Sec-Butyl Chloroformate: A Closer Look at Yesterday, Today, and Tomorrow
Historical Development
Sec-butyl chloroformate didn’t appear overnight. The history of this chemical traces back to the growing toolkit of organic chemists in the mid-20th century, when demand surged for reagents that enabled more efficient production of pharmaceuticals and agrochemicals. Before this, early esterification and peptide-synthesis techniques felt like navigating with half a map. Chloroformates like this one offered a critical shortcut, letting scientists build complex molecules without as many side steps. It ushered in new classes of drugs and specialty chemicals investors still rely on today. As newer methods for carbamate and ester synthesis developed, its popularity grew among labs looking for reliable chlorinating agents that managed reactivity without causing runaway side reactions. In my view, anyone who’s spent time in synthetic labs knows there are always a few reagents you remember for their reliability—sec-butyl chloroformate has definitely earned its place among them.
Product Overview and Synonyms
In the warehouse and on order sheets, you’ll find sec-butyl chloroformate listed under more than one name. Sometimes, it might turn up as “secondary butyl chloroformate” or “2-butyl chloroformate.” Each name refers to the same core structure: a chloroformate group connected to a secondary butyl ring, creating a versatile reagent for introducing carbamates and other protective groups. It's not a household item and doesn't show up on shopping lists. You mostly see it in the hands of chemists and production engineers. This stuff works mainly as a building block, not as a finished product. You don’t pour it on crops or swallow it for headaches, but plenty of medicines and agrochemical actives pass through it on their way to final formulation.
Physical & Chemical Properties
Anybody who’s ever handled sec-butyl chloroformate in the lab won’t forget its sharp, biting odor—like a cross between acetic acid and pool chemicals. It’s a clear, colorless liquid most days, with a moderate boiling point that doesn’t cause havoc under normal reaction conditions. It hydrolyzes when exposed to water, releasing carbon dioxide and hydrochloric acid, which means you have to keep it sealed tight and away from humidity. In practical terms, using it demands well-ventilated spaces, protective gloves, and eye shields. Reactivity runs high, especially with nucleophiles, and its decomposing byproducts are as unforgiving on lungs and skin as anything in the chemical cupboard.
Technical Specifications & Labeling
Bottles of sec-butyl chloroformate arrive with hazard symbols no one should ignore. The labels speak clearly: corrosive, harmful if inhaled, dangerous to aquatic life. Regulatory agencies insist on standardized pictograms for a reason, not only for safety but because it forces everyone to treat this chemical with respect. Purity typically pushes 98% or higher for research or fine chemical use, with limited allowable impurities to avoid unwanted reactions. Storage instructions usually involve a cool, dry place—never near open flames or strong bases. Even with so many safety reminders, the physical properties draw attention first, making it clear this isn’t a backyard experiment candidate.
Preparation Method
Industrial and laboratory preparation still sticks mostly to a tried-and-true route: reacting secondary butanol with phosgene, itself a chemical with a checkered past. An acid scavenger, often pyridine or a mild base, sweeps up the hydrogen chloride generated, helping to push the conversion along. Anyone who’s ever run a phosgene line knows the risks involved—strictly controlled ventilation and monitoring, continual checks for leaks, and clear procedures for emergencies. Handling phosgene demands respect for its toxicity and a commitment to responsible waste disposal. Despite constant searching for “greener” chlorinating agents, this method stands, largely because it works efficiently without producing a mass of byproducts. There’s a push in research circles to swap out phosgene for less notorious carbonyl sources, but commercial uptake moves slowly given how ingrained the old routes have become.
Chemical Reactions & Modifications
One of the main reasons this compound remains so valued in research and manufacturing comes from its knack for transferring carbamate and carbonate groups. Organic chemists use it to convert amines into carbamates with precision, introducing protective groups that survive other harsh steps and can be removed on command. The reaction isn’t just useful for drug design—peptide syntheses, agricultural chemicals, and even specialty polymers benefit from the fine-tuned control sec-butyl chloroformate offers. It reacts briskly with nucleophiles, forming bonds cleanly without excessive rearrangement or side production. Some labs experiment with alternatives like methyl or ethyl variants, but sec-butyl holds its ground by balancing reactivity and manageability. Tinkering with the structure—adding extra branches or swapping the butyl ring for something bulkier—alters both reactivity and solubility, leaving the original a favorite for general use.
Safety & Operational Standards
There’s no room for half-measures in safety with sec-butyl chloroformate. I’ve worked in labs where shortcuts ended in trips to the emergency shower. Prolonged exposure to even small amounts can burn skin and corrode airways—short bursts of inhalation leave throats raw for hours. Spills demand immediate cleanup, usually with specialized absorbents kept within arm’s reach. Staff run drills for handling chemical releases, reinforcing procedures like neutralization with sodium bicarbonate and evacuating if needed. Proper disposal means more than pouring leftovers down the drain; regulatory bodies require neutralization steps and collection by licensed chemical waste handlers. Direct dumping not only violates laws, but puts groundwater and downstream workers at risk. In highly-regulated industries, periodic audits keep everyone on their toes, lowering the risk of accidents that ripple outward beyond the lab.
Application Area
Beyond the academic settings, sec-butyl chloroformate punches above its weight in the pharmaceutical world. Drug developers turn to it during early-stage synthesis to install groups that shield more sensitive portions of a molecule, letting them pursue complex modifications step by step. Peptide chemists depend on it for crafting short-chain blocks that eventually fold into proteins or bioactive compounds. Agricultural companies run it through similar processes when building herbicides and pesticides that call for targeted activity and environmental resilience. Paints and coatings sometimes involve related chloroformates for crosslinking purposes, adding strength or resistance to environmental stress. Its utility persists because it saves time, cuts down on side products, and gives chemists a well-characterized toolkit for delicate transformations.
Research & Development
Research teams continue to probe both the uses and liabilities of this chemical. Every year, new papers wrestle with the balancing act: maximizing safe reactivity without overburdening waste streams with hazardous leftovers. Teams focus on swapping out phosgene where possible, but usually run into issues with efficiency or increased cost. Others dive into designing derivatives with improved safety profiles or targeted reactivity. Environmental chemists aim for “benign by design” reagents that won’t persist in soil or water, an effort that might reshape how this chemical fits into tomorrow’s synthesis plans. Years spent in academic settings taught me that grants flow most readily toward projects that combine synthetic innovation with clear public health benefits. Addressing regulatory pressure, especially in Europe and North America, drives innovation as chemists seek to balance industry demand with stricter environmental controls.
Toxicity Research
Health studies make clear that sec-butyl chloroformate poses significant hazards for workers and the environment. Animal studies and accident reports consistently point to respiratory irritation, skin burns, and risks of longer-term damage to internal organs. Some downstream hydrolysis products, especially hydrochloric acid, create dangerous conditions even after disposal in controlled facilities. Regulatory bodies catalog these findings and push producers to implement safer handling, personal protective equipment, and strict disposal protocols. Continuous monitoring of exposure levels—combined with toxicity testing in model systems—helps identify cumulative risks and shape better standards. Experience in various labs taught me that the best approach isn’t waiting for someone to get hurt; real safety means running constant safety seminars and never looking for shortcuts.
Future Prospects
The road ahead for sec-butyl chloroformate depends on technical innovation and changing regulations. Environmental concerns spur searches for alternatives that reduce the burden of hazardous waste, while industry needs continue to call for reliable, scalable reagents. Green chemistry principles—focusing on less toxic reagents, less hazardous waste, and renewable feedstocks—remain a guiding star for new research initiatives. Incremental improvements in process design, storage, and handling might extend the life of sec-butyl chloroformate in production pipelines. On the horizon, up-and-coming researchers keep an eye out for breakthroughs that minimize toxic exposure without stripping away the reliability and effectiveness that have kept it in the chemist’s toolkit for decades. The story of this compound reflects bigger questions facing the industry—how to preserve chemical innovation while stepping more lightly on worker health and the environment.
What is Sec-Butyl Chloroformate used for?
What Sets Sec-Butyl Chloroformate Apart
Sec-butyl chloroformate grabs attention in the world of chemical synthesis. Chemists and engineers turn to it as a strong reagent that helps make carbamates and other protected molecules. This compound steps forward in places where you need to modify and protect certain groups in a chemical reaction. By forming a barrier, it keeps reactive molecules from falling apart or reacting too soon.
Why Industries Reach for Sec-Butyl Chloroformate
Pharmaceutical labs dig deep into their toolkit and often land on sec-butyl chloroformate. This isn’t just about convenience; it’s about control. Picture a chemist shaping a complicated drug. One misstep, and months of work can vanish. Protection and deprotection steps using compounds like sec-butyl chloroformate let researchers tweak pieces of a molecule without ruining the rest.
Manufacturers producing active pharmaceutical ingredients keep tight schedules. With sec-butyl chloroformate, they get a smooth way to introduce carbamates, which helps with selective modifications. Those changes can boost bioavailability, stability, or make the drug easier to swallow and store. As someone who once worked alongside a process chemist on an anti-viral drug, I saw the gap that careful reagent choice can bridge—turning what looks like luck into predictable science.
Beyond Medicine: Other Uses in the Lab
Paints and coatings producers need chemical intermediates that behave predictably. They reach for sec-butyl chloroformate when making specialty polymers and agrochemicals. These applications don’t make the headlines, but they affect shelves in every hardware store. Synthetic chemists use this compound in peptide coupling, creating building blocks for vaccines or high-end research tools. It isn’t the only option, but it offers a balance between power and predictability.
Hard Numbers and Facts
The global chloroformate market continues to climb, topping $500 million in annual revenue by the early 2020s. Pharmaceutical demand leads the charge, since new drugs in development need more tools. Carbamates, made from sec-butyl chloroformate, form a part of countless medicines, because they help fine-tune how drugs work in the body. Research from J. Med. Chem. shows these protected molecules slow down breakdown, letting drugs last longer and act more precisely. Agrochemical companies push for new and safer insecticides, using sec-butyl chloroformate as an intermediate step, according to reports from the European Chemicals Agency.
Safety and Quality: No Corners to Cut
Sec-butyl chloroformate brings real hazards along with its benefits. Its fumes cause breathing issues and burns if handled without tight controls. I remember training sessions where the safety officer repeated: gloves, goggles, good ventilation, no excuses. Labs need robust safety protocols and emergency response plans to cut risks down to size. Production must take purity seriously to avoid nasty side reactions or dangerous byproducts.
Looking Ahead: What Can Change?
Regulatory pressure mounts each year as agencies watch over chemical storage, transport, and waste disposal. The industry invests in greener alternatives, aiming to reduce toxic byproducts and lower carbon footprints. Ongoing research tries to expand the list of reagents with similar benefits but fewer hazards. Until then, chemical makers and researchers have to rely on training, experience, and solid engineering to keep people and the environment out of harm’s way.
Sec-butyl chloroformate deserves respect, both for what it can do and for the discipline it demands from those who use it.
What are the handling and storage requirements for Sec-Butyl Chloroformate?
Understanding the Basics
Sec-butyl chloroformate never gets invited to easy-going workspaces. It’s one of those chemicals people watch closely, because getting careless often results in more trouble than the job’s worth. This colorless liquid brings a sharp smell and a reputation for causing headaches—literally and figuratively. Most who have worked around it know even a drop on the skin sends you running for water. Breathing in vapors or accidental splashes cause damage that no one forgets in a hurry. So, with real health consequences, not only are safety sheets required by law, but the team needs to remember them by heart.
Best Practices for Storage
An ordinary shelf in a storeroom can’t offer enough protection. This chemical should get its own well-ventilated spot. Warm air quickens decomposition and raises the chances bottles build up pressure. At room temperature, it slowly lets off corrosive fumes. Setting bottles away from heat sources, open flames, and direct sunlight makes a big impact on both safety and shelf life. Humidity in the air? Skip it. Exposure to water, even a drop, sparks a dangerous reaction and releases choking gases. Sealed, moisture-free storage is not optional—it’s life-saving. Most experienced chemists will double-check caps and stoppers whenever they walk past. Sturdy, clearly labeled containers with a corrosion-resistant lining help avoid mix-ups or leaks.
Proper Handling Methods
Working with sec-butyl chloroformate isn’t just about keeping your skin dry—it means always using chemical-resistant gloves, eye protection, and a high-quality lab coat. Fume hoods let you keep vapors from building up around your face. Small spills demand fast, careful action. Dry sand or an appropriate absorbent takes care of puddles, but starting cleanup without proper gear often leads to chemical injuries and ruined clothes. Keeping a spill kit on hand is as basic as taping shut the lid on a bottle. Remember the strong odor—if you smell anything sharp, ventilation needs an upgrade. Closing bottles tight after use is an easy habit, but it’s one that protects the whole crew.
Health Risks and Emergency Response
Most of the serious injuries tied to this chemical happen because people put off safety gear or let vigilance slide as pressure piles up. Breathing the vapors irritates airways quickly; exposure can leave headaches and rashes for days. Rapid decontamination limits damage. Running contaminated skin under cold water for fifteen minutes helps, but medical attention always comes next. Eye contact sometimes happens fast, so eyewash stations should stay open and clear at all times. Spilling even small amounts on an unprotected workbench will damage surfaces and could spark longer cleanups if ignored. Safety showers, emergency numbers, and clear signage around the workspace lower the odds of panic when trouble hits.
Sharper Oversight and Training
Supervisors who skip regular safety refreshers usually see shortcuts multiply. Practices learned at one job might not fit another. Ongoing training makes old hands and new hires more aware of changing protocols and chemical incompatibility. Making safety checklists routine keeps minds focused and storage tight. Building a work culture where people feel comfortable flagging risks means fewer accidents, more trust, and smoother shifts. Investing in thorough record-keeping, regular stock rotations, and simple maintenance pays off, day after day. In the end, those details turn a risky task into a manageable one.
What safety precautions should be taken when working with Sec-Butyl Chloroformate?
Understanding the Risk—And Respecting It
Sec-Butyl Chloroformate doesn’t rank high on folks’ lists of household chemicals. Still, it pops up in labs and chemical plants, and everyone working around it should know what makes it tick. If even a small amount gets out, eyes and lungs figure it out fast. I’ve seen chemists scramble when just a drop hit the air. It’ll sting eyes, set off coughing, and trigger headaches. That earned it a rep for causing more than minor trouble. The risk isn’t imaginary—Serious chemical burns, long-term lung sensitivity, and severe irritation are real possibilities. Hazard sheets say so for a reason.
Gear Up, Don’t Gamble
Pretending a lab coat counts as real protection just puts people in harm’s way. Sec-Butyl Chloroformate gets through standard gloves and clothing. Experience shows that nitrile gloves—checked for pinholes and changed regularly—work a lot better than latex. Chemical splash goggles go a long way towards saving vision. Face shields back up goggles, especially with splash-prone work. Cotton lab coats protect skin, but sometimes only a chemical-resistant apron will do. I’ve worked with old-timers who didn’t bother, then learned the hard way after reactions splashed so fast that they barely got to the shower.
Ventilation Matters—Hoods Always Win
This compound evaporates quickly. It gives off toxic fumes that attack eyes and lungs instantly. An open bench offers no defense. Fume hoods—or glove boxes if you’re dealing with big amounts—keep those toxic clouds away from faces. Getting lazy about the hood invites trouble. I wouldn’t open a bottle or pour it outside a working hood. I’ve watched someone do it, and the coughing fit that followed kept them away from direct work for weeks. Room fans, windows, and air conditioning don’t count for much—only real ventilation designed for chemicals does.
Know Your Spills and Plan For Them
Even careful folks drop flasks or knock over vials sometimes. Cleaning a spill with paper towels or sponges just spreads around the danger. Absorbent pads labeled for chemical spills do a much better job, and keeping a spill kit handy nearby is basic good sense. Vinegar or other weak acids won’t neutralize this stuff safely. Only use what standard operating procedures say works. I always point out the eye wash and shower these days before anyone starts work—losing seconds in an emergency costs people dearly.
Storage Smarts Save Lives
Sec-Butyl Chloroformate reacts with water and many common solvents, releasing gases that will choke a room. Dry, cool storage in tightly-sealed, compatible bottles beats improvising every time. Forgetting to label containers or leaving residues behind risks more than contamination. One slip-up with water or alcohol sitting nearby can spark fumes that fill a whole lab. That’s not an imagined story—I remember an emergency where half a work floor evacuated because of an unsealed vial left next to a leaking tap overnight.
Waste Doesn’t Just Disappear
Pouring leftovers into a sink or tossing wipes in the trash ignores the rules—and it’s how people force janitors or wastewater workers into danger. Organic waste cans labeled for hazardous chemicals give everyone a fighting chance. Disposal companies handle it from there. If anyone cuts corners or tries shortcuts, the risk spreads beyond the lab. Workers down the line deserve protection too.
Training and Reminders—Nobody Should Go It Alone
Reading instructions once isn’t enough. Training sticks best with hands-on practice and scenario drills. One thing my mentors always did—run through spill and exposure plans before new projects. Every person in the lab needs to feel confident about what to do. No one gets all the answers just from a PowerPoint. Everyday reminders—signs near hoods, labels, quick review sessions—help busy teams stay sharp. A culture where people look out for each other, asking “Hey, is that the right glove?”—that’s what keeps folks safe around chemicals like this one.
What is the chemical formula and molecular weight of Sec-Butyl Chloroformate?
Chemical Formula and Molecular Weight
Anyone who steps into an organic chemistry lab or flips through a chemical catalog probably sees the name Sec-butyl chloroformate. This compound’s chemical formula is C5H9ClO2. Scales, calculators, and a dash of arithmetic give a molecular weight of about 136.58 g/mol. At first glance, these two numbers might look like trivia, like details reserved for data sheets. In the real world, these figures tell us a lot about what this chemical can do, how it behaves, and what researchers need to watch out for.
Why Formula and Weight Matter
Small details, such as a single atom switch, take a molecule from gentle to hazardous. That’s not hype or scare tactics. Anyone who’s worked with compounds like sec-butyl chloroformate knows that formula and weight are directly tied to how a chemical reacts and breaks down. Every molecular gram makes a difference during reactions. A slight miscalculation leads to runaway effects, wasted funds, or worse, unsafe conditions. So those numbers go on labeling, material safety data sheets, and get embedded into research habits. Even when synthesizing in small amounts, weighing that 136.58 grams per mole with precision means fewer headaches—all the way from lab bench to factory floor.
Where Sec-Butyl Chloroformate Ends Up
This compound isn’t just another item in the supply cabinet. Chloroformates play crucial roles as reagents, especially in pharmaceutical labs and material science settings. Sec-butyl chloroformate activates carboxylic acids, helping chemists introduce new functional groups. So if someone is working on drug design, polymers, or fine chemicals, this formula shows up time and again. Its reactivity, shaped by those five carbons, nine hydrogens, one chlorine, and two oxygens, makes it useful—but also demands respect. Chemists stay alert to spills or vapor because chloroformates can release gases that bite lungs and eyes.
Getting Safety Right with the Right Data
My time in the lab showed me how tempting shortcuts can be. People forget that the properties of sec-butyl chloroformate notice even a slip of the hand or a fumble of the flask. With its molecular weight, if you work with a hundred-milligram scale and misread by just a milligram, reactions go off track. Waste piles up. Downtime stretches out. Some will say proper ventilation, personal protective gear, and double-checking numbers only matter in big operations. That view overlooks how often individuals pay the price for chemistry done in a rush. It’s much easier and safer to pause, trust the facts the formula gives, and handle every gram mindfully.
Smarter Solutions Going Forward
Sec-butyl chloroformate won’t disappear from the scene. Its uses are too important for that. Instead, smart labs build better workflows by training their teams to understand not just what the compound does, but how its formula and weight shape every task. Modern sourcing apps now chart each reagent from arrival to storage. More labs use digital balances that sync to quality control logs, minimizing manual error. As green chemistry grows, alternative reagents and safer analogs may edge into specific applications, but the backbone of safe, accurate reactions remains: know what’s in the flask, how much, and what it’s capable of. Molecular details prevent accidents and protect lives.
How should Sec-Butyl Chloroformate be disposed of?
What Makes Sec-Butyl Chloroformate Challenging?
Sec-Butyl chloroformate sits on the list of chemicals nobody wants loose in the environment. I’ve watched researchers handle it with care, gloves always on, fume hoods running to catch every fume. This compound reacts strongly with water, letting off hydrochloric acid – a gas that bites your nose and stings your throat. Pouring it down the drain looks fast but spells trouble for pipes, people, and whatever downstream water creatures come across it.
Chemicals like this have a way of showing up where they shouldn’t. In one lab I worked, someone poured solvent waste into a regular bottle. The fumes pushed the cap off overnight. By morning, the tangy, throat-burning smell hit everyone walking through the door. Since then, the lesson stuck: taking proper disposal seriously dodges unpleasant stories.
Safe Disposal Isn’t a DIY Project
People sometimes believe diluted chemicals pose less risk, but sec-butyl chloroformate stays hazardous in small amounts. In my experience, a little complacency invites disaster. Instead, safety standards grow from painful lessons learned by others. The Environmental Protection Agency and Occupational Safety and Health Administration recommend treating this compound as hazardous waste. Fact is, there’s no cheat code here.
The correct move—every time—calls for a chemical waste specialist. Labs collect sec-butyl chloroformate in tightly sealed glass bottles, label everything by full chemical name, and keep incompatible substances apart. Trained professionals manage neutralization and destruction, using methods that trap fumes, cool runaway reactions, and break down the dangerous ingredients in controlled steps.
Incinerators that handle chemical waste operate under strict regulations. The process destroys organic compounds above 1,000°C, reducing the odds that any part of the molecule survives. Hydrochloric acid off-gassing gets scrubbed by neutralizing agents, stopping acid rain before it forms. These protocols exist not out of paranoia, but from seeing what happens when shortcuts lead to fire, toxic exposure, or environmental fines.
What’s at Stake if We Skip Steps?
Disposing of sec-butyl chloroformate in unauthorized ways puts both human health and nature at risk. Hydrochloric acid attacks the lungs and eyes, while runoff can damage soil and water systems. Few want to fish or swim in water tainted with industrial waste. I once helped clean up a minor spill—just a small amount spread over a workbench, but the cleanup crew suited up and vented the entire room…just for a splash.
Ignoring regulations can trigger huge fines. Research shows the EPA issued millions in penalties against companies that mishandled hazardous chemicals. Besides fines, cleanup costs run sky high once a spill hits soil or groundwater. Responsible action beats damage control every time.
Solutions for Safer Labs and Cleaner Communities
Clear rules, hands-on training, and regular safety audits build a culture where proper disposal comes naturally. Reputable chemical waste contractors destroy hazardous substances safely and document the process, closing the loop. Anyone managing sec-butyl chloroformate should review safety data sheets, update spill kits, and make reporting easy.
Green chemistry, where possible, offers safer alternatives. Some research groups swap out sec-butyl chloroformate for milder reagents, lowering risk and shrinking waste streams. While not every substitute delivers the same results, the trend grows as environmental pressures mount.
Looking back at all the accidents avoided by following proper disposal channels, it’s easy to see why it matters. It’s not about red tape – it’s about keeping people and waterways safe for years to come.
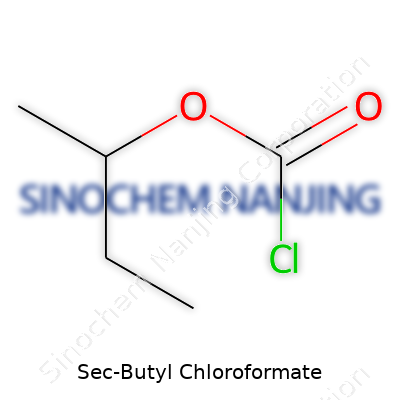
| Names | |
| Preferred IUPAC name | 2-Chlorocarbonylbutane |
| Other names |
SBCF sec-Butyl chlorocarbonate 1-Methylpropyl chloroformate Chloroformic acid sec-butyl ester Carbonochloridic acid, 1-methylpropyl ester |
| Pronunciation | /sek-BYOO-til klohr-oh-FOR-mayt/ |
| Identifiers | |
| CAS Number | 1673-89-8 |
| Beilstein Reference | 1738736 |
| ChEBI | CHEBI:132962 |
| ChEMBL | CHEMBL429613 |
| ChemSpider | 141607 |
| DrugBank | DB14442 |
| ECHA InfoCard | 100.007.942 |
| EC Number | 205-623-5 |
| Gmelin Reference | 9039 |
| KEGG | C19561 |
| MeSH | D017209 |
| PubChem CID | 12301 |
| RTECS number | CN9650000 |
| UNII | VL22E489B2 |
| UN number | UN2742 |
| Properties | |
| Chemical formula | C5H9ClO2 |
| Molar mass | 150.59 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | pungent |
| Density | 1.015 g/mL at 25 °C |
| Solubility in water | React slowly |
| log P | 1.93 |
| Vapor pressure | 10 mmHg (20°C) |
| Acidity (pKa) | 1.41 |
| Basicity (pKb) | Basicity (pKb): 12.85 |
| Magnetic susceptibility (χ) | -7.29 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.406 |
| Viscosity | 1.229 mPa·s (20 °C) |
| Dipole moment | 2.13 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 365.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -454.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -715.6 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V3B0 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H314, H331, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P273, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P307+P311, P312, P314, P337+P313, P363, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-2-W |
| Flash point | 54°F |
| Autoignition temperature | 510°C (950°F) |
| Lethal dose or concentration | LD50 oral rat 1,520 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 1,600 mg/kg |
| NIOSH | CD8225000 |
| PEL (Permissible) | PEL: 0.1 ppm (0.5 mg/m3) as ceiling |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
Methyl chloroformate Ethyl chloroformate Isopropyl chloroformate Isobutyl chloroformate |

