Sec-Amylamine: Deep Dive Into Its Journey and Role in Chemistry
Historical Development of Sec-Amylamine
Chemists stumbled upon amines in the 19th century, connecting natural alkaloids with basic nitrogen compounds. As the industry explored new organic bases, the spotlight gradually fell on Sec-Amylamine, a five-carbon secondary amine. By the 1920s, researchers had mapped out straightforward ways to produce secondary amines from alcohols and ammonia sources. Growing demand for specialized organic intermediates led chemical manufacturers to revisit these early discoveries. Sec-Amylamine's straightforward synthesis, low molecular weight, and predictable behavior bridged the gap between laboratory research and factory floors. Its rise tracks the shift from bulk petrochemicals to targeted building blocks that suit modern pharmaceuticals, agrochemicals, and specialty chemicals.
Product Overview
Sec-Amylamine, classified among secondary aliphatic amines, carries the formula C5H13N. It appears as a colorless to pale yellow liquid at room temperature, exuding a characteristic fishy odor typical of organic amines. Producers offer it in various grades, tuned for laboratory or industry needs. High-purity versions support stringent pharmaceutical synthesis, while standard varieties find homes in broader sectors, such as coatings, surfactants, and water treatment. Laboratories gravitate toward its predictable reactivity, and commercial outfits use its structure as a skeleton to hang specialty chemical functions.
Physical & Chemical Properties
Sec-Amylamine boasts a boiling point hovering near 97°C and carries a density just below water, at about 0.75 g/mL. It mixes well with most organic solvents and remains moderately soluble in water. The nitrogen atom, surrounded by alkyl groups, provides modest basicity; pKa values hover around 10.7. This boosts its ability to scavenge protons or form salts with strong acids. Storage conditions—cool, ventilated, and away from oxidizing agents—help maintain stability. The sharp vapor, low flash point, and reactivity with acids and halides keep safety managers vigilant, especially in large-scale operations.
Technical Specifications & Labeling
Regulators demand clear, accurate Sec-Amylamine labels. Purity levels often exceed 98%. Labels indicate molecular weight (87.16 g/mol), potential contaminants, and batch numbers. Manufacturers must highlight hazards such as skin sensitivity and flammability. Safety pictograms and concise handling instructions accompany every drum or bottle. Most producers comply with GHS guidelines, flagging health, fire, and environmental impacts. Consistent QC and batch testing create reliability for downstream users—a recurring point of feedback from process engineers who value stable starting materials.
Preparation Method
Sec-Amylamine synthesis rarely strays from traditional routes. Industrial makers often start with 2-pentanol, running it through amination with ammonia or suitable amine sources using metal or acid catalysis. Pressurized reactors encourage yields while curbing byproducts. Careful distillation sharpens product purity. Parallel lab-scale routes apply straightforward alkylation of ammonia with secondary halides, using less specialized equipment. No matter the scale, the key lies in tight process control—small shifts in temperature or reactant ratio skew the amine mix, forcing chemical engineers to fine-tune every batch.
Chemical Reactions & Modifications
Sec-Amylamine steps up as a building block for more complex molecules. Its lone nitrogen, flanked by hydrocarbon chains, bonds easily with acyl chlorides, sulfonyl chlorides, and aldehydes. Such reactions underpin the synthesis of amides, sulfonamides, and secondary amine derivatives. Organic chemists often reach for Sec-Amylamine in reductive aminations and N-alkylation projects, exploiting its predictable nucleophilic nature. Under oxidizing conditions, the molecule faces deamination, churning out lower-weight byproducts. Its compatibility with a wide range of functional groups attracts both pharma researchers seeking new drug profiles and industrial chemists creating bespoke surfactants or agrochemical products.
Synonyms & Product Names
Markets sometimes label Sec-Amylamine as 2-Aminopentane, pentan-2-amine, or secondary amylamine; local naming conventions appear on international safety data. Some catalogs list it under legacy or company-specific codes—for example, NSC 28246 or UN2435 in regulatory shipments. Research circles stick to IUPAC or straightforward shorthand, but seasoned buyers usually know to verify chemical structure on a data sheet to avoid confusion with primary or tertiary amines.
Safety & Operational Standards
Anyone who’s worked in chemical production knows safety isn’t just paperwork. Sec-Amylamine’s flammable liquid status, skin and respiratory irritation risks, and sharp odor put it on safety watchlists. Spill containment, solvent-resistant gloves, goggles, and effective ventilation cut down accident risks. Local authorities and global frameworks like REACH, OSHA, and EPA monitor permitted exposures, transportation packaging, and disposal. Emergency protocols call for swift cleanup with nonreactive absorbents, ample water flushing, and professional waste disposal. On the ground, the most effective teams treat routine handling as just as critical as major emergencies—a culture learned from past incidents and near-misses.
Application Area
Sec-Amylamine slots into a surprising range of uses. Pharmaceuticals draw on its backbone for antihistamines, anesthetics, and CNS-active molecules, tweaking its amine group to tune bioactivity. Agrochemical firms convert it into herbicides or plant growth agents, counting on its stability and reactivity. Laboratories use it as an intermediate to construct bigger, function-heavy targets—sometimes as a solvent, sometimes as a ligand, or even as a test substrate for biochemical assays. Paints, surfactants, and corrosion inhibitors benefit from its compatibility with other organic ingredients, making plant operation smoother and product engineering faster. Each industry brings its own quirks of use, influenced by local regulation, economics, and legacy process choices.
Research & Development
Research teams at universities and private labs often put Sec-Amylamine under the lens to unlock new transformations. Medicinal chemists like its balanced lipophilicity and metabolic stability, crucial traits as new drugs snake their way through trials. Process engineers install continuous flow or greener catalytic routes to lower energy and waste. Modern analytics—NMR, GC-MS, IR—trace every minor impurity, racking up insights into how changes in production influence downstream performance. Requests from end-users—higher optical purity, better shelf life, lower odor footprint—feed directly into R&D pipelines. The field grows as young chemists bring computational modeling and bio-catalysis into an area long dominated by traditional organic synthesis.
Toxicity Research
Animal studies and workplace monitoring steer the conversation around Sec-Amylamine toxicity. Acute exposure commonly brings eye and respiratory tract irritation; higher doses push toward CNS depression and systemic effects. Long-term studies indicate moderate organ risk, placing it below some legacy aromatics but above near-neutral solvents. Regulatory groups set occupational exposure limits, encourage medical surveillance, and push for pre-market toxicity screening. Researchers probe metabolic breakdown, biotransformation pathways, and cumulative toxicity, publishing data that strengthens worker protection programs. Individual chemistry labs keep MSDS files up-to-date and test air for amine vapors, taking no shortcuts on health.
Future Prospects
The push for cleaner, safer chemicals drives innovation in Sec-Amylamine use. Green chemistry finds new catalysts to cut waste, renewable feedstocks to replace petro-origin alcohols, and continuous reactors to limit batch loss. Demand from pharmaceuticals and fine chemicals expands as more complex molecules build from modular amines. Tighter environmental rules tighten the leash on hazardous chemicals, nudging specialty amines toward higher purity, lower toxicity, and safer handling profiles. Young scientists experiment with bio-based transformations and digital process control—lessons that promise to keep Sec-Amylamine in play as a flexible, adaptable tool across many fields.
What is Sec-Amylamine used for?
What’s Sec-Amylamine?
Sec-Amylamine, also known as 2-pentanamine, plays a quiet role in the world of industrial chemicals. Its structure, with a straight five-carbon backbone and a nitrogen at the center, gives it a spot among aliphatic amines. In plain terms, it’s a clear liquid with a fishy odor, and it’s reactive enough to make it useful for plenty of things. The story of sec-amylamine stretches into industries that shape everyday life — even if most people don’t ever hear its name.
Sec-Amylamine in Chemical Synthesis
Factories and research labs keep sec-amylamine around because it kicks off chemical reactions that help build much bigger molecules. Take pharmaceuticals: amines like sec-amylamine go into the recipes for drugs you’d find in any pharmacy cabinet. They help create painkillers, antihistamines, and blood pressure medications. Not all of those finished pills have sec-amylamine inside, but this chemical works behind the scenes, helping engineers test reactions and put together complicated molecules.
Agriculture gets a boost from sec-amylamine, too. Pesticides and herbicides rely on chemical building blocks, and amines stay popular choices. Sec-amylamine helps build the active ingredients that keep insects off crops or unwanted weeds out of fields. I’ve walked through modern farms and seen that behind neat rows of produce, there’s a chemical science that keeps things growing.
Special Roles in Polymers and Dyes
Sec-amylamine even finds a job in materials most folks overlook. The polymer industry — think plastics, foams, and synthetics — draws on amines to trigger reactions or finish off molecules. Without chemicals like this, tons of the materials around homes and workplaces would look and feel different.
Dye makers, too, turn to sec-amylamine as they work up colorants that will last on fabrics and products. This chemical helps bind dyes so vivid color stays put after a wash or two. Without well-chosen amines, colors run or fade faster. I remember seeing textile suppliers debate which additives work best, and sec-amylamine shows up on those lists for a reason.
Industrial Cleaning and Solvent Applications
Cleaning up tough grease or residue often takes more than elbow grease. Sec-amylamine works as a solvent for special jobs, helping break down oily build-up in industrial settings. I’ve seen maintenance crews reach for amine-based cleaners to keep machinery humming. It cuts through certain deposits that water alone can’t budge.
Risks and Safe Handling
Sec-amylamine poses a health risk if handled poorly. People exposed to its vapor can feel dizzy or suffer skin irritation. Factories that use it stress personal protection and proper ventilation. Many of these lessons trace back to past workplace accidents — there’s no room for shortcuts, and that urgency gets handed down from shift to shift.
Waste from amine use, including sec-amylamine, begs for careful disposal. No one wants runoff leaking into rivers or soil. Regulations require tracking, treating, and storing chemical waste, not just to follow rules, but to protect communities downwind or downstream. Chemical management shouldn’t be about checking boxes — it’s about safety for the long haul.
Paths Forward
Companies look for greener alternatives across all chemical supply chains. Sec-amylamine isn’t exempt. Better ventilation systems, safer packaging, and clear worker training all help shrink risks. Some research teams chase new production methods using bio-based feedstocks to boost sustainability. Long-term, reducing chemical hazards combines thoughtful design, investments in safety tech, and a culture that values prevention.
What is the chemical formula of Sec-Amylamine?
Understanding the Structure: Getting Closer to the Facts
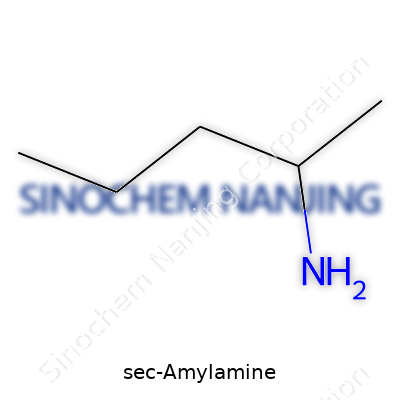
Sec-Amylamine stands out among organic amines. Its chemical formula holds the answer: C5H13N. Visualize a five-carbon chain, where the amino group connects to the second carbon. This secondary structure, rather than attaching at the edge of the chain, turns the story from straight to branched. Many confuse “amyl” with n-amyl or isoamyl, but sec-amylamine means the NH2 anchors at carbon two—yielding 2-aminopentane. That unique spot matters to chemistry students and industry chemists alike since structure guides both reactivity and use.
Why Secondary Amines Matter in Real Life
Picking out secondary amines from a lineup helps in pharmaceuticals, plastics, and even cleaning products. This format, with two alkyl groups hugging the nitrogen, gives different properties than its straight-chain siblings. Take boiling point: secondary amines show a drop compared to primary ones, thanks to changes in hydrogen bonding. These small shifts end up influencing everything from manufacturing setups in chemical plants to the kind of lab glassware needed for synthesis.
Safety Patterns and Hands-On Handling
Amines, especially the lower-chain types, often arrive with strong, fishy odors. In my own undergraduate days, opening a bottle of sec-amylamine could clear out a lab. Beyond the smell, these molecules can irritate skin, eyes, and lungs. Some people forget how fast they soak into gloves or waft across a fume hood. Proper training reduces risk. Reading up on materials safety data sheets became as familiar as flipping through textbooks. Anyone working with sec-amylamine should keep the goggles handy, gear up in nitrile instead of latex gloves, and make sure to vent the workspace.
Applications: From Reactions to Real-World Objects
Sec-Amylamine does not top the charts in fame, but its uses touch more corners of life than most expect. In industry, chemists lean on secondary amines during the making of surfactants, coatings, and pharmaceuticals. Some chemical reactions show more interest in branching—think about how amines become building blocks for drug molecules, epoxy hardeners, or rubber accelerators. I spent a semester tracking the outcome of an N-alkylation reaction that swapped out a primary amine for sec-amylamine. The results changed yield and purity of the final product, showing how the smallest tweak—just shifting the amine one carbon down the chain—transforms the outcome.
Thinking Ahead: Responsible Use and Disposal
Dealing with chemical waste brings up more than just environmental rules. Amines can form toxic byproducts, foul up waterways, and pose health threats far outside the lab. Regulations ask for careful collection and specific disposal methods. In my early research years, I saw the effect of ignored amine waste: rivers clogged with foam, wildlife showing odd behavior, and neighboring communities complaining of mysterious smells. Treating these compounds with acid, collecting the resulting solutions, and keeping them far from sinks—such steps echo across every lab worth its name. Strict procedures and regular inspections can bridge the gap between discovery and carelessness.
Learning from Sec-Amylamine: Chemistry That Connects
The story of sec-amylamine, with its simple formula and branched backbone, contains lessons for anyone in chemical science. Structure shapes function, safety requires vigilance, and even the humble amine leaves a mark on science and daily living. Paying attention pays off, not just in product yield but in safer, cleaner labs and cities.
What are the storage and handling recommendations for Sec-Amylamine?
The Real Reason Sec-Amylamine Deserves Respect
Sec-Amylamine isn’t just another chemical you leave sitting anywhere. My time in manufacturing taught me certain liquids like this one love to creep out of their containers if you don’t get storage right. The stuff brings vapor, odor, and spill headaches, and, left unchecked, won’t just disappear—it builds up, waiting for someone to make a mistake. NIOSH calls for vigilance: once inhaled, it can irritate airways, and direct skin contact burns or causes allergic reactions. These aren’t warnings for the paperwork.
Keep It Cool, Keep It Dry
Every veteran in a chemical warehouse knows the basics: avoid warm, steamy places. Sec-Amylamine does best at room temperature—or a bit cooler—out of the sunlight. Humid corners of a building might draw moisture, and anything wet increases the odds of chemical reaction. I remember a plant manager joking, “If you’re sweating, so is your inventory.” Warmth quickens evaporation, which means more vapors in the air and more risk for the people clocking in and out.
Containers: A Weak Link in the Chain
Any old drum won’t do. I’ve seen cheap lids warp after just a few months, letting vapors sneak past. Stainless steel or high-quality, tightly sealed containers cut down risk. Plastic types must stand up against amines—some soften and fail after a single season. Always check for corrosion or brittleness before moving anything, and label everything clearly along with hazard symbols. Labels don’t fix mistakes, but they stop half of them from happening in the first place.
Keep Away from Heat and Sparks
Sparks and open flames don’t mix with Sec-Amylamine, and I’ve watched more than one rookie learn this the hard way. Forklifts, static discharge, badly wired tools—in busy settings, mistakes happen fast. Grounding containers staves off static. A dedicated area, physically distanced from machinery, closes one more door on disaster. Add proper ventilation—industrial fans, chemical fume hoods—and you scatter vapors instead of letting them build up. I’ve met folks with stories about old warehouses that lacked airflow and became time bombs after months of neglect. Don’t be that workplace.
No Shortcut on PPE
Goggles, face shields, gloves, aprons: basics for any operation. Chemicals seem easy to handle after years of repetition, but bare skin or lungs can’t compete with Sec-Amylamine’s effects. Respirators or fume masks go from “nice to have” to “mandatory” where vapor levels climb. Safety showers and eyewash stations prove their worth after one splash, and regular drills make sure nobody forgets where to run. I’ve seen operators take shortcuts, but every shortcut piles up risk until someone gets hurt.
Training and Emergency Prep
OSHA doesn’t put out training standards just for paperwork: refresher sessions and morning briefings help catch errors before they roll downhill. Spills demand quick cleanup with absorbent pads or neutralizers. Not every crew knows the right way to handle sudden leaks, but regular drills plant that response deep. Emergency contacts, extra gear, and spill kits make the difference between a long story and a serious headline.
Good Habits, Fewer Surprises
Preventing problems beats cleaning them up—every time. Store Sec-Amylamine in a cool, dry, ventilated place, check containers, ban open flames, keep PPE close, and train teams for the unexpected. Teams that sweat these details see fewer accidents. It’s the difference between coming home safe and dealing with a mess nobody wanted.
Is Sec-Amylamine hazardous or toxic?
Daily Workplaces and Strange Smells
In labs, factories, or chemical plants, the mention of unfamiliar names always triggers memories of odd odors and tightly closed bottles stashed in storerooms. Sec-Amylamine lands right on that list—a chemical with a punchy smell, a clear formula, and a set of rules every worker learns sooner or later. It’s not a household topic, but for anyone handling chemical processing or research, knowing its hazards becomes as routine as donning a hard hat. Every time the seal on a bottle comes off, the risk isn’t far behind.
Health Hazards: More Than Just Irritation
Sec-Amylamine (also known as 2-pentylamine) sets off alarm bells for industrial hygienists and chemists with good reason. Short exposure can bring on eye and skin burns, and even breathing in its vapors can leave lungs raw. The US National Library of Medicine classifies it as both a skin and respiratory irritant. Some workers handling amines grow desensitized to the annoyance—at first. Over time, careless exposure piles up. Chronic inhalation isn’t just uncomfortable; it’s a road toward long-term respiratory issues.
Panic isn’t helpful, but a healthy dose of respect is warranted. The Centers for Disease Control and Prevention highlights the fact that this compound has a knack for causing chemical burns—painful, slow to heal, sometimes leading to permanent sensitivity. The effects happen quickly, which means the damage is done long before the typical symptoms reach their peak. Not everyone knows that even a splash on the skin can slip through cloth and gloves if they’re old or worn thin.
Fire Dangers in the Shadows
Flammability stands out as another concern. Sec-Amylamine catches fire with alarming ease, and its vapors roll along floors, collecting in low spots. I’ve watched fire marshals stage training demos where a single spark sent colorless vapors blazing across concrete. Standard fire extinguishers don’t always cut it—a chemical fire sometimes needs foam or dry powder instead of water, which can actually spread the flames. Anyone handling the substance keeps an emergency plan within reach. There isn't room for shortcuts.
Long-Term Exposure Concerns
Long exposure stories don’t always show up in glossy science journals. They’re in tales from the shop floor. After years around volatile amines, some workers report asthma, rashes, headaches, and chronic nosebleeds. Regulations try to keep airborne concentrations in check, but gap years in enforcement can lead to mystery health complaints. The European Chemicals Agency didn’t list Sec-Amylamine as a confirmed carcinogen, but many amines show troubling chemical similarities with others cataloged for cancer risk. The uncertainty here means extra caution, especially with repeat exposure.
Protection and Control Are Not Optional
In my experience, the difference between a near miss and a disaster often comes down to who’s watching out for everybody’s safety. Proper fume hoods, gloves that resist breakthrough, goggles, and quick access to showers and eye wash stations set the baseline. Good ventilation and training are part and parcel of chemical work, especially with things like Sec-Amylamine. Rushed handling is unforgiving. Frequent monitoring of air quality and keeping up a culture where speaking up about spills or leaks isn’t frowned on—that’s how you avoid tragedy.
Building Better Habits and Policies
Industry has improved, but accidents still pop up—sometimes due to old equipment, slipshod records, or forgotten SOPs. Solutions come from both the ground up and the top down. Workers need the authority to push back against unsafe practices. Management must see chemical safety as an ongoing investment. Regulators, meanwhile, bear the responsibility to keep updating public information and exposure limits as new research comes in. There’s progress, but much of it starts with admitting chemicals like Sec-Amylamine deserve unwavering attention—every shift, every bottle, every time.
What is the CAS number of Sec-Amylamine?
Understanding CAS Numbers in Chemistry
Every chemical has a story, and the Chemical Abstracts Service (CAS) number is often the key to understanding it. The CAS number acts as a fingerprint, cutting through language and regional naming confusion. Sec-Amylamine, for those of us who have spent long hours in a laboratory or scouting through research papers, carries the CAS number 107-18-6.
Why It Matters in the Real World
Looking at shelves in a chemical storeroom, similar names can trick even the most seasoned researcher. For example, sec-Amylamine sounds a lot like n-Amylamine or tert-Amylamine, but only one of them carries the number 107-18-6. Mixing up these chemicals, even accidentally, can cause serious headaches in synthesis work or analysis projects. Rather than debating names, I’ve always leaned on the certainty of CAS numbers.
Sec-Amylamine itself pops up in industrial settings, especially in organic synthesis. Explorers in pharmaceuticals, agrochemicals, and even dyes interact with it during development phases. Misidentifying this substance with a similar-sounding amine—not unusual in fast-paced labs—can cause project delays or, worse, unsafe reactions. In my experience, cross-referencing CAS numbers on data sheets and product containers saves both time and lives.
CAS Numbers and Scientific Transparency
Journals and regulatory agencies want clarity. Papers get rejected if substances aren’t described unmistakably. Regulators in the European Union’s REACH system and the United States EPA rely on CAS numbers to track data, impose restrictions, and perform risk assessments. Mistakes here create compliance problems, possible legal trouble, and input from auditors—the last thing companies want.
For companies shipping globally, the CAS number 107-18-6 for sec-Amylamine becomes a passport. Each regulatory market has different hazard warnings and labeling rules, but the CAS number clears up confusion. By focusing on this number, everyone involved—chemist, safety officer, shipper—speaks a common language. Every bottle arrives with the right documentation, which keeps waste to a minimum and cuts risk.
Supporting Safe Science
Lab safety doesn’t get enough attention until something goes wrong. If labels only mention names, students and new staff can reach for the wrong substance. CAS numbers help build the chain of custody for chemicals and reduce the odds of accidental exposure. After all, not every amine has the same toxicity, flammability, or reactivity. I’ve seen protocols where CAS numbers are mandatory for all orders and stock cards, and these labs report far fewer mix-ups.
Solving hazards in chemical management starts with standardizing identification. Digital inventory platforms make it easier to filter, track, and order chemical supplies using CAS numbers. For researchers at every level, learning to trust CAS numbers over common names should be part of their training early on.
A Clear Solution for Modern Chemistry
CAS number 107-18-6 for sec-Amylamine helps everyone from students to industry veterans keep their work accurate and their environments safer. As science gets more complex and collaborative, this unique code streamlines communication and supports reliability in everything from publishing research to moving chemicals across borders. Building a culture where CAS numbers are second nature sets up teams for long-term success and fewer preventable mistakes.
| Names | |
| Preferred IUPAC name | pentan-2-amine |
| Other names |
2-Aminopentane sec-Pentylamine |
| Pronunciation | /ˌsɛkˈæm.ɪl.əˌmiːn/ |
| Identifiers | |
| CAS Number | 124-40-3 |
| Beilstein Reference | 1738736 |
| ChEBI | CHEBI:15341 |
| ChEMBL | CHEMBL15315 |
| ChemSpider | 5658 |
| DrugBank | DB14096 |
| ECHA InfoCard | 17c2cc1d-69a2-4fc6-b1bb-7dfa066cf9ac |
| EC Number | 205-500-4 |
| Gmelin Reference | Sec-Amylamine: 5420 |
| KEGG | C06185 |
| MeSH | D000687 |
| PubChem CID | 8174 |
| RTECS number | AS9100000 |
| UNII | 2A56V127VW |
| UN number | 1110 |
| Properties | |
| Chemical formula | C5H13N |
| Molar mass | 87.16 g/mol |
| Appearance | Colorless to yellowish liquid |
| Odor | Ammonia-like |
| Density | 0.75 g/mL at 25 °C |
| Solubility in water | Soluble |
| log P | 0.92 |
| Vapor pressure | 4.4 kPa (at 20 °C) |
| Acidity (pKa) | 10.73 |
| Basicity (pKb) | 3.37 |
| Magnetic susceptibility (χ) | -53.0E-6 cm³/mol |
| Refractive index (nD) | 1.411 |
| Viscosity | 2.14 mPa·s (at 20 °C) |
| Dipole moment | 2.3137 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 101.1 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –53.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3515.6 kJ/mol |
| Pharmacology | |
| ATC code | N06AX05 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Danger |
| Hazard statements | Harmful if swallowed. Causes severe skin burns and eye damage. Toxic to aquatic life. |
| Precautionary statements | P210, P260, P280, P303+P361+P353, P305+P351+P338, P310, P370+P378 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 47.0 °C |
| Autoignition temperature | 285 °C |
| Explosive limits | 2–11% |
| Lethal dose or concentration | LD50 oral rat 200 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3,816 mg/kg (rat, oral) |
| NIOSH | SE9810000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Sec-Amylamine: "100 ppm (350 mg/m3) TWA |
| REL (Recommended) | S24 |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
n-Amylamine Isoamylamine tert-Amylamine |

