2-Chloropropionyl Chloride: Chemical Relevance and Future Pathways
Historical Development
Chemists have often sized up the impact a single compound can make on the world of molecules. 2-Chloropropionyl chloride delivers a striking example, tracing its early synthesis back through industrial chemistry annals by way of acyl chloride research. This compound first caught strong attention as a functional derivative of propionic acid in the middle decades of the twentieth century, when large-scale chlorination and acylation methods moved from exploratory lab benches into basic industrial usage. Around the 1950s and 1960s, advances in chlorination techniques drove up both access and application. As more researchers recognized its ability to kickstart reactions, 2-chloropropionyl chloride carved a niche in the manufacture of specialty chemicals and later, pharmaceuticals and fine chemicals. While the compound’s first roles leaned toward solvent modification and herbicide precursor chemistry, evolution came with new organic synthesis needs. Over several generations, it grew common to see this reagent on the benches of industrial research chemists, especially in Europe and North America.
Product Overview
Today, 2-chloropropionyl chloride stands as an important acyl chloride, usually appearing as a colorless to light yellow liquid. Chemists often reach for it where selective acylation or halogenation is required. It finds work in fine chemical production, with those seeking to introduce a two-carbon acyl chain topped by a chloride group considering it essential. Its volatility and reactivity mean it doesn't collect dust in storage; research and commercial users move it quickly from bottle to reaction. It rarely shows up in consumer-facing products, but plenty of everyday goods in pharmaceuticals, plastics, and crop protection trace their histories back to a plant or bench where someone used 2-chloropropionyl chloride along the way.
Physical and Chemical Properties
2-Chloropropionyl chloride doesn’t act shy around water or alcohols, reacting briskly to give off hydrogen chloride gas. Its molecular formula shapes out as C3H4Cl2O, with a boiling point typically situated around 120°C under standard pressure. Chlorine atoms bring about strong tendencies toward nucleophilic substitution, and the acyl chloride group is always ready to react. The faintly sweet, acrid smell warns of its aggressive chemistry. Even a seasoned chemist treats samples with care. On contact with even a trace of moisture in air, it fumes to form misty, corrosive clouds of hydrochloric acid vapor. Standard chemical gloves and ventilation help, but protective eyewear often comes out first.
Technical Specifications and Labeling
Factories and research labs label it following guidelines that reflect its toxicity and dangerous reactivity. You’re not likely to see this compound in high school labs due to the hazards, though university settings sometimes keep tightly controlled ampoules for advanced organic synthesis courses. Transport and storage require secure containers, and robust labeling provides hazard statements under regulations like the European GHS/CLP. Refrigeration isn’t always necessary, but low humidity environments slow down decomposition. Scientists sometimes note the need for tightly sealed containers—one loose cap can lead to nasty surprises. Technicians often rely on clear labeling of batch numbers and hazard warnings, and regular checks of material safety data sheets keep everyone alert to both the chemical’s identity and best handling practices.
Preparation Methods
Preparing 2-chloropropionyl chloride usually starts with 2-chloropropionic acid, which itself can be made by chlorinating propionic acid. The classic route to the acyl chloride runs through either thionyl chloride or phosphorus trichloride, where the reagents strip away water and tack on chlorine. The reaction throws off gaseous byproducts, often requiring condensation traps, fume hoods, or scrubbers to control hazardous emissions. Questions about scalability shape most production decisions; researchers seeking small quantities might still run classic glassware setups, while industrial sites lean on closed, continuous systems, favoring both yield and operator safety. Each method underscores process control and worker protection throughout—those whiffs of gas are never ignored.
Chemical Reactions and Modifications
This chemical’s main claim to fame traces to its ability to deliver the 2-chloropropionyl group to a wide selection of amines and alcohols. Peptide chemists sometimes use it for selective N-acylation. Industrial operators have long counted on its ready reactivity for manufacturing herbicides and intermediates in pharmaceuticals. Given its structure, reactions with nucleophiles like amines or phenols come quickly, attaching the acyl group and leaving behind hydrochloric acid as a byproduct. Functionalization can also extend beyond the parent group, with the ability to substitute the alpha-chlorine for other, more complex organic frameworks if conditions are right. The making of specialty polymers—incorporating functional handles for later reaction—draws on these characteristics, making it a building block for much larger molecules.
Synonyms and Product Names
You’ll find this chemical listed under several names, but 2-chloropropionyl chloride and alphachloropropionyl chloride pop up most often in research journals and catalogs. Other users call it 2-chloropropanoic acid chloride or, in some patent filings, simply “chloropropionyl chloride.” CAS Registry often helps resolve confusion, especially in international trade or regulatory filings. Despite these synonyms, material shipped around the world largely carries similar hazard statements and control requirements.
Safety and Operational Standards
Any lab or factory using 2-chloropropionyl chloride puts serious effort into worker safety. The potential for skin burns, eye damage, and respiratory irritation runs high. I’ve worked with chlorinated acyl chlorides enough to know that a quick slip-up, like knocking over a flask, doesn’t end in a small mess—it can mean evacuating a lab. Standard recommendations call for protective clothing, gloves, and chemical goggles, always paired with effective ventilation. Training walks newcomers through not just proper technique, but also what to expect if accidents occur. Emergency eyewash stations, absorbent pads, and spill neutralizers stay stocked. In tougher regulatory climates, government inspectors will spot-check safety protocols, and labs that slack off can face real penalties. Waste handling demands extra vigilance, as the hydrolysis byproducts corrode plumbing and harm wastewater systems.
Application Area
This compound doesn’t show up in home chemistry sets or general consumer markets—its territory stretches across professional research, chemical manufacturing, and industrial process streams. Herbicide development has relied on it since the 1970s, making it a backbone precursor for selective weedkillers. In pharmaceuticals, custom organic syntheses take advantage of its activation of difficult substrates, often building up key intermediates in just a few steps. Polymer chemistry benefits from its ability to introduce reactive functionalities at precise positions. For those working on specialty plastics, adhesives, or dyes, this acyl chloride enables unique structural tweaks not easily achieved with alternative reagents. By no means a commodity chemical, its value lies in creating structure and function—rarely the final stop in a synthetic journey but an essential one.
Research and Development
Academic chemists keep pushing the limits of how 2-chloropropionyl chloride can shape new molecules. Medicinal chemists have stretched its use to developing enzyme inhibitors and building blocks for biotech tools. Polymer scientists, drawing on its reactivity, engineer new macromolecules for medical devices and responsive materials. Sustainable chemistry researchers look to cut down on hazardous byproducts by swapping in greener methods, pushing for both higher yield and lower exposure risks. The constant search for new reactions, milder conditions, and alternative feedstocks asks companies to revisit established processes. Conferences routinely showcase unexpected application improvements, with teams reporting more selective catalysis or new downstream reactions enabled by the unique properties of this compound.
Toxicity Research
A big part of what separates this chemical from commodity substances traces back to its potential dangers. Toxicologists note that direct exposure can burn tissues, and repeated inhalation or skin exposure can cause lasting health effects. Studies dating back decades outline its acute toxicity, and more recent work extends into chronic exposure—occupational safety teams use these findings to set stricter handling protocols. Environmental scientists monitor emissions from plants making or using chlorinated acyl chlorides, recognizing risks not just to workers but to downstream water and soil. As global regulations toughen and detection methods improve, more companies invest in containment, monitoring, and alternative reaction routes to minimize off-site hazards.
Future Prospects
Looking ahead, 2-chloropropionyl chloride’s continued success links to a few trends. Chemists hope for greener and safer production routes, using either milder chlorinating agents or recyclable solvents. Industry groups invest in automation to lower exposure, putting more distance between operators and reaction sites. Academic groups explore how its structure can inspire the next generation of bioactive molecules or materials. As regulatory pressure builds, innovation in containment, neutralization, and remote handling may remake older plants and open new markets. My own experience hints that newer, multifunctional acyl chlorides may one day take some of its turf, but the base need for selective, controllable reaction chemistry keeps it in heavy rotation in advanced applications. Its story holds up as a reminder: one well-understood, carefully handled reagent still powers a surprising share of modern chemical advances.
What is the chemical formula of 2-Chloropropionyl Chloride?
Digging Into the Makeup of a Key Chemical
Chemicals bring an element of curiosity, especially for folks who like knowing exactly what's going into the products used in the lab, in industry, or even in household products. Take 2-Chloropropionyl chloride, for example. The name sounds pretty technical and maybe even intimidating, but the formula itself—C3H4Cl2O—has a logic behind it. This compound uses three carbons, four hydrogens, two chlorines, and a single oxygen atom.
Why Chemical Structure Matters
Looking at the features of 2-chloropropionyl chloride, the formula carries a practical benefit beyond memorization for science exams. Those chlorines and the acyl chloride group mean the molecule reacts quickly, which finds value in chemical synthesis. It helps make other chemicals, right down to pharmaceutical intermediates or agrochemicals.
Real-world experience tells me that knowing a molecule’s formula provides a kind of roadmap—not just for safe handling but also for realizing where risks could pop up and how to avoid trouble. The formula, C3H4Cl2O, tells you there's an acyl chloride group ready to react with alcohols, amines, or water. Those reactions often produce strong fumes and heat. In lab work or a production plant, preparation means the difference between smooth operations and emergency showers.
Health and Environmental Impacts
Chlorinated compounds can linger in the environment and pose dangers to health. 2-Chloropropionyl chloride isn’t something to spill or breathe in. The chemical formula gives a hint about those risks—the two chlorines signal both reactivity and toxicity. Chlorinated compounds show up time and again in incident reports from chemical spills, so it makes sense to pay attention before trouble shows up. Anyone working with this compound would want robust safety measures, like fume hoods, gloves, and splash goggles.
Disposal brings up another issue. Tossing chemicals like this down the drain risks harming water systems and the greater environment. Experienced chemists always follow local regulations and work closely with hazardous waste handlers. Knowing the formula is the starting point for predicting how it will behave if it escapes into water or soil, and planning what to do about that.
Improving Handling and Reducing Hazards
To lower risk, it takes training, not just rules. Seeing a chemical formula on a bottle turns abstract numbers and letters into an everyday safety reminder. Industry groups and chemical safety organizations regularly push for updated guidelines and constant education. In my time working with these kinds of compounds, safety meetings—backed up by real stories and not just dry rules—have stuck with me more than page after page of procedures.
So every time someone reads "C3H4Cl2O" on a label, it should spark thought about reactivity, safety, and responsibility. Knowing the formula is just the starting point for treating chemistry with respect and care, both for people and for the planet we all share.
What are the main applications of 2-Chloropropionyl Chloride?
Chemical Building Block in Pharmaceuticals
2-Chloropropionyl chloride shows up again and again in the pharmaceutical industry. Its main value comes from its ability to act as a starter molecule, letting scientists build more complicated compounds piece by piece. For example, many drugs in development and on the market have roots that trace back to derivatives made from this chemical. This is especially true for drugs targeting cancer or inflammation. Companies want ways to add specific chemical features to a molecule, and here, 2-chloropropionyl chloride earns its keep. Starting with this compound can improve yield and reduce byproducts, both goals every chemist appreciates.
Making Needed Agrochemicals
Farmers depend on pesticides and herbicides for reliable harvests, and many of these chemicals contain parts derived from 2-chloropropionyl chloride. Its role in making certain active ingredients matters for global food security. It allows for the introduction of chlorine atoms and specific carbon chains into larger molecules, which alters how crops fend off pests or weeds. A farmer in Iowa might never hear about this substance, but its fingerprints show up in the tools available to fight blight and increase yields. Trends in resistance mean chemists must keep tweaking existing formulas, and they often choose raw materials like this one for flexibility and accuracy.
Role in Polymer Manufacturing
Companies making specialty plastics often turn to 2-chloropropionyl chloride midway through the process. The substance lends itself to producing polymers that resist chemicals, heat, or physical stress. Think about factory machine parts, coatings for electronics, or even robust medical devices: many require plastics that keep their shape and properties under stress. In my own experience working with a supplier for automotive components, materials sourced from these processes proved critical for quality and safety. Productivity improved because parts didn’t deform or break easily, translating to savings for both manufacturers and customers.
Fine Chemical Synthesis and Custom Chemistry
Labs use 2-chloropropionyl chloride for a lot of ‘custom chemistry.’ It’s not flashy, but when making a unique dye, flavor compound, or research reagent, having a reliable starting point matters. Academics and industry experts alike value repeatable, well-understood reactions, and this chemical fits the bill. From textbook syntheses in graduate labs to small-batch specialty chemical production, its place is secure. I’ve seen this in collaborations between universities and startup companies aiming for next-generation sensors or new colors for paints. Failures to source or handle this reagent properly can stall entire R&D projects.
Safety, Regulation, and Sustainability
Handling 2-chloropropionyl chloride comes with real challenges. It reacts strongly with water, releases corrosive gases, and can harm skin or lungs. In industries where exposure risk exists, safety training and containment procedures aren’t optional. Regulatory oversight stands for good reason. Waste disposal and emissions get monitored closely, and governments regularly update rules as new research comes in. Looking to the future, researchers are searching for greener ways to make and use these chemicals, hoping to shrink the environmental footprint. Many companies are adopting closed systems, minimizing leaks and exposure, and developing alternatives based on renewable feedstocks to address long-term sustainability.
What are the storage and handling requirements for 2-Chloropropionyl Chloride?
Getting Real About Chemical Storage
2-Chloropropionyl chloride comes with a reputation that’s justified. Sitting in front of a drum of this stuff, you feel its sting in more ways than one. I remember my first encounter — that sharp, suffocating vapor, the reminder from the safety officer that this wasn’t your average solvent. Companies working with this chemical know that cutting corners in storage and handling spells trouble.
Why Control Matters
This is a moisture-reactive acid chloride, not something you want near water. Pop the lid in a humid room and you’ll get a noseful of HCl gas, probably a ruined product, and maybe a call to the hazmat crew. The law steps in here for good reason: exposures lead to severe burns and nasty lung troubles. Statistics from the CDC underline this, listing acid chlorides among the culprits behind accidental exposures every year.
The Real Storage Requirements
Corrosion eats through standard shelving, so shelve this chemical above solid, chemical-resistant trays. Stainless steel or polyethylene containers work best for leaks, and you can’t just toss these in any cabinet. Dedicated, ventilated acid cabinets keep vapors at bay. Those yellow cabinets with heavy locking bars you see in labs are not just for show — they’re built to keep volatile chemicals separated from vinegar and reagents that might trigger a runaway reaction.
Temperature swings make things worse. Keep everything cool and away from direct sunlight. There’s no need for a deep-freeze, but a steady, moderate climate goes a long way. Jugs and drums must stay tightly sealed, with clear hazard labels showing exactly what’s inside. It’s easy to spot where people mess this up: sticky, corroded lids and warning labels curling off with age give away neglected stocks.
Sensible Handling Practices
Before unscrewing anything, gloves and goggles go on. Splash-resistant aprons give added protection, and full-face shields come out for big batches. Fume hoods eat up the nasty vapors, keeping noses and lungs safe. Double-checking that sprinklers and eyewash stations work is non-negotiable. I’ve seen situations where one careless splash led someone to the ER in minutes — quick access can mean the difference between a close call and a disaster.
Training and Documentation
Every handler deserves real training, not just a once-over of the data sheet. Emergency drills let people practice what matters most: dealing with spills, accidental contact, and those rare, heart-pounding moments when alarms blare. Following OSHA standards means keeping records up-to-date and making those safety data sheets easily available. If containers ever leave storage, even for a transfer to production, chains of custody help prevent mix-ups or misplaced containers.
Pushing for Safer Alternatives
Some companies push for substitutes in research or pilot runs. Green chemistry lags on certain acid chlorides, but engineers keep hunting for drop-in replacements that cut down risks. In the meantime, strict storage and unbending attitude toward safety stay necessary. Skipping even one step in storage or handling sets up more than an accident; it sabotages everyone’s trust on the job.
What safety precautions should be taken when using 2-Chloropropionyl Chloride?
The Reality of Handling 2-Chloropropionyl Chloride
Anyone who has ever spent time in a research or production lab knows that chemicals like 2-Chloropropionyl chloride demand respect. Exposure to its vapors or liquid can burn skin, damage lungs, and cause eye injuries. That isn't just a warning from safety data sheets—chemists have experienced splashes and learned the hard way that comfort zones have no place near reactive chemicals.
Why 2-Chloropropionyl Chloride Is So Risky
This chemical reacts fiercely with water, releasing hydrochloric acid gas. That gas irritates the throat and lungs. Skin contact causes burns. If any gets in the eyes, you’re dealing with a race against permanent injury. Breathing in even small amounts leaves throats raw and sometimes sends folks home sick. Handling it without thinking through each step leads straight to accidents.
Personal Protection Always Comes First
Start every session in a proper lab coat. Lab mates tease about “looking like a surgeon,” but the stories about folks skipping goggles or gloves always end with regret. Eyes need chemical splash goggles, not just glasses. Face shields add another layer, especially when shaking, pouring, or working with larger amounts. For hands—go with thick nitrile or butyl rubber gloves, and double them up if possible. Splatters find their way through tiny holes or under cuffs faster than people imagine.
Modern hoods with strong airflow offer more than comfort—they clear away dangerous vapors. I’ve seen what happens when old hoods barely clear a chessboard-sized area on the bench: one slight breeze, and fumes drift to someone two meters away. Before using any hood, check the flow indicator. No airflow, no work.
Planning for Spills—and Preventing Them
Small bottles with tight lids beat oversized containers every time. Pour into secondary containment trays, not straight onto the bench. I remember a graduate student who knocked over a beaker reaching for lab tape; the secondary tray stopped a disaster. Kits with absorbent pads, calcium carbonate, and plenty of gloves belong nearby, not locked away in a cabinet down the hall. Everyone in the room needs to know exactly what to do if something spills.
Storage and Waste: Get Serious
Nobody wants to dig around a cluttered fridge for a bottle that’s leaking fumes. Keep 2-Chloropropionyl chloride in tightly sealed containers, labeled with clear hazard warnings. Store far from water sources, acids, and anything incompatible. I’ve seen bottles stored above eye level tip off shelves—never again. Heavy containers belong on lower shelves or in dedicated, ventilated bins.
Waste handling isn’t just about pouring leftovers into a bottle. Add neutralizers slowly, and never add water directly to the chemical; let it react in a controlled way. Use well-marked sealed waste drums, and call licensed hazardous waste disposal services promptly. Untrained hands and improper disposal wreck the work environment and risk community safety.
Training and a Safety-First Culture
No safety talk covers every curveball, but real experience sharpens instincts. Read the safety data sheet before each use. Practice drills for spills or accidental contact. Teamwork matters—a buddy system means someone’s there to call for help or rinse eyes if things go wrong.
Safe handling can’t be left to chance or wishful thinking. When everyone takes these steps seriously, labs produce more science and fewer injuries. The peace of mind that comes with a strong safety culture is something every chemist comes to value.
What is the shelf life of 2-Chloropropionyl Chloride?
Knowing What You’re Working With
2-Chloropropionyl chloride isn’t exactly a household name, but folks who use it in pharmaceuticals, agrochemicals, and specialty chemistry know its importance. This clear liquid doesn’t hang around forever, so remembering how it behaves on the shelf keeps people safe and saves companies a lot of trouble. Left too long, it becomes unpredictable—so nobody should toss a drum in storage and forget about it.
The Real Shelf Life—Not Just a Number
Manufacturers often say this compound, if sealed and stored in a cool, dry spot, lasts about a year. Some stretch it with special drums, low temperatures, and nitrogen blanketing, but in everyday conditions, you’re usually looking at 12 months. Temperature swings speed up decomposition; high humidity guarantees fuss. Even the smallest bit of water triggers hydrolysis, turning the compound into hydrochloric acid fumes and other nasty byproducts. One slip in a warehouse can mean production delays or safety scares down the line.
Why Stability Drops Fast in Bad Storage
Anybody who’s spent time with acid chlorides knows they act up with moisture. Humid warehouses or poorly sealed containers cut shelf life in half—not just on paper, but in surprise reactions and ruined batches. I once saw a small leak ruin an entire shipment before it hit the production line. The loss wasn’t just financial; every sealed container meant to outlast a year turned dangerous, backing up the line for weeks.
Verifying Quality Matters More Than Dates Printed
Expiration dates help, but the real story comes from testing. Analytical chemists often run gas chromatography or titration on stored material for good reason. Even if the label says ‘good through April,’ smart shops double-check purity. If there’s yellowing, weird odors, or pressure in the drum, the material’s likely gone south. Overlooking this step leads to loss in yield and unwanted side reactions that demand extra cleanup and money later.
Best Practices that Actually Work
Clean, dry, and cool storage beats fancy tech every time. Keep drums in well-ventilated sheds or temperature-controlled rooms. Use desiccants, keep air out with nitrogen, and open containers only when absolutely needed. Spills or leaks go straight to hazardous waste. Train every person who handles the drum; people rushing to save time often pay with ruined product or worse—injuries. Labels should list the date opened and the person responsible. It sounds tedious, but I’ve seen accountability cut waste by a third at my old job.
Safer Solutions Instead of Rushing Replacements
Some labs push alternative reagents or order smaller quantities more frequently, keeping stocks fresh. Just-in-time delivery reduces leftover material and disposal headaches. Larger firms sometimes use data loggers to track temperature and humidity at every step between factory and lab.
It Comes Down To Vigilance
Stability isn’t a guessing game. Understanding the limits of 2-chloropropionyl chloride helps avoid wasted stock, lost money, and safety violations. Real experience in the lab or warehouse exposes the truth behind the numbers, and that’s where the real risk—or savings—turn up. There’s no substitute for diligence when dealing with compounds that turn dangerous if ignored.
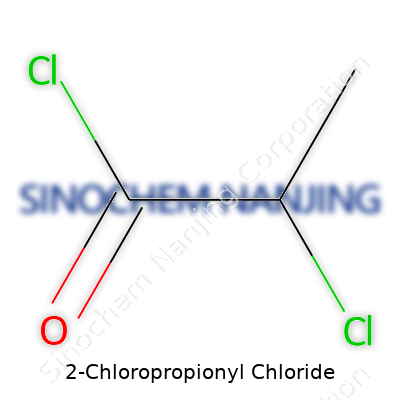
| Names | |
| Preferred IUPAC name | 2-chloropropanoyl chloride |
| Other names |
2-Chloropropanoyl chloride 2-Chloropropionic acid chloride alpha-Chloropropionyl chloride alpha-Chloropropionic acid chloride |
| Pronunciation | /tuː-klɔːr.oʊˈproʊ.pi.ə.nɪl ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 758-94-1 |
| 3D model (JSmol) | `JSmol.loadInline('set background [xffffff]; load data "mol" C1C(Cl)C(=O)Cl END "mol";')` |
| Beilstein Reference | 1438731 |
| ChEBI | CHEBI:85064 |
| ChEMBL | CHEMBL3337120 |
| ChemSpider | 87504 |
| DrugBank | DB14441 |
| ECHA InfoCard | 03b4b5de-ae40-4f40-80ed-5fbc1f9b6d51 |
| EC Number | 209-950-3 |
| Gmelin Reference | 8216 |
| KEGG | C19107 |
| MeSH | D017691 |
| PubChem CID | 69711 |
| RTECS number | UF3850000 |
| UNII | 1H24808D8E |
| UN number | UN 3265 |
| Properties | |
| Chemical formula | C3H4Cl2O |
| Molar mass | 142.44 g/mol |
| Appearance | Colorless to light yellow transparent liquid |
| Odor | Pungent |
| Density | 1.38 g/mL at 25 °C (lit.) |
| Solubility in water | Reacts with water |
| log P | 1.99 |
| Vapor pressure | 14 mmHg (20°C) |
| Acidity (pKa) | 1.8 |
| Magnetic susceptibility (χ) | -8.6×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.442 |
| Viscosity | 0.89 cP (20 °C) |
| Dipole moment | 1.85 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −319.1 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -686.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H314, H335 |
| Precautionary statements | Precautionary statements: P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362+P364, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1-W |
| Flash point | 43 °C |
| Autoignition temperature | 240 °C |
| Lethal dose or concentration | LD50 oral rat 626 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 769 mg/kg |
| NIOSH | BX5425000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 1 ppm (5 mg/m³) |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Acetyl chloride Propionyl chloride 2-Chloropropionic acid |

