N,N-Dimethylselenourea: Looking Beyond the Laboratory Bench
Historical Development: Roots in Curiosity and Demand
N,N-Dimethylselenourea told its own quiet story as chemists turned curiosity over selenium’s behavior into targeted action. Back in the days when selenium research moved from mining byproduct to laboratory reagent, organoselenium compounds gained a reputation for their unique reactivity. The search for gentle, selective selenium donors encouraged deeper study. The early synthetic routes took cues from sulfur analogs; pioneers looked for ways to push the envelope, realizing selenoureas could unlock fresh chemical transformations. Today, N,N-Dimethylselenourea owes much to these efforts, bridging a gap between basic selenium chemistry and applied science.
Product Overview: What Sets It Apart
Most organic labs dabbling in selenium try out selenourea derivatives at some point, but N,N-Dimethylselenourea stands out for the balance it strikes: manageable safety profile, solid stability, and noticeable influence on reactivity. The methyl groups aren’t just decorations. They prompt smoother solubility in common solvents and tweak both electronic and steric environments. The compound shows up as a handy reagent or intermediate, quietly playing its part in synthesis where other selenium sources overshoot or fall flat.
Physical & Chemical Properties: Under the Microscope
Talking about physical properties, N,N-Dimethylselenourea usually appears as a crystalline powder. This solid form means easier handling than some malodorous or volatile selenium chemicals. It holds together at room temperature and isn’t bothered by casual exposure in the lab. The signature selenium atom inside the molecule brings along a hefty atomic mass and full-bodied electronic cloud. With the methyl groups on top, the molecule resists water to some degree, leans toward organics, and answers quickly to heating. Chemically, it reacts as both a selenourea and an N,N-dimethyl derivative. Once added to a reaction flask, it can become a selenium transfer agent, a nucleophile, or even a building block for bigger, more complex tools.
Technical Specifications & Labeling: More Than Numbers
Buy any specialty reagent and labeling jumps out at you—purity, melting point, batch number. Anyone hoping for trouble-free reactions has learned to trust these tags and specifications. Purity gets checked by NMR or melting point consistency. Look for crystal clumping or off-smells as early trouble signs. That said, relying only on digits misses the bigger picture. Small impurities sometimes tell chemists what storage conditions worked best, or where supply chains cut corners. The right label on N,N-Dimethylselenourea means responsibility by everyone handling it.
Preparation Method: Recipes that Last
Most recipes blend methylamine or dimethylamine sources with selenium-based reagents, relying on controlled heating and solvent choice. Selenium can give surprises, so ventilation and slow addition reduce side products. Traditionally, selenourea skeletons grow from basic condensation reactions, but small tweaks in reactant ratios tune the process for better yields or cleaner product. Any venturing into scaling up must back up synthetic know-how with cautious material safety. Taking shortcuts or running blind risks ruining batches—or worse, creating selenium-laced waste that nobody wants to clean up.
Chemical Reactions & Modifications: The Chemistry Playground
What gives N,N-Dimethylselenourea its place in organic synthesis is flexibility. Add electrophiles, and the molecule can step in as a selenium transfer partner for constructing organoselenium units. Deprotonation under the right base can drive further transformation; exposure to halogenating agents unlocks another toolbox entirely. Some reactions tune the oxidation state, pushing selenium from its -2 state up a notch. In my experience, finding the right solvent and temperature gets half the job done—rushing these steps raises frustration more than yields.
Synonyms & Product Names: Navigating the Language Maze
Chemistry loves synonyms, and this compound pops up under labels like Dimethylselenourea, DMSeU, or 1,1-Dimethylselenourea. Chemical abstracts and journals don’t always agree, so researchers use CAS numbers or stick with systematic names to dodge confusion. There’s a lesson tucked in there—a shared vocabulary saves time and mistakes, especially where hazardous chemicals are concerned.
Safety & Operational Standards: No Cutting Corners
Few things matter more than safety in organoselenium chemistry. Compound design can soften risks, but selenium’s biological activity still deserves healthy respect. Standard good lab practices rise to the surface here: gloves, goggles, ventilation, and careful waste management. Rule of thumb says: treat every selenium compound as unpredictable unless proven otherwise. Inhalation and ingestion risks run high; selenium’s toxicity can sneak up and symptoms hit late. Cleaning up spills with attention—never dry sweeping, always using wet methods—reflects commonsense care learned the hard way. Waste disposal calls for special bins and often outside contractors; pouring selenium down the drain only invites trouble.
Application Area: From Synthesis to Science
N,N-Dimethylselenourea straddles several uses in research and industry. Folks in organic synthesis turn to this molecule as a mild, workable selenium donor. Medicinal chemists sometimes use selenourea derivatives to make enzyme inhibitors or antioxidant compounds. Materials scientists look for organoselenium units when developing new electronic materials or exploring nonconventional semiconductors. In my own lab experience, the value comes from how this compound can streamline synthetic steps—saving time, reagents, or even allowing transformations that sulfur analogues just won’t support.
Research & Development: Pushing the Boundaries
Every few years, new findings on organoselenium chemistry change the game. R&D with N,N-Dimethylselenourea circles around three main ideas: safer preparation methods, gentler reactions, and unique applications. High-throughput screening for drug candidates leans on molecules that can be easily built and modified, and selenourea analogs gain an edge here. Automation brings scale, and demand for reproducible, predictable results keeps standards high. Funding and time shape what gets tried, but breakthroughs in selenium chemistry often promise more elegant syntheses for complex targets.
Toxicity Research: Balancing Progress and Responsibility
Selenium holds a strange place in toxicity—needed for health in trace amounts, but dangerous when levels creep too high. Toxicity research around N,N-Dimethylselenourea and its relatives digs into the mechanisms of action within living cells. Testing goes past basic acute toxicity studies; researchers check for subtle effects on enzymes, gene expression, and organ systems over time. Chronic exposure studies matter because safety in the lab sometimes lulls people into forgetting real-world impacts. Data encourages chemists to refine protocols, reduce exposure, and approach new reactions with practical humility.
Future Prospects: Forging a Smarter Path Forward
Interest in organoselenium compounds keeps nudging the field forward. Green chemistry frameworks frame selenium use as both opportunity and challenge. Researchers seek new roles for compounds like N,N-Dimethylselenourea—not just as reagents, but as building blocks for more sustainable materials or advanced drugs. The next wave likely blends safety, environmental responsibility, and smart molecular design. Cross-disciplinary projects between biochemists, synthetic chemists, and toxicologists foster better insights and results with less risk. The path isn’t smooth, but each time a cleaner synthesis or safer method emerges, everyone benefits—from the benchtop to the broader community.
What is the chemical formula of N,N-Dimethylselenourea?
What’s Inside N,N-Dimethylselenourea?
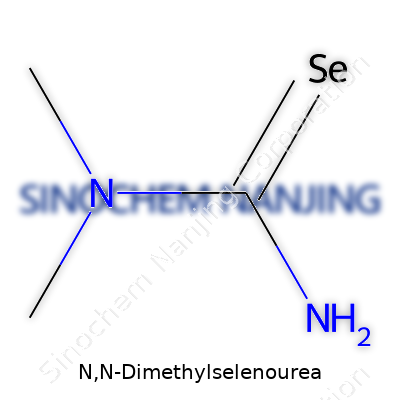
Let’s talk chemistry for a second. Mention N,N-Dimethylselenourea, and most folks outside of research circles draw a blank. Underneath this mouthful of a name, there’s a story of atoms and bonds that paints a clear structure. In plain terms, this compound brings selenium into an everyday molecular shape found with regular old urea, but with some strategic swaps: two methyl groups stuck onto nitrogen atoms, and a selenium replacing the traditional oxygen. The chemical formula reads: C3H8N2Se. That’s three carbons, eight hydrogens, two nitrogens, and a selenium.
Why Should We Care About This Compound?
Some might say it’s just another obscure lab chemical. But dig deeper and you realize selenium’s chemistry often has ripple effects. Selenium plays a picky role inside the body, required in tiny amounts for enzymes like glutathione peroxidase. Get too little, and troubles show up. Get too much in the wrong form, and that’s a different issue—selenium compounds can turn toxic fast. N,N-Dimethylselenourea itself isn’t found in nature. People in pharmaceutical labs build it on purpose. The attraction comes from the selenourea backbone, used in organic synthesis to introduce selenium atoms in smart, targeted ways.
Safety Takes the Front Seat
Experience shows that dealing with selenium stuff isn’t casual. Mishandling or ignoring protective gear can lead to symptoms: garlic breath, hair loss, nail changes, or even nervous system issues at high doses. This holds true for N,N-Dimethylselenourea and its chemical cousins. Factory and lab technicians rely on fume hoods, masks, and gloves for a reason. Not everyone on TikTok or YouTube gives the proper warnings. In the lab, reading the material safety data sheet always comes before opening a bottle—too many stories exist about folks learning the hard way.
Building Blocks for Research and Beyond
Why keep working with this molecule? Its main gig lies in the hands of researchers tailoring new pharmaceuticals or testing reactions where selenium’s unique chemistry pushes things forward. Some selenium-based drugs, like selenazole antifungals, start with molecules like these. Synthetic chemists use N,N-Dimethylselenourea as a stepping stone because the selenium enjoys making new bonds, opening doors to molecules not easy to reach using sulfur or oxygen. You’ll find this compound hanging around in research pipelines at places like the Max Planck Institute and specialist chemical suppliers serving global labs.
Responsible Handling and a Smarter Future
Putting a spotlight on this chemical means thinking both about its promise and its risks. Sustaining best practices means training young chemists early, making safe use second nature. Companies can adopt barcoded inventory and digital checklists, reducing risk of human error. Modern waste disposal and air clean-up systems matter just as much as clever chemistry. With stronger international cooperation between academic and industry players, keeping an eye on environmental impact goes hand-in-hand with discovery.
Final Word on N,N-Dimethylselenourea
Chemical advances rarely grab headlines. Still, the story of any substance—like N,N-Dimethylselenourea—depends on the people who use it, the rules that guide them, and the creativity behind each experiment. Chemistry’s not just for textbooks or patent filings. It’s a real-world responsibility, one that grows as researchers ask what’s possible and communities ask what’s safe.
What are the primary uses or applications of N,N-Dimethylselenourea?
Getting to Know N,N-Dimethylselenourea
N,N-Dimethylselenourea doesn’t show up in your neighborhood pharmacy or on hardware store shelves. It belongs to a set of chemicals mostly found in research labs or specialty manufacturing. This compound contains selenium—a chemical that’s both essential and toxic, depending on the dose and context. Scientists have turned to N,N-Dimethylselenourea for its role in organic synthesis and for the delicate touch it brings to certain chemical transformations that standard sulfur-based ureas just can’t match.
Making Chemistry and Life Sciences Move Forward
People talk a lot about unique building blocks in organic chemistry. N,N-Dimethylselenourea works as a selenium donor in synthesis. Researchers often use it to introduce selenium atoms into molecules where basic precursors won’t get the job done. Selenium chemistry can seem niche, but drop the right atom into the right place and you can create new classes of drugs, imaging agents, and even tools for exploring how our bodies fight off stress at the cellular level.
Getting selenium into drug-like molecules in a controlled way doesn’t come easy. This is where compounds like N,N-Dimethylselenourea find their calling. Selenium-based drugs, like certain anti-thyroid agents, need careful handling. Start with a compound like N,N-Dimethylselenourea, chemists can gain more precision in building up those targets.
Working Behind the Scenes in the Lab
Maybe you’ve worked in a chemistry lab, seeing how even tiny tweaks to a molecule change the whole outcome of an experiment. I’ve seen chemists struggle with old routes that depend on finicky, moisture-sensitive reactions that just won’t scale. Compounds with a selenium base sometimes offer a smoother ride. N,N-Dimethylselenourea speeds up certain organic reactions—namely cyclizations and insertions—so that other reagents perform better and you get improved yields where classic urea or thiourea would stall out.
Organic synthesis turns difficult without tools that let scientists swap, add, or rearrange atoms cleanly. Read old research articles, and you’ll find N,N-Dimethylselenourea cited as the difference-maker for new catalysts or for creating valuable intermediates in medicinal and materials chemistry. In practice, that makes for a more effective route toward new medicines or probes to track disease inside living cells.
Environmental and Safety Concerns
Selenium sits in a tricky spot. On one hand, our soil and diet need trace selenium. On the other, too much selenium becomes a health risk. Working with N,N-Dimethylselenourea, scientists must treat its waste and vapors with respect. The real challenge is balancing the benefits to research and industry with the risk that misuse or poor disposal could pollute water or harm people. That’s why people in the know stress closed systems, good filtration, and strong waste management routines. Regulations flag selenium-containing waste for strict tracking, and it pays off to use those rules as guidelines, not just requirements to check off.
Ways to Use It Responsibly
Some companies already take steps to design reactions that use lower amounts or recycle leftover selenium compounds. There’s also a push for “greener” chemistry, where researchers hunt for alternatives that give the same result but with safer byproducts and less impact on people and the planet. Educational programs also help young scientists learn not just synthetic tricks but habits that reduce harm outside the lab. Sharing more about successes and failures around N,N-Dimethylselenourea can only help others manage similar risks. Openness leads to smarter, safer research that doesn’t leave a mess for tomorrow.
How should N,N-Dimethylselenourea be stored and handled safely?
Why Respect for This Compound Matters
N,N-Dimethylselenourea doesn’t show up in most households, but for chemists, it holds real significance. The selenium atom in this compound holds both intrigue and a bit of menace—exposure risks you don’t take lightly. Even in a controlled lab setting, I remember the tension that came each time someone mentioned selenourea derivatives. Growing up in university research labs, folks taught me early on that many selenium compounds could pose unique toxicity, producing nasty volatile byproducts or irritating the skin and respiratory tract. Getting complacent never ended well.
What Real-World Storage Looks Like
Nobody grabs a plastic bin or an open shelf. N,N-Dimethylselenourea stays in cool, dry environments, always in air-tight amber glass bottles. Moisture causes trouble—many organoselenium compounds react with water or air and degrade over time. Heat speeds decomposition, raising the risk of releasing fumes you don’t want to breathe. People sometimes forget that stability studies aren’t just paperwork; a degree or two on the thermometer can alter exposure risk or accident potential. So, refrigerators or temperature-controlled cabinets become standard practice. An ordinary kitchen fridge will not cut it.
Any lab worth its salt keeps volatile and toxic chemicals within locked, well-ventilated chemical cabinets. Labeling jumps out in big letters—no smeared ink or abbreviations. I remember how easy it was for the labels to fade, which led to confusion more than once. In shared spaces, if a bottle isn’t crystal clear in its labeling, folks just don’t touch it. That’s basic self-preservation learned after seeing enough emergency drills and real cleanups.
Handling—Gloves, Guts, and Good Habits
Rarely does someone pick up a vial of N,N-Dimethylselenourea barehanded. Gloves—nitrile, never porous latex—sit ready by every workstation. Safety glasses shield against splashes; lab coats stop contamination from sneaking home. One professor I worked with once ended up in urgent care by ignoring a pinhole tear in his gloves and letting selenourea touch his skin. Lesson: mistakes happen fast, and recovery doesn’t always come easily.
Weighing and transferring take place inside fume hoods with the sash down low. Even well-trained researchers speak up if they smell anything funny. I also learned that spills create panic less through scale, more due to unpredictability: clean-up kits, absorbent pads, and selenium-specific neutralizers deserve a spot within arm’s reach. For surface decontamination, bleach won’t do; specialized selenium deactivants work better. Nobody corners themselves during work—easy access to exits and showers makes a difference if something goes sideways.
Disposal and the Path Toward Safer Labs
The dangers don't disappear down the sink. Disposal plans get filed with the local environmental office, following the rules for hazardous heavy metals and organoselenium waste. Containers labeled for selenium waste never double as trash bins. In university settings, routine safety meetings taught us the value of proper segregation and the risks of rushing through clean-up. In my circle, accountability means logging every disposal, even if the paperwork slows down the day.
Safer practices come from a blend of training, memory, and respect for these chemicals. Labs that choose transparency and regular risk reviews rarely find themselves in disaster. Mistakes spark changes—after each near-miss, our group overhauled protocols or invested in better containment. In the end, safe handling stands as a shared effort—it builds trust, protects people, and lets important research continue without unnecessary risk.
What are the potential health hazards associated with N,N-Dimethylselenourea?
What Happens with Exposure?
Encountering a chemical like N,N-Dimethylselenourea, it becomes important to know what risks sit in the bottle before trust builds around its use. This compound, related to the selenourea family, delivers both the unpredictability of organoselenium substances and the harsh track record of urea derivatives. People in labs, research, and some manufacturing industries cross paths with it during synthesis or application. The risks are not some faraway laboratory theory—the health hazards matter every day for real workers and communities.
Breathing or Touching It: The Reality of Acute Risks
Breathing dust or vapor from N,N-Dimethylselenourea can irritate airways quickly. Selenium compounds, even in small doses, set off coughing, sore throat, or shortness of breath. Skin or eye contact is not kind either—expect burning, stinging, redness. Skin absorption adds another layer, as selenium’s ability to pass through the skin gets documented in several occupational health studies. I remember a chemist friend warning about the metallic, garlic-like taste that pops up after skin exposure to organoselenium compounds, almost like the body waves a red flag itself before things get more serious.
Long-Term Effects and The Body’s Alarm Signals
Frequent or long exposures do more than just cause momentary discomfort. Studies from respected toxicology institutes have found that chronic exposure to selenium compounds, especially those containing methyl groups, can cause damage to the liver, kidneys, lungs, and nervous system. The U.S. National Institute for Occupational Safety and Health (NIOSH) points out that chronic, low-level exposure can bring on symptoms like headaches, fatigue, and digestive upsets—a clear sign the body struggles to process out the buildup. Continuous absorption leads to selenosis, where fingernails grow brittle, skin breaks out in rashes, and the breath turns persistently garlicky. It’s unsettling to see neglect of proper handling lead to these outcomes, knowing good protocols could have prevented them.
Are There Cancer Risks?
The question of cancer risk always comes up with unknowns like organoseleniums. The International Agency for Research on Cancer (IARC) still lacks enough data to lock in a verdict for N,N-Dimethylselenourea. Animal studies on similar compounds stir some worry—there, chronic high doses have led to increased rates of liver and lung tumors. While that doesn't guarantee danger for every person exposed, it signals caution should win over convenience until more human data lands on the table.
Prevention: What Works in Real Workplaces
Practical solutions matter more than paper rules. Laboratories and factories using N,N-Dimethylselenourea operate safer with proper air ventilation, secure containers, and routine air monitoring. Workers must wear gloves, goggles, and masks every single time; cutting one corner invites regret. Training workers on spotting signs of exposure and pushing a culture where reporting symptoms meets support (not discipline) makes a difference. Regular medical check-ups for those with long-term exposure give early warning if the body absorbs more selenium than it can handle. I’ve seen peer-driven safety programs in smaller labs catch problems management missed. Involving everyone, not just top-down instructions, brings the real change.
Why Attention Pays Off
N,N-Dimethylselenourea sits in an odd spot—important for niche chemistry, but rough for health if trust replaces caution. Regulatory gaps still exist, so responsible companies set their own limits based on global examples. History reminds us the small label on a bottle can’t compete with the lesson learned through harm. Fighting chemical hazards like this one needs both science and the steady mindset of people watching each other’s backs. It’s not just about rules; it’s about real people staying healthy at work and at home.
Is N,N-Dimethylselenourea available in different purities or grades?
A Close Look at Available Grades
Anyone who’s spent time in a research lab knows there’s no such thing as a “one size fits all” approach to specialty chemicals. Talking about N,N-Dimethylselenourea, it’s easy to focus on its name or its few appearances in research papers. The real question for most chemists and manufacturers is, “What am I actually getting in this bottle?” Purity shapes every outcome—whether we’re working on basic synthesis, high-precision electronics, or an obscure novel reaction.
I’ve stood in front of a stockroom shelf, squinting at chemical labels, wondering if 95% purity will do the trick or if it’s worth paying extra for the 99%-plus version. In many industries, cutting corners here makes no sense: contaminants can and will interfere. It’s not sensationalism; it’s based on lived experience chasing unexpected side-products or watching yields drop off just because trace impurities nudged a reaction somewhere else. Selenium chemistry brings special headaches, since many selenium compounds show high reactivity even in tiny quantities.
Where Grades Make a Difference
The research world splits chemicals into at least two broad camps. There’s “technical grade” material, which may work for industrial-scale processes or rough bench synthesis. Then, there’s “analytical grade” or “high purity”—the stuff we turn to when new methods, sensitive catalysts, or advanced measurements come into play. Looking across suppliers, N,N-Dimethylselenourea shows up with reported purities in ranges spanning from mid-90% to as high as 99.9%. These numbers aren’t just for show. A synthesis route for a pharmaceutical intermediate will grind to a halt if an impurity interferes with a key transformation or alters solubility. Researchers push suppliers with questions about elemental analysis, metal content, water, and even specific-byproducts from the synthesis method.
Publishing results in peer-reviewed journals or qualifying material for a batch destined for human consumption never leaves room for guesswork. Having run microgram-scale reactions and gram-scale pilot projects, I’ve learned that small differences in chemical grade mean real changes in project timeline, product safety, and cost. There’s only so much you can filter or purify in-house before your budget and patience run out. The phrase “good enough” doesn’t hold up when your desired effect only appears with material hitting high purity standards.
Supplier Transparency and Customer Responsibility
Folks buying N,N-Dimethylselenourea do themselves no favors by skipping the details. Suppliers serious about business provide certificates of analysis and methods for identifying trace impurities. The challenge pops up with newer, less common compounds: competing suppliers may cut corners, betting most customers won’t notice a missing percentage point or two. Unfortunately, every batch tells its own story, and a poor documentation trail quickly leads to wasted batches, failed experiments, and potentially unsafe outcomes.
Lab managers and project leads should keep a critical eye on paperwork and trusted supplier relationships. Before a bottle ever hits a bench, demanding clarity about grade, impurity profiles, and lot-to-lot consistency brings long-term savings. It also reduces the risk of chasing phantom problems caused by unlisted contaminants.
Where Do We Go Next?
The solution isn’t always pushing for the top grade every time, especially if budgets are tight and end-use applications allow a little compromise. The trick is knowing what the project can handle and sparing no attention in sourcing and verification. Higher up the value chain, chemical companies and distributors must do the hard work in pressing for rigorous QA/QC, transparent data, and quick communication when customers hit a snag. This builds an industry where researchers, process chemists, and buyers trust the labels and focus on innovation instead of cleanup.
| Names | |
| Preferred IUPAC name | N,N-dimethylselanamide |
| Other names |
Dimethylselenourea N,N-Dimethylselenourea 1,1-Dimethyl-2-selenourea N,N-Dimethylselenoformamide |
| Pronunciation | /ɛn ɛn daɪˌmɛθɪl sɪˌliːnoʊˈjʊərə/ |
| Identifiers | |
| CAS Number | [34456-61-2] |
| Beilstein Reference | 1099571 |
| ChEBI | CHEBI:38843 |
| ChEMBL | CHEMBL140723 |
| ChemSpider | 158390 |
| DrugBank | DB08157 |
| ECHA InfoCard | 100.166.311 |
| EC Number | 219-218-6 |
| Gmelin Reference | 83486 |
| KEGG | C15668 |
| MeSH | D017965 |
| PubChem CID | 4090902 |
| RTECS number | KV7525000 |
| UNII | 4R2Y3G6103 |
| UN number | 2811 |
| CompTox Dashboard (EPA) | `DTXSID0045442` |
| Properties | |
| Chemical formula | C3H8N2Se |
| Molar mass | 135.14 g/mol |
| Appearance | White to off-white crystalline powder |
| Odor | amine-like |
| Density | 1.42 g/cm3 |
| Solubility in water | soluble |
| log P | -0.28 |
| Acidity (pKa) | 13.6 |
| Basicity (pKb) | pKb = 12.26 |
| Magnetic susceptibility (χ) | -68.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.663 |
| Dipole moment | 3.25 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 207.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -38.7 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -487 kJ mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. Suspected of causing genetic defects. Toxic to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H301 + H331 |
| Precautionary statements | Precautionary statements: P261, P280, P301+P312, P304+P340, P308+P313 |
| NFPA 704 (fire diamond) | 1-2-2-W |
| Flash point | 86 °C |
| Autoignition temperature | Autoignition temperature: 450 °C |
| Lethal dose or concentration | LD50 (oral, rat): 215 mg/kg |
| LD50 (median dose) | LD50: 1650 mg/kg (rat, oral) |
| NIOSH | NIOSH: KL5950000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 25g |
| Related compounds | |
| Related compounds |
N,N-Dimethylthiourea N,N-Dimethylurea Selenourea Thiourea Urea |

