(S)-4-Phenyl-2-Oxazolidinone: Evolution, Properties, and Prospects
Historical Development
Tracing back to the roots of modern chiral chemistry, (S)-4-Phenyl-2-Oxazolidinone owes much to the work done in laboratories searching for effective and controlled routes to asymmetric synthesis. In the late twentieth century, researchers became engrossed with chiral auxiliaries—the helper molecules steering reactions towards single-enantiomer outcomes. Chemists like David Evans and peers played significant roles lifting oxazolidinones into the center of attention. The development of (S)-4-Phenyl-2-Oxazolidinone came amid increasing demands for sharper stereocontrol in homogenous catalysis and synthesis, especially when pharmaceutical regulations started sharpening their heads around enantio-purity for drug candidates. Over the years, its story weaves through the evolution of synthetic strategy, drawing together community experience from academia and industrial process innovation.
Product Overview
(S)-4-Phenyl-2-Oxazolidinone appears as a white, odorless crystalline solid that crystallographers quickly recognize. Its clean lines reflect the reliable asymmetric environment it introduces in organic synthesis. Chemists picking up a bottle notice a compound that delivers repeatable performance. Unlike many fleeting lab intermediates, (S)-4-Phenyl-2-Oxazolidinone’s shelf stability and bulk availability has long since moved it from niche to staple in synthetic methodology. It seems to cross boundaries as both a research staple in teaching labs and a workhorse in GMP facilities.
Physical & Chemical Properties
The molecular formula C9H9NO2 leads to a molecular weight around 163 g/mol. This compound melts between 93°C and 97°C, with impressive purity in commercial lots. The phenyl ring at the 4-position on the oxazolidinone core backbone brings both rigidity and a handy handle for further functionalization. The structure mitigates hydrolytic vulnerability, giving good air and moisture stability. In most solvents common in organic synthesis—ethers, chlorinated hydrocarbons, and polar aprotics—its solubility permits efficient incorporation into reaction mixtures. It stands up well under typical stoichiometric and catalytic conditions in synthetic workflows.
Technical Specifications & Labeling
Manufacturers often package (S)-4-Phenyl-2-Oxazolidinone to meet ISO and cGMP guidelines when destined for pharmaceutical production. Lot-specific certificates include chiral purity, usually reported with enantiomeric excess exceeding 98%. Analytical documentation features HPLC and NMR traces. Safety labels present CAS number 23329-41-5, hazard pictograms, and signal words as per GHS standards. Storage recommendations usually mention keeping the material tightly sealed, cool, and dry. Such specificity allows researchers to compare batches with confidence and follow up on any analytical questions directly with suppliers.
Preparation Method
The classic laboratory route begins with the condensation of (S)-phenylglycinol and ethyl chloroformate or similar activating reagents. The cyclization follows smoothly in mild basic conditions, with careful temperature control needed to suppress side reactions and racemization. Industrial processes often substitute reagents for cost, safety, and environmental reasons but preserve the stereochemical outcome. Each new generation of preparation builds on greener solvents and more scalable purification steps, responding over time to environmental stewardship and worker safety regulations. Chemists continually optimize this part of the workflow, reducing waste and tightening yields.
Chemical Reactions & Modifications
(S)-4-Phenyl-2-Oxazolidinone thrives as a chiral auxiliary for enolate-based chemistry, especially in aldol, alkylation, and Michael addition reactions. Its ring nitrogen coordinates metal cations and chaperones intermediate species, enforcing facial selectivity in product formation. Practitioners appreciate a method’s reliability, subtle reagent effects, and the ease of auxiliary cleavage post-transformation, often using simple acid or base hydrolysis. Researchers have also explored modifications to its phenyl group, adjusting reactivity or solubility based on downstream process needs. The core oxazolidinone motif resists many oxidants and reductants, which gives a broad operational window for further transformations.
Synonyms & Product Names
Beyond its IUPAC designation, (S)-4-Phenyl-2-Oxazolidinone shows up on shipping manifests and catalogs as (S)-4-Phenyl-1,3-oxazolidin-2-one, and less formally as (S)-Evans’ Auxiliary. Some distributors use variants like (S)-4-Phenyl-2-Oxazolidone. Pharmacological and regulatory bodies typically prefer the CAS registry for traceability. Product names may vary internationally, which can cause confusion during procurement. Professionals often cross-reference CAS and lot numbers in procurement systems, eliminating mix-ups about chiral form or chemical structure.
Safety & Operational Standards
Users follow lab and plant safety protocols honed through years of handling chiral intermediates. Avoiding inhalation of dust and contact with eyes and skin matters most. Material safety data sheets note low acute toxicity but list respiratory irritation among possible hazards. Gloves, goggles, and lab coats remain standard kit, and fume hoods see regular use for weighing and transfers. Waste solutions pass through collection for solvent reclamation or incineration, reflecting the wider industry shift toward responsible chemical lifecycle management. Training on safe handling and exposure controls usually forms a foundation step for all users, and periodic audits maintain compliance with local regulatory frameworks.
Application Area
Pharmaceutical synthesis absorbs more than half of global (S)-4-Phenyl-2-Oxazolidinone output, particularly in the construction of complex drug scaffolds with precise stereochemistry. Chemists value its utility in making beta-lactam antibiotics, antiviral agents, and enzyme inhibitors, among other classes. The compound also crops up in research developing small-molecule libraries, chiral ligand design, and even specialty material applications. As regulatory bodies intensify scrutiny of drug enantiomers, research teams return to oxazolidinone auxiliaries for pilot-scale and full manufacturing runs. Buyers from biotech and chemical suppliers alike support demand cycles tied to innovative clinical programs.
Research & Development
Despite decades of industrial use, the research pipeline keeps generating methods that use or modify (S)-4-Phenyl-2-Oxazolidinone. Chemists continue to push the boundaries of asymmetric catalysis, increasing reaction scope and reducing auxiliary load. Recent publications describe catalytic cycles that regenerate or recycle the oxazolidinone, slashing material costs. Analytical chemists develop methods for faster, more sensitive monitoring of chiral purity in process streams. Cross-disciplinary projects in green chemistry investigate more sustainable feedstocks and waste minimization. Young researchers cut their teeth designing derivatives with specialized solubility or reactivity profiles, aiming for niche applications in sustainable materials and biological labeling.
Toxicity Research
Most available animal and cell studies indicate low toxicity for (S)-4-Phenyl-2-Oxazolidinone under standard handling and use conditions. Acute exposure studies in mice and rats show only minor irritant effects at high doses. In vitro assays suggest little potential for mutagenicity or reproductive toxicity. Regulatory agencies keep these studies under review, especially as downstream pharmaceutical metabolites may carry different toxicological profiles. Environmental researchers examine breakdown products in wastewater and assess bioaccumulation risks. So far, findings support continued use provided good lab practice is maintained, but users stay alert to any updates from toxicology reports and environment agencies.
Future Prospects
Looking forward, the role of (S)-4-Phenyl-2-Oxazolidinone will shift as synthesis and sustainability priorities change. Automation in chemical manufacturing may further standardize auxiliary usage and recovery. Machine learning tools are already helping design new reactions that stretch the utility of these chiral backbones, guiding chemists towards more efficient routes with smaller carbon footprints. Regulatory guidance on genotoxic impurities and micro pollutant monitoring could drive next-gen auxiliaries to become more biodegradable or even bio-based. Each phase of development draws from current industrial experience while folding in academic breakthroughs—building on what’s known without standing still. Researchers, regulators, and manufacturers continue to share results in open forums, advancing the entire field’s understanding and safe, effective use of (S)-4-Phenyl-2-Oxazolidinone in science and industry.
What is (S)-4-Phenyl-2-Oxazolidinone used for?
What Makes (S)-4-Phenyl-2-Oxazolidinone Useful?
In the world of chemistry, a single molecule can open doors to unique products or solve long-standing problems. (S)-4-Phenyl-2-Oxazolidinone stands out as one of those building blocks that chemists keep within arm’s reach, especially those who tackle organic synthesis in the lab daily. You might not spot its name in major headlines, but talk to any synthetic organic chemist, and this compound likely has a spot in their playbook.
Why Chemists Reach for It
This compound is mostly known for its role as a chiral auxiliary. That means it helps chemists steer reactions toward making just one mirror image of a molecule, which matters a lot when working with drugs. I remember the first time I ran a reaction that relied on this class of auxiliaries. Watching a reaction favor the particular direction I wanted—while filtering out its mirror image—felt like a little magic trick, but there’s hard science and careful planning behind it.
The pharmaceutical industry leans hard on the ability to control which mirror image (or “enantiomer”) a reaction produces. That’s not some niche concern. Drugs like ibuprofen, for example, come in two mirror-image forms. One treats pain efficiently, the other brings no benefit and can even produce unwanted effects. Getting the right form the first time saves money, time, and resources. (S)-4-Phenyl-2-Oxazolidinone makes that possible, acting as a guiding hand for the reaction. Researchers use these auxiliaries for synthesizing antibiotics, antivirals, and medicines for chronic conditions.
The Broader Reach in Research
Apart from medicines, chemists use this oxazolidinone to build complex molecules required in new materials and agricultural science. Since it can steer reactions with such precision, it shows up in the synthesis of flavors, fragrances, and advanced polymers. I’ve read studies where researchers chose this molecule to solve challenges in building up intricate ring structures in natural product synthesis—a task known for its difficulty.
Reliability and Safety
Researchers prize chemicals with predictable behavior. (S)-4-Phenyl-2-Oxazolidinone delivers on that front. Its characteristics stay consistent from batch to batch, so results remain stable. It also holds up well during storage and, with appropriate training, can be handled safely in a standard laboratory. These aren’t small considerations. In fast-paced labs, inconsistent chemicals can derail projects, waste months, and inflate costs.
Challenges and Future Solutions
One topic that’s hard to ignore in the lab is waste. After a reaction, the auxiliary gets removed—sometimes recovered, sometimes discarded. That process can generate solvent waste or leftover chemicals. We saw some improvements when colleagues tried greener solvents or found ways to recycle the auxiliary. For research teams thinking long-term, finding ways to cut down on steps, recover auxiliaries efficiently, or switch to ones with even lower environmental impact should stay a priority.
Final Thoughts
My own experience reminds me that chemistry often moves fastest through these small, underappreciated molecules. (S)-4-Phenyl-2-Oxazolidinone has proved itself by helping both the research lab and the manufacturing floor build safer, more effective drugs and products. That reliability, paired with ongoing efforts for cleaner chemistry, keeps this compound relevant among chemists who care about quality and progress.
What is the molecular formula and weight of (S)-4-Phenyl-2-Oxazolidinone?
Molecular Formula and Weight
Sometimes a name carries more value to chemists than to anyone else. (S)-4-Phenyl-2-Oxazolidinone does more than sound technical. The molecule stacks up as C9H9NO2. On a scale, it lands at 163.18 g/mol. That number comes from adding up each atom’s mass—carbon nine times, hydrogen nine, one nitrogen, two oxygens.
Getting from Structure to Impact
On paper, a formula only tells part of the story. Out in the lab, this molecule finds use as a chiral auxiliary in synthesis. Chemists care about its structure. Its chirality shapes how other molecules form, letting them make complex substances with control over arrangement. Picture a chef slicing vegetables with the same knife every time to get perfect shapes for a dish. In the same way, this chiral oxazolidinone gives chemists precision in building block after block, especially if handedness matters.
Pharma and Chemistry Value
Molecules like (S)-4-Phenyl-2-Oxazolidinone matter most where control over stereochemistry turns a basic recipe into a life-saving drug. Plenty of medicines need the “right” version of a molecule to work without side effects. One hand works better than the other, and sometimes the wrong one triggers trouble. Having an auxiliary that creates that exactly—without waste or trouble down the line—means more efficient research, safer products, and less chemical mess.
Safety and Practical Concerns
You don’t pull a molecule like this off the shelf and jump into work. Good labs train chemists to track every risk: gloves, fume hoods, storage in cool and dry spots. Each part of the molecule can bite, even if it looks harmless. A nitrogen or oxygen by itself doesn’t seem dangerous, but inside a ring, in the right environment, things shift.
From my experience, it only takes one lax moment for a slip-up. Someone I worked with years ago, rushing a reaction before lunch, found out the importance of precise measurement after a lid popped. That’s a sharp reminder—handle even familiar molecules with the respect they deserve. Written protocols and a double-check can make that difference.
Balancing Progress and Responsibility
As research ramps up, demand for enabling molecules keeps rising. Companies and universities chase newer, greener methods to make and recycle chiral auxiliaries like this one. Waste disposal keeps creeping up as a concern since the chemicals used in separations and the by-products don’t always play nicely with nature. Fact: over a third of chemical synthesis waste can be traced back to methods that use chiral auxiliaries, so reducing or recycling them can mean a smaller footprint.
Making a switch to processes that use less hazardous solvents, or options that break down into safer by-products, doesn’t just tick an ethics box. It trims costs for clean-up and lets good science stand out for more than just its results. Some teams have started looking at biocatalysis, using enzymes to replace or complement classic auxiliaries. Those efforts could shrink the pile of spent chemicals and give more precise results, all with milder conditions.
Conclusion
Chemical details like the formula C9H9NO2 or a mass of 163.18 g/mol matter to some, but the effects ripple out. The combination of smart technique, daily caution, and greener solutions can keep chemistry both sharp and safe for whatever comes next.
How should (S)-4-Phenyl-2-Oxazolidinone be stored?
Understanding the Substance
(S)-4-Phenyl-2-Oxazolidinone gets a fair bit of use in the lab. Chemists value it as a chiral auxiliary and intermediate in organic synthesis, often putting it to work in the hunt for enantiomerically pure compounds. Working with chemicals like this taught me early on that proper storage isn’t a throwaway detail. It can make the difference between a productive workspace and a safety incident.
The Role of Environment
Inconsistent storage conditions can have a big impact on purity and handling safety. This compound handles room temperature pretty well, but not all “rooms” are created equal. Humid basements and sun-baked shelves can change things fast. Moisture is the enemy here—left unchecked, it can degrade sensitive molecules and taint results.
Many labs use tightly sealed amber bottles for (S)-4-Phenyl-2-Oxazolidinone. Light can mess with chemical stability, so darkness usually helps. Even if you think the lights in the storage room aren't strong enough to cause problems, it’s not worth the risk, especially when the answer is a simple opaque container.
Container Choice and Labeling
Relying on plastic containers might seem convenient. From personal experience, glass offers real peace of mind. No worries about permeability, leaching, or reacting with solvents, even by accident.
Proper labeling isn’t just bureaucracy. Date of receipt, date of opening, and the name of the substance all matter. I once saw a colleague have to start a project over because the contents of a mystery bottle hadn't been labeled. Retrospective guesses don’t fly in regulated environments or careful research.
The Problem of Contamination
Cross-contamination has ended more experiments than I care to admit. Keep spatulas clean and scoops dedicated; don’t fish out material with whatever tool is handy. Even a trace of water can cause trouble, so always dry equipment before using it with hygroscopic materials.
My own disasters with moisture usually came from leaving a cap off for just a few minutes, under the illusion it wouldn’t matter. Crystal clumping, sticky residues, and unexpected melting points all teach the same lesson: close the lid, every single time.
Temperature Issues and Stock Management
While (S)-4-Phenyl-2-Oxazolidinone tolerates room temperature, keeping stock away from direct heat sources is a low-effort habit. Save yourself headaches and use a secondary desiccator if your lab offers one. Silica gel does more than gather dust; it grabs stray moisture that would otherwise sneak into open containers.
Shortages and waste come from overordering. Supervising inventory helps avoid resentful glances at expired bottles. Chemicals tend to degrade whether you use them or not. Opened bottles should go toward the front of the shelf to get finished before new ones.
Personal Protective Measures
Donning gloves, goggles, and a lab coat before handling chemicals is routine for me now, but too easy to skip in a rush. No synthesized product is worth a splash or an accidental exposure. Storing personal protective items alongside chemicals isn’t safe, either. Give them a separate space.
Building Good Habits
Consistent care in storing (S)-4-Phenyl-2-Oxazolidinone creates a safer lab, more accurate experiments, and a less stressful clean-up later. Even with the demands of fast-paced research, the small pause before recapping, relabeling, or shelving can keep a project—and its participants—on track.
Is (S)-4-Phenyl-2-Oxazolidinone hazardous or does it have any safety concerns?
What This Compound Means for Safety
Anyone working in a research lab quickly learns that even a small white powder can pose big questions. (S)-4-Phenyl-2-Oxazolidinone looks harmless at first, but ignoring its story is a mistake. I’ve handled similar oxazolidinones, watching colleagues rely on simple gloves and goggles. Most chemists don’t worry unless something smells sharp or burns skin. With this compound, risks don't jump out, but that doesn’t mean there aren’t any.
What the Data Shows
Current safety sheets put (S)-4-Phenyl-2-Oxazolidinone in the “handle with care” box. It irritates the eyes and the respiratory system, so it’s smart not to breathe any dust. I’ve seen people sneeze after opening a fresh bottle. There’s no record of serious toxicity in public databases so far, but underestimating mild symptoms is a gamble. Small molecules like this creep into the air easily, especially when the powder is fine or the workbench is dusty.
The chemical’s structure gives a clue. Oxazolidinones come up in medicinal chemistry, and a few relatives end up as antibiotics. That points at potential for biological activity. Sometimes this kind of activity brings surprises down the road—just because scientists haven’t documented long-term effects doesn’t mean they don’t exist. In the past, I’ve read through enough Material Safety Data Sheets to know careful habits keep everyone out of trouble.
Taking Safe Practices Seriously
Most accidents start not because people ignore warning labels, but because they get too comfortable. Horseplay or ignoring fume hood use leads to real risks. I remember an incident where a friend, working with another ring-based compound, skipped proper ventilation for a quick step. He developed a cough that lingered for weeks. Though (S)-4-Phenyl-2-Oxazolidinone hasn’t made headlines for severe injuries, the possibility of inhaling fine dust stays real.
Splashing solutions can also harm skin or eyes. Basic chemistry courses don’t usually stress enough that any unknown effect—sensitization, mild burns, or reactions with other chemicals—could show up after months on the job. Relying on past experience can lull researchers into a false sense of security. Robust lab routines, checked and followed, matter, even if everyone feels overconfident.
Stepping Up Safety Culture
Scattered stories about headaches or mild coughing barely make the rounds in journal articles. These are warnings, not formal studies, but they still add up. Improving safety starts with sharing information—open lab meetings, honest discussions about near-misses, and a culture where reporting small incidents becomes the norm, not the exception.
Stocking spill kits, keeping clean benches, and double-checking that hoods actually work cuts down risk, even if it feels like overkill. Clear labeling, updated safety sheets, and encouragement from senior researchers do more good than a few pages of fine print rules. I’ve seen groups where young trainees look out for each other, and as a result, accidental exposures drop. This kind of support shapes the tone for everyone else.
What Needs Improvement
Manufacturers need to dig deeper into chronic health data, especially for compounds like this one used in experimentation. Universities and labs should keep revisiting and updating internal safety guidelines as more evidence comes out. It also helps if suppliers demand users acknowledge risk before every purchase—an extra nudge to keep respect for the less-obvious dangers top of mind.
Chemistry moves forward with curiosity, but curiosity works best with good habits and healthy skepticism. (S)-4-Phenyl-2-Oxazolidinone doesn’t scream hazard, but treating it with consistent respect prevents surprises none of us want in the middle of an experiment.
What is the enantiomeric purity or optical activity of (S)-4-Phenyl-2-Oxazolidinone?
Digging Into the Chemistry
Enantiomeric purity shapes the reliability of any chiral compound, especially (S)-4-Phenyl-2-Oxazolidinone. In the world of organic synthesis, a single mirror image molecule can spell the difference between a groundbreaking pharmaceutical and a useless byproduct. True chemical precision demands that the specific enantiomer of a compound, like the (S)-enantiomer here, holds its intended three-dimensional form, free from contamination by its counterpart.
Why Purity Matters Beyond the Lab Bench
Chemical processes drive innovation in drug design and crop protection. When producing (S)-4-Phenyl-2-Oxazolidinone, stakes run high because even traces of the opposite enantiomer can undercut a reaction’s selectivity or introduce risk in clinical uses. Look at thalidomide’s history. One enantiomer soothed morning sickness, while the other caused birth defects. Mistakes like these leave scars that run deep across generations. Precise control—measured by enantiomeric excess or optical rotation—keeps modern chemists honest and patients confident.
Getting a Grip on the Metrics
Quality labs use chiral HPLC or polarimetry to pin down enantiomeric purity and optical rotation. (S)-4-Phenyl-2-Oxazolidinone shows specific optical activity, often reported in deg·mL·g-1·cm-1, reflecting its ability to rotate polarized light one way due to the (S)-configuration. Pure samples can show more than 98% enantiomeric excess—enough to keep downstream reactions in line without constant second-guessing. Less pure material could send a synthesis sideways or require wasteful extra steps to purify.
Direct Impact: Research and Manufacturing
Folks working in pharmaceutical development trust enantiomeric purity to deliver reproducible results. Synthesis of fine chemicals or pharmaceuticals using (S)-4-Phenyl-2-Oxazolidinone leans on these standards the way athletes lean on training. A misstep, even at the molecular level, brings consequences that ripple through the industry. Families counting on effective medicines deserve that scrupulous attention. Regulatory bodies do their part, too, setting strict approval hurdles for chiral products. Regulators like the FDA will check data on optical activity and purity as closely as they would any other crucial quality.
Raising the Standard: Solutions and Progress
Investing in better chiral synthesis techniques helps chemists reliably prepare pure enantiomers. Enzymatic methods, asymmetric catalysis, and thoughtful crystallization tricks have all improved since the 1980s. Robust quality assurance, frequent batch checks, and traceable data keep mistakes in check. Advancing detection—like coupling mass spectrometry with chiral chromatography—offers even tighter control. Everyone in the chain stands to gain: research is faster, manufacturing costs go down, and the end products give patients and growers safer, more effective tools.
Experience at the Bench
My own hours with chiral compounds taught me that “almost pure” rarely cuts it. Running polarimetry after a tedious separation either lifts your spirits or sends you back to rethink an entire week’s work. Catching a hint of the wrong enantiomer late in the process means lost time and burned budget. The more labs commit to verifying every batch, the more trust the wider world will have in these subtle, vital differences.
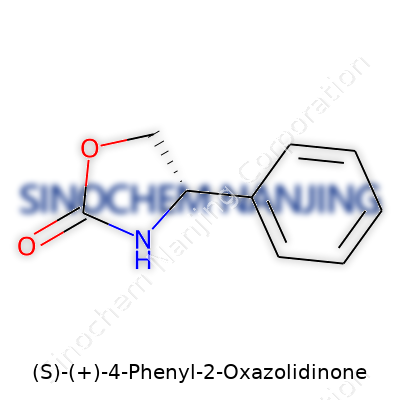
| Names | |
| Preferred IUPAC name | (5S)-4-phenyl-1,3-oxazolidin-2-one |
| Pronunciation | /ɛs fɔːr ˈfiːnɪl tuː ɒkˈsæzəlɪˌdaɪnoʊn/ |
| Identifiers | |
| CAS Number | 1443-25-6 |
| 3D model (JSmol) | ``` 4 JSmol structure C1[C@@H](NC(=O)O1)c2ccccc2 ``` |
| Beilstein Reference | 109203 |
| ChEBI | CHEBI:85152 |
| ChEMBL | CHEMBL146875 |
| ChemSpider | 10012420 |
| DrugBank | DB08796 |
| ECHA InfoCard | 06a33e8e-08e2-4427-8752-e5e2d2fc4cbc |
| EC Number | 626-48-2 |
| Gmelin Reference | 80536 |
| KEGG | C06353 |
| MeSH | D016679 |
| PubChem CID | 67504 |
| RTECS number | RG1870000 |
| UNII | 406Z6Q739Y |
| UN number | 2811 |
| CompTox Dashboard (EPA) | DTXSID2044408 |
| Properties | |
| Chemical formula | C9H9NO2 |
| Molar mass | 179.19 g/mol |
| Appearance | White to off-white solid |
| Odor | Odorless |
| Density | 1.2 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | 0.3 |
| Vapor pressure | 3.7E-6 mmHg at 25°C |
| Acidity (pKa) | 13.3 |
| Basicity (pKb) | 5.11 |
| Magnetic susceptibility (χ) | -79.3×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.570 |
| Viscosity | Viscosity: 98.3 mPa·s (20 °C) |
| Dipole moment | 4.74 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 359.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -458.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4266 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | N06AX16 |
| Hazards | |
| Main hazards | Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P261, P305+P351+P338 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 98°C |
| LD50 (median dose) | LD50 (median dose): >5000 mg/kg (Oral, Rat) |
| NIOSH | SR3500000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10g,25g,100g |

