Commentary on Propylarsonic Acid: A Closer Look
Historical Development of Propylarsonic Acid
Propylarsonic acid may not be a household name, but its background in the chemical industry traces back to a period when organoarsenic compounds caught the attention of agricultural chemists. My introduction to this field came through the stories of veterans who recalled the scramble for efficient animal feed additives and pesticide alternatives in the mid-20th century. It was during this search, with a lens on growth promotion and disease suppression, that propylarsonic acid entered laboratories. These early years saw researchers pushing the limits of food productivity, driven by a belief in chemistry’s ability to foster abundance. Focusing on ideas from predecessors like Salvarsan’s development, chemists saw organoarsenicals as a bridge between biological and mineral agents. Regulations did not loom as large as today, so the excitement over new performance benchmarks sometimes overshadowed questions about safety, an attitude that has shifted with time and experience.
Product Overview and Synonyms
In industrial circles, propylarsonic acid stands as an organoarsenic compound, often running under alternative names like propyl arsonic acid or 1-propyl-arsonic acid. Researchers working in veterinary applications would refer to it by these names, sometimes using legacy brand labels—though those have faded in global markets due to stricter controls. My own encounters with this compound surfaced when reviewing inventories of feed additives and legacy water treatment chemical records, and I realized the variety in nomenclature could confuse anyone outside specialty fields. It is important to highlight this diversity in product naming because it reflects how evolving scientific communication has changed both perceptions and usage patterns.
Physical and Chemical Properties
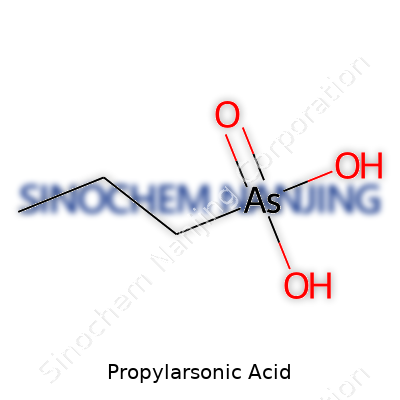
Chemically, propylarsonic acid features an organoarsenic backbone, substituting an arsenic atom onto a propyl group. In lab settings, the compound usually appears as a crystalline or powdery substance, soluble in water under most conditions. A typical sample produces no distinct odor, but the presence of arsenic rules out any lax attitude in handling. From my perspective, the chemical’s twin identity—organic in structure, metallic in toxicology—underscores a tension central to the history of synthetic agriculture. The acid’s stability under room-temperature conditions allowed it to fit into premixes for feed or water-soluble formulations. Its strong mineral acidity enabled reactivity, setting up further chemical transformations in research contexts.
Technical Specifications & Labeling
Labeling and handling requirements have grown much more thorough in recent decades. Any propylarsonic acid container today must feature hazard labels warning of arsenic toxicity and environmental risk statements. Looking back thirty years, the same chemical sometimes traveled under less rigorous documentation, but tightening workplace safety protocols changed that landscape. Packaging now must meet leakproof, corrosion-resistant standards set by transport regulators. Regulatory frameworks underline not only worker safety but also emphasize traceability in applications, especially with food-chain relevance. I have inspected several archived containers where faded labels and outdated handling instructions serve as reminders of how easily overlooked small lapses can snowball into major compliance problems.
Preparation Methods
The process for making propylarsonic acid involves a reaction between arsenic acids and propylating agents. In most documented protocols, chemists start with arsenic trioxide, dissolving it in acidic solution and introducing a propyl source such as propanol or propyl halides under controlled temperature. My own observation of such preparations reveals the importance of slow reagent addition—rapid temperature spikes or splash incidents can pose real dangers. Production-scale operations substitute more efficient continuous reactors, but even in these settings, the core principle holds: managing the exothermic reaction to prevent runaway. Such processes require ventilation, fume hoods, and emergency protocols that smaller labs sometimes struggle to maintain.
Chemical Reactions and Modifications
Propylarsonic acid does not lend itself to countless modifications but does offer some chemical flexibility. Its functional groups allow formation of salts and esters, and researchers working in analytical chemistry use this to tweak solubility or reactivity for detection techniques. For example, certain transformations increase the molecule’s compatibility with liquid chromatography or spectrographic analysis. The most striking aspect, to me, comes from the compound’s role as an intermediate—serving as a building block for other organoarsenicals. Yet this chemical flexibility comes with a burden, as improper disposal or mismanagement can produce persistent environmental traces. These risks persist in some old industrial sites, showing how even carefully planned syntheses create long-term impacts.
Safety and Operational Standards
Handling propylarsonic acid brings into focus the realities of chemical safety in an era increasingly intolerant of workplace risk. Today, facilities treat organoarsenicals with robust containment, ventilation, and personal protective equipment—gloves, goggles, separate storage. Long ago, workers sometimes got away with much less, and the consequences came into focus as arsenic’s health hazards became undeniable. Regulatory agencies now require training, exposure monitoring, and environmental controls, especially with materials involving drinking water or animal feeds. During site visits, I have seen the difference this makes—modern labs manage waste capture and decontamination far more thoroughly, often driven by fear of both regulatory and litigation consequences.
Application Areas
Historically, propylarsonic acid’s main use landed in animal nutrition, especially poultry and swine feed additives. The theory ran that small doses could ward off disease, boost feed efficiency, and stunt the growth of harmful microbes. This drew heavy use, especially in parts of Asia and North America, but concerns about arsenic residues and chronic toxicity started shifting attitudes. Some countries phased out its use altogether after studies linked routine exposure with carcinogenicity and ecosystem buildup. Beyond animal feeds, minor research uses touch on water treatment and analytical chemistry, but such uses never matched the scale of agriculture. Gradually, tightening regulations have pushed industries toward safer alternatives—reflecting a hard-learned lesson from environmental health research.
Research & Development Efforts
Research on propylarsonic acid now runs at a crossroads. Scientists who once aimed at optimizing its action for broader agricultural application now focus more on understanding legacy contamination and remediation methods. Analytical chemists work on better detection in soil and water, sometimes using advances in mass spectrometry or chromatographic separation. In my own engagements with academic groups, the drive to map distribution patterns in old livestock farmland reveals how stubbornly some compounds linger. Meanwhile, chemists developing new feed additives take lessons from the environmental side effects documented with propylarsonic acid, shifting innovation toward non-toxic growth promoters or immune enhancers. Collaboration between toxicologists, environmental chemists, and agricultural scientists defines current R&D, as knowledge about systems-level impacts expands.
Toxicity Research
Epidemiological and laboratory research paint a clear picture—propylarsonic acid belongs on the list of compounds demanding careful stewardship. Studies in animals link chronic exposure to a suite of problems, including cancer risk, organ damage, and neurological impacts. Data surfaced in the latter 20th century, spurring rounds of regulatory reviews and bans. Environmental studies uncovered residue buildup in soils, water tables, and even edible tissues, raising public health alarms. My own reading of toxicity reports highlights a pattern: lag time between adoption and regulation, often filled by heated debates in scientific and public forums. Efforts to replace or neutralize legacy deposits take time and money, underlining that safety must figure into both product launch and eventual disposal.
Future Prospects
Looking forward, the role of propylarsonic acid recedes rather than expands. New regulations in many countries limit or outright ban most organoarsenic compounds in agriculture and consumer products. The market for this chemical has shrunk, leaving research squarely focused on remediation, detection, and historical impact. Those traces left behind in agricultural fields still need to be addressed through soil remediation, water treatment, and continued monitoring. Alternative feed additives with lower ecological footprints are winning market share, driven by both consumer pressure and scientific consensus. Propylarsonic acid now serves as a case study in how technological optimism sometimes outpaces risk assessment. I take from this history a strong belief in the value of holistic chemical evaluation—before, during, and after introduction to the environment. Scientists and policymakers must keep pushing for safer options, and the lessons learned from propylarsonic acid will shape future decisions in agrochemicals and public health chemistry.
What is Propylarsonic Acid used for?
What Farmers Use Propylarsonic Acid For
Ask any livestock farmer what keeps animals healthy and eating, and most will mention growth promoters and feed additives. Propylarsonic acid falls into that line-up. Over the years, this chemical has popped up in the raising of poultry and swine, mainly added to feed. The goal is straightforward: help animals avoid gut infections and put on weight more efficiently.
Propylarsonic acid isn’t some miracle chemistry. It actually belongs to a family of organic arsenic compounds. The logic behind using it comes from a real struggle: farms everywhere have fought a long war against bacterial diseases in livestock. For example, coccidiosis—a gut-wrenching problem for broilers—shreds flock health and costs a fortune in lost birds. Early research showed that adding certain arsenic compounds helped fight these infections, which made animals grow better and use feed more efficiently.
Is It Safe to Use in the Long Run?
People worry about the chemicals involved in growing our food, and for good reason. Scientists have studied propylarsonic acid, and authorities across countries have set up rules around its use. In the United States, the FDA once allowed this compound as a feed ingredient, with certain limits. Over time, as evidence came out and as the public pushed for safer food, the rules changed. By 2015, most farm arsenicals got phased out.
Many older farmers remember these additives as just another item in the feed store. Back then, with less scrutiny, it was normal to aim for the most efficient animal and the fattest paycheck. Now, many are rethinking what goes into animal feed and how that affects people down the line.
What Science Says About Arsenic Compounds
The real worry isn’t just the pig or chicken munching on tainted feed. Eventually, low levels of arsenic can show up in the end product—chicken breasts, pork chops or even manure that gets spread on fields. Scientists have studied how these compounds break down in the environment. Some arsenic forms get flushed out, but others stick around in soil or water, building up over time. The trouble is, even low doses of inorganic arsenic can cause health problems in people.
Europe has long taken a stricter stance on these compounds in farming, while in the U.S. and parts of Asia, the ban caught on later. Growing consumer demand for cleaner food has forced everyone in the supply chain to find new solutions. For me, as someone who’s handled livestock and seen the difference in healthy and sick herds, moving away from unnecessary chemicals isn’t just a trend—it’s a return to what feels right.
What Could Take Its Place?
Without old-school chemicals like propylarsonic acid, producers are finding new strategies. Today, probiotics, vaccines, better hygiene, selective breeding—all play a bigger part. Yes, it costs more. There are hiccups getting everyone on board, especially for small farmers in tight markets. Still, the benefits show up over time: healthier animals, cleaner rivers and fields, better trust at the dinner table.
Using science-backed nutrition and management gives livestock a fighting chance without chemical shortcuts. Policy changes only work when backed by strong science and support for the people doing day-to-day farm work. If we keep listening to both farmers and consumers, we can shape food systems that don’t rely on risky additives and still feed everyone.
Is Propylarsonic Acid safe for animals or humans?
Understanding What Propylarsonic Acid Brings to the Table
Propylarsonic acid keeps popping up in farm supply catalogs and some agricultural chemistry reports. Its main use has been as an animal feed additive in certain countries, especially for poultry. Some see it as a growth promoter, others for disease control in livestock. The real question many folks—farmers, pet owners, even veterinarians—want answered: Is it safe for the creatures we care about, or for people who consume animal products?
Why People Started Paying Attention to Propylarsonic Acid
Many chemical additives in animal feed have attracted controversy. Stories of antibiotics and heavy metals in feed spark worries about food safety, pollution, or harm to livestock. Propylarsonic acid’s chemistry carries a red flag: it belongs to the family of arsenic-containing compounds. That’s not an accusation, just a fact from basic science. Arsenic in its pure or inorganic forms causes cancers, neurological issues, and problems with cardiovascular health in humans. Organic arsenic—that is, molecules like propylarsonic acid or roxarsone—has been used under the argument it’s less toxic or is quickly metabolized and removed from animal tissue. Whether that story holds up to current scrutiny is at the core of the safety debate.
Digging Into the Evidence
I’ve seen careful studies run by academic and government labs. Some research in the US and Europe found that animals fed organic arsenic compounds like propylarsonic acid could end up with residues in their tissues. These residues, often small, can turn into carcinogenic inorganic arsenic during processing or cooking. Even small amounts raise concern because chronic low-level exposure adds up. The World Health Organization has said there’s really no safe level of arsenic exposure, especially for long-term risks like cancers.
In the US, the FDA used to allow some arsenic additives in poultry feed. That changed. By 2015, products containing those compounds lost approval due to mounting concerns and improved testing technology. European regulators reached a similar conclusion, banning arsenic-based animal feed additives. China and other major agricultural producers are also re-examining arsenical use, reflecting a broader trend. This shift comes from solid data and a preference for erring on the side of caution.
Animal and Human Health Both in the Spotlight
Farmers look for ways to boost production and keep flocks healthy. It’s a tough job when global demand rises and disease pressures increase. But safety isn’t negotiable. Propylarsonic acid’s main risk doesn’t always show up as an obvious poisoning—rare for both animals and people in ordinary agricultural use. The real grit of the problem is hidden: kitchen tables become the site of long-term health risks for families eating contaminated meat or eggs. For farm workers handling feed, frequent exposure could mean gradual buildup in the body, increasing their risk as well.
What Could Replace Propylarsonic Acid?
Other feed supplements—probiotics, certain herbal additives, or carefully managed vaccination—can reduce disease in poultry without relying on chemical short-cuts. Regenerative farming practices and improved animal welfare standards help, too. But these solutions often carry higher costs or require changes to established routines. Transition isn’t instant; it has to make sense for both producers and consumers. Yet, watching how large food brands and responsible farmers adapt, it’s possible to see a future where risky chemical shortcuts lose their appeal.
Moving Forward With Caution and Care
Sitting at the dinner table, most people would rather not worry about what’s hidden in their chicken or eggs. As science makes it clear that even organic arsenic compounds can bring avoidable risks, the smartest move is to keep the food chain clean. This isn’t a shot in the dark—it’s about taking seriously what the research tells us. Policy and personal choice together can create a safer outcome for everyone with a stake in food and farming.
What are the recommended dosage and administration guidelines for Propylarsonic Acid?
Understanding the Substance
Propylarsonic Acid often finds its place in animal agriculture, particularly in the feed of poultry and swine. It serves as an organoarsenic compound designed to support growth promotion and disease resistance. Scientists and producers have paid closer attention to its usage due to health and regulatory concerns that spring from the potential buildup of arsenic residues. The dosage and application steps for this compound carry some nuance, and experience shows this isn’t one of those “add and forget” feed additives.
Recommended Dosage in Practice
Practical books, along with documents from the U.S. Food and Drug Administration and international authorities, commonly place Propylarsonic Acid dosages for swine and poultry between 22 and 45 parts per million (ppm) in finished feed. In my time on a poultry operation during a summer internship, the nutritionist explained that exceeding the threshold invites more than just regulatory scrutiny—birds exposed to higher levels showed a notable drop in feed efficiency. On top of that, some limits shrink as slaughter approaches; withdrawal periods help ensure that tissue residues fall within safe margins.
Administration Guidelines
Propylarsonic Acid comes premixed in feed-grade products, as bulk powders or liquids that staff blend directly into feed rations. Feed mills using manual batching need close measurement; even a small miscalculation leaves the whole batch off-spec. Most modern mills rely on automated systems to weigh and add the product, but I’ve seen a few older operations where scale accuracy still depends on the operator’s eye. Staff safety can’t take a backseat, so handling guidelines recommend gloves and dust masks due to the chemical’s toxic nature and potential for irritation.
Talking with veterinarians and extension agents, withdrawal times became a recurring message. For instance, broiler chickens fed 45 ppm need a minimum of five days on non-medicated feed before heading to slaughter. Hogs show a similar schedule. Farmers often tackle this by keeping detailed barn records covering start and stop points, adjusting the rations ahead of shipment. Missing a withdrawal period has real consequences, not just for regulatory fines but for market reputation as well.
Addressing Concerns and Moving Forward
Much of the concern around Propylarsonic Acid relates to food safety and environmental runoff. Studies from places like the National Academies press for both proper dosing and responsible use. Chicken growers I met stayed in touch with extension specialists to get early warnings about local guideline changes. Experience helps too—overyears, many have switched to alternative products as arsenic limits tightened worldwide. Producers choosing this compound for growth have weighed the costs against new consumer concerns about arsenic, often opting for enhanced biosecurity and new feed regimens instead.
Regulators such as the FDA and EFSA revise their advice as new data emerges. Staying connected to reliable sources—veterinarians, feed manufacturers, government bulletins—remains vital. Routine record-keeping, batch testing, and investment in automated feed delivery systems help avoid dosing errors. Education and diligence, more than anything, shape safe and responsible usage.
Potential Paths Forward
From what I’ve seen, the future for additives like Propylarsonic Acid leans on technology and transparency. Digital feed management, routine ingredient audits, and open lines with regulatory bodies give farmers confidence in the rations they deliver. Ongoing research might yield safer alternatives, but for now, following published dosages and working closely with veterinarians stand as the clearest route to both safe food and steady farm returns.
What are the potential side effects of using Propylarsonic Acid?
Agriculture’s Push for Yield Meets Chemical Realities
Farming feeds the world, but it also leans heavily on chemicals to guard crops against pests and disease. Tools like Propylarsonic Acid, an organoarsenic compound still found in some agricultural practices, show how the desire for higher yields can complicate farm safety and environmental health. My time spent helping on extended family farms gave me a close-up view of these tradeoffs. On one hand, pesticides and barn disinfectants drive away bugs and bacteria. On the other, they introduce worries, especially when residues show up in water or food down the line.
Understanding Human Health Risks
Looking at the research, Propylarsonic Acid isn’t just harmless runoff. Exposure, even at low levels, can trigger a host of side effects in humans. Most public health warnings tie back not just to acute poisoning, but to longer term concerns.
Short-term exposure can cause a burning throat, chest pain, and dizziness, which hospital staff recognize as symptoms of arsenic compounds. Rashes and skin irritation are common when solution comes in contact with unprotected skin. I once developed a prickly red rash on my arms after cleaning out an old barn, only to learn later the disinfectant included low-dose arsenic.
Long-term risks are more troubling. Chronic exposure to Propylarsonic Acid raises the risk of organ damage. The World Health Organization warns that low-level arsenic exposure over years can slowly harm the liver, kidneys, heart, and central nervous system. Cancer risk goes up, especially linked to liver and skin cancers. There's no denying that repeated contact, even through residues on food, can build up harm over time.
Children stand out as the most vulnerable. Their smaller bodies absorb toxins faster, and the effects stick with them as they grow. Families living near sprayed fields often wonder whether coughs or learning problems can be linked back to chemicals in the groundwater. Evidence from rural health clinics absolutely shows higher rates of illness where arsenic-based agents get regular use.
Wildlife and Waterways Caught in the Crossfire
Beyond human health, Propylarsonic Acid transfers to soil and water, affecting fish, frogs, and birds. Aquatic life struggles as arsenic builds up in rivers and lakebeds. Reduced reproduction and altered growth cycles show up fast in bioassays. Fisheries downstream from treated fields suffer, putting both animal and human diets at risk. My uncle’s community saw a drop in fish catches after years of using similar compounds on rice paddies. Locals noticed the change well before any scientists arrived with water test kits.
Runoff laced with organoarsenic doesn’t respect property lines. Rainwater can move chemical residues for miles, blending them into new ecosystems and food webs. Rural clinics and wildlife rehab centers often tell the same story: sick animals and unexplained illnesses spike during and after chemical applications on large monocrop farms.
Better Practices and Safer Alternatives
Communities that guard local food and health push for transparency on what lands in their soil and water. Switching to safer, less persistent pest control has made a difference. Biological pest management, crop rotation, and certified organic practices help reduce the demand for toxic metals. Recent studies show farms using these systems record fewer cases of pesticide poisoning and also maintain more stable wildlife populations. Agricultural groups in some regions now track chemical residues and flag dangerous spikes early.
Safer farming and careful monitoring don’t just protect workers and families; they keep whole landscapes healthy for future generations. Direct contact still carries serious risks, and the lingering effects in our water, soil, and bodies remind us to push for stronger, smarter solutions in agriculture—especially when old methods threaten lasting harm.
Are there any restrictions or regulations regarding the use of Propylarsonic Acid?
What Makes Propylarsonic Acid a Talking Point
Propylarsonic acid, a chemical once found in agricultural and animal feed industries, pops up in discussions about farm safety, food integrity, and environmental protection. This compound drew attention for its previous use as an animal feed additive, mostly for poultry. The science community and regulators flagged it years ago for its arsenic content. People started to question what side effects might turn up along the food chain or in water runoff from farms.
Oversight and Public Health Stances
Regulators haven’t gone easy on chemicals with arsenic, for good reason. Studies from the World Health Organization and the Centers for Disease Control have raised alarms about chronic exposure to arsenic leading to cancers, cardiovascular issues, and developmental problems in children. Those risks don’t sit idle in theory—they push governments to act. The European Union and the United States both took action on these kinds of feed additives. The FDA, for instance, withdrew approvals for most arsenic-based animal drugs a decade ago.
Rules on Use and Where They Still Matter
Folks in the farming industry can’t just buy or use propylarsonic acid as they once did. Most developed countries now ban or strictly control this chemical in animal feeds. If a farm gets caught using it, there can be stiff legal and financial consequences. There’s extra oversight for food producers as well. Crops or livestock tested for arsenic above safety limits get flagged; those products don’t make it to grocery shelves. If I look at labels and traceability reports from retailers today, I notice how much scrutiny surrounds products that might have even trace arsenic.
Leftover Risks in Water and Soil
Bans on current use don’t erase past impact. Runoff from old use or careless disposal contaminated some soil and waterways. Farmers working the same land decades later still have to test for legacy contaminants. Environmental groups and scientists recommend regular monitoring near areas once tied to these feed additives. It’s easy to forget that what gets dropped in a field or pen can show up in groundwater and rivers years later.
Addressing the Leftover Legacy
Better transparency and regular testing work as frontline shields. Community groups and public agencies often pitch in with soil and water checks. Where contamination persists, they might use soil remediation—removing and replacing contaminated dirt—or install filters on wells. It’s expensive and never instant, but these actions matter for safe food supplies and clean drinking water. While I’ve seen some frustration among small farmers over red tape, no one wants to risk their health or business selling a tainted product.
Moving Past Propylarsonic Acid
Research keeps pushing toward cleaner farming methods and animal feed alternatives. Safer feed additives have found footholds, and organic producers sidestep arsenic-based compounds entirely. Consumer demand for clear ingredient lists and food-safety data hasn’t faded, either. In every grocery trip, I see verification labels that mean someone checked for past and present contaminants. The story of propylarsonic acid shows how regulation, science, and public pressure mix to shape what lands on our tables and in our water glasses. Every step away from these risky chemicals means a step toward peace of mind on the farm and in the kitchen.
| Names | |
| Preferred IUPAC name | Propylarsonic acid |
| Other names |
4-Hydroxyphenylarsonic acid p-Hydroxyphenylarsonic acid ASA-214 Acide propylarsonique |
| Pronunciation | /ˌprəʊ.pɪˈlɑːr.sɒn.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 2457-96-7 |
| Beilstein Reference | 1469616 |
| ChEBI | CHEBI:38756 |
| ChEMBL | CHEMBL1614775 |
| ChemSpider | 14594 |
| DrugBank | DB14606 |
| ECHA InfoCard | 100.018.596 |
| EC Number | 212-171-4 |
| Gmelin Reference | 8781 |
| KEGG | C18718 |
| MeSH | D011376 |
| PubChem CID | 66247 |
| RTECS number | UD5950000 |
| UNII | V6OU564D6Q |
| UN number | UN3265 |
| Properties | |
| Chemical formula | C3H9AsO3 |
| Molar mass | 184.03 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.692 g/cm3 |
| Solubility in water | Soluble |
| log P | 0.38 |
| Vapor pressure | 0.01 mmHg (20°C) |
| Acidity (pKa) | 1.92 |
| Basicity (pKb) | 12.39 |
| Refractive index (nD) | 1.528 |
| Viscosity | Viscosity: 1.52 mPa·s (at 20 °C) |
| Dipole moment | 1.63 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 160.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -589.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1277.8 kJ/mol |
| Pharmacology | |
| ATC code | QOCA01 |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H301: Toxic if swallowed. H331: Toxic if inhaled. H373: May cause damage to organs through prolonged or repeated exposure. |
| Precautionary statements | P260, P262, P264, P270, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P314, P330, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-2-1-W |
| Autoignition temperature | 255°C |
| Lethal dose or concentration | LD50 (oral, rat): 2,500 mg/kg |
| LD50 (median dose) | LD50 (median dose): Mouse oral 482 mg/kg |
| NIOSH | Not established |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Propylarsonic Acid: Not established |
| REL (Recommended) | ADI 0-7 mg/kg bw |
| IDLH (Immediate danger) | IDLH: 250 mg/m3 |
| Related compounds | |
| Related compounds |
Arsanilic acid Roxarsone p-Aminophenylarsonic acid Nitarsone |

