Pyrazinamide: More than Just a Pill in Tuberculosis Treatment
From Discovery to Frontline Therapy
Back in the middle of the last century, doctors faced a tough battle against tuberculosis. The disease wore out patients and their caregivers alike. At that time, newly uncovered antibiotics fueled hope but also sparked skepticism. Pyrazinamide entered this scene in the 1950s, discovered after chemists looked for chemical cousins of nicotinamide. The early studies, mostly done in animal models, showed that pyrazinamide worked best not in isolation but when paired with other drugs, especially during the initial “intensive phase” of TB therapy. As treatment guidelines evolved, pyrazinamide became an integral part of the standard triple or quadruple regimens, leading to shorter and more effective courses. Old journal articles often point to the surprise among clinicians at how pyrazinamide managed to kill persistent TB bacteria in tissue environments that other agents barely touched. Its introduction shortened therapy for many patients, changing not just outcomes but daily routines and even public health projects worldwide.
What Is Pyrazinamide, Chemically Speaking?
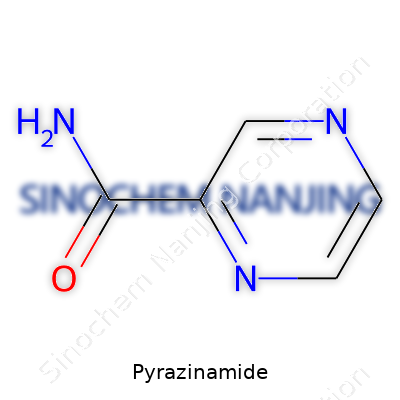
In my own work with pharmaceutical samples, I came across pyrazinamide as a fine white crystalline powder. The structure looks simple at first glance: a pyrazine ring with a carboxamide group at the third position. This places it in the group of synthetic analogues similar to pyrazine, but the amide group gives it the edge for antitubercular action. It dissolves modestly well in water, and even better in organic solvents like ether or alcohol. This dual solubility profile helps during both testing and tablet formulation, which partly explains why many generic manufacturers stick with the age-old crystalline form. I remember reading that the chemical stability of pyrazinamide helps during storage, especially in environments with fluctuating temperatures. That trait counts when distributing TB medicines in regions where supply chains stretch over deserts or up mountain passes.
Technical Specs & the Ways Scientists Label It
Pyrazinamide tends to show up in research or clinical formulary texts listed with a formula of C5H5N3O and a molecular weight in the low 120s. The melting point usually falls in the 189-191°C range, easy to check in a student lab with a cheap capillary tube. Since the drug’s activity depends on the unchanged molecule getting inside mycobacterial cells, strict standards direct how pure it should be, with pharmacopeial samples tested for moisture, pH in suspension, and absence of breakdown products. Technical sheets always mention the same CAS number and sometimes synonyms like PZA, pyrazinoic acid amide, or even its assignation as an “oral antituberculosis agent." Each tablet on the shelf carries distinct batch numbers to track manufacturing runs, and the labeling flags both its main indications and cautions about potential side effects.
How Pyrazinamide Is Made
The main production method I’ve come across starts with the nitration of pyrazine, followed by hydrolysis and amidation. Some manufacturers use different starting materials or tweak solvents and reaction times depending on local availability and regulations. Core steps include precise control of temperature during hydrolysis, which avoids side products that could end up weakening the batch’s potency. Chemical engineers who design these syntheses talk about the value of streamlined methods for maintaining purity while cutting costs. Once, during a tour of a pharmaceutical factory, I saw how the quality control team ran repeated chromatography checks to catch any unreacted starting materials or new impurities before the product ever reached final packaging.
Chemical Reactions & Tweaks for Better Medicines
Beyond its original form, pyrazinamide’s structure offers chemists a platform for creativity. Some labs have experimented by changing the substitution on the pyrazine ring or by attaching groups that improve absorption. The lab down the hall from mine once looked at various esters and prodrugs that might boost delivery to lung tissue or cut down on adverse effects. Most changes try to make dosing easier or to fight against strains of Mycobacterium tuberculosis that resist the usual drug. A few papers I read pointed to research on new analogues with improved activity under different pH conditions, pulling from the insight that pyrazinamide works especially well in acidic environments inside immune cells. Each new twist in the molecule brings hurdles in both safety trials and regulatory approval, which stops more ideas than it enables, but researchers keep looking for the next breakthrough.
Product Names & Synonyms in Clinical Use
Walk into any pharmacy stocked for TB, and pyrazinamide doesn’t hide under obscure names. You’ll see “PZA” in big letters or simple generic branding like “Pyrazinamide Tablets." A few regions have local names, but they always include “pyrazinamide” somewhere in the label. In old textbooks or earlier patents, synonyms like “pyrazinoic acid amide” show up, and some technical monographs list it as part of “first-line antitubercular agents.” Generics dominate the market now, so the names might differ by country, but the basic molecule rarely changes.
How Safety & Handling Rules Shape Its Use
No one in the field takes pyrazinamide for granted, since its benefits come with risks. The main safety worry relates to liver health. Cases of drug-induced hepatitis appear in the literature every year. My own time in clinics taught me that liver enzyme monitoring needs to go hand in hand with prescription, and that even mild jaundice means the team rethinks each medication in the regimen. Pharmaceutical practice guidelines short-list pyrazinamide with warnings about joint pains, since some patients experience gout-like flares from uric acid build-up. The practical side of operation means handlers wear gloves during bulk production, and manufacturers follow strict ventilation and dust control measures. Operational standards get reinforced every time a new adverse event pops up, keeping clinicians, pharmacists, and patients ready for quick adjustments.
Who Depends on Pyrazinamide?
The primary role of pyrazinamide shows up in tuberculosis care. Physicians add it to the “RIPE” regimen alongside rifampin, isoniazid, and ethambutol. Health workers in parts of Asia and Africa regularly rely on this combination to clear up even difficult or long-standing TB infections. In my visits to infectious disease wards, I’ve watched patients turn a corner after weeks of combination therapy, with pyrazinamide acting early to cut down lingering bacteria in tough-to-reach spots inside the body. Recent research circles back to its possible value in non-tuberculous mycobacterial diseases, but most prescribing sticks with confirmed TB. Drug-resistance trends push some specialists to ask whether new indications might be found, especially in coinfection settings or for shorter therapies.
Research & What Still Needs Solving
Although decades have gone by since its introduction, pyrazinamide remains a research target. Scientific teams keep asking why the drug works best in certain environments – especially within macrophages where tuberculosis bacteria hide. Some genetic studies have pinpointed the enzyme pyrazinamidase as the key to activating the drug inside bacteria, and mutations in its gene explain much of the emerging resistance problem. Labs race to develop faster tests for resistance, since delays keep the wrong drug in regimens, winding up the risk of treatment failure. At the same time, clinical investigators wonder if modified forms of pyrazinamide could clear up treatment or limit side effects. That tension – between fighting resistance and making therapy safer – shapes much of the current dialogue in TB drug research.
The Tough Side: Toxicity Under the Microscope
Safety worries shape every conversation about pyrazinamide, especially as stories about liver toxicity crop up in clinical records. I’ve seen patients pause or swap therapy as soon as signs of hepatitis appear. Clinical trial data reported elevated liver enzymes in a noticeable fraction of people, especially when combined with other drugs that also put stress on the liver. Other cases document acute gout-like attacks due to shifts in uric acid processing, often leading to frustrating joint pain and swelling. Younger patients and those with pre-existing liver problems face the highest risks. Toxicology studies in lab animals also track down possible links between high doses and damaging changes in liver tissue, even at levels above what most clinicians would ever prescribe. This reality makes both surveillance and patient counseling an ongoing priority at every level of use.
What Comes Next: Can Pyrazinamide Keep Its Place?
The world keeps changing, and tuberculosis continues to evolve in step. Multidrug resistance now complicates what used to seem a routine prescription, and the push for shorter, simpler regimens continues gathering steam. Scientists work on new pyrazinamide analogues and tech-driven ways to monitor resistance in real time. The next chapters for this old drug likely involve harnessing its unique strengths – rapid action against persistent bacteria – while countering its limits on safety and durability. Global health planners puzzle out ways to make therapy more accessible and less toxic, sometimes pinning hopes on new research into lower dosages or targeted delivery methods. From what I see in both the literature and in fieldwork, pyrazinamide’s story remains far from finished. Its value hangs on innovation as much as on respecting the lessons of its long-standing use.
What is Pyrazinamide used for?
Pyrazinamide's Role in the Fight Against TB
Back in medical school, tuberculosis felt like history. Textbooks showed dusty photos and old stats. In reality, TB still affects millions every year, even in places with modern medicine. Pyrazinamide helps turn the tables in this fight. Doctors worldwide reach for pyrazinamide because it targets Mycobacterium tuberculosis, the bacteria behind tuberculosis. Unlike other antibiotics, pyrazinamide attacks the bacteria in a "dormant" state, inside the cells and difficult to target.
Why Pyrazinamide Matters in TB Treatment
The significance of pyrazinamide hits home in countries with high TB rates, where families can’t afford long hospital stays or expensive drugs. Before its use, TB treatment stretched up to eighteen months, pushing patients to quit early—inviting drug resistance. Adding pyrazinamide to the drug regimen changed the game. The therapy dropped from eighteen months down to six in many cases. That saves resources and lives. The World Health Organization regularly includes pyrazinamide as a first-line drug for TB, alongside rifampicin, isoniazid, and ethambutol.
Missing doses or stopping early leads to drug-resistant TB, a nightmare for public health. Proper use of pyrazinamide as part of directly observed therapy (DOTS) helps prevent these dangerous strains from spreading. I’ve seen patients struggle to keep up with long, harsh drug regimens. Anything that makes treatment shorter and more tolerable means more people complete treatment, pushing TB closer toward control.
Challenges: Side Effects and Resistance
Pyrazinamide isn’t without problems. Patients often report joint aches, gastrointestinal upset, and rarely, liver trouble. The trade-off: the high reward of shortened therapy. Doctors regularly check liver function and uric acid levels during treatment, a step patients sometimes see as a hassle, but it’s necessary. The problem doesn’t end there. Bacteria change, and resistance to pyrazinamide grows in some settings, especially where drug surveillance falls short or medication isn’t properly taken. Resistant TB strains demand more drugs, greater costs, and longer recovery.
Solutions Start on the Ground
Real progress comes from solid patient education and robust drug supply chains. Community health workers who deliver TB meds know the troubles their neighbors face better than anyone. Supporting DOTS programs helps keep TB under control, especially in rural or low-income regions. Pharmaceutical innovation helps too. Rapid diagnostics to catch resistance early give patients better options. Governments and global organizations investing in research speed up the search for alternative drugs and improved treatment regimens.
Pyrazinamide may not grab headlines, but its story shows the power of focused science and global teamwork. For many facing TB, it transforms months of illness into a real chance at recovery. That impact matters—both in statistics and in families breathing easier across the world.
What are the common side effects of Pyrazinamide?
Experience With Tuberculosis Treatment Brings Lessons
Pyrazinamide shows up in most tuberculosis treatment plans. Doctors add it to the mix early on because it works well against bacteria hiding in spots where other drugs struggle to reach. Yet, as someone who has worked alongside infectious disease teams and listened to patients navigating tough drug regimens, it’s clear this pill demands respect. No drug comes free of risks, and Pyrazinamide delivers its fair share. Understanding them means less fear and a better shot at catching problems early.
Liver Concerns: Not Just a Statistic
Ask any doctor about Pyrazinamide, and most will mention liver troubles before anything else. Liver enzymes can spike—sometimes quietly, sometimes with warning signs like yellow skin (jaundice), dark urine, or right-sided belly pain. In one study from the American Thoracic Society, hepatitis showed up in close to 15% of patients on four-drug TB therapy, with Pyrazinamide playing a big role. Every patient I’ve seen on this drug gets routine blood draws to catch any sign of liver stress before it gets out of hand. If you look tired all the time and feel nauseous after starting this medication, don't chalk it up as a small thing—tell your provider.
Gout Flare-Ups: More Than Just an Ache
Few folks realize Pyrazinamide can raise uric acid levels. Joints, especially toes and ankles, may start to ache without warning, swelling up from a classic gout flare. Some patients find this out the hard way, with sharp pain waking them up in the night. In people with a history of gout, close monitoring is key. A blood test will often reveal the problem before symptoms explode. Staying hydrated, choosing lighter meals, and alerting your doctor about old joint pain stories make a real difference.
Stomach Upset and Appetite Loss Are Hard to Miss
Most people on Pyrazinamide mention queasy stomachs—nausea, less interest in food, or occasional vomiting. Missing a meal here or there adds up across weeks, making it tough for anyone already beaten down by TB’s weight loss. I’ve watched patients lose weight fast, not because the disease is winning, but because the drug makes eating tough work. Taking the medicine with a bit of food (if your doctor says it’s okay), sipping water, and reaching out for help if appetite tanks can all keep energy up and recovery on track.
Skin Rashes and Other Surprises
Sometimes, the body pushes back with a rash—anything from a mild itch to angry red patches across arms or chest. Some report mild fevers, sore joints, or a sense of feeling just off. Allergic reactions are less common, but since these can get serious fast, getting checked out quickly matters. Telling your care team about new bumps, swelling, or trouble breathing means they can act before things escalate.
Why Paying Attention Pays Off
Pyrazinamide can save lives, but only if its risks don’t overshadow its benefits. Honest conversations between patients and their providers help make this possible. Frequent lab checks, honest talk about odd symptoms, and tracking changes in mood or body all let people keep treatment on track. People who understand what to expect from their medication often finish therapy in better health. And that's what everyone wants at the end of a tough fight against tuberculosis.
How should Pyrazinamide be taken?
The Everyday Reality of Tuberculosis Treatment
Every year, millions face the rigors of tuberculosis therapy. Anyone who has sat with doctors and heard the strict instructions for taking tuberculosis drugs knows it’s a marathon, not a sprint. Pyrazinamide sits right at the center of this effort. It’s one of the key medicines brought in during the early months of treating TB, and getting it right means a lot—shorter recovery time, fewer complications, and a much better shot at kicking TB to the curb. Pyrazinamide isn’t some miracle fix; the way someone takes it matters more than most folks think.
The Right Way to Take Pyrazinamide
Here’s the heart of the matter: swallow pyrazinamide on an empty stomach at the time the doctor tells you. Why empty? Food can mess with how well the body absorbs the medicine, and nobody wants to draw this out any longer than necessary. If nausea barges in—something that isn’t rare with TB drugs—a doctor might give the green light to take it with a bit of food to calm the stomach, but that decision rests with the healthcare team.
Missed doses create real problems. Skipping a day here or there lets TB bacteria regroup and toughen up, which means more risk of drug resistance. A big part of any TB treatment story is sticking to the routine. Some clinics set up directly observed therapy—someone actually watches as pills go down—to keep folks on track. In busy cities and rural clinics where logistics get in the way, families or friends sometimes help out as “treatment supporters.” The point: every single dose matters.
Why Dose and Duration Matter
The usual dose depends on body weight and a doctor’s call based on the specific case. Most adults get one daily dose, folded into a larger treatment plan that usually includes isoniazid, rifampicin, and ethambutol. This combination knocks out TB from multiple angles. For the sake of safety, regular blood tests and checkups should follow. Pyrazinamide stresses the liver. Anyone who loves beer or lives with hepatitis needs careful attention. Liver troubles are rare for most, but not unheard of.
Taking the Big Picture Seriously
My own work with TB patients showed me how rough the first weeks can get—side effects hit hard, and motivation sinks. Some tried splitting up pills, hoping to dodge nausea, but that sabotaged their own recovery. The few times I watched people stop therapy halfway, they almost always ended up back in the clinic, sicker and facing tougher bacteria. The World Health Organization’s numbers back this up: interrupted treatment feeds drug-resistant strains and fuels TB’s comeback.
Practical hurdles need honest talk. Complicated dosing schedules, language barriers, low health literacy—these can all wreck a good start. Mobile apps, reminders, family involvement, and easier-to-swallow combinations have made a difference. In some hard-hit communities, health workers do daily pill-drop check-ins or bring treatment to someone’s home. Access to friendly, nonjudgmental support keeps folks coming back for every dose.
Moving Toward Fewer Barriers
Pyrazinamide remains a key weapon in the fight against TB, but it only works as intended if people can stick to the schedule. Simpler instructions, less stigma, and creative support go further than any new formulation. Every missed dose opens the door to a tougher TB problem, so the real work lies in keeping treatment simple, supportive, and human.
Can Pyrazinamide be taken during pregnancy or breastfeeding?
The Real-World Risk of Tuberculosis
Tuberculosis still knocks hard in many corners of the world. Facing this infection during pregnancy or while breastfeeding pushes families and doctors to tough crossroads. Pyrazinamide, a mainstay in tackling active tuberculosis, sits at the center of a difficult question: Is it safe for pregnant or nursing mothers?
Understanding What’s at Stake
Expecting mothers already juggle enough concerns. Adding tuberculosis throws in higher risk of complications for both mom and baby. Delayed or incomplete TB treatment threatens lives—early action saves them. That sends most care teams hunting for safe, effective medications. Many people might assume if a medicine works, it’s fine to use. Doctors can’t be sure quite so easily, especially with a drug like pyrazinamide.
What We Know About Pyrazinamide in Pregnancy
Doctors worldwide lean on guidelines built by experience, research, and public feedback. Large agencies, including the World Health Organization, agree that treating active TB during pregnancy matters more than the discomfort of unknowns. They include pyrazinamide in recommended regimens. That said, solid research about its specific safety for pregnant women remains thin.
Animal studies didn’t turn up worrying birth defects or clear signs of harm when using typical TB doses. Years of clinical experience haven’t flagged big patterns of birth complications either. Still, most information comes from small groups or case reports, not huge studies. No one can claim complete certainty. Most doctors factor in the mother’s health, TB severity, and stage of pregnancy before writing the prescription. They turn to specialists and shared decision-making rather than acting alone.
Breastfeeding and Pyrazinamide: What’s Known
Many medications make their way into breast milk. With pyrazinamide, the amount showing up is low—much smaller than the dose given directly to treat TB. No strong evidence links breastfeeding during pyrazinamide therapy with harm to babies. Routine breastfeeding is not discouraged if a mother takes pyrazinamide for TB. Still, doctors prefer to keep an eye on infants for rare or unusual reactions. Drug-resistant strains and complicated TB situations prompt even closer supervision.
Some experts suggest vitamin B6 (pyridoxine) supplementation for both baby and mother to cut down on possible side effects tied to TB medicine, not just pyrazinamide. Personal doctors, nurses, and pharmacists offer guidance here, based on local conditions and updated research.
Why Clear Communication Matters
Typical TB drugs carry health warnings, sometimes printed in language that leans toward the worst-case scenario. That leaves many pregnant and nursing mothers feeling confused and anxious. Uncertainty can breed fear, and some may stop treatment early. This step can actually put babies and mothers at greater risk. Talking straight about benefits and trade-offs means fewer surprises and better decisions. Each person’s story—medical history, family setting, risk of catching TB again—matters for planning the safest approach.
Looking Ahead: What Still Needs Doing
Most research on drugs like pyrazinamide doesn’t focus on pregnant or breastfeeding women. There’s space for global health agencies and researchers to push for more targeted studies. Real-life stories from women who went through TB treatment during pregnancy or while breastfeeding offer insights that charts and numbers can’t always show. Healthcare teams that listen as much as they prescribe help families face tough times with knowledge, not just hope.
What should I do if I miss a dose of Pyrazinamide?
No Need to Panic, But Don’t Shrug It Off
Missing a dose of Pyrazinamide, a medicine used in treating tuberculosis, can feel intimidating. I understand the anxiety of realizing your routine slipped. Tuberculosis medications demand consistency because the bacteria behind TB don’t give you second chances so easily. Sticking to a steady schedule controls the infection, protects folks around you, and lowers the risk of the bacteria getting smart and developing resistance. As someone who’s had to juggle long pill routines during a health scare, I know how easy it is for a day to get away from you. Still, one missed pill isn’t the end, though it does deserve respect and a little action.
Why Skipping a Dose Isn’t Just About You
Pyrazinamide plays a unique role in tuberculosis treatment. Together with other meds, it chips away at bacteria hiding deep in tissues. Each dose is like a brick in a wall that keeps TB from spreading—both in your body and out into the world. Ignoring a missed dose can let the bacteria regroup, multiply, and possibly learn how to dodge treatment the next time. As TB remains a significant public health risk, especially in crowded living situations, every person’s drug adherence matters. Data from the CDC show that even a few missed doses can tilt the odds toward treatment failing.
What You Should Actually Do Next
Daily life throws curveballs—a missed alarm, busy schedules, or sudden changes all can make a dose slip your mind. If you realize within a few hours that you skipped Pyrazinamide, take it as soon as you remember. If it’s close to your next scheduled dose, though, skip the make-up and carry on as normal. Doubling up isn’t worth the risk. Too much Pyrazinamide can hurt your liver and cause side effects like nausea, joint pain, or yellowing eyes and skin. Skipping double doses isn’t just advice from doctors; it’s backed by solid safety data in standard TB guidelines.
Talk to Your Health Team — They’re On Your Side
Many people struggle to talk honestly to their medical team when they slip up, often out of embarrassment or fear. Doctors see patients miss doses all the time, and they know what tweaks to make in your plan. I’ve been in that seat, thinking a single mistake would get me scolded or judged. In reality, sharing these lapses can help your provider spot patterns or adjust your therapy before the situation grows harder to handle. Sometimes healthcare workers even line up practical supports—like pillboxes, reminder calls, or friends who keep tabs on you.
Long-Term Game Plan: Build Habits, Not Excuses
Fighting TB takes months. Habits shape that journey far more than willpower. Setting an alarm, linking medication to another daily habit (like brushing your teeth), or using a smartphone reminder really helps. If cost or transportation makes picking up refills tricky, most health departments step in with support. Modern TB care leans heavily on making sure people finish the full course, using approaches like Directly Observed Therapy (DOT). My own medicine streaks improved once I stopped being harsh on myself over an error and, instead, made the next dose a priority.
Final Thought: Missing Isn’t Failing—but Fix the System
Nobody sails through long treatment courses without a hiccup. One missed dose is an opportunity to notice what tripped you up and change something up, not a red mark. What matters most is jumping back on the schedule, connecting with your doctor, and making it a little easier for your future self to stick with it.
| Names | |
| Preferred IUPAC name | Pyrazine-2-carboxamide |
| Other names |
Aldiazid Pyrazinecarboxamide PZA Pirazinamida |
| Pronunciation | /paɪˌræzɪˈnæmɪd/ |
| Identifiers | |
| CAS Number | 98-96-4 |
| Beilstein Reference | Beilstein Reference: 112670 |
| ChEBI | CHEBI:8644 |
| ChEMBL | CHEMBL1404 |
| ChemSpider | 1047 |
| DrugBank | DB00339 |
| ECHA InfoCard | ECHA InfoCard: 100.004.670 |
| EC Number | 6.3.1.25 |
| Gmelin Reference | 31166 |
| KEGG | D08336 |
| MeSH | D011713 |
| PubChem CID | 1046 |
| RTECS number | UR8400000 |
| UNII | Z76Q289Y0R |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C5H5N3O |
| Molar mass | 123.11 g/mol |
| Appearance | White to almost white crystalline powder |
| Odor | Odorless |
| Density | 1.4 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -0.6 |
| Vapor pressure | 2.97E-11 mmHg |
| Acidity (pKa) | 14.3 |
| Basicity (pKb) | 2.9 |
| Refractive index (nD) | 1.613 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.47 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 160.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -104.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3744 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | J04AK01 |
| Hazards | |
| Main hazards | May cause allergic skin reaction; harmful if swallowed; may cause liver damage. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | Keep out of the reach of children. If medical advice is needed, have product container or label at hand. |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 1, Instability: 0, Special: - |
| Flash point | 79.6°C |
| Autoignition temperature | > 570 °C |
| Lethal dose or concentration | LD50 (oral, mouse): 4,090 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Pyrazinamide: "3,290 mg/kg (oral, rat) |
| NIOSH | PY0525000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Pyrazinamide: "15 mg/m3 (total dust), 5 mg/m3 (respirable fraction) |
| REL (Recommended) | 25 mg/kg |
| IDLH (Immediate danger) | IDLH: 2,000 mg/m³ |
| Related compounds | |
| Related compounds |
PZA metabolite pyrazinoic acid nicotinamide isoniazid ethyl pyruvate |

