Selenic Acid: A Deep Dive into an Uncommon Oxoacid
Historical Development
People have studied selenic acid’s story since the first spark of selenium chemistry. Jöns Jacob Berzelius spotted selenium in 1817 while investigating sulfuric acid contaminants, and researchers quickly grew curious about its broader chemical family. Selenic acid itself didn’t take center stage right away, mostly because selenium research lagged behind the wave of sulfur’s popularity in industry. In the fading years of the 1800s, improved methods for oxidizing selenium dioxide nudged selenic acid into scientific catalogues. Selenic acid never drew huge industrial crowds like sulfuric acid or other mineral acids, but as scientists pushed into rare and niche compounds, its profile started nudging higher. Early chemical texts reference labor-intensive preparations, noting its similarity to sulfuric acid while warning about its high toxicity.
Product Overview
Selenic acid, known chemically as H2SeO4, forms as a powerful oxidizing agent in the family of selenium compounds. Chemists think of it as a strong mineral acid, although its demand stays limited due to toxicity and cost. In the laboratory, it lands as a syrupy liquid or in crystalline form, always highly hygroscopic—so it soaks up water from the air rapidly. Producers aim for pure, colorless product, but most lab samples take on a slight pink tinge due to trace selenium dioxide presence. Its oxidizing power beats out even sulfuric acid, and the rare times factories manufacture it intentionally, they usually plan for small-scale, specialized applications.
Physical & Chemical Properties
You’ll spot selenic acid as a colorless, nearly oily liquid at room temperature. Its chemical formula, H2SeO4, gives away its close relationship to sulfuric acid, and their similar behaviors in water highlight that connection. Boiling happens at roughly 140°C for the hydrated form, but pure selenic acid will decompose to selenium dioxide and oxygen before you hit that point. Lab handling requires careful attention: it’s both highly corrosive and extremely hygroscopic, clinging to water vapor in any environment. Mix it with water and the acid dissolves outright, forming a strongly acidic solution. Selenic acid oxidizes everything from manganese(II) salts to hydrochloric acid—sometimes aggressively so, kicking up chlorine gas. In the crystal state, it adopts a unique tetrahedral structure, which makes it useful as an analytic reagent despite its risks.
Technical Specifications & Labeling
Regulations demand precise labeling for any industrial- or laboratory-grade selenic acid. Labels typically reflect concentration, batch number, production date, and corrosive hazard pictograms. Chemical grade information also appears clearly: most samples claim 99% purity or above when used for analytic purposes. Manufacturers list molarity for aqueous solutions, commonly ranging from 0.1 M for educational use to 6.0 M or higher in technical settings. Storage instructions always call out the need for airtight containers, glass, or PTFE, along with secondary containment if possible. Safety data sheets outline potential hazards, required PPE, and emergency response measures, with global harmonized standards (GHS) classifying the acid as highly toxic and environmentally hazardous. Laboratory storage rules treat it like other highly corrosive substances, storing away from organic materials and reducing agents.
Preparation Method
You don’t find selenic acid growing on trees, and you rarely ever see bulk production. Most common preparation starts with oxidizing selenium dioxide (SeO2) using strong oxidants like hydrogen peroxide (H2O2) or chlorine gas in controlled conditions. Some researchers start from elemental selenium and pass it through stages: they oxidize selenium to selenium dioxide, dissolve the dioxide in water to form selenious acid, and then push it further with nitric acid or potassium permanganate. Recovery and purification involve evaporation under reduced pressure, careful crystallization, and thorough removal of volatile byproducts. Process operators need training and protective equipment—hydrogen selenide and selenic acid byproducts cause acute poisoning, so proper ventilation and remote handling serve as non-negotiables.
Chemical Reactions & Modifications
Selenic acid stands out as a strong oxidizer. It can oxidize hydrochloric acid to chlorine gas, reduce to selenous acid under the touch of reducing agents, and even displace fluoride from potassium fluoride samples. Chemists use it to prepare selenate salts by neutralizing with various metal hydroxides or carbonates. Mixing with organic compounds quickly gets dangerous, as it can spark violent oxidation. In redox titrations, selenic acid occasionally replaces sulfuric acid when a stronger agent is needed. It doesn’t just react: it modifies. Labs create derivatives by adding alkali metals to make soluble selenates, or treat it with ammonium salts for precipitation studies. Modifications remain rare outside analytic chemistry, as risks and toxic byproducts usually outweigh most commercial benefits.
Synonyms & Product Names
Selenic acid shows up under the synonyms “selenic(VI) acid” and “hydroselenic acid.” Academic literature leans on the formula H2SeO4, especially in reaction schemes or analytical protocols. Catalogues sometimes use “selenium oxyacid” or “selenium(VI) acid,” though these names rarely stick in everyday lab conversation. “Dihydroxidoselanium” appears in more technical chemical registries. Trade names for commercial grade selenic acid remain rare, as direct sales avoid mass markets; most vendors refer to it by formula or standard IUPAC name.
Safety & Operational Standards
Workplace safety standards rank selenic acid as a highly hazardous substance. Inhalation, ingestion, or contact causes acute and sometimes severe symptoms including burns, respiratory distress, and systemic selenium poisoning. The acid penetrates skin—corrosive burns form rapidly and healing takes weeks. Many countries treat laboratory handling of selenic acid as restricted, with worker training requirements and regulated waste disposal. Laboratories must run ventilation systems to handle vapors and keep selenic acid in tightly sealed glassware away from incompatible chemicals like organics and reducing agents. Spills require evacuation, acid neutralization (using sodium carbonate or similar), and professional cleanup. Transport involves UN shipping codes and hazardous materials markings. In my own experience, gloves, full-face shields, and a working emergency shower count as absolute essentials when handling even milligram quantities. Waste requires labeling for “toxic selenium compounds” and must go to specialized disposal, avoiding any entry into general aqueous waste streams.
Application Area
Selenic acid doesn’t headline any mainstream production process, but holds a place in analytic chemistry and precise oxidation reactions. Researchers use it in trace metal determinations, sometimes in wet digestion protocols where a strong enough oxidizer is needed to clear tough organic matrices. In organic synthesis, it rarely sees action except for specific challenging oxidations, such as turning certain hydrocarbons into carboxylic acids. Mineral analysis sometimes calls for selenic acid’s power, especially where sulfuric acid stalls. Labs occasionally draw on it to prepare selenate salts for agricultural or biochemical studies, but most industry turns to milder, less risky alternatives. Specialized sectors—including some electronics research and certain pigment production—see rare small-scale needs. Use cases get documented more in research journals than in annual industry reports.
Research & Development
Recent years haven’t pushed selenic acid into household or heavy industry use, but R&D teams haven’t stopped exploring it. Analytical chemists keep testing its value for oxidizing resistant compounds and forming selenate standards. Environmental chemists look at its behavior as a possible soil or water contaminant, especially since selenium’s impact on ecosystems grows as mining and electronics waste increase. Pharmacological researchers study its role in selenium metabolism—a tricky topic, since selenium flips from essential micronutrient to dangerous poison depending on the form and dose. Experimental labs examine selenic acid as a model to study high-valence non-metal oxoacids. Many patents mention it in passing, as a reference compound or in procedures aiming for other selenium-based materials.
Toxicity Research
Selenic acid stakes out dangerous territory on toxicity charts. Animal data says LD50s for related compounds fall in the microgram-to-milligram-per-kilogram range, which means even tiny exposures threaten life. Selenic acid’s oxidizing power leads to tissue destruction on contact—skin, eyes, respiratory tract, or digestive tissues show damage after brief exposure. Systemic effects come from selenium absorption, leading to symptoms like garlic breath, gastrointestinal stress, central nervous system disturbances, and chronic selenosis over time. Environmental risks matter, too: selenic acid converts easily to selenates in water, which, depending on concentration, can accumulate in plants and move up food chains. Toxicity studies call out the sharp difference between trace dietary selenium as an essential nutrient and selenic acid as a potent poison. Animal and cell culture models help reveal underlying biochemical mechanisms—including how selenic acid disrupts enzymes and damages DNA—but safe handling trumps any promise of benefit.
Future Prospects
Looking forward, selenic acid’s mass adoption faces big barriers. Its toxicity and environmental risks keep it sidelined compared to safer oxidizing agents. Still, researchers keep their eye on possible uses in advanced oxidation projects, high-end synthesis, and as a tool for systematic studies in analytical chemistry. With rising attention on selenium pollution from industry and electronics, environmental scientists may end up studying selenic acid more closely than most manufacturers ever plan to produce it. Any hope of mainstream adoption would need new breakthroughs in safe containment, use, and disposal—none of which has reached practical stage yet. As science keeps refining methods for tough oxidations or selenate analysis, selenic acid claims its niche as a compound for those who respect its hazards and know their way around high-risk chemistry.
What is Selenic Acid used for?
What Makes Selenic Acid So Useful?
Selenic acid, rarely found on your medicine cabinet or supermarket shelf, wields serious clout in the world of chemical synthesis and industry. With a structure similar to sulfuric acid but a selenium core, it doesn’t show up in front-page headlines, yet it carries a big role in the lab. From my own chemistry background, I remember opening selenic acid only under a hood, gloves tight, because this isn’t something handled lightly. Its oxidizing character grabs attention: in reactions where even sulfuric acid throws up its hands, selenic acid pushes forward.
Oxidizing Power in the Lab
I’ve seen selenic acid step up to the plate when chemists need to turn tough, reluctant compounds into something new. It can turn elemental selenium into selenium dioxide, or break down routine substances most other acids can’t touch. A paper from the Journal of the American Chemical Society shows chemists using it to produce peroxides and transform manganese compounds. Its action doesn’t just help academics – the results feed into processes making semiconductors, glass, and specialty metals.
Industrial Impact and Rare Usage
On the industrial front, selenic acid surprisingly pops up in the thin world of selenium refining. Selenium, used for colored glass or certain electronics, comes out of ore in a mix of states. To get it pure enough for high-tech uses, selenic acid cleans things up. The process burns out the impurities and leaves behind the selenium wanted for solar panels, rectifiers, and pigments. Companies rely on this step, and while it sounds niche, the solar industry’s growth means more demand. The Environmental Protection Agency in the US tracks these uses closely, given the toxicity risks both for workers and the environment.
Biological Interest and Research Frontiers
Researchers also chase selenic acid as a way to study how selenium affects the body and the environment. Unlike the trace amount in multivitamins, free selenic acid hits with much more strength and toxicity. Some studies look at trace exposure’s link to antioxidant enzymes, while others use selenic acid as a tool to understand how selenium cycles through plants and soil. At this level, the acid serves as more than just a bottle on a chemistry shelf—it becomes a lens into understanding environmental selenium and its risks.
Health and Safety Concerns
No one calls selenic acid safe. Its reactivity means a splash will burn skin and tissue, while breathing fumes damages lungs. Industrial and lab safety guidelines, like those from OSHA and NIOSH, keep risks in check with strict exposure limits, fume hoods, and sealed containers. I’ve seen what happens when these steps get skipped—a minor spill leads to a major clean-up, and the stuff can corrode metal shelving in days. Proper labeling, good ventilation, and training aren’t just rules—they protect real people from potent harm.
How to Manage the Risks
So, handling selenic acid really demands a culture of safety and strong oversight. Process engineering can limit exposure by automating steps and isolating the acid from hands-on work. Waste treatment and recycling methods recover valuable selenium and neutralize harmful leftovers. Implementing regular training and audits, along with using robust personal protective equipment, makes sure mistakes stay rare. These practices echo what the best-run chemical plants and research labs already do, with good outcomes for both people and the planet.
Looking Forward
As tech and green energy expand, selenic acid’s relevance will only increase. Improvements in containment, detoxifying waste, and worker safety can let industry tap selenium’s power without the downsides. Responsible handling, backed by solid science and clear regulation, allows us to keep its benefits flowing without regrets down the road.
Is Selenic Acid dangerous or toxic?
What Selenic Acid Really Brings to the Table
I’ve worked in labs where safety signs mean business, and selenic acid always drew a cautious respect. This isn’t some basic bottle in the chemical store. Selenic acid, H2SeO4, stands out because it isn’t just corrosive—it's one of the strongest oxidizers you'd ever meet in a beaker. Picture a clear, syrupy liquid that can go toe-to-toe with sulfuric acid for strength, only it brings selenium to the mix. And selenium, while essential at tiny levels, gets nasty quick when you get too much. So, people who underestimate selenic acid often end up learning the hard way about chemical burns and toxic fumes.
Health Risks That Deserve Attention
Most people hear about selenium as a dietary trace mineral. In this form, it’s harmless, even helpful. Selenic acid lives in another league. This chemical will burn straight through your skin, and vapors can damage lungs in minutes. Overdosing on selenium compounds leads to something called selenosis—hair loss, fatigue, bad breath that smells like garlic and, in serious cases, nerve damage or death. Selenic acid gets into the body easily through contact, inhalation, or swallowing, so even small spills turn into emergencies fast.
Damage Doesn’t Only Happen in the Lab
Selenic acid isn’t common in households or small businesses. Its dangerous properties make it a specialty product for industry and research. The places that use it—specialty glassmaking, rare chemical synthesis, metallurgy—carry strict rules for handling. Spill response usually means chemical showers, full hazmat suits, emergency shutdowns, and a call to environmental cleanup teams if even a small amount goes astray.
The Broader Environmental Impact
As soon as selenic acid escapes into the water supply or soil, it stops being just a workplace concern. Selenium compounds interact with organisms at all levels. Fish, livestock, and crops take up selenium easily, but too much leads to mass die-offs. The infamous 1980s Kesterson Reservoir disaster in California comes to mind—selenium runoff from irrigation changed a thriving ecosystem into a wasteland. Selenic acid carries that kind of risk. There isn’t a cleanup shortcut once it saturates an environment. Even the byproducts of a selenic acid spill stay dangerous for years.
Building a Responsible Culture in Chemical Handling
After years working around volatile compounds, I vouch for training as the top line of defense. Anyone near selenic acid needs face shields, chemical gloves, and airtight procedures. Safety data sheets must stay within arm’s reach. Fume hoods and sealed containers—never open-air storage or handling. You can’t treat this chemical casually and expect to avoid disaster. At the corporate level, reviews and upgrades for safe storage pay for themselves. If a company can afford selenic acid, it can afford real protective barriers.
Better Solutions Mean Fewer Disasters
People in science lean on technology for safer labs. Automation keeps human error out of the equation during risky steps. Sensors that detect rising selenium vapors or chemical leaks act as early warnings, giving people a chance to act before disaster strikes. Green chemistry chases less aggressive alternatives, searching for ways to avoid selenic acid entirely in routine processes. Reducing both exposure and transportation limits the odds of accidents, and often cuts costs along the way.
Selenic acid is as dangerous as reputation suggests and deserves respect every step of the way. Real safety doesn’t happen by accident—it starts with open eyes, the right equipment, and a promise not to cut corners.
How should Selenic Acid be stored?
Looking Straight at the Hazards
Selenic acid hits harder than some of the more common acids folks might keep in a lab or industrial setting. This one’s got teeth. Corrosive as anything and a powerful oxidizer, it chews right through skin, metal, and concrete if given the chance. Many people underestimate just how reactive it is, especially compared to sulfuric or nitric acid. The stories of leaky containers and chemical burns keep piling up for a reason.
Don’t Trust Anything But Glass or PTFE
Some folks remember the first time they saw a brand-new lab bottle eaten right through by selenic acid. Polyethylene, even so-called “chemical-resistant” labware, turns brittle. Metals? They do even worse—iron, steel, copper, all start to corrode or pit within days of exposure. Borosilicate glass or PTFE, on the other hand, stands up to the challenge. Anyone with years in the chemical industry has learned to keep selenic acid as far from metal shelves, drains, and fittings as possible.
Check the Cap, Double Check the Label
No one benefits from a mix-up. Walking into a storage room and grabbing the wrong acid turns a long day into a dangerous one. Clear, tough, chemical-resistant labels refuse to peel or blur after a spill. Heat-sealed or etched labels on glass give peace of mind. Keeping selenic acid in its original packaging is a habit that protects people far down the line—from delivery drivers to waste handlers. In my own work with unfamiliar stockrooms, I’ve found that the most organized labs see the fewest accidents—it’s not a coincidence.
Cool, Dry, Well-Ventilated— No Shortcuts
I’ve worked in buildings where the heat rises, air gets thick, and storage cabinets sweat. Storing selenic acid under those conditions raises risks for everyone in the building. Humid air lets the stuff suck up water, making it even easier to spill or splash. High heat speeds up decomposition; that means the bottle might start building pressure. Proper chemical storage cabinets—dedicated to oxidizers—may look expensive on the invoice, but they’re cheaper than medical bills and lost work days.
Fire Risks Need Real Action
Piling other chemicals near selenic acid, especially organics or anything flammable, can turn a single accident into a full-out lab fire. An old colleague once shared a story about a minor spill reacting with sawdust from a shipping crate. Ten minutes later, the room filled with smoke. Keeping incompatible materials apart works better than any fire extinguisher. Large-scale users should always review inventory for surprise risks—paper labels, wood shelving, cleaning rags, and solvents in the same room spell trouble.
Training Isn’t Optional—Knowledge Saves Skin
Every staff member, from interns to custodians, should know what to do in case of a selenic acid emergency. Real emergency showers, gloves rated for strong oxidizers, face shields, and fast access to SDS information save lives and eyesight. I remember labs where regular drills made people comfortable, not complacent. Even experienced chemists can freeze up under pressure, so rehearsing those steps matters.
Takeaway—Responsibility Starts With Storage
Anyone handling selenic acid owes it to colleagues, lab neighbors, and even folks outside the building to respect the risks. Good habits, right gear, and proper training don’t just keep the acid contained—they protect every part of the workflow, from storage to disposal. The extra attention feels like work today but turns into peace of mind tomorrow.
What are the safety precautions when handling Selenic Acid?
Working With Selenic Acid: Real Risks, Real Responsibility
Selenic acid isn’t a chemical most people come across every day, but those who work with it in labs or industry know it means serious business. It’s up there among the more aggressive acids—much more reactive and more toxic than sulfuric or hydrochloric. Its strong oxidizing power doesn’t just break down organic matter; it can put your health on the line with just a splash or a whiff.
I’ve seen the aftermath of a careless mistake with chemicals like these. A glove that wasn’t up to scratch, a fume hood left off for “just a minute”—small choices with big consequences. Selenic acid attacks skin, eyes, and lungs without pause. Even a diluted solution can burn through layers of tissue, and inhaling the fumes leads to lung damage you might not fix. Ignoring safety isn’t just risky; it could wreck a life.
Protecting Yourself Starts With the Right Gear
No one handling selenic acid should rely on thin latex gloves or street clothes. Disposable nitrile or neoprene gloves, face shields, and lab coats are a must. Good footwear keeps accidents from soaking feet. The acid will chew through cheap gear quickly, so investing in actual chemical-resistant protection pays off. Handling concentrated forms? Double up on gloves and sleeve cuffs.
Eye protection goes beyond basic goggles. Splash-proof face shields give extra defense—you only get one set of eyes. Emergency eyewash stations should never be across the room. Nearest exit, every time. I always make sure everyone with me knows where to run in an emergency. Familiarity cuts response time and saves damage when a moment goes sideways.
Ventilation: Keeping the Bad Stuff Out of Your System
Selenic acid fumes pack a punch. Standard ventilation won’t cut it. Using a real fume hood takes most of the threat out of the air and keeps it away from your lungs. At home or work, keeping the workspace contained, sealed, and ventilated is the baseline. On top of that, anything accidental gets dealt with straight away.
Even after hours of exposure, the body holds onto selenium. It can build up and poison you slowly. Headaches, nausea, a metallic taste—these are not normal. Any hint of a problem should mean dropping work and getting medical help. Turning a blind eye risks chronic health issues that nobody needs.
Cleaning Up and Disposal: No Shortcuts Allowed
Spills happen. I’ve cleaned up enough to know that paper towels and hope won’t cut it. Acid-resistant spill kits need to be in arm’s reach before you even touch a bottle. Absorbents help, but getting the residue up, neutralizing it, and bagging everything for hazardous waste disposal is non-negotiable. Pouring leftovers down the drain means putting everyone downstream at risk. Selenic acid doesn’t just disappear; it lingers, ready to harm wildlife or water supplies.
Anyone new to handling toxic oxidizers should learn the ropes from someone experienced. Training means more than a checklist. Understanding why each step matters changes how seriously it gets taken. I’ve found that clear communication and a culture of speaking up stop accidents before they start. People protect each other when they know mistakes aren’t just possible, but inevitable unless respect for the risks stays high.
Building a Culture of Caution
Complacency shortens careers and lives. Following the rules around selenic acid isn’t overkill; it’s practical self-preservation. It doesn’t matter if someone’s a student, seasoned chemist, or working in industry for decades. Respecting the power of chemicals like selenic acid protects health, protects peers, and keeps the lab up and running. Mistakes often teach hard lessons—sometimes permanent ones. Shaping habits that never skip steps is the real win in a world with selenic acid.
Where can I purchase Selenic Acid?
Selenic Acid: Not Your Average Chemical
Most folks searching for selenic acid, known for its strong oxidizing properties, probably don’t run a local hardware store. This stuff isn’t something that sits in a dusty bottle behind the counter; it requires special handling, and you won’t run across it on Amazon or at a garden center.
Strict Controls for Good Reason
Selenic acid stands out because even a small mistake during handling can cause harm. Think chemical burns, toxic fumes, and consequences for both people and the environment. Regulators, seeing the danger, set up tough controls. Chemical supply companies—like Sigma-Aldrich, Alfa Aesar, and Fisher Scientific—strictly sell selenic acid only to validated professionals. Regular individuals get denied, and for good reason.
Laws in many countries have teeth. In the US, the Environmental Protection Agency and Occupational Safety and Health Administration demand proof of safe storage, safety training, and a legitimate business or research purpose. Europe, Canada, Australia—all draw pretty much the same red lines. Online orders require registration, company background checks, and documented intent. This isn’t Big Brother gone wild, this is common sense in action.
Why It’s So Rare in the Open Market
There’s barely any room for negotiation on these rules. People ask, “Can I just buy it off eBay?” Some sites advertise “laboratory reagents,” but legitimate sellers still demand proof—a university address, business verification, even a purchase order. Anything less is a huge red flag. Risk goes both ways: sellers don’t want lawsuits or criminal charges, customers don’t want to risk health or land themselves on a government watchlist.
Alternatives and Safer Routes
Not all applications need such a dangerous tool. Depending on your need, safer selenium compounds might handle the job. Sodium selenate or selenious acid pop up more often in laboratories for teaching or less risky experiments. If it’s about scientific research, reach out through an established institution, work under supervision, and respect the protocols.
I once worked next door to a school chemistry lab. Even with basic acids, everybody treated safety as law, not suggestion. Selenic acid demands even more respect. Even skilled chemists don’t handle it casually, always suiting up and double-checking everything.
The Bottom Line
Anyone looking for selenic acid needs to ask why. If it’s for private tinkering, step back—there are real dangers and legal troubles waiting. If you’re part of a legit research institution or business, talk with established chemical suppliers. Document everything, stay up-to-date on safety regulations, and talk to safety officers before you think about placing an order.
Chemicals like selenic acid remind us that not every reagent belongs on a public shelf. They serve a purpose for science and industry, but that comes with real obligations—accountability, training, and respect for safety above all else.
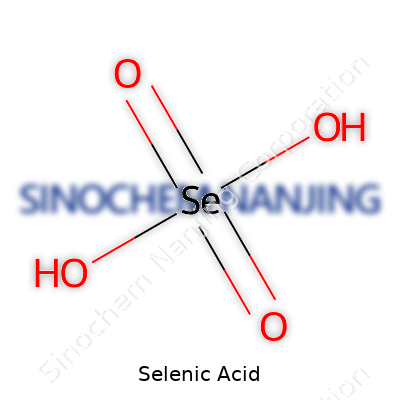
| Names | |
| Preferred IUPAC name | seleniumic acid |
| Other names |
Selenic(VI) acid Selenium(VI) oxide dihydrate |
| Pronunciation | /ˌsɛˈliːnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 7783-08-6 |
| 3D model (JSmol) | `JSmol.loadInline("data/mol:Selenium(=O)(=O)(O)O");` |
| Beilstein Reference | 1201865 |
| ChEBI | CHEBI:27156 |
| ChEMBL | CHEMBL1233486 |
| ChemSpider | 20843935 |
| DrugBank | DB11135 |
| ECHA InfoCard | 100.029.165 |
| EC Number | 231-974-7 |
| Gmelin Reference | Gmelin 1321 |
| KEGG | C02762 |
| MeSH | D013074 |
| PubChem CID | 24616 |
| RTECS number | VS8575000 |
| UNII | 7J8E17EJ8R |
| UN number | UN1818 |
| Properties | |
| Chemical formula | H2SeO4 |
| Molar mass | 143.07 g/mol |
| Appearance | Colorless, oily liquid |
| Odor | odorless |
| Density | 2.036 g/cm³ |
| Solubility in water | Miscible |
| log P | -4.48 |
| Vapor pressure | 0.07 mmHg (20 °C) |
| Acidity (pKa) | -4.28 |
| Basicity (pKb) | -15.0 |
| Magnetic susceptibility (χ) | -46.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.54 |
| Viscosity | 14 cP (20 °C) |
| Dipole moment | 2.18 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 157.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −844.5 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -713 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | A12CE02 |
| Hazards | |
| Main hazards | Corrosive, causes severe burns, toxic if swallowed, inhaled, or in contact with skin, strong oxidizer, may cause fire or explosion. |
| GHS labelling | GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H300 + H310 + H330: Fatal if swallowed, in contact with skin or if inhaled. H314: Causes severe skin burns and eye damage. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | H260, H314, P210, P223, P231, P280, P303+P361+P353, P305+P351+P338, P310, P370+P378, P390, P403+P233 |
| NFPA 704 (fire diamond) | 4-0-2-OX |
| Lethal dose or concentration | Lethal dose or concentration: "LD50 oral rat: 85 mg/kg |
| LD50 (median dose) | LD50 (median dose): 5.17 mg/kg (rat, oral) |
| NIOSH | SEF |
| PEL (Permissible) | 0.2 mg(Se)/m3 |
| REL (Recommended) | 0.2 mg |
| IDLH (Immediate danger) | 1 mg/m3 |
| Related compounds | |
| Related compounds |
Selenium trioxide Selenous acid Sulfuric acid Telluric acid |

