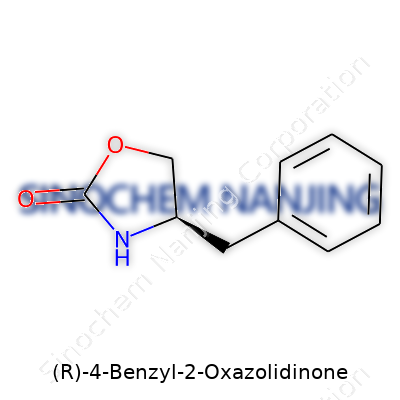
(R)-4-Benzyl-2-Oxazolidinone: An In-Depth Look
Historical Development
Back in the 1980s, chemists searching for new ways to build complex molecules turned their attention to oxazolidinones. What drew them in was the unique ring structure that promised both stability and flexibility for developing different drugs. (R)-4-Benzyl-2-oxazolidinone emerged as a standout, not just because of its stereochemistry but also thanks to its compact core. By the late 1980s, this molecule found its home in university laboratories, where chirality made the difference between a promising medicine and a dud. The discovery of this specific enantiomer reflected a larger trend, with researchers realizing that the “right” version of a molecule could mean everything. As more people experimented with asymmetric synthesis, (R)-4-Benzyl-2-oxazolidinone built up a reputation as a staple, especially for developing new pharmaceuticals and building blocks in organic chemistry.
Product Overview
On the surface, (R)-4-Benzyl-2-oxazolidinone looks simple, with a five-membered ring and a benzyl group sticking out. But in the world of synthetic chemistry, it’s a quiet workhorse, enabling controlled reactions that make otherwise tricky processes easier. Scientists usually buy it as a white to off-white solid, often in pure enantiomeric form. For chemists in the lab, holding a vial means holding weeks or months of careful development by chemical suppliers who nail both quality and stereochemistry. This compound gets pulled off the shelf for use in enantioselective syntheses, acting like a guiding hand to control the 3D shape of new drugs and chemical intermediates. In an industry where small changes can mean different outcomes, this consistency matters.
Physical & Chemical Properties
This molecule weighs 205.24 g/mol, melts at 117–119°C, and dissolves best in organic solvents like dichloromethane. The oxazolidinone ring gives (R)-4-Benzyl-2-oxazolidinone its chemical backbone, while the benzyl group lends extra weight and reactivity. Although it might look like just another white powder, what matters are its hydrogen bond acceptors and donors, the way it resists hydrolysis under standard lab conditions, and the smooth way it forms crystalline solids. It’s stable enough for storage, provided you keep it away from strong acids, bases, or sources of high heat. NMR spectroscopy confirms its structure and confirmed purity, with a signature set of peaks that organic chemists recognize in moments.
Technical Specifications & Labeling
Suppliers lay out clear technical sheets for (R)-4-Benzyl-2-oxazolidinone. Looking at a label, you’ll find the chemical name, purity (often above 98%), CAS number (14290-13-4), molecular formula (C10H11NO2), and stereochemistry. There’s a batch number for tracking, hazard statements for safe handling, storage recommendations, and sometimes, certificates of analysis. This level of transparency reflects what both industry and academia expect now: traceability, reliability, and clear communication. Having seen mishaps due to poor labeling in crowded labs, it makes sense to spend effort ensuring every bottle tells its own story.
Preparation Method
Synthetic organic chemists rely on established methods to prepare (R)-4-Benzyl-2-oxazolidinone. Starting from (R)-phenylglycinol, the amino alcohol cyclizes with phosgene derivatives (traditionally phosgene or safer modern substitutes) in controlled conditions. After this intramolecular ring closure, purification steps follow—usually recrystallization or chromatography. Since the starting material is optically active, the product retains the desired 'R' configuration. Every chemist learns to respect the dangers and importance of phosgene alternatives, which require careful management. The choice of enantiopure starting materials drives up cost but pays back in downstream reliability, especially for pharmaceutical synthesis.
Chemical Reactions & Modifications
In the lab, (R)-4-Benzyl-2-oxazolidinone acts as a chiral auxiliary. It attaches to carboxylic acids or similar groups through straightforward acylation reactions, setting the stage for highly selective reactions like alkylation or aldol additions. The auxiliary then gets cleaved off, leaving behind a new chiral center. This strategy played a key part in countless drug syntheses, giving chemists control over a drug’s biological activity. The benzyl group itself can also take part in hydrogenolysis, freeing up routes for further chemical modifications. Over the years, creative minds have used this scaffold as a starting point for new derivatives, enhancing water solubility or opening up different pharmaceutical profiles.
Synonyms & Product Names
This compound goes by several names, reflecting both historical routes and modern cataloging. It’s known as (R)-(+)-4-Benzyl-2-oxazolidinone, (R)-4-Benzyl-2-oxazolidone, or sometimes simply as (R)-BOx. Catalogs and research literature may also refer to it using system numbers or proprietary codes assigned by chemical suppliers. Regardless of the name, the chemical fingerprint makes it easy to distinguish from its 'S' enantiomer or closely related oxazolidinones, both in the bottle and during analytical workup.
Safety & Operational Standards
Safety matters every day in the lab, and (R)-4-Benzyl-2-oxazolidinone requires the usual caution. Wearing gloves, goggles, and working inside a fume hood keeps accidental exposure low. According to established guidelines, inhalation or ingesting the dust should be avoided, although the acute toxicity profile sits at manageable levels compared to more reactive intermediates. Waste gets collected separately due to nitrogen-containing heterocyclic groups. Storage away from acids and oxidants preserves the compound. Standard first aid measures remain important, with safety data sheets outlining steps in case of accidental contact. For anyone who’s ever seen a small spill turn into a safety meeting, these protocols clearly matter.
Application Area
Drug discovery teams and process chemists reach for (R)-4-Benzyl-2-oxazolidinone as a fundamental tool in asymmetric synthesis. Medicinal chemistry leans on its ability to help create enantiomerically pure compounds—the backbone of modern pharmaceuticals like antibiotics, antivirals, and central nervous system drugs. Beyond pharma, it pops up in the synthesis of agrochemicals and specialty chemicals that demand strict chiral control. The industrial sector adopted it not just for its usefulness but because it lowers barriers to scaling up complex syntheses. Academic researchers depend on its reliability for teaching chirality and new synthetic approaches.
Research & Development
Active R&D keeps expanding the toolkit for oxazolidinone chemistry. Research teams keep testing new ways to synthesize (R)-4-Benzyl-2-oxazolidinone using cheaper, greener methods. Excitement builds around catalytic asymmetric synthesis, which promises to cut costs and limit hazardous byproducts. Teams studying reaction mechanisms use this compound to probe selectivity and unravel how chiral environments guide chemical transformations. Chemoinformatics groups catalog its reactions, opening up faster ways to predict outcomes of new syntheses. These developments echo a trend where collaboration between academic and industry chemists drives faster improvements, saving resources, and improving safety.
Toxicity Research
Compared to high-risk chemicals, (R)-4-Benzyl-2-oxazolidinone finds itself lower on the hazard scale, but that does not mean neglecting toxicology testing. Early screening focused on acute oral and dermal toxicity in model organisms and cell cultures. Results generally point to low acute toxicity, provided contact remains limited. Chronic exposure data stays less complete due to modest commercial volumes and targeted lab uses. Researchers monitor for signs of allergic reactions or organ-specific effects, with new work expanding into metabolic breakdown products in humans and the environment. Even with lower risk, regulatory agencies advise ongoing monitoring, especially as scaled-up syntheses for pharma manufacture become more common.
Future Prospects
The future of (R)-4-Benzyl-2-oxazolidinone looks set to follow broader trends in green chemistry and efficiency. With the pharmaceutical industry demanding cleaner, more selective synthesis routes, this chiral auxiliary could see upgrades in reusability or integration with flow chemistry setups, reducing waste and cycle times. If advances in asymmetric catalysis continue, some applications may start bypassing traditional chiral auxiliaries, but the reliability and ease of (R)-4-Benzyl-2-oxazolidinone keep it relevant for tough reactions. AI-driven route scouting in drug development highlights its use for new scaffolds, and regulatory pressures encourage safer production methods and thorough toxicology. For those shaping the science behind tomorrow’s medicines, practical, well-understood tools like this compound remain essential, balancing innovation with the realities of daily bench work.
What is (R)-4-Benzyl-2-Oxazolidinone used for?
Meet the Unsung Hero of Chemical Synthesis
Every tool kit has a favorite wrench, something reliable that never lets you down. For organic chemists, (R)-4-Benzyl-2-Oxazolidinone takes up that spot. This molecule fits right into the workflow for building complex drug ingredients and specialty chemicals. If you walk into a pharmaceutical lab or a university chemistry department, chances are someone has used it recently to help piece together a new compound with the right shape and function.
Not Just Another Laboratory Compound
Most people might not get excited about oxazolidinones, but they play a real role in shaping medicines and crop protection agents. What makes (R)-4-Benzyl-2-Oxazolidinone stand out is its chiral center. Chemists lean on chirality because the left- or right-handedness of a molecule changes how it works in the body. For new medicines or fungicides, getting exactly the correct orientation can mean the difference between fixing a problem and causing a side effect.
Back when I spent weekends helping out in a university research group, I watched researchers use this compound as a chiral auxiliary. It might sound fancy, but it’s basically a molecular steering wheel. They hooked it onto their starting material, steered the reaction in the direction they wanted, and then took it off again—like temporary training wheels for building complex targets. Other chemists rely on it for creating beta-lactams, a giant family of antibiotic drugs. The oxazolidinone helps shape the core of these molecules, which continue to save lives around the globe.
Why It Matters Beyond the Lab
People might not think about the building blocks behind their prescriptions or the food on their plate, but these starting materials lay the groundwork for safer, more effective medicines and agrochemicals. Getting highly selective reactions cuts down on waste and lowers costs, both financial and environmental. Too much trial and error ends up wasting expensive raw materials—often derived from petroleum—and puts extra pressure on the environment.
Peer-reviewed journals point out that molecules like (R)-4-Benzyl-2-Oxazolidinone help bring efficiency and greater safety, especially in drug design. For example, using the right chiral auxiliary can lead to fewer by-products, less need for complex purification, and an easier route from test tube to patient. The FDA and regulatory bodies often ask for high purity and precise control over chirality in pharmaceuticals, making these molecules a quiet but crucial part of compliance as well.
Building a Better Future in Chemistry
The push for better chemistry keeps driving researchers to design even more specialized and user-friendly auxiliaries. Academic labs and companies test new methods to recycle or reuse oxazolidinones, cutting down on resource waste. Cost and environmental impact always stay in the back of minds, especially as society asks for more sustainable solutions. We all benefit from streamlined synthesis where these tools do the heavy lifting, making new treatments or safer pesticides more widely available.
While most folks outside the lab may never hear its name, (R)-4-Benzyl-2-Oxazolidinone keeps working behind the scenes. It doesn’t grab headlines, but its contribution echoes through every disease treated with a new antibiotic, every crop field protected from fungal attack, and every scientist striving for the next breakthrough medicine.
What is the molecular formula and molecular weight of (R)-4-Benzyl-2-Oxazolidinone?
What Stands Behind the Name?
Chemistry likes precision. Each molecule gets branded by its structure and elements, not just what’s easy to say in conversation. (R)-4-Benzyl-2-Oxazolidinone tells a story: a five-membered oxazolidinone ring sits at the center, with a benzyl group linked to the fourth carbon and a specific ‘R’ chirality steering its three-dimensional behavior. The naming may sound dry, but it defines how this molecule interacts with the world—how it fits inside pharmaceutical research, how it chooses which chemicals to bond with, and sometimes how it alters the lives of those who depend on fine-tuned medicines.
The Molecular Formula at Its Core
Looking at the bonds and atoms in (R)-4-Benzyl-2-Oxazolidinone, you count each piece: a benzyl group brings a C7H7 tail; the oxazolidinone ring carries two nitrogens, three carbons, one oxygen, and rounds out with five hydrogens. Add all of them:
Molecular formula: C10H11NO2
This formula isn’t just a string of numbers and letters, but the fingerprint of the molecule. Lab work depends on this. Weighing out reactants for a synthesis? This formula tells you how much to use. Double-checking a substance’s purity or testing for contaminants? Labs aim for the precise amount of carbon, hydrogen, nitrogen, and oxygen noted here.
Molecular Weight Tells You More Than a Number
Sticking the word ‘weight’ after ‘molecular’ usually means grams per mole, calculated by adding up the atomic weights of each atom present. For C10H11NO2:
- Carbon (C): 10 × 12.011 g/mol
- Hydrogen (H): 11 × 1.008 g/mol
- Nitrogen (N): 1 × 14.007 g/mol
- Oxygen (O): 2 × 15.999 g/mol
Add those: 120.11 (C) + 11.088 (H) + 14.007 (N) + 31.998 (O) = 177.20 g/mol (rounded to two decimal places).
People in science use this number every day, sometimes without thinking about it. Making solutions, dosing, running chromatographic analyses—molecular weight is the yardstick. It’s the difference between a successful experiment and one where nothing works because the math got skipped.
Why These Details Matter
Some might roll their eyes at basic compositional data, but these numbers sit at the bottom of research and manufacturing. Drug development stumbles if a compound’s formula gets mistaken. Custom catalysts, chiral auxiliaries, or new synthetic routes—these all depend on pinpoint accuracy. I’ve watched colleagues troubleshoot failed reactions for hours, only to find someone made a decimal error calculating the mass.
Want fewer mistakes? Double-check the numbers. Pull references from trusted databases, like PubChem or Sigma-Aldrich, and match experimental outcomes with calculated mass spectra. Accurate basics guard against costly failures.
Now, if you’re stuck with the task of making or analyzing (R)-4-Benzyl-2-Oxazolidinone, ice-cold calculations pay off. Whether you’re scaling up in a chemical plant or hunting for new drug leads, it always comes back to knowing your compounds, down to the last atom.
How should (R)-4-Benzyl-2-Oxazolidinone be stored?
Understanding What We’re Dealing With
(R)-4-Benzyl-2-Oxazolidinone isn’t your everyday chemical. It helps chemists design complex molecules, and it plays a part in a range of pharmaceutical creations. Without proper storage, even a great reagent like this one can lose effectiveness, posing risks for both output and safety. Anyone who’s worked in a research lab knows the sting of pulling out a degraded sample right when a crucial reaction demands consistency.
Keeping It Safe: Storage Conditions You Can Trust
This molecule doesn’t like moisture, and it shows sensitivity to strong sunlight. I learned early on that a tightly sealed vial makes a huge difference. Leave the cap loose, even briefly, and you’ll notice clumping or discoloration—not the signs you want in a high-purity molecule. Store (R)-4-Benzyl-2-Oxazolidinone in a cool, dry space. My team always favored a traditional laboratory refrigerator, as this slows down breakdown and limits humidity exposure. Set it away from acids, strong bases, and abnormal heat. Placing it near exothermic reactions once cost our group a whole batch.
Direct UV exposure can change its character too. Whether you label yourself a synthetic organic chemist or a process engineer, clear amber bottles and solid cabinets go a long way. At home or at the bench, use a secondary container. I once lost half a gram to a careless spill—one slip, and the powder clung to every surface. Double containment not only saves your material, but limits headaches for lab mates, too.
Why Quality Storage Matters
Beyond maintaining chemical purity, correct logistics protect people. (R)-4-Benzyl-2-Oxazolidinone rarely poses hazards alone, but its interactions can turn unpredictable. Moisture might launch unwanted hydrolysis, shifting the product profile. Tidy shelves, clear labeling, and rotating stock don’t just keep things looking neat—they actively cut down wasted resources. Labs with poor procedures run into expired batches far too often, costing time and funds that no grant covers.
Personal experience showed me how just a few missed storage checks prompt emergency meetings. Inventory tracking sounds boring, but a simple spreadsheet updated every two weeks keeps surprises to a minimum. Rushing never pays off. More than once, a researcher on my floor tried to speed things up by storing new intermediates “temporarily” on their bench. Shortcuts led to confusion, accidental mix-ups, and tricky cleanups.
Small Habits Go a Long Way
Wearing gloves and goggles goes without saying. Wipe work surfaces both before and after handling: dust and residue love to stay where you least expect it. I encourage every lab—no matter the budget—to post storage reminders right where chemicals are kept. Quick reference cards next to the cabinet door spark good habits and keep everyone on the same page.
Suppliers and safety data sheets offer valuable tips. Yet, no amount of paperwork beats routine. Make it a rule: every new shipment gets a fresh label, a close check for damage, and a prime spot in the correct storage. If the bottle ever looks off or the label fades, flag it for review. It only takes a few minutes to get these things right, and those moments protect the investment—the research, the people, and the results.
Well-maintained stocks of (R)-4-Benzyl-2-Oxazolidinone add certainty to scientific work. That reliability balances risk and fuels discovery long after the experiment ends.
Is (R)-4-Benzyl-2-Oxazolidinone hazardous or does it require special handling?
Looking Beyond the Name
The chemical (R)-4-Benzyl-2-Oxazolidinone often pops up in research labs, especially those working on organic synthesis or pharmaceutical intermediates. Researchers know the deal: just because a compound sounds complex doesn’t guarantee danger, but ignoring risk never helped anyone. No matter how familiar or harmless a compound appears, treating every bottle with respect makes for fewer problems in the long run.
Digging Into the Hazards
I've spent years handling a range of laboratory compounds, and it pays to keep perspective. (R)-4-Benzyl-2-Oxazolidinone isn’t on any mainstream government watchlist as a notorious poison, but that doesn’t mean it's risk-free. Data from chemical safety databases shows that oxazolidinones, as a class, can sometimes pose hazards—skin and eye irritation stands out most. Inhalation may bring a sore throat or respiratory discomfort if the dust gets out of hand. That alone calls for attention, because repeated exposure or sloppy technique builds up safety problems fast.
Accidents rarely make headlines unless someone ends up in trouble, but those small allergic reactions, rashes, or mishandling events add up. The Environmental Protection Agency and international regulators highlight that lab work brings unique risks, even with chemicals that aren’t flagged as high-hazard. Take the simple route: gloves, safety glasses, and lab coats protect more than just your clothes. We’re talking about avoiding emergency showers and unwanted doctor visits, not just ticking boxes.
Why Responsible Handling Matters
My own lab days taught me that complacency shows up the minute you skip the basics. A splash in the eye, a poorly labeled vial, or a missed hazard label may seem like minor mistakes—that is, until someone loses hours to paperwork and first aid. (R)-4-Benzyl-2-Oxazolidinone often comes as a white powder. Even if it doesn’t smell funny or eat through gloves, dust can hang in the air, sneak onto benches, and end up on lunch breaks. Hand washing and surface cleaning matter, and so does proper chemical storage.
Most safety data sheets underline these points: store the compound dry and cool, keep containers tightly sealed, and avoid heat. Good ventilation does more than cut down on odor; it makes sure airborne particles don’t travel from bench to lungs. Disposal shouldn’t get lazy either—dumping chemicals down the sink creates bigger headaches for the environment and everyone downstream.
Better Habits, Safer Work
Training and regular updates for lab teams save time and money. Every person in a lab, no matter the experience, benefits from reminders about spill kits, eyewash stations, and basic protective equipment. For every compound, including (R)-4-Benzyl-2-Oxazolidinone, asking for the latest safety data and reviews before use becomes second nature over time. Technology can help: digital inventories track expiration dates and flag hazards faster than a spreadsheet ever could.
Good habits grow from these commitments. Sharing real cases of mistakes, not just theory, sticks with people better than lists of rules. Chemists, technicians, and even visitors all play their part in building a safety culture. At the end of the day, treating each bottle as if it might cause harm keeps everyone on their toes. Whether the chemical belongs in a research breakthrough or basic training kit isn’t as important as arriving home healthy, with every story just another memory of a careful day’s work.
What is the enantiomeric purity of (R)-4-Benzyl-2-Oxazolidinone?
Why Enantiomeric Purity Holds Real-World Weight
Standing at the bench or reading a batch report, chemists know that a molecule’s handedness matters. (R)-4-Benzyl-2-Oxazolidinone is a building block in chemistry, especially for those developing pharmaceuticals or intricate organic compounds. Enantiomeric purity speaks to how much of this substance shows up in just one enantiomeric form (the “R” version), as opposed to its mirror image. The wrong handedness often means the difference between a life-saving drug and an inactive powder — or far worse, an unexpected side effect. In my lab days, finding a trace of the unwanted “S” enantiomer in a supposedly pure sample could send an entire project back to the drawing board.
How Chemists Measure Enantiomeric Purity
Spotting which enantiomer dominates takes practice and sharp tools. Chemists often use chiral chromatography, polarimetry, or advanced techniques like NMR with chiral shift reagents. For (R)-4-Benzyl-2-Oxazolidinone, something like chiral HPLC easily distinguishes between enantiomers, giving a clear ratio. Drug makers and research labs rely on these results not just for regulatory compliance, but to maintain trust — nobody wants to explain unexpected side effects traced to low purity. Published data on this compound usually puts enantiomeric excess (ee) above 98%, since modern syntheses and purification methods efficiently weed out the wrong type. Suppliers who cut corners may list lower ee, but reputable vendors compete on purity levels above 99%, since downstream applications depend on consistency and safety.
Enantiomeric Purity: Why Cutting Corners Never Pays
Years back, I saw a colleague’s pain when an impurity slipped through: months of careful work ruined by a racemic contaminant. One percent of the wrong enantiomer may seem tiny, but with large batches or sensitive reactions, that margin causes headaches. In pharmaceuticals, regulatory agencies like the FDA demand rigorous characterization. For some medications, patients get benefits only from the right-handed molecule, and the incorrect enantiomer might deliver no help or introduce risks. Scientists can’t just assume a synthesis spits out pure “R” each time; rigorous checks at every stage remain the only way forward.
How to Raise Enantiomeric Purity and Avoid Surprises
The pursuit of high enantiomeric purity drives innovation. Standard methods include asymmetric synthesis with specialized catalysts or using chiral auxiliaries. I saw teams adopt kinetic resolution — turning only one enantiomer into a product and washing away the rest. For (R)-4-Benzyl-2-Oxazolidinone, asymmetric hydrogenation and enzymatic approaches both offer routes to dauntingly high selectivity. But books and journals rarely warn that scale-up might throw these processes off, making robust analytical routine a must. It’s not enough to rely on literature values; each batch deserves scrutiny.
On the supplier side, transparent reporting of purity levels and analytical methods helps customers avoid bad surprises. Trust builds through data, not guesses. If a bottle doesn’t come with its chiral HPLC trace or at least an ee value, odds are it isn’t worth adding to your project.
Clear Communication and Quality Data Support Safer Science
Every chemist I’ve known values three things: accuracy, reliability, and the safety of end users. Enantiomeric purity reporting isn’t some academic flourish — it’s a baseline requirement for drugs, catalysts, and advanced materials alike. In the age of tight timelines, it’s tempting to accept “good enough,” but science and health demand more. Chasing numbers above 99% enantiomeric excess might seem obsessive, but it keeps bad batches out of research and wrong-handed chemicals out of medicine cabinets. That’s a goal worth pursuing, every single time.
| Names | |
| Preferred IUPAC name | (4R)-4-benzyl-1,3-oxazolidin-2-one |
| Pronunciation | /ɑːr fɔːr ˈbɛnzɪl tuː ɒkˌsæzəˈlɪdɪnəʊn/ |
| Identifiers | |
| CAS Number | 112096-22-1 |
| 3D model (JSmol) | ``` 3Dmol.js:load={pdb:'C1CN(C(=O)O1)[C@@H]2CCCCC2'} ``` |
| Beilstein Reference | 146026 |
| ChEBI | CHEBI:65707 |
| ChEMBL | CHEMBL1221242 |
| ChemSpider | 137348 |
| DrugBank | DB08399 |
| ECHA InfoCard | 100.124.710 |
| EC Number | 69795-06-0 |
| Gmelin Reference | 74433 |
| KEGG | C11236 |
| MeSH | D021281 |
| PubChem CID | 709708 |
| RTECS number | VA8400000 |
| UNII | U4DJJ3R89T |
| UN number | 2811 |
| Properties | |
| Chemical formula | C10H11NO2 |
| Molar mass | 179.22 g/mol |
| Appearance | White to off-white solid |
| Odor | Odorless |
| Density | 1.2 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | 0.02 |
| Acidity (pKa) | 13.7 |
| Basicity (pKb) | 4.74 |
| Magnetic susceptibility (χ) | -70.91·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5700 |
| Dipole moment | 4.49 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 395.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4845.7 kJ/mol |
| Pharmacology | |
| ATC code | N06AX17 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS07, GHS08 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P273, P280, P302+P352, P305+P351+P338, P337+P313, P362+P364 |
| Flash point | Flash point: 227.7 °C |
| NIOSH | LV8225000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 10-30 mg |

