Sec-Butyl Propionate: Tracing a Modern Industrial Chemical
Historical Development of Sec-Butyl Propionate
The story of sec-butyl propionate is less about headline-grabbing moments and more about steady scientific progress. In the years when chemists first started to explore the universe of esters, it wasn’t obvious which molecules would show real staying power. Propionate esters, with their blend of fruity odors and solvent power, attracted interest for everything from perfumery to paint. Sec-butyl propionate emerged in this crowd thanks to its particular mix of volatility, solvency and stability. Early work likely started in research labs where chemists mixed butanol isomers with propionic acid, searching for compounds that could handle resin chemistry or boost performance in coatings. Over decades, the focus shifted from bench-top curiosities to industrial scale. Sec-butyl propionate’s rise mirrored changing attitudes toward workplace safety, environmental regulation and a need for more adaptable chemicals in manufacturing. That long memory survives not just in dusty journals, but in the choices formulators make today when they reach for this clear, flammable liquid.
Product Overview: Uses in the Real World
Sec-butyl propionate has carved out a niche in manufacturing, painting, printing, and cleaning industries. Anyone who’s spent time in a coating facility or print shop has met this solvent, even if they didn’t memorize its name. This liquid makes it easier to dissolve varnishes and paints, thin inks and resins, and clean up equipment. Labs choose sec-butyl propionate because it combines decent evaporation rates with a lower odor than many cousins. In daily work, that matters—not just for production speed, but for worker comfort and air quality. Formulators know sec-butyl propionate can blend well with nitrocellulose, acrylics, and other tough polymers, helping increase gloss in finishes or flexibility in films. Its role as a solvent backbone reaches across applications: cleaning, degreasing, removing printing inks, and catalyzing smooth finishes in automotive topcoats.
Physical and Chemical Properties: What Sets It Apart
With a clear appearance and a faint, pleasant odor, sec-butyl propionate fits into the suite of medium-boiling esters valued by coatings chemists. Its molecular weight of 130.19, flash point near 32°C and moderate evaporation rate give it some safety advantages over more volatile solvents. Water solubility stays low, helping it pair up with oil-based systems. The liquid generally feels light and spreads quickly without leaving residues or deposits. Low reactivity under normal conditions means sec-butyl propionate holds up well in storage, as long as tanks stay dry and cool. It resists breaking down under sunlight and, compared to much older solvents, poses less risk of contributing to photo-oxidant smog—though it’s by no means a green product. Consistent performance in resin modification, ink application and pigment dispersal—these features explain its broad acceptance in industries aiming to balance safety, speed, and finished product quality.
Technical Specifications and Labeling Reality
Every chemical gets boxed in by rules and specs, and sec-butyl propionate is no exception. Industrial suppliers rely on standardized purity benchmarks, usually in the range of 99% or greater, to guarantee reliable results in sensitive formulations. Labels cover boiling range, density, refractive index, evaporation rate and residue analysis. Transport and storage get regulated too, since this is a flammable liquid. Packaging usually means well-sealed drums or totes, built to stay leak-free as they move across borders. In my experience, operations managers and safety officers spend just as much time scanning those labels for hazard pictograms as they do for technical details—it’s all about reducing surprises in storerooms, on the loading dock, and in environmental audits.
Preparation Method: Chemistry in Practice
Production lines for sec-butyl propionate run on a simple esterification reaction. Mix a batch of sec-butanol with propionic acid, introduce an acid catalyst, and apply heat. The system kicks out sec-butyl propionate and water, demanding a water separator to keep the process moving forward. Efficient facilities rely on continuous processes for steady output, using distillation to strip off the ester and recycle unreacted starting materials. Down the line, purification steps make sure the product meets tough purity standards. Industrial chemistry often runs up against problems with waste management and emissions here, so operators look for ways to minimize acid fumes and water treatment loads, aiming for both regulatory compliance and smooth operation.
Chemical Reactions and Modifications: What Chemists Do With Sec-Butyl Propionate
Once isolated, sec-butyl propionate behaves like many esters—it resists breaking apart in neutral or mildly basic solutions, but hydrolyzes under strong acid or base. This stability supports its use in harsh paint-stripping blends and complex topcoats. Plants can modify the molecule further, using hydrogenation or other reactions to tweak performance. Saponification brings it back to propionate salts and sec-butanol, which recyclers eye for solvent recovery. Its limited reactivity keeps it attractive for those seeking reliable, repeatable behavior in blends, especially compared to lower molecular weight esters that require more handling caution due to volatility or rapid breakdown. Rarely, research teams use sec-butyl propionate as a stepping stone for building more elaborate esters, though for most real-world tasks its value lies in stability and predictability.
Synonyms and Product Names: Navigating the Terminology
Ask around factories or chemical distributors and you’ll hear a laundry list of names for this single molecule. Some stick to the formal registry: sec-butyl propionate, 1-methylpropyl propanoate, or 2-butyl propionate. Elsewhere, “SBP” pops up on order sheets. Some product lines brand it for use in coatings or inks, but for the end-user the important thing is spotting the correct chemical identity and CAS number on shipping papers. This helps prevent mix-ups with similar-sounding esters or isomers, especially in global operations that trade with partners across continents. For plant personnel, these synonyms can be a source of frustration or humor, depending on the day's level of confusion.
Safety and Operational Standards
Sec-butyl propionate carries clear-cut risks: flammable in both liquid and vapor forms, capable of causing irritation on contact, and known for harmful effects if inhaled in large concentrations. I’ve seen facilities build storage areas with solid grounding and ventilation, never taking shortcuts when working with ignition sources. Spill protocols require absorbent materials rated for organic solvents, not just any mop and bucket. Workers don gloves, goggles, and flame-resistant clothing out of habit, not just regulation. For operators, health and environmental exposure limits shape daily routines, from shifting ventilation patterns to using closed transfer systems. Fire marshals and safety committees watch inventory levels, knowing confined spaces or hot work sites can turn a small leak into a serious incident. In shipping and handling, all it takes is one failure to remind a team why these protocols matter.
Application Areas: Where the Chemical Goes
Sec-butyl propionate finds fans in industries that live or die by solvent quality. In coatings, paint, printing inks, and adhesives, it solves two big challenges: keeping mixtures workable and drying at just the right speed. Paint manufacturers gain from its ability to dissolve resins easily, deliver smooth flow during spray applications and reduce the “blushing” problem in humid conditions. Printers add it for faster, cleaner runs, especially in high-productivity operations that switch colors or jobs all day. Some cleaning and degreasing formulas make use of its balanced solvency, especially for stubborn grease or hardened residues. I’ve seen formulators pick sec-butyl propionate not as a one-stop fix, but as a team player among other esters, glycol ethers, or aromatic solvents, each with their own strengths. Some niche markets, such as electronics cleaning and specialty coatings, have also explored its use, though regulatory pressures often nudge users to look for lower-impact alternatives.
Research and Development: New Frontiers and Ongoing Questions
R&D teams continue to ask tough questions about sec-butyl propionate’s limits and potential. Environmental chemists target lower-impact solvent systems, weighing sec-butyl propionate against other esters and bio-based solvents for tasks like dissolving resins or delivering coatings. Lab researchers build up databases of evaporation rates, odor thresholds and reactivity profiles, aiming to optimize formulations for safety and performance. Some work goes into streamlining the esterification route, with greener catalysts or closed-loop recovery of byproducts, to meet sustainability goals. In performance testing, trials investigate how sec-butyl propionate interacts with new polymers or cross-linkers, looking for improvements in gloss, flexibility or durability. Others hunt for blends that keep the benefits of sec-butyl propionate but reduce VOC content, responding to shifting air-quality rules. Real breakthroughs tend to happen incrementally—so most innovation unfolds over years of quiet lab work, rather than with much fanfare.
Toxicity Research: Understanding Health and Environmental Impact
While sec-butyl propionate is less notorious than some older solvents, its safety record still owes plenty to careful study and regulation. Animal testing and exposure modeling suggest irritant properties, especially for eyes and the respiratory tract. At higher doses, central nervous system effects become possible, which hasn’t gone unnoticed by industrial hygienists. Regulators in North America, Europe and Asia set workplace exposure limits that companies must follow, balancing production needs with worker health. Some environmental persistence questions linger, such as the impact of accidental release into waterways, where breakdown products may interact with aquatic life. So far, research hasn’t flagged sec-butyl propionate for extreme acute toxicity or chronic harm in low-exposure settings, but ongoing surveillance continues. Companies invest in closed system handling, spill management and air scrubbing, not just because of regulations, but because nobody wants to see a chemical accident headline trace back to their shop floor.
Future Prospects: What Lies Ahead for Sec-Butyl Propionate
The future for sec-butyl propionate rides on the evolving balance between performance, cost and regulation. Solvent formulators keep looking for blends that keep work sites safer and deliver top-notch finishes while cutting emissions. As governments tighten rules on VOCs, pressure grows to adjust formulations, seek biobased sources or completely re-engineer classic paints and inks. Sec-butyl propionate may stay in the toolkit for specialized high-performance systems where reliability trumps speed or green credentials, but big-volume users will keep a close eye on regulatory trends and alternatives like esters from renewable feedstocks. From my perspective, this chemical tells the story of modern industry: progress measured in thicker safety binders, cleaner plant air, fewer waste drums and smarter chemistry—always one eye on what works today, and another on what’s coming next.
What is Sec-Butyl Propionate used for?
Understanding Sec-Butyl Propionate
Sec-butyl propionate sounds technical, but its role lands pretty close to home for folks who care about reliable products and safe workplaces. You see it listed as a solvent in the coatings, paints, and printing ink you might find in any well-stocked hardware store or print shop. Its chemical structure makes it a favorite for getting the right mix in products that need quality and performance, not just cost savings.
Why Industries Rely on It
Walk into a modern car factory or a commercial printing operation. Quality finishes don’t just happen by luck. The solvents in the coatings drive how evenly the paint spreads, how quickly it dries, and whether it holds up to years of wear. Sec-butyl propionate makes a real difference here. Its evaporation rate sits in a sweet spot—not too fast, not painfully slow—which means paints and coatings can be applied smoothly, level out just right, and leave a finish you actually want to see.
In the printing business, clear, crisp images matter. Smudges or streaks mean wasted materials and lost money. The right solvent blend helps inks dry at the right rate, so pages don’t stick together and graphics stay sharp. Sec-butyl propionate helps lower viscosity in these inks, making them work well in high-speed printers without clogging things up. Years ago, printers might have relied on older, more hazardous chemicals. Switching to something with a lower odor and toxicity profile has improved conditions on shop floors and made meeting safety guidelines less of a headache.
Health and Safety
No two solvents are created equal when it comes to health risks. Some are harsh, leading to headaches, skin irritation, or even long-term health issues if the workplace gets careless. Sec-butyl propionate doesn’t carry an “all clear” label, but compared to tougher solvents like xylene or toluene, it creates fewer air quality issues and handles more safely under routine protocols. Ventilation and gloves matter, but with this solvent, the risk of chronic exposure drops, so companies see fewer workplace complaints and incidents.
Addressing Environmental Concerns
Solvents, by nature, raise environmental flags. Companies and regulators rightfully worry about air emissions, groundwater contamination, and hazardous waste. Sec-butyl propionate breaks down more predictably in the environment compared with some chlorinated cousins. That doesn’t mean dumping it is okay, but disposal and treatment processes tend to be more straightforward. Facilities can manage waste more efficiently, which saves money and headache down the line.
Finding Better Solutions
Folks look for alternatives all the time. The push for water-based products has picked up steam, and, in some cases, waterborne coatings give the same results with less solvent content. Still, some applications need that extra level of performance, especially in automotive and heavy industry. Using sec-butyl propionate, with proper engineering controls, makes sense as a middle ground—good balance between performance, safety, and cost until cleaner technologies catch up on all fronts.
Experience on shop floors and in plant management shows progress doesn’t mean flipping a switch. It’s practical—better training, better equipment, and gradually switching to safer chemicals where it counts. In my own hands-on work with facility upgrades, we've cut down on spills and air complaints, simply by swapping out harsher solvents and tightening process controls. Sec-butyl propionate sits in that realistic zone: safer than old-school chemicals, pretty versatile, and better for workers who want to finish a shift without headaches.
Manufacturers who value both the bottom line and worker health have real incentive to pick their solvents carefully. Sec-butyl propionate offers dependability and a strong safety margin compared to what came before. The push for safer plants and responsible products isn't slowing down, and making smarter solvent choices stands at the center of that push.
What are the safety precautions when handling Sec-Butyl Propionate?
Why Sec-Butyl Propionate Matters in the Workplace
Sec-Butyl Propionate pops up in paint shops, surface coatings, and industrial cleaning. It’s got the punch to lift grease and dissolve tough spots, making it useful on the line. That same power brings its fair share of risks if care takes a backseat. Most folks overlook how strong solvents pack invisible dangers. Rushing through a job, skipping a glove, or not checking the label—these shortcuts leave burns, headaches, and long-term trouble.
Trouble Signs with Sec-Butyl Propionate
Sec-Butyl Propionate evaporates fast, filling up an enclosed space with fumes. A half hour with poor ventilation, and lungs start to burn or the room spins. Even brief skin contact dries skin and can trigger allergic reactions. Spills on unprotected skin tingle, then sting. Rub your eye after touching it, and redness follows. Long exposure? Inhalation brings on nausea, coughing, and headaches—the kind that lingers for hours. Working with hot plates or welding torches? Don’t forget: a small spark hits a spill and fires start.
Personal Experience with Solvents
Working in a repair workshop years ago, I learned to respect chemicals. My boss hammered in glove use, face shields, and always propped open doors. One day, a co-worker ignored warnings and worked in a half-ventilated room with a small bottle of solvent. Halfway through, he felt dizzy, left work early, and stayed home sick. After that, nobody skipped a respirator. Safety talk felt annoying at first, but watching someone pay the price sharpened everyone’s focus.
Common-Sense Steps for Safer Handling
Ventilation stands as the best friend in any chemistry job. Open windows, use fans, or install local exhaust systems. A chemical-resistant glove set works far better than cheap latex pairs that melt or tear. Nitrile gloves hold up, and long sleeves keep splashes away from skin. Splash-proof goggles or a full face shield cuts down on fear of accidental sprays or unexpected mist.
Workplaces run best when spills and tools stay tidy. A labeled, sealed container goes a long way—no open cans or mystery bottles near the bench. Use trays or spill mats to catch drips. Everyone should know where to find eyewash stations and showers. Run practice drills. In an emergency, panic drops when muscle memory kicks in.
Static and sparks don’t mix with this solvent. Avoid open flames, don’t smoke, and check wiring in workspaces. Clean up rags and waste in metal bins with lids. A cluttered workspace encourages mistakes.
Information Makes a Difference
Most safety gaps come from missing information or skipping the basics. Read the safety data sheet—each step, from storage to disposal, matters. Proper labeling sets expectations and reduces guesswork. Waste should never pour down the drain. Approved hazardous waste containers keep pollutants out of water systems.
People sometimes act like safety rules belong to someone else, but the after-effects land on the worker, the team, and the environment. Shortcuts rarely pay off. A few extra steps, the right equipment, and steady awareness shape a healthier, safer workspace, every single time.
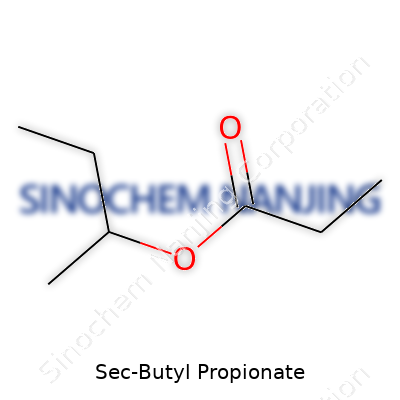
What is the chemical formula of Sec-Butyl Propionate?
What Is Sec-Butyl Propionate?
Sec-butyl propionate doesn’t exactly sound like a household name, but it plays a quiet role in many industries. Its roots run deep in the world of solvents, especially for coatings and industrial cleaning. The chemical formula grabs the attention of both students in labs and professionals in factories: C7H14O2. You might catch a whiff of it in certain paints and cleaners. What matters isn’t just the science behind it—it’s the way this chemical fits into supply chains and safety protocols.
Breaking Down the Formula
Plugging the name into chemical knowledge, sec-butyl propionate forms by connecting the propionic acid molecule (C3H6O2) and sec-butyl alcohol (C4H10O). The reaction shapes an ester, explaining the –O2 at the end of the formula. The “sec” term means the butyl part hooks up at the second carbon. For folks who like drawing structures, the formula C7H14O2 gets mapped out with each carbon, hydrogen, and oxygen accounted for. Those small differences in structure make a big impact on how this solvent behaves.
Why This Compound Shows Up
Sec-butyl propionate often shows up where a balance between performance and safety is needed. It dissolves coatings thoroughly without adding the sharp hazards of more potent solvents. Many coatings, inks, and resin blends rely on this chemical to dry smoothly. My own days in chemical storage made me closely aware of just how essential clear labeling and formula knowledge become. There’s no room for error when dealing with substances like this.
Health and Environmental Focus
Exposure to solvents creates real concern about workplace health. Sec-butyl propionate isn’t the most toxic, but repeated inhalation or spills can lead to headaches and irritation. Safety data sheets stress ventilation and skin protection—and from experience, gloves and lab ventilation aren’t negotiable. The chemical catches fire easily, so smart storage becomes a routine, not a suggestion. Fire marshals and safety inspectors often want to know the specific formula before approving a site for operation.
Many companies look for less volatile, more biodegradable versions of classic solvents. Some teams keep a close eye on emissions and groundwater impact, given the organic nature of compounds like sec-butyl propionate. Keeping the formula in mind helps chemists tweak manufacturing processes to cut down on waste. Industry groups often encourage collecting and reusing solvent wastes to limit fresh use and lower environmental load.
Unearthing Better Practices
Switching to safer alternatives can’t always happen overnight. CRC Handbook of Chemistry and Physics lists physical properties of many compounds, including sec-butyl propionate, so manufacturers can make informed choices. Regulatory groups and scientific panels recommend keeping exposure below certain thresholds, emphasizing real-world monitoring. Solvent substitution remains a slow process, but public awareness keeps the search going. Sharing solid facts, not just chemical jargon, will keep workers, neighbors, and consumers on the same page.
It’s easy to overlook the importance of learning the correct formula for chemicals like sec-butyl propionate. In my experience, knowing the difference between similar compounds prevents mix-ups, and following safety rules keeps damage at bay. Chemistry isn’t just about formulas—it’s about the responsibility that comes with every bottle or barrel.
How should Sec-Butyl Propionate be stored?
Understanding the Risks of Sec-Butyl Propionate
Sec-Butyl Propionate helps create coatings, inks, and adhesives—useful stuff, but you might not give its storage a second thought until you realize how volatile it can be. This liquid evaporates easily, and a simple spark can turn a careless moment into a disaster. The fumes catch fire fast, and over years, industry reports keep showing fires and spills that trace back to sloppy storage. For folks managing chemicals, a safe spot to stash this solvent matters as much as getting the work done right.
Why Ventilation and Temperature Control Count
Heat turns Sec-Butyl Propionate from a liquid into vapor, and those vapors just love to find an ignition source. So, a cool, well-aired room does more than keep the workplace comfortable—it protects lives. If storage sits next to sunlight or near machinery that’s always running hot, the risks go up. The Environmental Protection Agency points out that proper ventilation and cooler temperatures help reduce the chances of explosive vapors gathering. These steps aren’t a luxury—they guard workers and property.
Fire Safety Calls for Respect, Not Luck
Forget wishful thinking. Keeping a drum of Sec-Butyl Propionate next to an exit or break room just because it’s convenient is asking for trouble. I’ve watched staff in smaller shops stack paint thinners in busy hallways, and realize one static spark could change everything in a second. Fire codes exist for a reason. National Fire Protection Association (NFPA) guidelines underline clear rules—approved metal drums, flame arresters, and no open flames nearby. If someone skips these rules, it’s not just a citation from the fire marshal; it could be a call to the fire department after the fact.
Labeling and Training Aren’t Optional
On a recent walk-through at a facility, I spotted containers marked only with faded masking tape. Honest mistakes happen, but mixing up chemicals exposes everyone to new hazards no one expected. Labels matter. Regulations like OSHA’s Hazard Communication Standard demand clear, up-to-date signs—ingredients, hazards, handling instructions. Clear training means staff know what they’re handling and what to do in an emergency. Sometimes it feels like overkill, until one day a confusing label stops a bad mix-up just in time. Data backs this up: better training and signage cut down on workplace accidents.
Spill Response and Secondary Containment
Most people think of “leaks” as slow drips, but I remember a case with an old valve that let out a flood in five minutes. Secondary containment—like spill pallets or bunded areas—catches everything before it spreads. The EPA and state agencies list secondary containment as a top safeguard, because once spilled solvent hits the ground or drains, cleanup costs skyrocket, and local waterways can take a hit. Absorbents, spill kits, and emergency protocols need to stay close to storage, so teams react right away without scrambling.
Simple Habits Make the Difference
Storing a chemical safely isn’t a big mystery. Reliable habits—checking seals, updating inventory, and checking room temperature and airflow—stop dangerous shortcuts. I’ve seen places skip these steps and end up dealing with avoidable drama. Sec-Butyl Propionate will always have its hazards, but consistent care and honest respect for the real risks—following proven rules and using basic common sense—help everyone get home safe at the end of the day.
Is Sec-Butyl Propionate hazardous to health or the environment?
Understanding The Chemical and Its Everyday Presence
Sec-butyl propionate finds a place in countless products from industrial cleaners to paints. It brings its unique solvent power to manufacturing floors, workshops, and even art studios. Its presence often goes unnoticed, hiding behind product labels and chemical names most people never stop to read.
Health Risks That Don’t Stay at Work
The moment I picked up a can of industrial primer in my first warehouse job, I felt that sharp, almost sweet chemical scent. I remember the headaches after a day around open containers. That was sec-butyl propionate and its cousins at work. The facts back up the experience. Short exposure to high levels can cause eye, skin, and respiratory tract irritation. Longer exposure or higher concentrations may lead to more serious symptoms: dizziness, drowsiness, or even loss of consciousness in poorly ventilated rooms. Years in the paint shop taught me that gloves and goggles aren’t suggestions—they’re shields. The U.S. National Institute for Occupational Safety and Health lists this chemical as potentially harmful if inhaled or absorbed over time, so the risk isn’t theory.
Some folks feel the effects faster because of asthma or sensitivity to solvents. Expecting mothers or younger workers stand at a higher risk, since developing bodies react more to many chemicals. These aren’t just warnings for those mixing paints on a factory floor. Anyone spraying paint, stripping metal, or cleaning with industrial solvents could get exposed unless they stay careful.
Environmental Footprint Stretches Beyond the Factory Gate
Improper handling and disposal mean sec-butyl propionate ends up in soil or waterways. Its vapors travel, especially from large-scale industrial use. Once in the air or water, it doesn’t sit still: this solvent evaporates quickly and may harm aquatic life if enough reaches rivers or streams. Fish and insects in lab tests stopped moving or died when concentrations crept up. Larger spills exhaust bacteria that break down waste, leaving damage that lingers for months or longer. Waters near industrial zones have shown fluctuations in species numbers after routine dumping of solvents like this one.
Factoring in the Broader Impact
No workplace operates in a vacuum. Workers bring chemical dust home on shoes and clothes, passing tiny amounts to their families or pets. Hobbyists in garages risk the same dangers as the pro. Neighborhoods near plants get the legacy: trace amounts in the air, slightly off tap water, reports of dying weeds at the edge of property lines. Although most products using this solvent follow strict legal thresholds, these measures only offset risk—not eliminate it. In some countries without solid regulations, the burden grows heavier for residents and ecosystems alike.
Reducing Harm Through Sensible Steps
I’ve seen small shifts make a lasting impact. Opening windows in a garage, using a respirator, or switching to less hazardous alternatives can drive exposure way down. Companies with active ventilation and waste management actually save money, avoid health lawsuits, and protect workers. Regulations push manufacturers to use safer substitutes, recycle solvents, and limit air releases, but enforcement often follows public pressure and education.
Anyone using sec-butyl propionate should stay aware of its risks, follow safety labels closely, and advocate for safer chemical options when possible. Real safety isn’t just rules on paper—it’s the everyday actions people choose, from engineers down to DIYers in suburban garages.
| Names | |
| Preferred IUPAC name | Propyl(propan-2-yl)acetate |
| Other names |
1-Methylpropyl propionate Propanoic acid, sec-butyl ester SBP sec-Butyl propanoate |
| Pronunciation | /ˌsɛkˈbjuːtɪl prəˈpɒneɪt/ |
| Identifiers | |
| CAS Number | 105-64-6 |
| Beilstein Reference | 626720 |
| ChEBI | CHEBI:89939 |
| ChEMBL | CHEMBL517872 |
| ChemSpider | 185513 |
| DrugBank | DB13845 |
| ECHA InfoCard | 44a17106-ce12-4917-9414-222d79f5cfe6 |
| EC Number | 607-043-00-7 |
| Gmelin Reference | 7935 |
| KEGG | C19808 |
| MeSH | D017644 |
| PubChem CID | 12312 |
| RTECS number | UJ1050000 |
| UNII | 99QXI6I8X6 |
| UN number | UN2338 |
| Properties | |
| Chemical formula | C7H14O2 |
| Molar mass | 130.18 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Sweet |
| Density | 0.866 g/cm3 |
| Solubility in water | 0.95 g/100 mL (20 °C) |
| log P | 1.94 |
| Vapor pressure | 2.2 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 25 |
| Basicity (pKb) | pKb ≈ 15.97 |
| Magnetic susceptibility (χ) | -7.78×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.401 |
| Viscosity | 1.1 mPa·s (25°C) |
| Dipole moment | 3.07 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 383.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -471.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4690.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P340, P312, P305+P351+P338, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-2-1 |
| Flash point | 43 °C (Closed cup) |
| Autoignition temperature | 420°C |
| Explosive limits | Explosive limits: 1.1% - 10.1% |
| Lethal dose or concentration | LD50 oral rat 13,400 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 9,400 mg/kg |
| NIOSH | WZ4725000 |
| PEL (Permissible) | PEL: 100 ppm (OSHA) |
| REL (Recommended) | 24 Months |
| IDLH (Immediate danger) | 800 ppm |
| Related compounds | |
| Related compounds |
Butyl acetate Isobutyl propionate Methyl propionate Ethyl propionate n-Propyl propionate |

