Selenious Acid: A Practical Look at a Powerful Chemical
Historical Development
Selenious acid has traveled a curious path through history. It made its mark back in the 19th century, when chemists isolated selenium from ores and explored its strange behavior. Over time, this acid found its purpose in industry and research, shaped by the discoveries and needs of each era. The early days saw scientists using it mostly for qualitative analysis, but that changed as technology advanced. In the 20th century, demand for pure selenium compounds for glassmaking and electronics grew, which pushed chemical manufacturers to refine their methods. Today, selenious acid’s story intertwines closely with new fields like photovoltaics and medicine.
Product Overview
Selenious acid stands out as one of the more versatile selenium-based chemicals. Most labs ship it as a clear, colorless solution, strong and ready for use. Bulk suppliers keep it flowing for a host of industries—think glassmakers looking for deep red hues or pharmaceutical researchers beckoned by selenium’s unique qualities. Anyone handling selenious acid quickly figures out that it calls for careful storage and smart handling because of its strong, oxidizing nature. Its reach extends beyond the bottles on the shelf, touching everything from photography to advanced medical diagnostics, and even the refinement of other selenium chemicals.
Physical & Chemical Properties
Selenious acid, H2SeO3, shows itself as a crystalline solid or, more often in industry, a concentrated water solution. Its crystals dissolve well in water, producing a strong acid. Touching or inhaling this stuff is not a good idea, which speaks to its power. It stands out as a solid oxidizer and acts a lot like sulfuric and sulfurous acid, which isn’t surprising given selenium’s place in the periodic table. Chemical reactions involving selenious acid often run cleanly and quickly because of its high reactivity. The acid itself slowly evaporates, so seals and air management matter in storage.
Technical Specifications & Labeling
Production standards for selenious acid tend to lean on international benchmarks for safety and performance. Industrial buyers pay attention to selenium content by weight, solution concentration, and levels of trace impurities like heavy metals. Containers always wear a range of warning symbols—corrosive, toxic, and environmental hazard. Accurate labeling saves lives and keeps workflows humming, so compliance experts double-check batch numbers, expiry dates, and shipping hazard codes before the acid leaves the warehouse. MSDS sheets point out proper gear and emergency measures, staying a step ahead of any spills or accidents.
Preparation Method
Making selenious acid isn’t rocket science, but it does demand attention to detail. Most producers start by dissolving selenium dioxide in water, a process that works best under controlled temperatures, away from organic materials that might spark unwanted side reactions. Careful monitoring helps keep purity high and waste low. On a larger scale, companies sometimes use selenium metal, reacting it with strong nitric acid to create the dioxide before adding water. Waste management plays a big role—unreacted selenium and byproducts can cause trouble downstream, so efficient trapping and recycling make a real difference.
Chemical Reactions & Modifications
Selenious acid doesn’t just sit on a shelf. Chemists use it to oxidize other compounds, turning up in everything from analytical chemistry to producing specialty selenium derivatives. It reacts with reducing agents to form elemental selenium, a reaction that produces a distinctive red color. In organic chemistry, it helps install selenium into molecules, a trick that biochemists find useful. With a strong acid, selenious acid generates selenium trioxide, which feeds into making pure selenium metal for semiconductors and alloys. Reaction pathways vary, letting manufacturers adapt depending on what they want to make.
Synonyms & Product Names
Walk into a chemical catalog, and selenious acid might go by a few names: selenium dioxide solution, selenium(IV) acid, or even just "selenious solution." In trade, CAS numbers (7783-00-8) provide a hard anchor for those ordering worldwide. Its aliases mostly come down to translation differences or conventions in academic versus industrial circles, but everyone working with it recognizes its signature bottle and safety warnings. Consistent naming across markets keeps confusion at bay and lets emergency responders act quickly if something goes wrong.
Safety & Operational Standards
If you keep selenious acid in storage or use it in processes, serious safety routines come into play. Unprotected contact with skin or inhalation causes burns or lung damage. Facilities invest in heavy-duty ventilation and chemical-resistant gloves, along with splashproof goggles. Emergency showers and eye wash stations sit nearby. Spill kits and containment bins stand ready because selenious acid reacts quickly in the open air. Regular training for all staff helps reinforce high standards and makes sure the acid ends up where it should—not in local drains or the wrong containers. Regulatory bodies, from OSHA to the European Chemicals Agency, keep rules strict and audits regular, driving home a culture of compliance. Environmental teams work in parallel, tracking air and wastewater outputs to guarantee that community health stays safe.
Application Area
Selenious acid’s reach feels broad. In glassmaking, it colors glass deep red—a signature look, hard to achieve any other way. Laboratories rely on it as a reagent in analytical chemistry, picking up trace metals or modifying organic molecules. Medical researchers eye it warily but eagerly for its role as a selenium supplement, since selenium is an essential trace element for human health, albeit in minuscule doses. In electronics, the acid contributes to the production of photoconductors for photocopiers and specialized solar cells. Its oxidizing strength gives it punch where precise changes to complex molecules are needed. Photography and electroplating also lean on its properties. Storage and handling practices differentiate its use—lab work usually calls for smaller bottles and tighter controls, while bulk industry settings focus on cost and volume.
Research & Development
Research keeps opening new doors for selenious acid. Teams studying alternative energy sources test its reactivity for quantum dots and solar panels that run on selenium alloys. Pharma scientists keep digging into ways to balance its toxicity with its potential benefits in metabolic therapies or as trace nutrient supplements. Materials scientists explore coatings and compounds that rely on selenious acid’s quick chemistry for next-generation batteries, screens, and medical imaging. Even environmental cleanup researchers consider it as a way to process mercury-laden waste. Each experiment demands rigorous control. Benefits draw attention, but risk management walks side-by-side at every stage. Collaboration between academic researchers and industry outfits speeds up innovation but often runs into the twin barriers of cost and regulatory complexity.
Toxicity Research
Toxicologists don’t take selenious acid lightly. Even small exposures mess with cell membranes and metabolic processes, making careful dose control a must. Chronic or acute poisoning in humans leads to serious lung and organ damage. Industrial accidents have prompted a lot of safety reform in the workplaces that use it. For environmental scientists, selenious acid shows up as a culprit in some water pollution cases, especially where selenium-based industrial waste has escaped controls. Animal studies back up these concerns, listing a tight margin between useful and harmful doses. Thanks to this, daily handling routines, labeling, and storage all look different from those used for less hazardous reagents. Regulatory notifications and ongoing workplace monitoring help keep risks in check, but a zero-tolerance approach to spills and improper disposal persists.
Future Prospects
Looking ahead, prospects for selenious acid look both promising and challenging. New uses in green technology, such as in super-efficient solar panels, tend to pull in research funds and commercial interest. At the same time, increased scrutiny over chemical toxicity and environmental impact spurs companies to look for safer alternatives or to design processes that recycle and reuse byproducts. Potential breakthroughs in medicine keep research going, even if practical applications take a while to pass through safety and approval checks. Improved containment and smarter process engineering may make large-scale, safe use easier, keeping selenious acid on the roster for high-value manufacturing and research. Above all, the story of selenious acid teaches that careful management and restless innovation make it possible to tap into some of chemistry’s strongest powers without letting safety or sustainability slide.
What is Selenious Acid used for?
Everyday Utilities and Why Selenious Acid Matters
Whenever a conversation drifts to lesser-known chemicals, selenious acid rarely shows up. Yet, it holds a surprisingly strong role in industrial, medical, and research settings. Think of it as the quiet worker helping other elements shine—literally, in some cases.
Glass Tinting and What I’ve Seen
Working at a glass factory fresh out of college, I watched skilled hands mix colorants for specialty glass. Selenious acid played a part in the creation of fiery red and delicate pink glassware. This compound gives glass that particular tint you’ll see in old car tail lights or commemorative drinking glasses. Its unique chemistry forms a red hue when combined with certain metals. Artisans didn’t talk much about safety, but I learned quickly that proper gloves and ventilation matter: selenious acid comes with health risks if ignored.
Electroplating and Equipment Protection
Electroplating isn’t as glamorous as glassblowing, but it keeps machinery durable and protected against corrosion. Selenious acid stands out in this process, especially for copper and nickel plating. It adjusts the structure of the metal surface, making plated layers more even and more adherent. In my hands-on days at a metal-finishing shop, small tweaks in the selenious acid concentration made a difference—shiny, smooth finishes instead of uneven, flaky ones. Less product waste means lower costs. This fact alone draws big companies to trust it, despite the hazardous label.
Making Selenium Supplements—Yet Playing with Fire
Science and nutrition journals mention selenium as an essential trace mineral. Selenious acid gets used to manufacture selenium supplements. Getting the right dose of selenium matters—a little too much, and you’re in trouble. I once followed a dietary supplement company’s production process; every step used automated monitoring. They aimed to hit that delicate balance: just enough selenium to aid metabolism and immunity, without crossing the line into toxicity. Strict safety protocols and quality checks shaped every batch. Even so, the tightrope walk between health benefit and risk creates constant tension.
Laboratory Workhorse
Researchers appreciate how selenious acid reacts in controlled experiments. It serves as a critical oxidizing agent in chemical analysis—testing minerals, checking purity in samples, or prepping for more complicated reactions. Those working in research labs know their stockroom shelf always includes a bottle, tightly labeled, for its precision and consistency. Years ago, during a stint in an environmental testing lab, our team depended on its reliability to measure selenium content in soil and water samples. The right result could reveal if farmland needed new management, or if water supplies risked contamination.
Tackling Risks and Moving Towards Safer Use
Handling this acid comes with responsibility. It is both toxic and corrosive. Exposure can cause burns or more serious health issues if inhaled or ingested. Transparent safety data, proper label warnings, and updated job training make a real difference; I’ve seen attitudes shift simply by bringing seasoned safety officers to the shop floor. Some industries explore alternative compounds, but few replacements deliver the same results.
Selenious acid often gets overlooked, but in the right hands, it plays a crucial role—whether that’s creating glass art, strengthening machine parts, supporting nutrition, or advancing scientific knowledge. Respect for its hazards, along with a clear understanding of chemistry, keeps workplaces safe and products reliable.
Is Selenious Acid safe to handle?
What Selenious Acid Brings to the Table
Selenious acid crops up in labs, glass manufacturing, and the metal industry. Its role often revolves around helping create certain types of pigments, glass, or even as a chemical intermediate for producing selenium compounds. The chemical carries strong oxidizing properties and plays a part in critical reactions. I once saw a glass artist handle a batch of pigments formulated with this compound—one whiff of the fumes was enough to leave every bystander alert to its punch. These aren’t ingredients for classroom science experiments.
Understanding Selenious Acid’s Risks
A big challenge with selenious acid shows up when people treat it like everyday chemicals. The compound holds serious risks for the skin, eyes, and lungs. A splash on your hands stings fiercely and can burn. Breathing in vapor can bring on a sore throat, coughing, and more serious lung problems if exposure continues. It’s more toxic by ingestion, attacking internal tissues, and selenium compounds like this one build up in the body. Chronic exposure leaves people open to garlic breath, stomach pain, skin rashes, and even nerve damage.
I remember my first training in a small industrial plant. Our mentor kept repeating stories about workers who cut corners. One turned his gloves inside out because he forgot fresh ones—the guy missed several months of work. Nobody wanted to be the next warning story.
Safety Counts: Gloves, Goggles, and More
Working with selenious acid safely boils down to straight-up preparation. Anyone handling this needs solid barriers—nitrile gloves plus eye shields—since skin and mucous membrane contact spell trouble fast. A staff member in a white coat isn’t automatically safer unless they check their gear for tiny rips and fit. Respirators come out if you can’t guarantee a fume hood. Good ventilation, sealed storage in glass containers, and quick access to running water or eyewash all matter. I never saw experienced operators handle this compound alone; somebody nearby kept a watchful eye, ready to help if something splashed.
Regulatory Oversight and Health Guidance
Rules from OSHA and NIOSH lay down strict exposure limits for selenious acid and related compounds. Facts show that respiratory irritation can kick in at very low concentrations—just a tenth of a milligram per cubic meter in workplace air. Long ago, some regions kept looser rules, but accidents and health tracking changed that. Companies adopt rigorous checklists, workers get regular health checks, and labeling stands out in bold colors. Without that structure, statistics show higher accident rates and more health claims.
A Culture of Caution & Training
Safety culture comes from steady training—nobody learns how to handle risky chemicals from a quick read of the label. Real practice takes time. Every workplace should revisit procedures for storing, transferring, and cleaning up after selenious acid. Spills don’t wait; training should make response automatic, not a panic-driven scramble. I’ve seen a well-drilled team prevent a minor splash from turning into a serious injury, just by knowing exactly what to do without hesitation.
Better Solutions and Practical Steps
Safer alternatives get attention in labs and industry, but so far, selenious acid sticks around when nothing else can do the job. Upgrades in ventilation and equipment have cut accident rates, and some workplaces mix less concentrated solutions—another smart move. Frequent refresher training sticks better than a one-time seminar. Even outside the workplace, proper disposal stops the compound from sneaking into water supplies.
It’s not the kind of acid anybody handles lightly, and nobody who cares about their team’s health should let shortcuts slide. Sharing stories, learning from accidents, and building habits take the fear out of working with selenious acid and replace it with respect.
How should Selenious Acid be stored?
Getting Real About Selenious Acid Hazards
Selenious acid doesn’t belong on a shelf next to everyday chemicals. In my years working alongside industrial chemists and safety professionals, no one overlooked the sharp risks tied to this compound. Even opening a bottle in an unventilated room feels dangerous. Why? It releases toxic fumes and causes nasty burns. Without good storage routines, selenious acid turns from a laboratory staple into a health nightmare. Let’s call things what they are—turning a blind eye in this area leads directly to hospital visits and environmental fines.
The Basic Rules Aren’t Optional
Put selenious acid in a cool, dry spot—never close to heat sources. This isn’t just about following a rulebook, it’s about stopping accidental releases that can poison air and water. I’ve watched colleagues wipe down containers with double gloves and wish everyone took that much care. Place the substance in a tightly sealed glass container, never something reactive like metal. The end result? Fewer leaks and a better shot at keeping fingers and lungs safe.
Lock It Up and Keep It Marked
No teenager or curious staff member should lay hands on selenious acid outside of controlled supervision. I store hazardous compounds like this in locked cabinets that open only with special keys. Maybe it sounds paranoid. But a single splash causes chemical burns or, worse, ends up in the wrong hands entirely. Every bottle begs for a bright label with warnings, date received, and who last checked it out. No one should ever reach for a bottle and wonder what’s really inside.
Why Inventory Tracking Saves Lives
I've seen labs lose track of what sits on their shelves for years. Overdue inventory checks let selenious acid collect dust until it corrodes lids or containers break down. Once, I found a leaking bottle that had rotted through plastic shelving. That memory sticks. So my advice: log every bottle, record what comes in and goes out, and schedule monthly checks. Digital inventory systems cut mistakes and reveal missing stock fast.
Don’t Forget Ventilation and Spills
Storing selenious acid in a well-ventilated area matters as much as anything else. Fumes build up fast and cause breathing problems—nobody wants to be the one who reported to an emergency room because the ventilation system failed or someone skipped routine checks. Keep spill kits right next to storage. I always make sure everyone knows how to neutralize and clean up spills. The kits stay within arm’s reach, never tucked in a closet.
Responsible Disposal Always Matters
Holding on to old selenious acid “in case it’s needed someday” doesn’t pan out. Expired materials raise risks. I arrange routine pickups with hazardous waste contractors who handle this stuff safely. Look for contractors certified by your local environmental safety board—cutting corners turns a lab into a liability.
Better Habits Start With Training
No one can guess their way through chemical storage rules. Regular training sessions make safe habits stick. In workshops I’ve attended, even seasoned chemists admitted forgetting critical details. Written procedures posted next to storage areas help everyone double-check themselves.
The Bottom Line: Don’t Gamble With Safety
Selenious acid asks for respect and careful attention. Safe storage goes beyond checklists—it’s about respect for those sharing the workspace and the environment outside your lab walls. Neglected safety steps don’t just risk fines or broken bottles—they put real lives on the line. That’s something every professional, hobbyist, or anyone handling chemicals should remember.
What are the hazards of Selenious Acid exposure?
Working with a Hazard Few Have Heard Of
Selenious acid rarely grabs headlines, but its dangers deserve attention. Many people working in labs or certain industrial settings might come across this chemical without fully understanding the risks. I remember my early days in a university research lab—a misplaced drop of a corrosive substance taught me how unforgiving chemistry can be. Every bottle on a shelf held more than a mystery—it packed consequences for carelessness, and selenious acid is a prime example.
Damaging to Skin, Eyes, and Lungs
Selenious acid burns. Skin contact starts with irritation and can quickly worsen to blisters, long after the sting fades. I met a colleague who wondered about a patch of angry red skin after accidentally handling residue on glassware. By lunchtime, that patch hurt far worse, and we realized selenious acid had been the culprit. Eye contact takes the danger up a notch—this stuff can literally blind. The fumes may not always announce themselves, but breathing them in irritates the throat, damages the lungs, and triggers a cough that lingers.
Toxic by Ingestion or Inhalation
This chemical isn’t just an irritant. Swallowing selenious acid spells disaster for the gut: nausea, vomiting, diarrhea, and—if the dose runs high enough—life-threatening damage to internal organs. Selenium compounds in general show their toxicity through a narrow exposure window. Selenious acid damages red blood cells, upsets the nervous system, and can lead to death in serious cases. There’s no downplaying the consequences for unprotected exposure.
Chronic Hazards and Long-Term Risk
Selenious acid doesn’t need large amounts to build up problems over time. Long-term exposure can turn fingernails brittle and hair thin, even trigger nerve and kidney problems. Selenium toxicity, or selenosis, reminds me that some hazards in the workplace creep up silently. Studies show workers near selenium compounds can develop garlic breath—strange but telling evidence of a chronic hazard. The World Health Organization lists selenium poisoning as a genuine occupational danger, and selenious acid occupies a top spot among culprits.
Protection and Prevention
Risk isn’t inevitable. I used to think gloves and goggles felt like overkill, until a splash nearly cost me an eye. Now I reach for safety gear before even popping a lid. Laboratories and industries can rely on fume hoods, chemical aprons, and splash-resistant eyewear. Good ventilation cuts down airborne risk. Spill kits and proper storage practices mean fewer nasty surprises and less chance for skin or eye contact. Regular training turns careful habits into second nature—something I learned through hard experience and have watched pay off more than once for others.
Emergency Response Matters
If something bad happens, fast action helps. Rinse skin and eyes for at least fifteen minutes. Move to fresh air right away if breathing issues start. After any accidental exposure, call emergency services and share the details—treatment depends on knowing selenious acid played a role. No substance in the lab or factory should ever be underestimated, but few punish lapses like selenious acid.
What is the concentration of Selenious Acid in this product?
Why Selenious Acid Gets Attention
Selenious acid isn’t a household name, but it matters in labs, chemical plants, and certain specialized industries. You’ll find it in products where selenium’s unique properties play a role—think supplements, glass production, and electronics. But concentration is no small detail here. The stakes get higher with chemicals that interact with human health and the environment. It pays to look closer before anyone pops the cap or pours the bottle.
Unpacking the Label: Where to Look
My early years in the lab taught me to never assume anything from product names or front labels. Manufacturers sometimes display concentration right up front, often citing something like ‘2% solution’ or ‘0.5 mol/L’. But sometimes the answer hides in the fine print—listed among composition statements, safety data sheets, or back-panel details labeled “active ingredients.” If it’s not easily found, I hunt down the product’s Safety Data Sheet, or SDS, which spells out exact percentages or gram-per-liter data.
Why Concentration Matters
Selenious acid isn’t just another chemical. At low concentrations, it helps as a micronutrient source. Doses step higher, and its oxidative power becomes hazardous. As little as 0.1 milligrams per kilogram can shift from nutritional to toxic. Overexposure risks severe health effects, including gastrointestinal and respiratory harm. If a product lists 4% selenious acid, that’s four grams per hundred milliliters—a dose that needs careful handling and accurate measuring.
Random searches online show selenious acid concentrations from below 1% in nutritional uses up to 10% in industrial solutions. Without clear labeling, there’s real risk: Using the wrong strength for what looks like “the same product” means someone could be swapping a safe solution for one that damages equipment, threatens safety, or worse.
Regulatory Standards and Industry Oversight
Agencies like OSHA, REACH, or the FDA put careful rules in place for chemicals such as selenious acid. The FDA caps selenium content strictly in supplements and animal feeds because the gap between “enough” and “too much” stays narrow. That regulatory oversight pushes manufacturers to publish concentration details on packaging and in technical sheets. I’ve seen failed spot inspections where missing or mislabeled concentrations led to recalls or fines—so reputable suppliers rarely skip this step.
Finding Answers and Staying Safe
Over years in science, I grew used to never trusting a package alone. Email the supplier for more information if labels or sheets fail to clarify things. Most responsible vendors provide detailed technical documents within a few hours. For imported chemicals or generics bought online, lack of a reliable SDS stands out as a red flag. I’ve had colleagues run assumed “safe” tests only to discover the solution was several times too strong because the supplier cut corners on documentation.
Better Choices and Solutions
Transparency protects people, equipment, and the environment. The culture in chemical supply circles is shifting—clients expect proper documentation, and suppliers that cut costs by skimping tend to lose repeat business. For those working with selenious acid, double-checking sources, demanding documentation, and choosing suppliers who back up claims with third-party analysis all pay dividends. Lifting the hood on what’s actually in a bottle or barrel protects work from both short-term accidents and long-term harm.
Summary Table: Key Info Points
| Where to Check | What You'll Find |
|---|---|
| Front Label/Panel | Sometimes percent or molarity listed directly |
| Safety Data Sheet (SDS) | Exact concentration, handling, and toxicology data |
| Technical Data Sheet | Breakdown by chemical, batch analysis |
| Direct Supplier Contact | Clarification for ambiguous products |
| Names | |
| Preferred IUPAC name | Dioxoselenic acid |
| Other names |
Selenic acid Dihydroxoselenol Selenious acid |
| Pronunciation | /ˌsɛˈliːni.əs ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 7783-00-8 |
| Beilstein Reference | 4038044 |
| ChEBI | CHEBI:26671 |
| ChEMBL | CHEMBL1233090 |
| ChemSpider | 52921 |
| DrugBank | DB11131 |
| ECHA InfoCard | 100.027.944 |
| EC Number | 231-974-7 |
| Gmelin Reference | 775944 |
| KEGG | C18616 |
| MeSH | D012704 |
| PubChem CID | 24562 |
| RTECS number | VS7175000 |
| UNII | 2U8C4325D0 |
| UN number | UN1871 |
| Properties | |
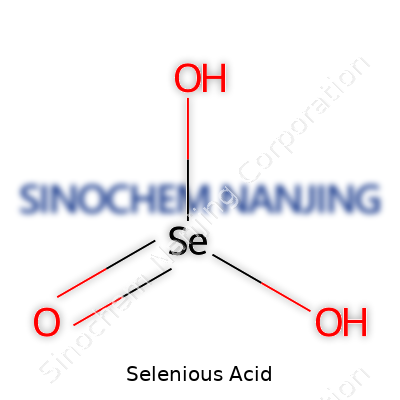
| Chemical formula | H2SeO3 |
| Molar mass | 128.97 g/mol |
| Appearance | Colorless to slightly yellow, clear liquid |
| Odor | Odorless |
| Density | 3.098 g/cm³ |
| Solubility in water | very soluble |
| log P | -1.2 |
| Vapor pressure | 0.03 mmHg (20°C) |
| Acidity (pKa) | 1.92, 7.52 |
| Basicity (pKb) | 1.78 |
| Magnetic susceptibility (χ) | -49.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.509 |
| Viscosity | 15 cP (25°C) |
| Dipole moment | 1.57 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 150.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -540.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -606 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | A12CE02 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes severe skin burns and eye damage; may cause respiratory irritation. |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P260, P264, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P314, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Lethal dose or concentration | LD50 oral rat 7 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 7 mg/kg |
| NIOSH | NIOSH: WB6300000 |
| PEL (Permissible) | 0.2 mg/m3 |
| REL (Recommended) | 0.3 mg Se/mL |
| IDLH (Immediate danger) | 1 mg/m3 |
| Related compounds | |
| Related compounds |
Selenium dioxide Selenic acid Selenate Selenite |

