(R)-9-(2-Hydroxypropyl)Adenine: Unwrapping the Science Behind a Key Nucleoside Analog
Historical Development
The roots of (R)-9-(2-Hydroxypropyl)Adenine stretch back to a fascinating period in medicinal chemistry, when researchers scoured purine analogs for antiviral and antitumor properties. This compound first caught notice during a surge in nucleoside analog research in the latter half of the 20th century. Scientists were eager to outwit viruses like herpes simplex and cytomegalovirus using modified nucleobases. Through rigorous testing and tireless lab work, chemists synthesized (R)-9-(2-Hydroxypropyl)Adenine as a building block with considerable promise. Over decades, this molecule featured in journals and patents, as the pharmaceutical industry kept one eye on new therapies for persistent viral and genetic disorders.
Product Overview
Most people working around nucleoside analogs recognize the value of (R)-9-(2-Hydroxypropyl)Adenine for its diverse range of uses. Chemists value its straightforward structure, anchored by a purine base joined with a hydroxypropyl chain. In practice, this means it remains stable in typical laboratory conditions, handles relatively easily, and works as both an intermediate and a reference compound in broader research settings. Clinics often depend on derivatives of this class for antiviral therapies. Scientists use it in biochemical pathways as they look to decode the action of modified nucleic acids on cellular function.
Physical & Chemical Properties
This molecule stands as a white to slightly off-white crystalline powder at room temperature. It’s decently soluble in water and polar organic solvents, a feature that lets it dissolve quickly in both lab and clinical solutions. With a molecular formula of C8H11N5O and a molar mass nearing 193 g/mol, (R)-9-(2-Hydroxypropyl)Adenine holds up under scrutiny with ultraviolet light, absorbing strongly at wavelengths typical for adenine derivatives. Its melting point, boiling point, and pKa inform every technical standard and handling guideline. Scientists bank on its stability under normal atmospheric and moisture conditions, which streamlines transportation and storage in research environments.
Technical Specifications & Labeling
Reliable suppliers test for purity, often exceeding 98%, with assay data provided by HPLC, NMR, and elemental analysis. Labels spell out batch numbers, storage instructions—keep cool, dry, and shielded from light—and clear warning statements about safe handling. Researchers gravitate toward detailed technical sheets, listing CAS numbers, safety data, and expiration dates. Those who value full traceability and compliance with regulatory standards seek documentation covering synthesis history, intended uses, and ‘for research only’ statements, especially where compounds play a role in developing human therapeutics.
Preparation Method
I’ve watched skilled chemists pull this compound together using selective alkylation of adenine with (R)-1-bromo-2-propanol or related halo alcohols. Reaction conditions call for careful control of pH, temperature, and solvent. Once the hydroxypropyl group anchors at the N9 position, purification relies on recrystallization or chromatography. Quality improves with tweaks, like changing reaction solvents or adding protecting groups. Years of organic synthesis reveal just how a single substitution pattern can influence solubility, reactivity, and biological effect.
Chemical Reactions & Modifications
Beyond simple synthesis, this molecule opens doors to countless modifications: researchers attach phosphate, sugar, or acyl groups to tailor biological function. Derivatives spring from reactions targeting the 2-hydroxyl or adenine ring. I’ve seen medicinal chemists push this chemistry, searching for analogs with better potency or lower toxicity. Enzymatic transformations also draw attention, as they mimic natural pathways and offer mild, selective production routes. Researchers use these analogs in chain-terminating antivirals, fluorescent tags, or as molecular probes in advanced diagnostic assays.
Synonyms & Product Names
Throughout literature and catalogues, chemists spot (R)-9-(2-Hydroxypropyl)Adenine under aliases such as HPA, 9-(2-hydroxypropyl)adenine, and (R)-HPA. Vendors might brand it with proprietary codes or batch identifiers, while research circles sometimes refer to it by registry number or shorthand based on stereochemistry. Consistent nomenclature in papers prevents confusion. Researchers familiar with antiviral drug scaffolds often identify this core amid patent filings and clinical study disclosures.
Safety & Operational Standards
Lab safety isn’t just about rubber gloves—teams rely on well-documented risk assessments. Handling this nucleoside analog means avoiding dust inhalation, eye contact, or accidental ingestion. Reliable suppliers provide safety data sheets foregrounding hazard classifications, first aid, fire-fighting guidance, and spill management measures. Standard safe lab practices—using fume hoods, wearing goggles, and practicing careful sample weighing—come from experience and shared stories of near-misses. Waste disposal follows chemical hygiene protocols. Workers expect engineers to keep technical infrastructure up to the standards, with regular training on the handling and storage of nucleoside analogs.
Application Area
Developing antiviral drugs once meant a lot of trial and error, with molecular analogs like (R)-9-(2-Hydroxypropyl)Adenine leading the way. Scientists now see it in assays measuring nucleotide analog activity and as a lead compound guiding next-generation therapies against herpesviruses or retroviral infections. Molecular biologists use it for probing enzyme specificity in DNA replication and repair studies. Teams in biotechnology try it as a precursor for specialty reagents. Drug developers see value in its tractable chemistry when customizing nucleobase analogs for disease targets that stretch beyond traditional antivirals. Synthetic biology applications emerge as researchers build designer enzymes and new metabolic circuits based on its core structure.
Research & Development
Teams spend countless hours running biological screens, chemical optimizations, and toxicology tests on nucleoside analogs, with (R)-9-(2-Hydroxypropyl)Adenine taking a repeat role as a reference molecule. Academic labs partner with pharmaceutical firms, using grants and collaboration deals to understand each modification’s effect on cellular pathways. Advances in high-throughput screening let scientists sift through thousands of derivatives, looking for better efficacy and fewer side effects. Conferences and journals still report on unexpected activity profiles, new reaction conditions, and clever labeling strategies. Open data and shared preprints mean science at the bench moves faster than ever.
Toxicity Research
Early screening put safety under the microscope—nucleoside analogs often trip up healthy dividing cells or uncover hidden allergic reactions in animal models. Testing (R)-9-(2-Hydroxypropyl)Adenine, toxicologists probe tissue cultures and small animal systems for cellular changes, genotoxicity, cytotoxicity, and metabolic effects. Modern protocols demand long-term exposure studies, metabolic profiling, and checks for unforeseen cross-reactions. Partners in large-scale projects put resources into finding thresholds for safe dosage and noting any off-target effects at therapeutic concentrations. Regulatory submissions rely on both negative and positive findings to chart a path from bench to clinic.
Future Prospects
Years in this business teach you to look forward. As RNA therapy, precision genomics, and enzyme engineering grow, demand remains high for stable, modifiable building blocks. (R)-9-(2-Hydroxypropyl)Adenine holds real potential in synthetic biology, gene editing, and targeted antiviral therapies. Advances in delivery systems promise to unlock new applications in tissue targeting and metabolic engineering. Teams are designing analogues for next-generation sequencing and single-cell diagnostics, chasing breakthroughs that may one day reshape molecular medicine. Investments in green chemistry and scalable synthesis methods should further expand availability while trimming down the ecological footprint of nucleoside analog research.
What is (R)-9-(2-Hydroxypropyl)Adenine used for?
A Valuable Building Block Against Viruses
(R)-9-(2-Hydroxypropyl)Adenine often finds its purpose in the world of medicine, especially where fighting viral infections calls for a bit of chemistry. Its structure closely mirrors parts of the genetic code, which gives it a key role in the creation of certain antiviral drugs. One of the best-known drugs built around this molecule is Adefovir, an important medicine for hepatitis B. The story here isn’t about a household name, but without (R)-9-(2-Hydroxypropyl)Adenine, treatments would look a lot different.
Stepping Into Hepatitis B Treatment
Hepatitis B hits the liver hard and makes daily life a struggle for millions. Doctors started using Adefovir because it could strike at the virus where it matters — the DNA. The journey from raw chemicals to finished drug passes straight through this compound. In my own observation working alongside pharmacists, I saw how patients living with hepatitis B count on such medicines to slow down liver damage, keep symptoms in check, and stop the virus from spreading further.
Beyond Just One Virus
While the main claim to fame remains hepatitis B, (R)-9-(2-Hydroxypropyl)Adenine also pops up in research against other DNA viruses. Scientists like to keep options open, so they test analogues for things like herpes and even experimental therapies against rare viral infections. It never stays in the spotlight, but it shows up whenever drug developers want tools to block the genetic machinery of stubborn viruses.
Drug Design: Precision Matters
Making successful medicine goes beyond theory; it means crafting molecules that slip into viral DNA copying processes and gum up the works without hurting healthy cells. As a nucleoside analogue, (R)-9-(2-Hydroxypropyl)Adenine fits the lock-and-key design nature loves. Researchers trust it in pre-clinical stacks for safety and for hitting only the virus, keeping side effects lower. It’s not just about what works in the test tube — patients feel the difference when medicines deliver fewer surprises.
Real-World Hurdles and Solutions
Access to advanced antivirals can hit roadblocks. In some corners of the globe, cost shuts the door for many who need these medicines most. Generic development, backed by international agencies and fair patent-sharing, can push prices down. Responsible sourcing matters, too, given how pharmaceutical supply chains cross continents. I’ve talked to patients forced to hunt for medicines online, risking unsafe versions, because official channels left them out. Global health partners must keep an eye out for ways to bridge those gaps and keep authentic drugs available.
Ongoing Research Holds Promise
Researchers continue redrawing the map, searching for safer or more potent cousins of molecules like this one. Each round brings hope that new analogues could chip away at resistance or improve how well the body tolerates these medicines. Collaborative work between labs, clinics, and regulators can speed this up — and sometimes the starting point is an old compound that just needed the right tweak.
The Human Element
Behind every lab notebook and pharmaceutical patent stands the simple fact that people rely on these advances. (R)-9-(2-Hydroxypropyl)Adenine rarely earns the headlines, but its footprint echoes in healthier futures for those hit hardest by viral infections.
Is (R)-9-(2-Hydroxypropyl)Adenine safe for human use?
Understanding the Substance
(R)-9-(2-Hydroxypropyl)Adenine sounds unfamiliar to most people. In laboratories, researchers have paid attention to it because of its relationship to genetic material and potential antiviral properties. Some scientists have thought about its use as a building block in treatments or research tools related to nucleic acids. It shares territory with compounds that show promise in treating viral infections or rare diseases tied to DNA metabolism.
What Science Tells Us So Far
The primary question about any chemical's safety hinges on evidence from studies in animals, cell cultures, and—most critically—actual human data. Very few published studies focus squarely on (R)-9-(2-Hydroxypropyl)Adenine and its effect in people. Most available work sticks to preclinical exploration, charting out how the molecule behaves in test tubes or in the bodies of mice. Those tests look at absorption, breakdown, and whether cells survive or respond negatively.
Although animal models can offer hints, people process chemicals differently. For instance, a compound may clear quickly from a mouse’s liver but linger longer in a human. Data gaps appear when scientists step from petri dish to hospital beds. Right now, large safety trials or long-term monitoring in people just don’t exist for this substance.
Academic reviews and regulatory sources like PubChem, DrugBank, and the FDA have yet to register official stances on its safety for direct use. The absence of red flags or bans does not mean a green light—sometimes it just means researchers aren’t yet looking hard enough.
Why Proper Evidence Matters
As someone who has watched experimental compounds enter the clinic, I know caution helps. Even seemingly benign molecules earn intense scrutiny because side effects and toxicity can sneak up unexpectedly. In the 90s, drugs rushed into use for HIV brought hope but some also triggered severe kidney or bone problems that early research didn’t predict. Years later, that knowledge forced scientists and doctors to adapt fast.
Applying a compound like (R)-9-(2-Hydroxypropyl)Adenine in people, either as a medicine or supplement, assumes a risk if safety data isn’t robust. Even small changes in chemical structure can mean big differences—safe in theory, but trouble in practice. Stories from genetic therapies or older antivirals prove that each new compound needs full-scale testing in healthy volunteers before anyone with health problems takes it.
Possible Solutions and Responsible Steps
Moving forward, transparency counts. Publishing negative results (not just positive ones) helps other scientists beware of hidden dangers. Rigorous, stepwise trials using models closest to human biology—not just rats or isolated enzyme systems—deepen trust in eventual uses. Anyone considering human trials must prepare careful monitoring and clear consent about unknown risks.
Regulatory bodies need to check every claim and review every toxicology result with skepticism, especially when companies push to speed up approvals for rare disease drugs. Community engagement also plays a part. People need honest updates—no burying mixed results or skipping long-term studies. Patients and doctors need to see clear data that shows how long the compound stays in the body, any signs of organ damage, and impacts on fertility or immune systems.
The promise of novel molecules matters, but the public deserves full truth about safety before headlines tout wonder drugs or biotech startups race to market. As history shows, strong data is the best shield against unintended harm.
What is the recommended dosage of (R)-9-(2-Hydroxypropyl)Adenine?
Thinking About Dosage in Real Life
Most folks outside pharmacy circles don’t bump into the name (R)-9-(2-Hydroxypropyl)Adenine unless medicine or research brings them together. This compound, once you dig into it, mainly shows up in antiviral work, especially under the nickname “HPMPA.” Doctors and researchers see promise for tackling DNA viruses, which isn’t something over-the-counter pills usually handle. So right away, this topic draws a line between regular folks and the world of experts.
Why Dosage Matters
At the doctor’s office, dosage never looks like guesswork. Too little, and the infection keeps rolling. Too much, and side effects punch harder than the sickness. With HPMPA, scientists run their tests in tidy, controlled environments. For example, published studies and animal models use doses ranging from several milligrams per kilogram of body weight, sometimes going as high as 30 mg/kg per day in certain animals. Real people, though, have bodies far more complicated than a petri dish or even a laboratory rat—livers that clear drugs at different speeds, older folks with tired kidneys, kids who process everything differently. None of this fits into a perfect formula.
No Approved Use, No Guessing
Nobody’s writing prescriptions for HPMPA capsules at the corner drugstore. No health authority rolled out guidelines, so there’s no chart to check for the “right” dosage for humans. That’s not just red tape; that’s a safeguard. Without years of testing in people, hundreds of volunteers, and lots of back-and-forth between researchers and watchdogs like the FDA, we’re left without a safety net. Mistakes with experimental antivirals cost real outcomes—think kidney injury, long-term organ strain, or resistance that makes future viruses tougher.
Lessons from Experience: Don't Shortcut the Science
Trust plays a big role in handling medicine. I remember folks clamoring for new antivirals in a hurry during the pandemic or when a rare virus pops up in the news. Hope runs high, and desperation can push anyone to consider medicines still stuck in the research stage. But hope without careful, slow science lets the door open for harm. In my own family, a relative with a rare infection thought about trying experimental options. We dug through every bit of information, but in the end, the doctor pointed straight back to evidence and the lack of real, approved dosage details. Health carries no shortcuts worth taking when risk still looms large.
Supporting Safe Solutions
Legitimate progress for HPMPA or any similar drug won’t come from guesswork or internet hearsay. It happens in the open—long, closely tracked studies, transparency from drug makers, and tough questions from regulatory agencies. If one day HPMPA earns approval for human use, honest facts about doses, benefits, and dangers will fill medical journals and pharmacists’ shelves. Until then, no internet search or forum can hand out a safe recipe. True safety comes from patience, research, and respecting the barriers designed to keep treatments both effective and safe.
Clear Fact: Ask the Experts
Anyone who hears about HPMPA and wonders if it can help should always circle back to medical pros. They know not only the science behind a compound but the reasons for every limit, every trial, every caution sign along the way. That’s less about holding back hope and more about making sure today’s decision doesn’t cause tomorrow’s regret.
Are there any side effects associated with (R)-9-(2-Hydroxypropyl)Adenine?
What Kind of Drug Are We Talking About?
Doctors and patients sometimes talk about (R)-9-(2-Hydroxypropyl)Adenine, or HP-Ade, as a potential antiviral molecule. Chemists built it with the hope that it could bring real help for fighting tricky viruses in humans. If you've heard of acyclovir or other antiviral drugs, you know how important this class of treatment remains. The real question here—are there any drawbacks or risks connected to HP-Ade?
Known Side Effects and What to Watch Out For
HP-Ade doesn’t flood the headlines, but a few medical studies and research papers do mention reactions linked to its use. Most of the time, these side effects seem similar to the ones people notice with other antiviral agents like acyclovir or ganciclovir. The list usually kicks off with digestive discomfort. Nausea, upset stomach, or even loose stools crop up sometimes. These things might sound minor, but anyone who’s tried to make it through a workday with a stomachache knows how much of a deal-breaker that can be.
Doctors bring up liver strain with many antivirals, and HP-Ade does land on that radar. Some studies noticed notable increases in liver enzymes, a sign that the liver is working harder than usual. If the liver gives out, everything else falls apart, so blood tests during treatment matter. A few researchers say kidney strain looks possible in a tiny slice of people, though nowhere near as common as with the classic antivirals.
I haven’t seen as many allergic reactions in my look through published case reports. Rashes, itching, or more serious signs like facial swelling have barely surfaced in trials so far. That doesn’t mean the risk is zero—people with a track record of drug allergies need to step carefully. No one wants to be the rare person who winds up in the ER with trouble breathing because of a medication.
Chronic Use and Long-Term Effects
A lot of info still sits behind closed doors, locked up in labs. Most available data comes from short-term studies, not years of use. There's some early animal data suggesting that dosing over months might stress organs, but that isn’t much comfort for people who’ve dealt with long COVID or chronic hepatitis. Anyone facing long-term use should expect regular checkups and bloodwork.
How Can Patients Lower Their Risk?
Doctors have tools to help lower the odds of serious trouble. Good hydration makes a big difference for the kidneys—plain water usually does more than fancy sports drinks. People taking HP-Ade should look for liver-friendly diets and avoid alcohol. It’s not glamorous, but keeping lines of communication open with a healthcare provider helps spot early warning signs—think yellowing eyes, a sudden rash, or unrelenting stomach pain. No one should ignore odd changes while on a new drug.
The Importance of Accurate Information
I sometimes look at online discussions and get a bit worried. Misinformation about side effects moves fast online. Anyone considering a medication, especially new or less widely used antivirals like HP-Ade, needs real facts from trusted sources. Peer-reviewed journals, doctors, and established medical panels still hold more weight than Reddit threads or viral tweets.
Continued Research Matters
Science doesn’t stop at approval. Side effects can take years to fully show up. Every new patient adds to what doctors learn, and reporting any strange reactions remains a duty for everyone involved. This way, what starts as a trickle of info can turn into clear advice for others down the line. HP-Ade might start as a small player in antivirals, but smart, careful use and honest feedback will shape its future care profile.
How should (R)-9-(2-Hydroxypropyl)Adenine be stored?
Understanding Proper Handling
Anyone working in a lab or pharmaceutical setting knows how often the outcome of an experiment or a drug formulation rides on details. Small lapses in storage can turn a promising compound into a waste of money and effort. (R)-9-(2-Hydroxypropyl)Adenine, or HP-Ade, serves as a perfect example of this reality. HP-Ade, essential in antiviral research and sometimes used to synthesize prodrugs, reacts readily with oxygen, heat, and moisture. Forgetting this costs time, results, and even career credibility. I’ve seen what happens when someone just leaves a sensitive compound like this on a shelf. The lab fridge ends up with a useless, yellowed sample instead of something that powers research or patient care.
Ideal Storage Conditions
HP-Ade wants a cool, dark, and dry home. High humidity or light exposure kicks off the kind of chemical changes that no one wants. Storing this compound at 2–8°C— inside a tightly sealed container—keeps it from degrading. That means avoiding containers with loose lids or worn seals. A desiccator or a refrigerator shelf away from any condensation works best. Everyone says “room temperature” is fine for some chemicals. Not for HP-Ade. Heat from a nearby incubator or even lights above a countertop can speed up hydrolysis or oxidation. Once that happens, reproducibility vanishes.
Container Choice and Labeling
Glass bottles or high-grade polypropylene vials protect the compound from unwanted chemical reactions. Ordinary plastic won’t always cut it. Air, moisture, and UV light filter in through cheap containers and before long, the HP-Ade starts to look— and act—wrong. Every experienced technician I’ve worked with double checks all labels and lot numbers. Mislabeling means more than just confusion; it sends months of work down the drain if someone draws from a degraded batch by accident. Adding the date of opening serves as an insurance policy, since even tight storage cannot guarantee indefinite purity.
Staff Training Sets the Standard
Facilities with rotating staff or students run into issues more often. New hires or interns might not recognize why HP-Ade needs such careful handling. Training makes the difference. Written protocols, demonstration storage runs, and clear signage inside cold rooms or storage cabinets help keep everyone on the same page. I remember a new technician placing a bottle by the autoclave once. Quick action saved it, but that mistake spelled out how vital hands-on training remains for protecting specialty reagents.
Solutions for Better Consistency
Reliability in research springs from small decisions. Controlling who can access storage, conducting regular inspections, and logging in-out records reduce mistakes. Some labs invest in automated temperature monitors and humidity sensors, alerting staff before conditions slip outside the safe range. These little investments stop problems before they start. Productive labs rarely suffer quality losses because they treat storage as a foundation, not an afterthought. From my years on the bench, I’ve learned the best researchers obsess just as much over how they store their compounds as how they use them. HP-Ade reminds us that science builds on details— and smart storage shows respect for the work itself.
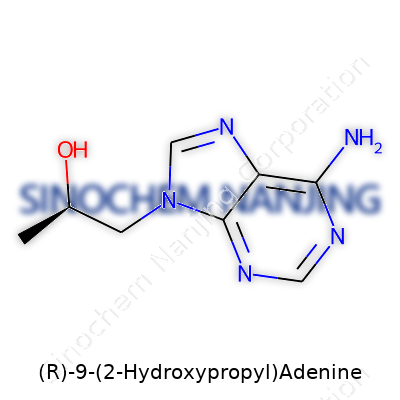
| Names | |
| Preferred IUPAC name | (9R)-9-(2-hydroxypropyl)-9H-purin-6-amine |
| Other names |
DHPG Ganciclovir Impurity A Adenine, 9-[(R)-2-hydroxypropyl]- (R)-9-(2-Hydroxy-1-methylethyl)adenine |
| Pronunciation | /ɑːr naɪn tuː haɪˈdrɒksiˈprəʊpɪl ˈædənɪn/ |
| Identifiers | |
| CAS Number | 51930-13-3 |
| Beilstein Reference | 1722184 |
| ChEBI | CHEBI:28262 |
| ChEMBL | CHEMBL582 |
| ChemSpider | 237279 |
| DrugBank | DB06145 |
| ECHA InfoCard | EC 629-818-6 |
| EC Number | 4.3.2.26 |
| Gmelin Reference | 497840 |
| KEGG | C07327 |
| MeSH | D03106 |
| PubChem CID | 72281 |
| RTECS number | SC8392000 |
| UNII | 7CVX666Q9B |
| UN number | Not assigned |
| CompTox Dashboard (EPA) | DTXSID3039246 |
| Properties | |
| Chemical formula | C8H11N5O |
| Molar mass | 167.17 g/mol |
| Appearance | White to off-white solid |
| Odor | Odorless |
| Density | 1.49 g/cm³ |
| Solubility in water | soluble |
| log P | -0.81 |
| Vapor pressure | 8.1E-9 mmHg at 25°C |
| Acidity (pKa) | 12.1 |
| Basicity (pKb) | pKb = 3.47 |
| Magnetic susceptibility (χ) | -61.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.640 |
| Dipole moment | 2.33 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 206.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | J05AB07 |
| Hazards | |
| Main hazards | Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07; Warning; H315, H319, H335 |
| Pictograms | GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. H312: Harmful in contact with skin. H332: Harmful if inhaled. |
| Precautionary statements | P261, P264, P270, P271, P272, P273, P280, P301+P312, P302+P352, P304+P340, P308+P313, P312, P314, P321, P330, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | > 228.2 °C |
| LD50 (median dose) | LD50: 760 mg/kg (oral, mouse) |
| NIOSH | UR7890000 |
| PEL (Permissible) | There is no specific PEL (Permissible Exposure Limit) established for (R)-9-(2-Hydroxypropyl)Adenine. |
| REL (Recommended) | 10 mg/mL |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Adenine 9-(2-Hydroxypropyl)adenine Vidarabine Acyclovir Penciclovir |

