The Many Faces of (S)-3-(1-Methylpyrrolidin-2-Yl)Pyridine: Tracing Its Roots and Future Trajectory
Historical Development
Any good discussion about (S)-3-(1-Methylpyrrolidin-2-Yl)pyridine owes something to the tangled history of alkaloid research in the 1800s. This compound—better known as S-nicotine in both research and industry circles—emerged from the efforts to untangle the chemistry of tobacco. Early chemists tackled isolation and structure with crude tools, yet their persistence paved the way for stereochemical studies that let us separate the S-isomer from its R-twin. By the late 20th century, more refined chromatographic and synthetic methods handed researchers reliable access to the target molecule. That opened a floodgate of neuropharmacological work and drove an entirely new appreciation for how stereochemistry shapes biology. The race to map its properties and implications wound its way through the pharmaceutical industry, public health policy, and regulatory debates around stimulant use.
Product Overview
Scientists and manufacturers know (S)-3-(1-Methylpyrrolidin-2-Yl)pyridine by many names; S-nicotine tops the list. The compound takes center stage in both natural products and synthetic chemistry, and its chiral purity can carry a price tag that tells you everything about the technical challenge involved. Synthetic versions show up in pharmacological studies as standards or starting points for drug design, and the compound’s legacy as the active form of nicotine cements its relevance in any tobacco research or replacement therapy program. In the lab, you can buy milligram to kilogram quantities, generally with strict traceability and high-purity certification to match the material’s use in regulated settings.
Physical & Chemical Properties
In pure form, (S)-3-(1-Methylpyrrolidin-2-Yl)pyridine stands as a colorless to pale yellow oily liquid with a pungent, unmistakable odor. This compound dissolves readily in water and most organic solvents, thanks to its polar nitrogen atoms and five-membered ring. The molecular formula, C10H14N2, points to a compact and hydrophobic core. Careful handling is a must: the liquid oxidizes easily, turning brown over time, and its volatility poses both inhalation risks and the challenge of keeping everything sealed tight in storage. The (S)-enantiomer rotates plane-polarized light to the left, which stands in contrast to the R form. The melting and boiling points—usually logged around -79°C and 247°C—show how the molecule shifts between phases with small changes in temperature.
Technical Specifications & Labeling
Regulated sales mean bottles of this material arrive with professional labeling, often indicating the enantiomeric excess, batch number, purity (often exceeding 98%), and a detailed certificate of analysis. Any reputable manufacturer will put the chemical’s CAS number front and center, along with recommended storage conditions and warnings regarding toxicological hazards. Labels usually steer you to consult full safety data sheets, as exposure risks extend beyond simple irritation: (S)-3-(1-Methylpyrrolidin-2-Yl)pyridine interacts strongly with the nervous system.
Preparation Method
Anyone who’s tried to synthesize enantiomerically pure alkaloids knows the frustration that comes with separating one mirror image from the other. For (S)-3-(1-Methylpyrrolidin-2-Yl)pyridine, production routes split into natural isolation from tobacco or total synthesis. Large-scale extractions treat cured leaves, drawing out complex alkaloid mixtures, then resolving the enantiomers using chiral chromatography or chemical resolution agents. Chemists also build the molecule from simple building blocks—often using asymmetric catalysis or stereoselective reduction—carefully steering the reaction to favor the S form. Yields and costs depend on the chosen method; isolation from plants can never match the consistency and purity of modern synthetic approaches.
Chemical Reactions & Modifications
The pyridine nitrogen and the pyrrolidine ring both open doors for chemical modifications, so S-nicotine doubles as both a research probe and a synthetic intermediate. Laboratory work often converts the base to salts by treating the free amine with acids, which improves stability and solubility for pharmaceutical use. Researchers target the nitrogen atoms for N-alkylation, or alter the ring by oxidation and reduction to probe receptor activity. Photochemical reactions and cross-coupling techniques further widen the range of analogs on offer, letting medicinal chemists test binding and efficacy across a range of biological targets. Chemical modifications can erase or amplify biological effects, so every change needs careful tracking through both analytic and biological assays.
Synonyms & Product Names
The most widely accepted synonym for (S)-3-(1-Methylpyrrolidin-2-Yl)pyridine is S-nicotine, underscoring its status as the physiologically active isomer of nicotine. Doug through chemical catalogs, and you’ll turn up labels like (S)-Nicotine, (S)-(-)-Nicotine, or simply levorotatory nicotine. Whenever regulatory agencies reference this substance, they use systematic names to avoid confusion with the racemic blend or the R isomer. This effort to keep terminology clear supports both safety and compliance, especially as regulatory scrutiny of nicotine has tightened worldwide.
Safety & Operational Standards
Folks working with (S)-3-(1-Methylpyrrolidin-2-Yl)pyridine must treat it with deep respect: Absorption through skin or by inhalation poses acute poisoning risks. Every serious lab keeps gloves, goggles, and proper ventilation in play. Standard operating procedures insist on spill kits and poison control details at hand, because even trace exposure can bring on nausea, dizziness, tremors, or worse. Training and restricted access shore up these defenses, and waste management rules tightly control both residue and solvents. Advances in engineering controls—like glove boxes and remote handling—reduce the odds of accidental exposure, yet personal vigilance anchors all safe practice.
Application Area
The lion’s share of interest in S-nicotine comes from its activity as a nicotinic acetylcholine receptor agonist. That property underpins work in neuropharmacology, addiction science, and toxicology. Pharma researchers seize on its chirality for drug screening or as a scaffold for new treatments aimed at neurodegenerative disease, psychiatric disorders, and pain modulation. Plant scientists probe its role in pathogen defense and pest resistance. Behavioral scientists track its effects in animal models to parse out the complex tangle of dependence, tolerance, and cognitive modulation. Product developers in smoking cessation reach for the S-isomer to improve nicotine gum and patch formulations, aiming for predictable absorption and reduced side effects. Its reach in science and medicine can’t be boxed in by just one industry.
Research & Development
The last five decades showed a dramatic uptick in research articles dedicated to (S)-3-(1-Methylpyrrolidin-2-Yl)pyridine. Analytical chemistry teams raced to quantify trace levels in blood and environmental samples, fueling big studies on population exposure and metabolism. Drug discovery jumped on S-nicotine for testing new receptor modulators or imaging agents for PET scanning. Refinements in chemical synthesis keep pushing down costs, letting labs explore larger screens of derivatives. Increasingly, research teams lean on computational modeling tied to in vitro and in vivo work, tying together binding data with real-world pharmacological output. All these trends make clear that S-nicotine remains a hub for technical innovation.
Toxicity Research
Toxicologists know how little margin for error exists with S-nicotine. Experiments in animals trace the dose-response curve with fine precision, and human toxicology underscores the speed of absorption and the severity of overdose. Acute exposure symptoms—vomiting, tremors, respiratory distress—match what many smokers learn the hard way, but purified S-nicotine delivers effects faster and with more punch than crude tobacco. Chronic studies raise worries about mutagenic and teratogenic effects, especially with repeated dosing. Newer cell culture and molecular techniques keep chipping away at the details, but the basics remain unchanged: S-nicotine acts as a potent neurotoxin, with a safety threshold narrower than most pharmaceutical agents. Toxicity data push regulators and industry to continually re-examine exposure limits for workers, researchers, and the general public.
Future Prospects
Fields as diverse as neuroscience, public health, and drug development show no sign of losing interest in (S)-3-(1-Methylpyrrolidin-2-Yl)pyridine. Research pipelines for new neuroactive drugs keep drawing on the molecule’s strong receptor targeting. Advances in analytical testing promise to pick out ever-smaller exposures in biological and environmental samples, fueling new waves of epidemiological work. Safety engineers look for better barriers and early-warning monitoring as handling protocols tighten. Synthetic chemistry still aims for greener, more efficient pathways to both the S and R isomers, reflecting pushback against the environmental footprint of large-scale syntheses. As regulatory standards for nicotine shift globally, S-nicotine stands as both a model and a challenge—an old molecule constantly folded into new science. The ground beneath it keeps shifting, and only continued vigilance, curiosity, and open collaboration will shape where this story goes next.
What is the primary use of (S)-3-(1-Methylpyrrolidin-2-Yl)Pyridine?
Understanding Its Place in the World
(S)-3-(1-Methylpyrrolidin-2-yl)pyridine may not sound familiar to most people, but nicotine certainly does. This chemical isn’t just a tongue-twister—it’s the scientific name for the nicotine molecule in its “S” form. Out in the field, farmers know this compound as the potent natural alkaloid in tobacco plants. Smokers recognize nicotine by the buzz it gives or the challenge it creates during a quit attempt. Inside the lab, chemists see possibilities beyond addiction.
Nicotinic Focus: The Chemistry Behind Habit and Health
The primary use of this compound centers on its role as the main active ingredient in tobacco. Almost every cigarette, vape device, or chewing tobacco product delivers nicotine to the body, where it binds to nicotinic acetylcholine receptors in the brain. This connection produces stimulating effects—wakefulness, focus, even mood elevation. People have grown, processed, and smoked tobacco for centuries, but nicotine’s biological pull drives those habits. Researchers have pinpointed the “S” enantiomer as the form mostly found in tobacco and absorbed by users.
Beyond Smoking: The Value in Medicine and Research
Nicotine has a notorious reputation, but its story doesn’t stop with addiction. In the last decade, scientists started looking closely at (S)-3-(1-Methylpyrrolidin-2-yl)pyridine for potential benefits. Pharmacologists explore its effects on memory and attention, particularly for neurodegenerative conditions like Alzheimer’s and Parkinson’s. Some laboratory studies suggest that nicotine—at controlled low doses—can boost cognitive function and possibly protect brain cells. The goal isn’t to encourage nicotine use, but to isolate helpful effects from harmful delivery methods like smoking.
Nicotine in the Lab: A Critical Tool
Every major medical school and many pharmaceutical labs work with this compound. It serves as a reference standard for studying addiction pathways and neurological function. Scientists use it to test new drugs that target brain signaling, since nicotine’s molecular action is well-charted. For example, a group of researchers I met at a university in Boston measure how nerve cells fire in response to nicotine, comparing healthy brains with those coping with disease. Their findings could lead to treatments that mimic nicotine’s benefits without the harmful effects of tobacco.
Societal Impact and Regulatory Debate
Decades of public health campaigns, cigarette taxes, and regulatory changes all center on nicotine’s power. Nearly every major effort to curb smoking has focused on reducing access to this compound or helping people break free from its grip. The rise of vaping, nicotine patches, and gum reflects society’s struggle to both manage addiction and reduce harm. Authorities have tried lowering the nicotine content of cigarettes to make them less habit-forming, but results remain mixed. Some folks quit entirely, others switch to alternative products, and a portion simply keeps smoking.
Opportunities and Challenges
Future solutions should combine strong science with compassion. Reducing smoking rates will always matter, but there’s promising territory in research aimed at new therapies for brain health. Public education must walk a tightrope—discouraging recreational use, while supporting scientific progress. Honest science keeps doors open to treating illness while shutting out needless harm. The story of (S)-3-(1-Methylpyrrolidin-2-yl)pyridine isn’t just about chemistry. Its wider impact touches health, policy, and even hope for those struggling with addiction.
Is (S)-3-(1-Methylpyrrolidin-2-Yl)Pyridine safe for human consumption?
Looking Past the Chemical Name
Ask most people about (S)-3-(1-Methylpyrrolidin-2-Yl)Pyridine, and you'll probably get some blank stares. Most folks know it as S-nicotine, the same nicotine found in tobacco, vaping products, and sometimes in smokeless alternatives. The chemical jargon hides an everyday reality: this molecule is behind addictions, livelihoods, debates in public health, and family troubles at the dinner table.
The Everyday Impact of S-Nicotine
Nicotine slips into people's routines quietly. Cigarettes often show up in stories from my dad’s generation. He thought they made him look grown up, and the urge never really left. Now, with vaping everywhere, the old nicotine problem pops up in fresh packaging. The delivery changes, but the molecule stays the same.
Researchers have studied nicotine’s role in the body for decades. It kicks the nervous system into gear, raising heart rate and blood pressure. It’s no urban legend—withdrawal makes some folks agitated, distracted, or even depressed. Hard to ignore, especially when watching friends bounce between quitting and relapsing. Until you've seen it, you might underestimate the grip of this molecule.
Science and the Data
Reports connect nicotine to risks for heart disease, stroke, and other illnesses. The U.S. Surgeon General highlighted nicotine’s addictive power years ago, and new research often circles back to dependence as a key concern. Kids and teens draw extra concern. Their brains keep developing through their twenties, and nicotine seems to mess with this process. The CDC points to links between adolescent exposure and later life problems—memory, attention span, and risk of later addiction.
Controversy flares around products claiming to be “pure” or “safer” forms of nicotine. Whether inside a cigarette, a vape cartridge, or synthetic pouches, S-nicotine doesn't lose its habitual nature. The method changes, not the chemical punch. Imagine seeing someone pick up a vape after finally dropping cigarettes. The sense of relief gets tangled up with worry. My own friends swap one dependency for another, thinking they’ve picked a lesser evil—but their bodies don't always agree.
The Legal View and Safety Debate
Governments step in where companies don't tread. Nicotine restrictions change from country to country. In the U.S., the FDA regulates tobacco and vaping products but draws criticism for not moving fast enough. Some countries ban nicotine pouches or restrict sales to certain ages. None claim it’s harmless.
Many folks still believe nicotine by itself isn’t the enemy—some blame the chemicals added to cigarettes or dangers of smoking itself. But evidence piles up: even without tar or smoke, nicotine tags along with a long list of risks. Healthcare professionals keep hammering home that “safe” or “natural” doesn’t mean risk-free.
How to Move Forward
Education matters. Young people benefit from frank conversations, not just lectures or scare tactics. Parents, teachers, and community leaders hold more power than they think in shaping attitudes. Regulation plays a role, but informed choices pack the real punch.
Support networks push people forward when quitting feels impossible. State quit-lines, group therapy, and apps can tip the scales. Harm reduction makes a difference for some, but only because it meets folks where they are. The hope lies in combining public health messages, community support, and access to science-backed resources—not in chasing the next best “safer” product.
What are the storage conditions for (S)-3-(1-Methylpyrrolidin-2-Yl)Pyridine?
Understanding the Importance of Proper Storage
Chemicals, whether for research, industry, or medical use, don't handle neglect well. Getting the storage right for something like (S)-3-(1-Methylpyrrolidin-2-Yl)Pyridine isn't just about following the rules—it's about caring for safety and the quality of what you’re working with. I remember back in my undergraduate days, one simple mistake with chemical storage led to a batch of degraded reagents. That ended up wasting precious time and money for everyone in the lab.
Temperature Control: No Shortcuts
(S)-3-(1-Methylpyrrolidin-2-Yl)Pyridine, better known to some as S-nicotine, remains stable at lower temperatures. Aim for a cool, dry space, ideally with temperatures kept between 2°C and 8°C. In practical terms, this usually means a refrigerator built for laboratory chemicals—never the same fridge you use for food or drinks. At room temperature, the risk goes up for oxidation and decomposition, which likely leads to subtle (or not so subtle) changes in chemical structure. That’s a disaster for any kind of precise scientific work.
Avoiding Moisture and Light
Many pyridine derivatives attract water. Moisture can cause hydrolysis or simply promote unwanted reactions over time. Desiccators come in handy for that reason. Use a tight-sealing vial, and toss in a desiccant packet for extra protection. This habit kept my own research from running into unexpected results more than once. Light sensitivity often gets overlooked, but prolonged exposure tends to slowly tweak the compound as well. Store bottles in amber-glass containers and keep them away from direct sunlight or the harsh fluorescent bulbs common in labs.
Label Everything and Secure the Area
I’ve seen what happens when chemicals end up mislabeled or get stored next to incompatible substances. It doesn’t end well. Clear, dated labeling on every bottle—include not only the name but the concentration, date received, and any hazard warnings. Even minor nicotine derivatives demand careful handling. Store these away from acids, bases, and oxidizers. Lock cabinets or secured chemical storage rooms act as more than just compliance with guidelines; they keep curious hands and unsuspecting staff from dangerous accidents.
Understanding Daily Use and Access
Anyone who works hands-on with research chemicals can tell you: Accidents happen fast. Secondary containment trays catch leaks and spills. Spill kits on the shelf become a daily necessity rather than a formality. I once had to rely on one after a bottle slipped from someone’s hands—without quick action and the right backup, that could have caused harm and cost a week’s worth of research.
Disposal: Not an Afterthought
Expired or unused (S)-3-(1-Methylpyrrolidin-2-Yl)Pyridine doesn’t belong down the drain or in general trash. In regions like the US, strict hazardous waste disposal regulations apply. Following these rules prevents environmental contamination and keeps labs in good standing with both local and federal agencies.
Fact-Based Solutions for Safe Storage
Solid training programs and a disciplined approach to documentation cut down on mistakes. Lab managers should review safety data sheets (SDS) with researchers regularly. Regular checks on temperature logs show up potential problems before the chemical degrades. In my experience, a strong culture of safety—built on careful training and well-maintained equipment—keeps both people and chemical supplies firmly out of trouble.
How should (S)-3-(1-Methylpyrrolidin-2-Yl)Pyridine be handled in the laboratory?
Understanding the Substance: More Than Just a Name
(S)-3-(1-Methylpyrrolidin-2-Yl)Pyridine sounds like a nightmare from organic chemistry class. In reality, it’s another name for nicotine in its S-enantiomer form. Most folks running a lab recognize nicotine as a powerful and toxic alkaloid, not something to take lightly. Even if you think you know all the habits and short-cuts, this is stuff that finds a way to surprise the careless. It packs neuroactive effects on animals and humans, and brief skin contact can send grown researchers scrambling to the sink, heart pounding and head swimming.
Personal Safety: You’re Not Invincible
I remember grabbing a bottle once, thinking the gloves I wore were tough enough. Minutes later, my palms tingled, and the room spun. Lesson learned, for good. Strong, chemical-resistant gloves beat thin latex every time, and safety goggles block the splash no one expects until it happens. Wearing a lab coat makes sense, but that’s only part of the gear. Dedicated fume hoods matter, too; breathing nicotine fumes will mess up your day faster than you’d think. No one should ever pipette by mouth. That sounds obvious now, but it took decades—and hard-learned lessons—for labs to stop, so double-check your habits.
Spills and Storage: Thinking Ahead Prevents Chaos
Nicotine evaporates. A careless spill leaves vapors hanging in the air, ready to race into someone’s bloodstream through their lungs or skin. Spills turn into emergencies because lab mates often freeze up or grab whatever is handy, only making it worse. In my old lab, we had a protocol taped above the bench: ventilate, evacuate, and call in trained staff if you see more than a droplet. Lint-free wipes, absorbent pads, and sealed sharps containers ate up sudden leaks without sending dust everywhere. A chemical-resistant tray under all liquid stocks kept bottles from tipping and breaking. Secure containers with tight-fitting caps, and label everything, no matter how pressed for time you feel.
Disposal: Ethics and Laws Over Convenience
Sink disposal looks tempting after a long day, but nicotine counts as hazardous waste. It harms aquatic life and pollutes water in ways that stick around for decades. I always used specialized disposal bins. We logged every gram for our institution’s hazardous waste tracking. Never dump diluted solutions down the drain, even if someone claims, “Everyone does it.” Lab audits pick up on shortcuts and leave teams looking for excuses. Following environmental protections means more than guarding your own workspace; it keeps research institutions out of legal and ethical trouble that can end careers.
Training and Accountability: No One Works Alone
Shifting these habits to the next generation of lab workers takes hands-on demos and open conversations about near-misses. Coworkers should feel safe raising their hands if something feels off, or if memory fails on which waste container to use. Too many labs operate with quiet fear of embarrassing questions, but overconfidence proves costlier than simple mistakes. Official material safety data sheets offer specifics, but routines grow safe through community, not just paperwork.
Solutions to Keep Labs Safer
Automatic chemical-handling systems boost safety, but smaller labs manage with careful human attention. Scheduling regular checks on storage, setting up spill kits in plain reach, and building a culture that values questions all make more difference than expensive equipment. Supervisors who correct carelessly stored samples or missing labels invest in people, not just process. These lessons root in experience, not bureaucratic reminders.
Are there any known side effects or hazards associated with (S)-3-(1-Methylpyrrolidin-2-Yl)Pyridine?
The Chemical Behind the Scene
(S)-3-(1-Methylpyrrolidin-2-Yl)Pyridine doesn’t pop up during daily life, but its close cousin, nicotine, most definitely does. Chemists call this compound S-nicotine, the natural form found in tobacco leaves. A cigarette in hand or a nicotine patch on skin both deliver this very substance into the bloodstream, so any discussion of its risks needs to get honest about everyday exposure.
Human Health Hazards We Can't Dismiss
Nicotine in this form acts fast. After inhalation or absorption, it heads straight to the brain and messes with nerve signals. People who have tried tobacco products know that racing heart and a sharp kick of alertness. This happens because nicotine binds to acetylcholine receptors, confusing the body’s regular messaging system. It gets addictive in a hurry—this comes from how the brain starts craving that kick and wiring itself to chase repeated doses.
Health authorities from the Centers for Disease Control and Prevention and the World Health Organization link this chemical to multiple concerns. Doses high enough, regardless of delivery method, can bring on nausea, vomiting, dizziness, increased blood pressure, and rapid heartbeat. Larger accidental or deliberate doses bring more serious effects—convulsions, respiratory paralysis, and sometimes death. Every year, cases pop up where pets or children chew on nicotine gum, patches, or e-cigarettes and end up in emergency rooms.
Chronic Exposure: The Hidden Toll
People who smoke or use nicotine regularly shoulder bigger risks long-term. Habitual nicotine increases the chances of developing heart disease and blood vessel issues by narrowing arteries and raising blood pressure every single day. Type 2 diabetes becomes more likely because of insulin resistance. Mental health often gets tangled up too, with more intense withdrawal, anxiety, and mood swings. The facts are blunt: Big tobacco companies have poured years into researching just how much this chemical can grab hold of the brain and body.
Not Just Smokers: Children, Pets, and the Collateral Damage
No talk about this chemical would be honest without mentioning accidental exposures. Kids getting into e-liquids or adults using concentrated nicotine for DIY vape juices face real threats. The stuff absorbs through skin, and sometimes only a splash leads to symptoms. Calls to poison control centers climbed once flavored, colorful “juice” bottles became more common in homes. Pets, especially curious dogs, can suffer poisoning by chewing discarded patches or containers.
Workplace and Environmental Risks
Growers and workers in the tobacco industry sometimes report “green tobacco sickness.” After handling wet tobacco leaves, the skin absorbs enough nicotine to cause headaches, nausea, and muscle weakness. Protective gloves and washing up keep workers safer, but the risk sticks around if precautions lapse.
Paths Toward Safer Use
Doctors, pharmacists, and regulators agree: keeping nicotine—no matter the form—far from kids and pets matters most. Lock up all products containing it. Labels need to be clear, tamper-proof, and include warnings. Researchers keep working to develop less addictive alternatives for nicotine replacement therapies, using gradual tapers to limit withdrawal and reduce cravings. Public health campaigns stress not just the dangers of tobacco, but the chemical heart of dependence itself.
Making Informed Choices
Plenty of folks underestimate the hazards of pure S-nicotine just because they grew up around cigarettes. Experience tells me that after seeing family members wrestle with heart disease and addiction, it’s easy to wish everyone had more knowledge about these side effects earlier in life. Honest conversations, smart regulation, and personal caution can chip away at the dangers wrapped up in this familiar, but risky, molecule.
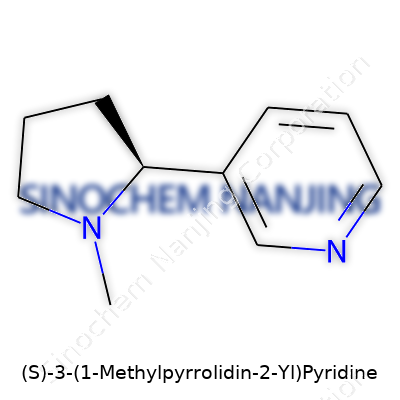
| Names | |
| Preferred IUPAC name | (2S)-3-(1-methylpyrrolidin-2-yl)pyridine |
| Other names |
S-Nicotine (–)-Nicotine |
| Pronunciation | /ɛs θriː wʌn ˈmɛθɪl pɪˌroʊlɪˈdiːn tuː aɪl paɪˈrɪdiːn/ |
| Identifiers | |
| CAS Number | 34624-72-5 |
| Beilstein Reference | 3598734 |
| ChEBI | CHEBI:31944 |
| ChEMBL | CHEMBL12306 |
| ChemSpider | 123802 |
| DrugBank | DB01381 |
| ECHA InfoCard | 12bbd40a-2d8f-41a0-a268-1ba91999d5d7 |
| EC Number | 872-81-5 |
| Gmelin Reference | 81638 |
| KEGG | C08355 |
| MeSH | D009538 |
| PubChem CID | 13709911 |
| RTECS number | UJ8750000 |
| UNII | 0Y780IA44Y |
| UN number | UN1993 |
| CompTox Dashboard (EPA) | DJ6PQ7Y3LO |
| Properties | |
| Chemical formula | C10H14N2 |
| Molar mass | 176.26 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | strong |
| Density | 1.12 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.03 |
| Vapor pressure | 1.7X10^-5 mm Hg at 25°C |
| Acidity (pKa) | 7.77 |
| Basicity (pKb) | 5.85 |
| Magnetic susceptibility (χ) | -68.4e-6 cm³/mol |
| Refractive index (nD) | 1.553 |
| Dipole moment | 2.59 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 362.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4427 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | N07BA01 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | CC1CC[N@H](C1)c2cccnc2 |
| Signal word | Warning |
| Hazard statements | H302, H312, H332, H351 |
| Precautionary statements | P261, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-0-🛇 |
| Flash point | Flash point: 96.9 °C |
| Lethal dose or concentration | LD50 (oral, rat): 50 mg/kg |
| LD50 (median dose) | 50 mg/kg (Rat, oral) |
| NIOSH | 0003 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 100 mg |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Nicotine Nornicotine Anabasine Myosmine Cotinine |

