
| log P | -15.8 |
| Vapor pressure | Negligible |
| Basicity (pKb) | – |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 2.01 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 160.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -155.6 kJ/mol |
| Pharmacology | |
| ATC code | V03AX04 |
| Hazards | |
| Main hazards | Toxic by inhalation, ingestion, or skin contact; carcinogenic; environmental hazard. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P260, P262, P264, P270, P271, P273, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P313, P330, P501 |
| NFPA 704 (fire diamond) | 2-3-2-OX |
| Lethal dose or concentration | LD50 oral rat 45 mg/kg |
| LD50 (median dose) | LD50 (median dose): 20 mg/kg (oral, rat) |
| NIOSH | SN4210000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Silver Arsenite: "0.01 mg/m3 (as Ag) |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | IDLH: 5 mg/m³ |
| Related compounds | |
| Related compounds |
Silver arsenate Silver phosphite Silver phosphate Sodium arsenite |

