Salicylaldehyde: Insights, Applications, and the Road Ahead
Historical Development
Salicylaldehyde carries a reputation that stretches back to the 19th century, when it caught the interest of chemists looking to unravel the secrets of phenolic compounds. Kolbe and Lautemann set the foundation for its discovery while pioneering the synthesis of salicylic acid from phenol, but it was the German chemist August Wilhelm von Hofmann who took things further by isolating salicylaldehyde in the laboratory. This compound offers a window into the kind of progress chemists made during the days when organic chemistry struggled to develop reliable ways to turn simple starting materials into more complex ones. Over the decades, as the understanding of aromatic compounds deepened, so did the technical application of salicylaldehyde. By the 20th century, it found fresh appeal as a precursor for a slew of industrial chemicals and dyes, influencing synthetic strategies and expanding the toolkit for what chemical engineers and researchers could build in a lab.
Product Overview
Salicylaldehyde shows up as a pale yellow, oily liquid, with a distinctive almond-like smell that makes most folks think of benzaldehyde but with an earthy twist. Whether sitting on a shelf in a research lab or stored in commercial drums, this compound finds demand in both fundamental research settings and large-scale manufacturing alike. Its structure—a benzene ring bearing both a formyl and a hydroxyl group—gives it a unique flavor for chemical reactivity and serves as a critical building block for a range of valuable derivatives. Chemists know it as a handy intermediate that makes complex synthesis just a little bit easier, especially if one’s chasing after Schiff bases, pharmaceuticals, or certain colors and flavors.
Physical & Chemical Properties
Salicylaldehyde weighs in with a molecular formula of C7H6O2 and a molecular weight of about 122.12 g/mol. It boils at around 196 °C and melts close to -8 °C, making it easy to handle in liquid form under most room temperature conditions. In water, it offers poor solubility, but finds itself at home dissolving in organic solvents like ethanol, ether, and chloroform. Its most telling chemical trait stems from both the aldehyde and hydroxyl group positioned ortho to each other. This arrangement sets it up for hydrogen bonding, making it able to take on chelation roles, and pushes its reactivity in directions not seen in benzaldehyde or phenol alone.
Technical Specifications & Labeling
Quality standards and technical specifications play an outsized role, especially for anyone sourcing salicylaldehyde for industrial or research use. Purity levels typically sit around 98–99%, with the compound sold either neat or in solution, depending on the end-user’s requirements. Detailed safety data sheets follow each shipment, outlining storage conditions, recommended handling practices, and labeling according to GHS standards. Transport regulators classify it as a hazardous substance, so certified containers and correct marking remain non-negotiable. Rigorous batch testing for residual solvents, water content (often measured by Karl Fischer titration), and heavy metal contamination underline the compliance demanded in pharmaceutical and specialty chemical sectors.
Preparation Method
Producers use a couple of routes to make salicylaldehyde, but the ortho-formylation of phenols often takes center stage. The Reimer-Tiemann reaction gets the nod in many teaching labs and industrial settings, where chloroform reacts with sodium hydroxide and phenol, delivering the aldehyde function ortho to the hydroxyl group. Industrial-scale operations might pull out the Duff reaction, leveraging hexamethylenetetramine and acid treatment, or play with the Gattermann-Koch reaction, using carbon monoxide and hydrochloric acid with an aluminum chloride catalyst. Process engineers keep a sharp eye on reaction conditions, as selectivity can tank if you don’t manage stoichiometry and temperature with care. Waste streams can’t be ignored either—chlorinated byproducts and strong alkaline effluent force operators to invest in treatment systems to stay within environmental limits.
Chemical Reactions & Modifications
Salicylaldehyde’s blend of aldehyde and phenolic hydroxyl opens it up to a wide set of chemical transformations. The aldehyde group reacts smoothly with primary amines, spawning Schiff bases—chelating ligands that underpin everything from homogeneous catalysis to medical diagnostics. Cyclization with other functional groups yields coumarins, a scaffold treasured by both perfumers and pharmaceutical researchers. The formyl group tolerates oxidants, giving rise to salicylic acid, a backbone for anti-inflammatory drugs. Under selective reductions, the aldehyde shifts down to a benzyl alcohol, while the hydroxyl sets up O-alkylation and acylation, building ever-more elaborate organic molecules. The push for greener chemistry drives modern labs to re-imagine these reactions with less toxic reagents or with catalytic conditions that lower the bar for waste and energy usage.
Synonyms & Product Names
You’ll run into a variety of names for this same chemical, depending on the context. Chemists track it as 2-hydroxybenzaldehyde in technical literature. Trade names and catalogues might list it as ortho-hydroxybenzaldehyde or just O-Hydroxybenzaldehyde. In industrial circles, abbreviations like SalAld pop up, especially in shorthand lab notes or purchasing documents. Despite the multiple names, the structure remains the same—one of nature’s more versatile aromatic intermediates.
Safety & Operational Standards
Anyone working with salicylaldehyde has to respect its hazards. Skin and eye irritation pop up as routine risks if proper PPE—gloves, goggles, lab coats—aren’t used. Fumes can irritate respiratory tracts, making good ventilation a must, and storage practices must lock the chemical away from acids, oxidizers, and open flames. Facilities lean on standard operating procedures, regular safety training, and emergency spill kits. Regulatory agencies like OSHA and the European Chemicals Agency keep a close watch, flagging safe exposure limits and enforcing rules on workplace monitoring. Waste from synthesis and post-use cleaning heads to specialized collection points, adding another layer of operational discipline to a chemist’s daily workflow.
Application Area
Salicylaldehyde finds its way into countless sectors. In the world of organic synthesis, it forms the backbone of many ligands, dyes, flavors, and fragrances. The food industry leans on derivatives in artificial flavors. Agrochemical producers tap into its chemical ancestry, using it to build pesticides and fungicides with broad-spectrum activity. Dye makers reach for salicylaldehyde-based molecules to tune color properties in both textiles and analytical reagents. In pharmaceuticals, its coupling reactions help synthesize anti-tuberculosis compounds and non-steroidal anti-inflammatory drugs. Developers of liquid crystals and specialty polymers count on its chelating abilities to engineer new materials. The reach of this compound owes as much to its reactivity as to the creativity of the folks working with it.
Research & Development
Researchers constantly probe new uses and modifications, looking to add value to the compound. Advances in green chemistry aim to swap out classic but polluting reagents for catalysts that use air and light to drive similar transformations. Nanotechnology groups exploit salicylaldehyde’s ability to bind metals, opening up applications in biosensing and targeted drug delivery. Analytical chemists develop sensor materials that change color or conductivity with the presence of heavy metals or biological toxins, all hinging on properly modified salicylaldehyde backbones. Intellectual property grows as labs patent new synthetic pathways or product applications, carving out specialized markets only reached through inventive chemistry grounded in this humble aldehyde.
Toxicity Research
The health impacts of salicylaldehyde, while not as notorious as those of some solvents or heavy metals, still draw concern from toxicologists. Acute ingestion causes stomach pain and headaches, and high vapor concentrations irritate mucous membranes. Animal studies on chronic exposure show some liver and kidney effects, though not at the same intensity as more reactive aldehydes. Ecotoxicity tests signal moderate risks to aquatic organisms if effluent isn’t tightly managed. The usual approach boils down to handling the compound with care—using fume hoods, gloves, goggles, and regular monitoring—and disposing of waste responsibly. Ongoing studies track metabolic breakdown in mammals, hoping to answer questions about bioaccumulation and long-term environmental footprints that only come up when chemicals become building blocks for larger-scale production.
Future Prospects
Salicylaldehyde stands at a crossroads shaped by demand for greener synthesis and engineered materials. With the push to replace petroleum-based feedstocks, scientists look to biosourced aromatic compounds and re-engineered yeast that crank out useful aldehydes straight from plant matter. Modern catalysis—photoredox, biocatalysts, electrochemistry—offers routes to make and use salicylaldehyde under milder conditions, with fewer emissions and less hazardous waste. In advanced electronics, researchers investigate its benzoin-type condensation products for next-generation sensors, solar cells, and optical materials. Synthetic biology teams rewrite microbial genomes, nudging algae and bacteria to make salicylaldehyde in fermenters, setting up future economies where specialty chemicals come from renewable sources instead of oil refineries. Breakthroughs will hinge on regulatory approval, process efficiency, and the talent of researchers keen to solve tomorrow’s material and environmental challenges by leveraging the humble, reactive core of salicylaldehyde.
What is Salicylaldehyde used for?
A Closer Look at Salicylaldehyde
Salicylaldehyde might sound like something only chemists care about, but its impact goes well beyond science labs. It’s a colorless oily liquid with an almond-like odor, and it shows up in many unexpected places. The roots of its importance run deep, especially in manufacturing, medicine, and materials science. Years ago, during a summer research internship, I helped run chemical reactions using salicylaldehyde, thinking the applications stopped at academic curiosity. I was wrong.
From Lab Bench to Life-Saving Drugs
Salicylaldehyde helps build molecules in the pharmaceutical industry. Chemists use it as a building block for complex drugs. For example, it’s key in creating coumarin derivatives, which have anticoagulant properties. Blood thinners like warfarin owe their success to processes that start with chemicals like salicylaldehyde. In recent decades, researchers have discovered methods for making better antibiotics, anti-inflammatories, and even anti-cancer agents using this chemical. The story of aspirin’s development also comes back to compounds that relate closely to salicylaldehyde’s chemical structure. These examples underscore the value of basic chemicals for improving public health.
Scent, Flavor, and Fine Chemistry
At home, folks may not realize there’s a connection between the makeup of perfumes and the science behind chemical synthesis. Salicylaldehyde helps craft scents and flavors. Perfumers blend it to mimic the sweet smell of almonds or give an extra depth to floral notes. Food chemists sometimes use traces to create almond-like flavors in candies and drinks. I remember in organic lab classes, the aroma of almonds would linger when synthesizing salicylaldehyde, making the lab a bit more pleasant for a day.
Industrial and Research Applications
In industry, salicylaldehyde acts as a basic ingredient in making resins and dyes. It also helps drive reactions that pull out heavy metals from wastewater. This is significant for environmental cleanup efforts. Coordination compounds made from salicylaldehyde can bind to metal ions, pulling toxins from contaminated streams or soils. The environmental role can’t be overlooked. As someone who’s spent time in environmental testing labs, I’ve seen firsthand how these processes speed up water treatment.
Challenges and Safer Solutions
Handling chemicals like salicylaldehyde brings challenges. It’s flammable and toxic in large amounts. Strict safety protocols matter. Protective gloves, splash goggles, and well-ventilated spaces are non-negotiable. Regulatory agencies, such as OSHA and the European Chemicals Agency, keep a close eye on safe handling. Research efforts now focus on greener alternatives and processes that use less toxic reagents, so workers and the environment both see fewer risks. Green chemistry, an approach I’ve advocated for in workshops, aims to limit harmful waste and cut exposure for people on the production line.
Looking Ahead
The role of salicylaldehyde in developing medicines, fragrances, and industrial goods shows how basic chemicals touch nearly every aspect of daily life. Its future usefulness depends on ongoing research into safer production and smarter recycling. Industry leaders, research institutions, and regulatory bodies can keep making progress by supporting better safety standards and encouraging eco-friendly alternatives. Decisions in chemical management today shape the innovations and safety practices of tomorrow, fueling both industry growth and environmental protection.
What is the chemical structure of Salicylaldehyde?
Seeing Molecules Beyond the Textbook
Salicylaldehyde shows up as a clear, slightly oily liquid with a bitter almond smell. Chemists point to its formula: C7H6O2. It looks simple on paper, but digging into its chemical structure reveals why it draws interest across labs and industries.
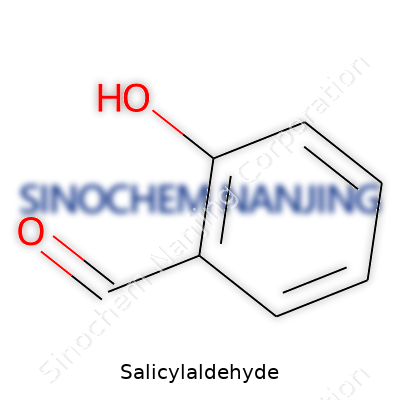
Its setup rests on a benzene ring, so you picture a loop of six carbons, each with its own hydrogen except where something replaces it. Salicylaldehyde swaps out one hydrogen for an aldehyde group (–CHO) and another for a hydroxy group (–OH). These two groups sit right next to each other at the “ortho” or 2-position of the ring. This gives it the name “2-hydroxybenzaldehyde” among chemists.
Function Shapes Its Place in Chemistry
The real twist comes from how those extra pieces—the aldehyde and the hydroxy—nestle side by side. That simple change makes the molecule a lot more reactive than plain old benzaldehyde. It’s not just about swapping atoms; the spot and the nature of each group create new ways for this molecule to interact. That’s why it forms such strong bonds in making ligands for metal complexes or in synthesizing dyes and flavor molecules.
My years in the lab taught me how location and small tweaks matter. With the hydroxy group so close to the aldehyde, they team up, forming hydrogen bonds. That makes this compound a key ingredient in more than just chemical reactions. It stands out in analytical chemistry, especially in chelating agents or as a building block for bigger, more complicated molecules. Some of the earliest lessons in organic chemistry point to mirroring these patterns in natural compounds, like salicylic acid, the backbone for aspirin.
Facts That Matter for Everyday Science
Industry keeps turning back to salicylaldehyde due to this distinct setup. Its chemical structure allows it to snap onto metals—think nickel or copper—so it plays a big part in extracting and separating metals from complicated mixtures. Anyone reading about environmental cleanup or mining technology might not spot its name, but its fingerprint is there.
Health workers notice it popping up in fragrances or even some pharmaceutical processes. Because of its reactive structure, it sometimes finds use as an intermediate for ingredients in perfume and flavor chemistry. Students often try to draw its structure and miss the detail that gives it these useful powers—the proximity of the two functional groups.
Paths to Safer and Cleaner Chemistry
With increased regulation around aromatic aldehydes, industry needs to keep an eye on exposure and safe handling. Recent research pushes for greener syntheses, with less waste and fewer harsh solvents. Catalytic methods take inspiration from nature—enzymes in willow bark, for instance—showing how a little biology can coax the right structure without the pollution.
It’s easy to skip over the fine points when meeting a molecule in a textbook. In the real world, salicylaldehyde’s chemical structure shapes its role across chemistry, health, and environmental science. Paying attention to these details guides us to smarter manufacturing, safer labs, and better products for everyone.
Is Salicylaldehyde hazardous or toxic?
My Experience with Open Chemical Cabinets
I remember my first introduction to salicylaldehyde during a college lab. Lab gloves on, ventilation fans humming, my professor handed me a small bottle of pale yellow liquid. Its almond-like scent carried a warning—a gentle nudge to take the material seriously. That moment stuck with me. Plenty of chemicals come with hidden risks, but some, like salicylaldehyde, announce themselves upfront. People use it in synthesis and research, so understanding its hazards isn't just academic.
What We Know from the Research
Salicylaldehyde comes from the world of aromatic aldehydes. It finds use in making dyes, plastics, and pharmaceuticals. The safety data sheets make the dangers clear. Inhaling its vapors can irritate the nose, throat, and lungs. Studies on rats show reactions at moderate doses. Not severe toxicity like cyanides, but enough to remind you that the lab coat is not just for show. Spilled salicylaldehyde can sting your eyes and leave a rash on your skin if you touch it. In my own lab, accidental splashes sometimes happened. Redness and brief discomfort passed only after a proper rinse. Those quick responses meant less harm—but the threat was real.
Getting Real about Safety Concerns
Chemicals deserve respect. Many people overlook subtle hazards because they only see the strong alerts, but chronic exposure to irritants can wear a person down. Repeated low-level contact with salicylaldehyde does not generate drama, but it could lead to persistent headaches or skin problems. We learn that the hard way: rare but real cases of occupational asthma connect with aldehyde exposure. Real stories exist about people missing early warning signs—my old coworker in pharma started developing skin problems after repeated handling of aromatic chemicals, including this one, without proper gloves.
EPA and European safety agencies rank salicylaldehyde as harmful upon contact, but less dangerous than top-tier toxins. The bigger risk emerges for those who ignore the basics: tight bottle caps, working fume hoods, and regular glove changes. Any chemical in an open-air workshop gets harder to control. One small spill on a hot plate, and everyone knows from the sharp, sweet scent cutting through the air.
How We Can Do Better
Some hazards feel abstract—this one earns respect by showing up right away. Relying on ventilation and standard personal protection can prevent nearly all minor exposures. Reliable labeling and staff reminders work in both academic and industrial spaces. Too often, I see distracted students skip the safety check or tight-lid closure in a rush. Shortcuts raise the odds of trouble. Teams that pause and review updated safety cards, practice spill drills, and swap gloves as soon as one breaks tend to avoid those unnecessary scares.
Modern labs owe their safety to culture as much as to technology. Salicylaldehyde is a sharp reminder: every chemical, no matter how common, requires mindfulness. Many learn from close calls. Sharing these stories and emphasizing vigilance can keep everyone safer than relying on warnings printed on a bottle alone.
How should Salicylaldehyde be stored?
The Risks Hiding in a Simple Bottle
Salicylaldehyde looks harmless enough in a brown bottle, but that aromatic liquid does not take kindly to careless storage. I remember stepping into an undergraduate lab and catching that distinct almond smell wafting from a loose cap. Someone figured a simple twist would do. A headache and some fuming later, the importance of proper handling stuck with me for good.
No Room for Guesswork
This chemical oxidizes easily when exposed to air, losing both purity and effectiveness. Anyone handling it knows the liquid turns yellow and thick if left open. Worse, it becomes more hazardous as decomposition sets in. Direct exposure to sunlight speeds up this breakdown, making sunlight a real enemy. Leaving it on a windowsill is just about the worst call—heat punches up volatility, and ultraviolet rays jumpstart spoilage.
Moisture isn’t a friend, either. Humid air creeping in through an unsecured seal sets off slow but steady degradation. Fumes not only compromise future experiments by contaminating the product—breathing them in serves up eye, skin, and respiratory irritation. Storing this compound in a cool, dry spot isn’t just lab dogma; it cuts down on risk at every level.
Why the Right Container Matters
Salicylaldehyde breaks down plastics and soft seals if given the chance, which means only glass bottles with airtight, chemically resistant stoppers are worth the investment. Even small leaks can lead to sticky residue and difficult cleanups. Glass doesn’t just slow down vapor escape; it stands up to the solvent’s bite over time. Every experienced chemist I know keeps these bottles away from heat sources, storing them on low shelves to prevent accidents. Over the years, I’ve seen what a toppled bottle does to flooring—one spill and the acrid scent lingers for weeks.
Labeling as a Safety Tool
Labeling goes past scribbling a name. Keeping dates of receipt and opening right on the label lets everyone track shelf life. After six months, most labs run purity checks to catch invisible changes. I learned quickly to trust fresh stock for anything critical.
Leaks Happen—Preparation Helps
Accidents don’t make exceptions for veterans. One time in a research lab, a cracked cap allowed slow leaks, contaminating the chemical cabinet and nearby glassware. We traced eye irritation among techs back to that hidden source. Regular inspection caught the culprit, but that experience hammered home the value of scheduled checks.
Putting Good Storage into Practice
Securing salicylaldehyde in a ventilated, fire-safe chemical cabinet makes a world of difference. Many institutions keep compatible absorbent material on hand—vermiculite or sand—right in the cabinet for quick response. Fire extinguishers and eye wash stations belong close by; even quick exposure burns or causes harm. I’ve seen teams run annual safety drills around these chemicals, which helps etch safe habits into muscle memory more than any warning sign.
Practical Solutions for Safer Labs
Digital inventory systems now flag expiry dates and track when bottles open, alerting staff before chemicals pass their prime. Training new users on chemical-specific hazards—rather than blitzing through generic safety videos—keeps attention sharp. Updating standard operating procedures to include checks for vapors, cap condition, and label clarity catches most minor issues before they turn major. A tight web of responsibility grows trust in the lab, reducing slips and sloppiness.
Every bottle of salicylaldehyde holds potential for new discoveries, but only where storage keeps risks on a tight leash. Those everyday choices, from proper shelving to airtight closures, shape not only safer labs but healthier people.
What is the molecular formula and weight of Salicylaldehyde?
Getting to Know Salicylaldehyde
Salicylaldehyde draws the attention of many chemists and lab technicians for its unique structure and fragrance. Its molecular formula, C7H6O2, reveals a small yet intriguing molecule. People who have spent time in a laboratory, watching reaction colors shift and compounds distill, know this compound’s distinct almond-like aroma. This scent doesn’t just appeal to the nose; it provides a quick clue about its chemical identity. Anyone who has ever used it as a starting material for synthesizing more complex compounds comes to appreciate its practical value as well as its chemical individuality.
Why the Molecular Formula Matters
With C7H6O2, each atom counts. Seven carbon atoms, six hydrogen atoms, two oxygen atoms form the backbone for a range of behaviors in organic chemistry. For researchers, these numbers connect directly to yield calculations, purity checks, and predictions about how salicylaldehyde will interact with other substances. Any mistake in these basics can muddy experimental results, lead to unsound conclusions, or, even worse, throw a project off course entirely.
Looking at Molecular Weight
The molecular weight of salicylaldehyde stands at 122.12 g/mol. On the bench, this number plays a big role. I’ve watched students fumble calculations, confusing molar mass with actual weight measured on the balance. Such errors create real problems down the road, especially in scale-up work or analytical chemistry. In classrooms and professional labs alike, the molecular weight ensures that volumes, concentrations, and stoichiometries work out as planned. Imagine preparing a solution for a reaction or an assay; without knowing this figure, consistency in results disappears.
Supporting Facts and Practical Uses
Salicylaldehyde falls into the class of aromatic aldehydes, and it’s no stranger to dye manufacturing, fragrance development, and pharmaceutical synthesis. In my own experience, using C7H6O2 as an intermediate for complex organic structures allowed tight control over side reactions. Published studies back up its popularity, highlighting robust yields in Schiff base formation and ligand synthesis for metal complexes. Chemists reach for it when they need reliability, and journals frequently cite its role in multi-step organic processes.
Challenges and Solutions in Handling
Lab safety takes precedence, especially since salicylaldehyde can irritate skin or eyes. Forgetting to use gloves or fume hoods can make a workday uncomfortable very fast. Proper labeling and airtight storage also prevent evaporation losses, protecting everyone in a shared lab space. Clear documentation about its formula and weight ticks the boxes for laboratory compliance—nobody wants to repeat a synthesis due to inattention to detail. These habits don’t just keep people safe; they build credibility with colleagues and maintain the integrity of data produced in the lab.
Building Confidence
Knowing salicylaldehyde’s molecular details elevates trust in scientific reporting and reduces costly mistakes. Experienced researchers understand this, and entry-level learners pick up the lesson through trial and error. Over the years, using and teaching the correct molecular formula (C7H6O2) and molecular weight (122.12 g/mol) has proven crucial for smooth workflows and dependable outcomes. Anyone serious about chemistry owes it to themselves—and their peers—to respect these fundamentals.
| Names | |
| Preferred IUPAC name | 2-Hydroxybenzaldehyde |
| Other names |
2-Hydroxybenzaldehyde o-Hydroxybenzaldehyde Salicylic aldehyde |
| Pronunciation | /ˌsæl.ɪ.sɪlˈæl.dɪ.haɪd/ |
| Identifiers | |
| CAS Number | 90-02-8 |
| Beilstein Reference | 1209223 |
| ChEBI | CHEBI:28201 |
| ChEMBL | CHEMBL14635 |
| ChemSpider | 5031 |
| DrugBank | DB03826 |
| ECHA InfoCard | 100.012.629 |
| EC Number | 203-002-1 |
| Gmelin Reference | 63633 |
| KEGG | C01599 |
| MeSH | D011055 |
| PubChem CID | 6947 |
| RTECS number | VO5075000 |
| UNII | I88P60P7JP |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | DTXSID9020719 |
| Properties | |
| Chemical formula | C7H6O2 |
| Molar mass | 122.12 g/mol |
| Appearance | Colorless to pale yellow oily liquid |
| Odor | Aromatic; Bitter almond |
| Density | 1.163 g/mL at 25 °C (lit.) |
| Solubility in water | slightly soluble |
| log P | 1.65 |
| Vapor pressure | 0.049 mmHg (25°C) |
| Acidity (pKa) | 7.40 |
| Basicity (pKb) | 13.38 |
| Magnetic susceptibility (χ) | -46.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.589 |
| Viscosity | 3.14 cP (20°C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 248.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −94.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3226 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D02AE14 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H402 |
| Precautionary statements | H302 + H312 + H332, H315, H319, H402, P261, P273, P280, P301 + P312, P305 + P351 + P338, P337 + P313 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 86.0 °C |
| Autoignition temperature | 160 °C |
| Explosive limits | 1.1% - 9.9% |
| Lethal dose or concentration | LD50 oral rat 978 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 520 mg/kg |
| NIOSH | WMP |
| PEL (Permissible) | PEL: 5 ppm (TWA) |
| REL (Recommended) | 1 ppm |
| IDLH (Immediate danger) | IDLH: 175 ppm |
| Related compounds | |
| Related compounds |
Salicylic acid Salicylamide o-Vanillin 2,4-Dihydroxybenzaldehyde 3-Hydroxybenzaldehyde |

