Silver Arsenate: Deep Dive into a Classic Inorganic Compound
Historical Development
Silver arsenate first showed up in chemistry labs in the late 1800s, back when scientists chased down new inorganic salts for mineral analysis and basic research. They started with simple precipitation reactions, jotting down colorful precipitates and puzzling over their structures. Chemists figured out that combining silver nitrate with sodium arsenate in water sparked a brick-red solid—a real giveaway for silver arsenate’s identity. Over time, researchers put the compound through the wringer. They checked its solubility, learned about its thermal stability, and explored what role it played in analytical chemistry. This curious salt wasn’t just another entry in a chemistry catalog; it helped push early chemical analysis and mineral separation, back before the digital age rewrote how labs do routine tests.
Product Overview
On the shelf, silver arsenate appears as a brick-red, crystalline powder. It’s not a household name, but it often pops up in laboratories that deal with heavy metals, mineral testing, or complex ion precipitation. Its chemical formula, Ag3AsO4, hints at three silver ions for every arsenate group, giving it a structure that stays fairly stubborn in regular water. No fancy multipurpose ingredient here—just a well-known compound with deep roots in test tubes.
Physical & Chemical Properties
You rarely see silver arsenate outside specialty labs, and its color immediately stands out. The powder carries a deep, reddish hue. Once you lay it out, it feels dense—almost gritty—and resists dissolving in cold water. If you try to heat it or drive it into solution with strong acids, some decomposition crops up. The compound melts down only at fairly high temps compared to its nitrate or chloride siblings. In regular conditions, it stays solid, pointing to low solubility—a property useful for its role in classic chemical tests. Silver ions tend to snatch up opportunities to latch onto anions like arsenate, forming a lattice that’s tough to break.
Technical Specifications & Labeling
To use silver arsenate safely and effectively, labs stick with strict labeling: “Silver Arsenate, Reagent Grade, Ag3AsO4, CAS 7784-46-5.” Purity hovers above 98%, with trace metals kept at bay using stringent controls. Labs focus on crystal size as well, making sure powdered forms average around 100-200 microns for consistent precipitation and mixing. Stockists list the molecular weight, melting point, and solubility limits in standard data sheets, along with storage instructions: dry, dark, and away from acids or reducing agents. Reliable traceability helps protect against batch variability, a headache for anyone running sensitive chemical assays.
Preparation Method
Making silver arsenate doesn’t call for complicated tricks. You shake up a solution of silver nitrate, then slowly add sodium, potassium, or ammonium arsenate, drop by drop, stirring all the while. The red precipitate appears on cue as silver ions and arsenate anions lock together. Filtration, washing, and drying give a fine powder ready for use. Folks working on industrial scales keep tight control over temperatures and mixing speeds, dialing in the reaction to avoid impurities. Every step needs steady hands and clean glassware; one stray contaminant throws off purity.
Chemical Reactions & Modifications
Silver arsenate’s real power lies in how it reacts. In basic or neutral environments, the brick-red solid barely budges—ideal for selective ion analysis. Bring in concentrated acids, and the salt starts breaking apart, releasing arsenic acid and silver ions. Drop in reducing agents, and silver metal can plate out, while arsenate converts into forms like arsenite. Researchers sometimes tweak the basic compound by adding dopants or using it as a bridge in silver-catalyzed or arsenic-driven reactions. Compared to silver chloride or silver phosphate, this compound offers a tighter grip with distinct color cues, perfect for confirming the presence of arsenates in unknown samples.
Synonyms & Product Names
On order forms or chemical catalogs, silver arsenate might turn up under different names. “Trisilver arsenate,” “silver(I) arsenate,” or “argentum arsenicum oxydatum” all mean the same thing. Some older texts may list it under its German moniker or by shorthand like Ag3AsO4, just to puzzle students or historians. Whatever the label, buyers check for the right CAS number or supplier codes to avoid confusion.
Safety & Operational Standards
Every experienced chemist approaches silver arsenate with a healthy dose of respect. Both silver and arsenic compounds require gloves, dust control, and smart ventilation. Inhalation, ingestion, or skin contact can lead to health issues, especially from the arsenic portion, which easily ranks high among toxic elements. U.S. and European safety agencies enforce strict exposure limits, so getting careless around the dust or solutions risks more than just ruined lab work. Disposal means collection as hazardous waste. Nobody flushes leftovers or sends runoff into regular trash; cleanup needs strong operational discipline. Standard practice calls for spill kits, labeled containers, and up-to-date safety data sheets on hand at every station.
Application Area
The market for silver arsenate never grew as large as for silver nitrate or silver chloride, but its specificity lands it small yet vital roles. Analytical chemists use it for qualitative spot tests for phosphate and arsenic. It also shows up in geological studies when tracing mineral samples containing silver or arsenate veins. Teachers may bring it out in teaching labs when guiding students through classic precipitation reactions, though modern safety rules sometimes push its use into more controlled demonstrations. Silver’s role in antimicrobial coatings led to brief explorations of silver arsenate in medical research, but toxicity usually closes that door fast.
Research & Development
The compound’s value in R&D matches its selectivity. Some researchers look into its ion exchange properties, hunting for new ways to separate metals using solid-state filters. A handful of teams explored silver arsenate as a component in composite materials or as a color standard. Lately, its distinctive precipitate has come in handy as a point of comparison when testing new analytical reagents or troubleshooting anomalies in legacy lab procedures. Researchers watch for cross-interference, learning how silver arsenate’s stubbornness can both help and hinder complex multi-element analysis.
Toxicity Research
Arsenic’s reputation for danger turns any study of silver arsenate into an exercise in risk assessment. Acute exposure brings the usual suite of arsenic symptoms: gastrointestinal distress, weakness, and possible organ damage. Chronic low-level uptake raises cancer risks and neurological deficits. Scientists working on silver arsenate’s toxicity rely on cell tests, animal models, and historical accident records. Based on my own reading, the risks mesh with broader arsenic hazards; silver may add additional irritation but rarely tips the scales by itself. Regulations and best practices torpedo any efforts to use it outside carefully supervised environments, and ongoing reviews tighten those standards as new data arrives.
Future Prospects
What comes next for silver arsenate? Its primary laboratory role probably won’t expand much. More modern, less toxic compounds keep pushing into the market. Still, its high specificity and clear color changes ensure a place in mineralogical or forensic labs. Some see opportunities in microanalysis or in calibrating instruments built for legacy procedures. The reality—its toxicity and environmental impact will always keep use limited, but specialists working in heritage chemical analysis or heavily regulated testing likely won’t give it up completely. The story of silver arsenate becomes part of a larger debate in chemistry: how to balance sharp analytical tools with safety and environmental stewardship, and when it’s time to set aside old friends for new ones.
What is Silver Arsenate used for?
Real Uses and Practical Value
Silver arsenate doesn’t draw a crowd like gold or platinum, but it has a steady role, especially for folks who work in labs. You find it in old-school and some modern chemical analysis. Take water-testing. Many labs trust silver arsenate during tests that check for phosphate ions. If you add a solution containing silver nitrate to something with phosphate, silver arsenate shows up as a distinctive yellow precipitate. That color tells the chemist, usually right away, that phosphate is there. This isn’t a trick for beginners. Testing water for phosphates guides how drinking water is treated in a city, how farms manage runoff, and how scientists track pollution in rivers and lakes.
Education Has a Place for It Too
Schools and universities rely on silver arsenate in hands-on chemistry classes. I still remember the first time I saw those yellow crystals form in a small glass tube. The reaction offers a straightforward way for students to see chemistry in action. Complex molecules can be intimidating for newcomers. Watching silver arsenate form helps students actually see the invisible world of atoms and ions. It’s not just theory anymore; it’s something you can hold. These real-world connections stick in a student’s mind.
The Darker Side: Toxicity and Handling Risks
I’ve worked with plenty of silver compounds, and arsenic always got a warning sign on the storage bottle. Silver arsenate is no different. Its risks come mostly from the arsenic it contains. Getting careless—spilling it on a bench, or worse, splashing it on your hands—can turn a science lesson into an emergency. Long-term risks involve more than a rash or a cough. Chronic exposure to arsenic links to nervous system problems, even cancer. I’ve seen labs enforce strict rules: gloves on, fume hoods running, waste labeled and double-bagged. If you ignore those precautions, you put yourself and others in harm’s way. It’s one of those chemicals that demands respect, not shortcuts.
Other Industrial Angles
Outside of labs and classrooms, there isn’t a long list of silver arsenate uses. Some niche industries use it in high-grade photographic processes. Photographers and engineers exploring specialty film have used silver arsenate for its unique chemical behavior. But this market faded in size with the rise of digital photography. I’ve met some folks in mining and metallurgy who run into silver arsenate as a byproduct when extracting silver from ore. It’s rarely the main attraction—just a piece of the larger process, often treated as hazardous waste and handled with care to avoid polluting groundwater.
Safer Alternatives and the Road Ahead
These days, more labs ask if there’s a safer way to test for phosphate. Advances in digital sensors and non-toxic reagents give options, but cost and reliability matter. Old tests like the silver arsenate method bring a level of simplicity and trust that automated machines sometimes can’t match, particularly in rural or underfunded labs. It’s wise for schools and industries to keep evaluating their choices, using safer chemicals where possible, keeping protocols sharp where toxic substances are still required. Even so, silver arsenate keeps its purpose in places that prize clear, reproducible results.
What is the chemical formula of Silver Arsenate?
Understanding Silver Arsenate
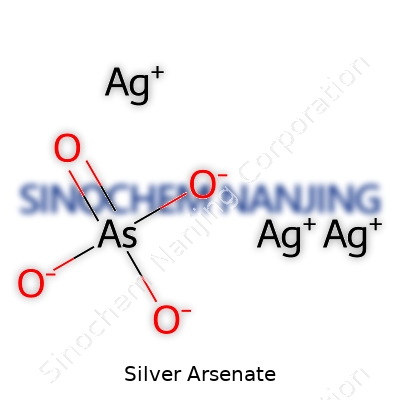
The chemical world overflows with hidden stories, and silver arsenate—with the formula Ag3AsO4—brings more to the table than just an arrangement of atoms. It stands as a compound formed between silver ions and the arsenate ion. For those who work around laboratories, this reddish-brown powder draws attention during classic qualitative analysis to detect the presence of arsenic.
The Details Behind Ag3AsO4
Breaking down the formula, three silver (Ag) atoms pair up with one arsenate (AsO4) group. Chemically, silver prefers a single positive charge, and arsenate carries a triple negative charge, causing them to link in this fixed ratio. This clear composition makes it possible for chemists to understand purity, reactivity, and possible interactions.
From my own time working in college chemistry labs, I remember the surprise when our group watched it form. One moment, you have a clear solution—add the right reactant, and out falls a dense, brick-red precipitate. This visual marker made it simple to spot arsenates, letting us specifically identify certain ions, even in a messy sample.
Why Knowing This Formula Matters
Facts build trust. In environmental science, understanding what makes up silver arsenate gives insight into its environmental impact. Both silver and arsenic have strong profiles—silver is well-known as an antimicrobial, and arsenic carries notoriety as a serious hazard. Combining the two creates a compound whose risks and benefits depend on exact conditions of use.
Take water testing as an example. Arsenic contamination ranks among the most serious public health issues worldwide. Some detection routines in the past made use of silver arsenate formation to uncover even trace amounts of arsenic in drinking water. The chemistry behind its production, and more importantly its formula, sets the stage for responsible handling. Labs can prepare the right solutions, calculate concentrations, and manage waste with safety in mind once the formula is clear.
Exploring chemical formulas sidesteps vague warnings and offers direct knowledge. I’ve seen firsthand how misunderstandings about formulas lead to accidental exposure or mishandling. Instead, properly naming Ag3AsO4 helps students, scientists, and safety officers to recognize the stakes in each bottle and plan accordingly. No need for guesswork or outdated charts.
Tackling Risks With Practical Solutions
Chemicals like silver arsenate don’t belong in untrained hands. Better signage, reliable chemical databases, and clear labeling all start with accurate formulas. There’s a growing movement to offer digital access to detailed material safety data sheets (MSDS) so researchers can check hazards on their phones before opening a cabinet. From high school labs to professional research sites, I’ve watched the lightbulb go on when people see the link between good practice and a simple, well-understood name.
Silver arsenate might seem like a dry textbook example to some, but its precise identification—Ag3AsO4—anchors real-world lab work and public health. Formula knowledge arms people with the details they need, whether the goal is discovering, diagnosing, or defending against environmental risks.
Is Silver Arsenate toxic or hazardous?
Staring Down the Risks
A childhood spent poking around school labs and curiosity cabinets taught me one thing: a chemical’s name rarely gives away how dangerous it might be. Take silver arsenate. The word silver adds a layer of intrigue, but arsenate sends a chill if you know basic chemistry. Arsenic compounds carry a long reputation for toxicity, stretching from folklore to cold hard medical cases of poisoning. Handling any arsenic-based material deserves real respect.
What Makes Silver Arsenate Concerning?
The dangerous side of silver arsenate comes from arsenic. Chemically, silver arsenate contains arsenic in the form of arsenate ions, which can cause severe health issues if inhaled, ingested, or if dust touches the skin too often. Just a quick look into toxicity studies reveals how even a small amount can trigger symptoms — headaches, confusion, gastrointestinal distress, and far worse if exposure continues. The World Health Organization has drawn a line between arsenic exposure and higher rates of skin, bladder, and lung cancers, with inorganic arsenic matching the profile found in silver arsenate.
In practical settings, contact with this compound can happen through accidental spills, poorly ventilated spaces, or in science classrooms where staff and students remain unaware of the risks. Proper handling rules and strong safety culture aren’t just window dressing around arsenic compounds. In my student days, one careless moment with an arsenic-laced mineral left a classmate hyperventilating until the emergency team arrived. They never let us forget that scare.
Why the Hazards Matter Beyond the Lab
This isn’t only a problem for chemists. Arsenic shows up in electronics, mining, pigment production, and even historic wood treatments or glassmaking. Forgetting about silver arsenate’s hazards invites trouble in storage rooms, waste bins, and industrial sites. Sometimes people downplay the problem because silver arsenate’s bright color or familiar metal content tricks folks into thinking it’s as safe as the coins in their pockets. But no one should relax their guard just because a material seems uncommon or because it’s tucked away in a sealed jar.
Tough Standards and Smarter Handling
We need real diligence every step of the way. Safety experts point out that gloves, lab coats, masks, and good air circulation drop the odds of toxic exposure. Labeling and secure storage matter, but so does a no-nonsense understanding among all workers. Supervisors juggle enough already, but missing a step with toxic dust or a spill can wreck someone’s health for years. Lessons from past accidents show that regular checks and clear communication reduce mistakes.
Disposal brings its own headaches. Hazardous waste rules from the EPA and similar agencies get stricter year by year for good reason. People can’t just dump silver arsenate down the drain or in regular trash. Specialized hazardous collection keeps these toxins from mixing into water or soil. There’s always debate about the cost and convenience of safe disposal, but a shortcut here can turn a forgotten container in a high school closet into a neighborhood emergency down the line.
Building a Safer Culture Around Chemicals
No one gets peace of mind by ignoring or underestimating substances like silver arsenate. Educating every new batch of students, lab techs, and workers remains crucial. Going beyond printed warnings means conversations, real-life stories, and practical drills. A curious nature and a love for chemistry can open the door to discovery, but a healthy respect for the risks keeps that door open for everyone, today and tomorrow.
How should Silver Arsenate be stored?
Getting Serious About Chemical Safety
Silver arsenate is not something people should mess around with or just toss onto a shelf. It raises two big concerns: the arsenic compounds found in it are toxic, and silver compounds aren’t exactly harmless either. Anyone who ever worked in a lab or handled chemicals knows that a single slip-up can turn into a big problem. I remember the nerves in the early days, holding containers labeled with skulls and crossbones, heart beating a bit faster. It pushes a person to double-check every move.
The weight of chemical storage hit me most during an undergrad lab cleanup. We moved bottles—often dusty—for disposal. Some bottles had cracked or leaking caps. One of those, with an arsenic compound, leaked onto the shelf. The realization came fast: everyone within reach could have been exposed, all because the storage was sloppy.
Straight Answers: Where and How?
Silver arsenate isn’t explosive or highly reactive like sodium or potassium, but it does give off arsenic dust if it dries up or spills. Toxicity comes mostly from airborne particles or accidental contact. Lab safety experts agree—use a tightly sealed glass or plastic container, keep it clearly labeled with warning phrases, and stash it in a dedicated poison cabinet or chemical storage area with proper ventilation.
Humidity raises the risk. Moisture can help leach arsenic compounds, increasing the odds of contamination. I’ve seen old basement storerooms where humidity corroded metal labels, blurred instructions, even cracked plastic lids. Chemical containers need a dry spot—no excuses. Away from heat sources, too, so that temperature swings don’t crack or warp the container.
No Room for Carelessness
Once a container cracks, it’s game over: particles escape. I always recommend secondary containment—a small bin or tray beneath the bottle, to catch any solid or liquid leaks. Wearing proper gloves and goggles isn’t just for handling spills; it matters every time you open the cabinet. Accidents rarely announce themselves.
Chemical compatibility also matters. Silver arsenate does not belong with acids, alkalis, or reducing agents. Once, a careless organizer stuck incompatible containers side by side. Later, a small spill caused a cloud. Sorting chemicals by risk, not just alphabetically, dodges a chemistry disaster.
Stop Problems Before They Start
Training and clear signage make the biggest difference. Most accidents I’ve seen start from someone either guessing at the rules or ignoring them. No two storerooms should work on guesswork. Standard operating procedures, regular audits, and a clear inventory list—these are steps everyone should use.
Let’s be honest: real safety demands a little paranoia and a lot of consistency. Silver arsenate will never be the most exciting bottle on a shelf. But the risks—chronic poisoning, environmental spills, frantic phone calls to poison control—are too real to ignore. Handling it with respect matters, not just because the books say so, but because lives and health are on the line. Everybody deserves to head home healthy at the end of their shift.
What safety precautions are required when handling Silver Arsenate?
The Real Hazards Behind Silver Arsenate
Silver arsenate isn’t a compound that many people encounter every day, but anyone working in a research lab or handling old scientific collections probably knows how dangerous it can be. Both the arsenic and silver elements raise red flags, but the arsenic side deserves the bigger spotlight. Chronic arsenic poisoning pulls no punches — breathing in the fine dust or letting it touch your skin causes serious health issues, including cancer and nerve damage over time. Having spent years in chemical labs, I’ve watched experienced chemists take ten minutes just to suit up before opening a tiny vial. There’s a good reason for all that caution: mistakes with arsenic compounds can ruin your health, long after you leave the lab bench.
The risks aren’t limited to the person handling the substance. Residue on gloves, contaminated lab benches, or powder drifting through the air creates hazards for everyone nearby. A careless moment can lead to headaches, nausea, chronic tiredness, or worse, just from breathing in what looks like a bit of dust. I’ve seen protocols tighten across universities as more information about the dangers surfaces — accidents from false confidence push change faster than any manual ever could.
What Makes a Difference: Safety That Actually Works
Preparation starts well before you open any container. Checking safety data sheets, confirming the correct labeling, and reviewing disposal plans gives everyone a fighting chance at staying healthy. Ventilation matters most. I’ve never worked with arsenic compounds outside of a well-maintained fume hood. Not just any fume hood, but one that pulls a strong airflow and uses proper filters. Even the best habits in a poorly ventilated space don’t offer real protection. Blowing air out through a window or using a kitchen-style fan seldom works, since airborne arsenic lingers and builds up where you least expect it.
Gloves don’t just protect hands, they keep residues off the rest of your body. Splash goggles or a face shield, plus a lab coat or protective gown, aren’t extras — they’re basics. Wearing two layers of gloves works well with arsenic, so if powdered material leaks onto the outer glove, you can peel it off safely. Changing gloves often and washing hands thoroughly after any chemical work matters, even if you think your hands stayed clean. Mistakes show up wherever curiosity runs ahead of training. I learned early on that eating or drinking in a lab, or using the same gloves to open a door, spreads toxins further than anything else.
Dealing With Spills and Waste
Most labs keep dedicated arsenic spill kits on hand: absorbent pads, neutralizers, sealable bags, clearly labeled waste containers. Some compounds dissolve in water, but not this one. Dry cleanup prevents powders from riding on puddles or splashing in ways that create hidden problems. Cleaning up with a standard broom or vacuum just moves the danger elsewhere. Wet wipes, dedicated vacuums fitted with HEPA filters, and disposable towels all help collect every trace.
Why Training and Culture Beat Rules Alone
Reading the rulebook won’t help much if people don’t believe the risks matter. Hearing first-hand stories about accidents sticks better than lectures. In places where I’ve seen real safety, someone always double-checks, people speak up if they see a shortcut, and everyone feels free to ask questions. Long-term health depends on habits, not just gear. That’s the wisdom I wish every new chemist picked up on day one: respect dangerous compounds not because someone told you to, but because you want to retire healthy, with all your memories and your well-being intact.
| Names | |
| Preferred IUPAC name | silver(I) arsenate |
| Other names |
Disilver arsenate Silver(I) arsenate |
| Pronunciation | /ˈsɪlvər ɑːrˈsiːneɪt/ |
| Identifiers | |
| CAS Number | [7784-88-7] |
| Beilstein Reference | 35868 |
| ChEBI | CHEBI:86345 |
| ChEMBL | CHEMBL1201604 |
| ChemSpider | 20230813 |
| DrugBank | DB14636 |
| ECHA InfoCard | 100.971.241 |
| EC Number | 215-375-4 |
| Gmelin Reference | 89857 |
| KEGG | C18613 |
| MeSH | D014022 |
| PubChem CID | 24554 |
| RTECS number | XH8390000 |
| UNII | 1Y6CO42S0B |
| UN number | UN3312 |
| Properties | |
| Chemical formula | Ag3AsO4 |
| Molar mass | 437.737 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 5.22 g/cm³ |
| Solubility in water | insoluble |
| log P | -43.71 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 16.21 |
| Basicity (pKb) | 11.54 |
| Magnetic susceptibility (χ) | `+83.0·10⁻⁶ cm³/mol` |
| Refractive index (nD) | 1.89 |
| Dipole moment | Zero Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −909 kJ/mol |
| Pharmacology | |
| ATC code | V09AX02 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin. May cause cancer. Environmental hazard. |
| GHS labelling | GHS07, GHS08, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P261, P264, P270, P273, P301+P312, P302+P352, P304+P340, P305+P351+P338, P311, P330, P362, P405, P501 |
| NFPA 704 (fire diamond) | 2-0-3-OX |
| Lethal dose or concentration | LD50 oral rat 45 mg/kg |
| NIOSH | WH6650000 |
| PEL (Permissible) | 0.01 mg/m3 |
| REL (Recommended) | 0.01 mgAg/m3 |
| IDLH (Immediate danger) | IDLH: Unknown |
| Related compounds | |
| Related compounds |
Silver orthophosphate Silver nitrate Sodium arsenate Potassium arsenate |

