Thinking About Propiolic Acid: Science, Safety, and the Path Forward
Looking Back: How Propiolic Acid Entered Chemistry’s Toolbox
The story of propiolic acid offers more intrigue than it first lets on. Chemists in the nineteenth century noticed a sharp, almost biting scent coming from tubes where they worked with acetylene and carbon monoxide. That tang belonged to propiolic acid, an unsaturated carboxylic acid with a triple-bonded carbon at its core. It became clear quite early that this compound deserved attention, not only for its reactivity but for its position in the broader sweep of organic synthesis. The names “propiolic acid” and “propynoic acid” echo out of older German and French laboratories, reminders of a time when tracking down a new acid meant breathing its fumes and recording a physical feeling rather than just running a chromatogram.
The Basics: What Propiolic Acid Does and What It Looks Like
Checking a bottle of propiolic acid in any well-equipped lab, the clear or slightly yellow liquid inside will announce itself with a strong, sometimes choking odor. With a molecular formula of C3H2O2, this acid turns up as a colorless liquid or sometimes forms as crystals if left undisturbed below room temperature. For those who thrive in the world of chemical detail, the structure sits as HC≡C–COOH. The triple bond right next to a carboxyl group hands this molecule a striking set of properties. It brings sharp acidity, volatility, and enough reactivity to warrant careful handling. Boiling at about 80°C and melting around –23°C, the acid doesn’t stick around long in open air—it tries to escape, sometimes making itself known before anyone even uncaps the container.
How it’s Made: Crafting Propiolic Acid in Today’s Lab
Preparing propiolic acid takes more thought than just mixing a few common reagents. Most routes today start from derivatives like propargyl alcohol or by modifying molecules such as acetylenic compounds with oxidants. For instance, chemists take propargyl alcohol and push it through oxidizing agents like potassium permanganate, gradually stripping away hydrogens, and bringing the triple bond closer and closer to forming the acid. These reactions demand close control over temperature and pressure, with skilled hands ensuring the product does not decompose or react further. Making this acid on a useful scale involves patience: purification usually goes through distillation under reduced pressure, given the tendency for the liquid to vanish or react with whatever it touches if simply boiled in open air. This is real chemistry—part skill, part patience, and always a dash of respect for materials that can surprise even a seasoned chemist.
The Science of Reactivity: What Propiolic Acid Can Do
Propiolic acid holds a triple bond beside an acid group, which puts plenty of power into this compact molecule. For those in synthesis, that means opportunities. Chemists use the acid for preparing esters, amides, anhydrides, and can send the triple bond through addition reactions, tapping the carbon-carbon bond’s keen appetite for new partners. This opens up the world of click chemistry, small-molecule coupling, and preparation of building blocks for pharmaceutical or polymer work. Taking advantage of its reactivity, researchers target modified amino acids, specialty ligands, and even explore routes toward advanced electronic materials with this humble acid in the mix.
The Names and Faces of Propiolic Acid
Propiolic acid answers to a few aliases—propynoic acid, 2-propynoic acid, and, less often these days, monoynecarboxylic acid. Chemical abstracts and data sheets tend to prefer the first two, but historical literature sometimes leans on structure-based names. As with many classic organic molecules, the name itself points to the acid’s makeup: a triple bond in a three-carbon chain capped by a carboxyl group.
Safety and Working Practices: Handling a Sharp and Reactive Acid
Anyone with real lab experience understands that even small bottles of substances like propiolic acid require rigorous respect. Exposure brings immediate irritation to the nose, throat, and lungs; it easily seeps through nitrile gloves if not changed out regularly. Fume hoods aren’t optional—they’re required. Beyond basic irritation, improper exposure can produce more serious problems, so professionals train new chemists with a hands-on approach and plenty of supervision. Emergency wash stations stay close at hand. Labels on bottles must list not only standard hazard phrases but also the particular risks associated with acids containing carbon-carbon triple bonds, since they’re more aggressive than one might expect. I recall a graduate student—distracted and in a hurry—opening a vessel in open air, with the room filled moments later by a choking, skunklike odor. Experiences like that drive lessons home.
Where Propiolic Acid Gains Value: Key Application Areas
Downstream, propiolic acid rarely makes headlines, but it occupies surprising niches. The pharmaceutical sector draws on its triple bond to install acetylenic motifs, valuable in anti-infectives, antivirals, and enzyme inhibitors. The world of advanced materials finds use for its sharp reactivity too, tuning properties of polymers or acting as a building block for specialty coatings. In university labs, the acid often turns up in undergraduate exercises to demonstrate electrophilic addition, with students learning just how much energy a little triple bond can hold. Most practical applications live behind the scenes—direct use is rare, but its fingerprints show up in countless experiments and synthesis campaigns.
Technical Details and Labeling: What Experienced Chemists Look For
Lab veterans know to check a bottle for specific concentration, impurity profile, and thermal stability. Detailed labeling matters—not only due to regulations, but for tracking storage life and knowing when to order fresh material. Triple-bonded acids degrade, and even a small impurity can wreck delicate synthetic plans. Most suppliers include warnings about water sensitivity, volatility, and light sensitivity. Experience teaches that sketching proper storage practices into standard operating procedures keeps everyone safer and reduces waste.
Fieldwork and the Push for New Discoveries: R&D in Propiolic Acid
Front-line researchers often chase new uses for this acid, pushing boundaries where other feedstocks have let them down. Chemists working at the interface between organic and organometallic chemistry tinker with propiolic acid—racing to find faster coupling reactions, greener oxidation methods, or new catalysts to improve efficiency and cut toxic byproducts. As new pharmaceutical scaffolds call for acetylenic groups with ever more precise patterns, this little acid remains a popular raw material. Teams focused on sustainable chemistry ask: How can we make propiolic acid from renewable feedstocks, not just oil-derived starting points? It’s a search for pathways that cut carbon footprints, trim waste, and keep the price affordable enough for wider use. The search continues, as every new process stumbles across unpredicted challenges—the tools of modern chemistry are sharp, but the puzzles keep coming.
Toxicity Research: Understanding Risks to Solve Real Problems
Propiolic acid’s toxicity sits front and center for those in industrial hygiene and public health. The acid irritates mucous membranes at low concentrations, raising real concerns for accidental release in both laboratories and industrial settings. Regulatory oversight has pushed providers to document acute toxicity figures, but long-term effects remain less thoroughly mapped out. There’s plenty of evidence for corrosive action on skin and tissues—a direct hazard in small, open labs with less experience or fewer resources. Training and monitoring offer the only reliable barrier to careless mistakes, with research groups turning toward safer derivatives whenever applications allow.
What Lies Ahead: Propiolic Acid in Tomorrow’s Chemistry
Looking ahead, the future for propiolic acid ties into the hunger for smarter, cleaner processes. Renewable chemistry circles hover near the acid as a target for greener synthesis, with bio-based feedstocks and electrochemical methods gradually gaining traction. That drive doesn’t just spring from environmental trends: companies and researchers sense that the triple bond—a motif full of energy and potential—can feed into new drugs, smart materials, and selective catalysts promising real commercial and medical impact. Scalability, safety, and sustainability will keep setting the pace, making smarter process control essential. Propiolic acid might not be glamorous, but it serves as a perfect example of a workhorse molecule that keeps pushing science and industry forward, one careful experiment at a time.
What is Propiolic Acid used for?
Digging Into a Sharp Chemical
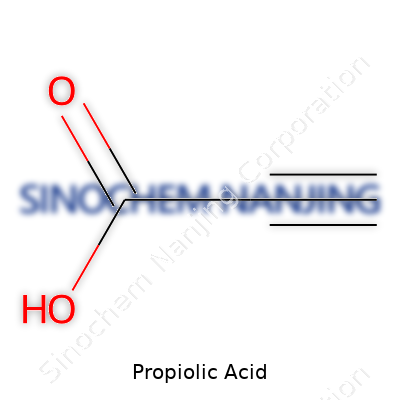
If you’ve ever stepped into a modern chemistry lab, certain peculiar smells and warning signs are impossible to ignore. Propiolic acid, with its distinct aroma and reactivity, definitely sets off those senses. It rarely pops up in everyday conversation, but in the research world, it matters quite a bit. With the formula HC≡C–COOH, it brings together both an alkyne and carboxylic acid, making it a versatile tool for chemical makers.
Why Industry and Labs Reach For It
Researchers often use propiolic acid as a starting point for more complex molecules. The triple bond of its alkyne group brings high reactivity, letting it link up with all sorts of other molecules. This property serves organic chemists who want to build structures that drugs, dyes, or advanced materials rely on.
Once, while supporting a small startup developing new anti-cancer agents, I saw propiolic acid help create experimental building blocks that could attach to proteins in controlled ways. It combined with other small molecules to craft these “handles,” which then connected with targeting agents—complex work, but with huge potential for the right medicine.
Manufacturers who need to prepare functionalized chemicals, like propargyl derivatives, often call for propiolic acid. A good example comes from the development of synthetic flavors and pharmaceuticals. This compound helps introduce “alkyne tags” for tracking cellular changes in biochemistry studies. Instead of relying only on classic markers, scientists click these special groups onto enzymes or DNA, helping reveal new details about how our bodies work.
So Why Should We Care?
People sometimes dismiss chemicals like propiolic acid as just tools for chemists in white coats. Yet the ability to build, modify, and understand molecules underpins entire industries. Medicines, agrochemicals, new energy materials—all rest on advances like those stemming from this tiny compound.
The health sector sees clear gains thanks to the molecule's flexibility. Cancer research, for example, depends a lot on discovering new ways to target cancer cells without hurting healthy ones. With the "click chemistry" facilitated by propiolic acid, scientists create pathways that home in on rogue cells. The Nobel Prize in Chemistry 2022—awarded for advances in click chemistry—reminds us how precision tools like propiolic acid propel medicine forward.
Agriculture also draws on propriolic acid’s talent for building protective chemicals to keep crops safe. And in the material sciences, folks push it to shape innovative polymers or coatings that give fabrics or plastics improved qualities, from fire resistance to biocompatibility.
Handling Hazard, Finding Solutions
Not everything about propiolic acid fits the glowing innovation script. The compound has real safety hazards: Strongly corrosive, easily absorbed through skin, and potentially toxic if mishandled. I recall colleagues once evacuating a section of the lab after a bottle fell and broke. Fast action, careful planning, and training prevented any disaster, but stories like this underscore a truth—progress and safety go hand in hand.
Solutions for responsible use have moved well beyond splashy warning labels. Gloves rated specifically for corrosives, improved classroom training, easy-access fume extraction, and smart sensors that flag vapor leaks all strengthen protection. Companies who use it at larger scales now tap into digital tracking for inventory, so danger spots can’t be overlooked.
Respect for what goes into our discoveries—both the promising and perilous sides—builds trust and keeps genuine breakthroughs moving forward. Propiolic acid, rarely in the headlines, is a quiet force underpinning progress in labs, factories, and hospitals. Recognizing its role pushes everyone to work smarter and safer, all while pursuing big ideas.
What is the chemical formula of Propiolic Acid?
Chemical Formula and Structure
Propiolic acid carries the chemical formula HC≡C-COOH. In another way, it can be written as C3H2O2. This compact formula tells a surprising story about the molecule. One hydrogen atom, two carbon atoms linked by a triple bond, a carboxylic acid group tacked on the end. Chemists sometimes call it prop-2-ynoic acid. You hear this a lot in labs that handle organic compound synthesis, especially those working with alkynes.
Physical and Chemical Properties
With its sharp, biting odor and tendency to irritate the nose and eyes, Propiolic acid announces its presence quickly. The triple bond in its structure gives the molecule an unforgiving reactivity. Spills on lab benches don’t get ignored—corrosive materials like this make short work of skin or surfaces. The molecule dissolves easily in polar solvents like water, but contact isn’t something you want to risk. Many organic acids present hazards, but the combination of acidity and an alkyne triple bond makes Propiolic acid especially attention-worthy.
Real-World Uses and Why It Matters
Propiolic acid hides behind the scenes in chemical labs and pharma research. This molecule helps researchers build more complicated materials. Chemists favor it to make compounds used in adhesives, coatings, and some advanced pharmaceuticals. Its triple bond creates a reactive starting point—the launches for further chemical transformations in the lab. If you need to add an alkyne group to a molecule, Propiolic acid shows up as the go-to raw material.
As a researcher, I have encountered Propiolic acid mostly as an intermediate for larger syntheses. Small amounts, strictly controlled—mistakes come with immediate consequences. Spills create harsh smells and the area clears quickly. The molecule’s potential lies in its willingness to connect and build, bridging to larger, more useful compounds.
Health and Safety Concerns
No story about Propiolic acid feels complete without tackling safety. Exposure puts skin, eyes, and lungs at risk. Researchers keep gloves on, goggles tight, and ventilation moving. Because it burns and corrodes on contact, labs that use this compound store it well labeled and locked away. In my time with organic synthesis, even a drop gives a harsh stinging sensation on skin—a memory not quickly forgotten. The fact that it even finds limited use, in spite of hazards, underscores its value in certain chemical processes.
Environmental Impact and Handling
Disposal raises concerns. Propiolic acid does not mix well with public water systems. Designed containers, neutralization steps, and vigilant oversight make sure it stays out of groundwater. The molecule’s triple bond, so useful in controlled conditions, could cause havoc in the wrong setting. Chemical waste handlers know this well, and respect for these risks keeps accidents at bay.
Solutions and Safer Chemistry
One path forward lies in improved engineering controls. Fume hoods and closed systems let scientists tap into the reactivity they need without facing direct exposure. Chemists also work on alternative pathways using less hazardous alkyne sources, though none match Propiolic acid’s efficiency in certain syntheses. Continued research pushes for safer chemical handling and possible replacements. Labs do not treat this compound casually—every person on site, from interns to principal investigators, knows the respect and caution required.
The Future for Propiolic Acid
Strong chemical tools like Propiolic acid build the foundation for innovation in medicine and materials. The molecule’s story reminds us that power often carries risk, and smart chemistry combines deep respect for those risks with a commitment to safety and responsibility.
Is Propiolic Acid hazardous or toxic?
Propiolic Acid: More Than a Chemical Name
Every day, lab workers and chemical handlers come across a long list of substances. Propiolic acid, a compound with the formula C3H2O2, turns up now and then, mostly in research circles or specialty synthesis. It’s a colorless liquid with a sharp, vinegar-like smell, the kind that makes you want to quicken your pace past an open bottle. This stuff is not lying on a grocery shelf. It finds a place in some specific reactions, sometimes used to make other chemicals. So, I’ve learned that most folks walking near it are people trained for the weight of responsibility.
Hazardous Nature: What Experience Shows
Propiolic acid’s hazards show up quickly. Small-scale chemists know it can irritate the skin and eyes right away, even if you try to work clean. Breathing it in could give you a burning throat or a wheeze that sticks around at least until you get fresh air. Animals exposed to very concentrated vapors got symptoms in some old research papers I read while prepping for a safety seminar. There’s little mystery in the fact that chemical burns can happen if you spill it on yourself.
Looking through the documentation—SDS sheets, CDC notes—it’s clear that this acid is more than a mild nuisance. It can cause corrosive damage on contact with tissue. Ingesting a swallow would cause serious internal damage fast. From my years pulling gloves on and cleaning up, I learned to treat strong acids with respect, even if the name sounds soft or unfamiliar.
Toxicity and Chronic Risk
Beyond the obvious immediate hazards, the question lingers: Is propiolic acid outright toxic in the long run? The big regulators—OSHA, NIOSH, the EU’s chemical safety authorities—don’t have a mountain of public studies on this particular molecule. What does exist tells a plain story. No acceptable exposure limit has been carved out because most people will rarely meet this chemical in any real quantity. Those who do work with it, though, face risk of chemical-induced asthma, chronic respiratory irritation, and scarring if they let a little recklessness creep into the work.
The close chemical relatives of propiolic acid, like acetylenic acids, bring their own toxicity along, too. Prolonged exposure or careless mixing with other reactive chemicals can lead to hazardous byproducts—and I’ve heard more than one story about near-accidents sparked by ignoring stale safety data.
Practical Safety: Real Protections in the Workplace
Old-fashioned experience wins out here. Chemical goggles, gloves with reliable resistance, long sleeves, and—most importantly—good ventilation help cut down risk. I’ve worked in a couple of bench labs where getting lax with hood ventilation once meant headaches and eye burn for days. I learned to double-check labels and keep a neutralizing agent nearby, so if something splashed, I could contain it fast.
Training matters most. Nobody remembers pages of warnings; they remember the time their mentor sank a hand into a cleanup bucket and came out cursing. The buddy system, regular review of spill protocols, and keeping the emergency shower within reach all build up to a culture where people use these chemicals wisely. Waste handling, too—small drums marked for acid only, vented properly, kept folks out of trouble during waste pickups.
Room for Improvement: Solutions That Stick
For chemicals like propiolic acid, clear labeling and easy-to-find hazard symbols give a leg up. Electronic safety monitoring—such as sensors for fumes—has become cheaper and faster to set up. Mandatory safety training refreshers every year help keep everyone sharp. Sharing real incident reports in meetings brings the point home far better than a dozen rules on the wall. Take the time to know the chemicals on your bench, listen to those who’ve handled a spill or a bad incident, and make these stories part of the training for everyone who steps into the lab. The trouble comes from surprise—first-timers and old hands alike benefit from real stories and practical guidance.
How should Propiolic Acid be stored?
Looking Beyond the Label
Propiolic acid might not be a household name, but it holds a spot in quite a few chemical stockrooms, mostly for research or manufacturing. If you’ve seen it up close, you know the stuff gives off a sharp, choking odor that makes your eyes water. No one wants that leaking into a workspace. I once saw a cracked cap let those vapors drift across an entire bench and start a domino effect of headaches and chaos—no wonder safety folks practically sprint to double-check acid bottles.
Why Storage Choices Matter
The chemical formula tells part of the story—propiolic acid brings together a carboxylic acid group and a triple bond. That means it doesn’t just smell fierce, it reacts with bases, metals, and sometimes even moisture from the air. Ignoring that spells trouble. Acids like this one can chew through flimsy plastics, corrode soft metals, or eat away at wood shelving when spills happen.
Regulatory bodies like OSHA and the NFPA spell out their warnings: Propiolic acid rates high in health hazard, brings flammability risks, and can damage eyes and skin in a flash. Just last year, a tech in a teaching lab ignored proper protocols, left a bottle on an open shelf, and discovered it had cracked from light exposure—turns out, the vapor started eroding nearby aluminum foils and gaskets. Lucky no one got hurt that day.
Facts From Experience
Direct sunlight doesn’t just discolor glass bottles, it speeds up decomposition of light-sensitive compounds inside, and that can build up pressure. I’ve learned the hard way—no clear glass on open wire racks near a sunny window. Shatterproof amber containers and opaque storage cabinets add a line of defense.
Room temperature can swing wildly in poorly ventilated storerooms, especially in older buildings where HVAC barely keeps up in summer. Temperature controls matter. I always put propiolic acid in a chemical fridge set below 15°C, not the kitchen fridge. Food contamination horror stories travel fast in research circles, and acids like this don’t belong within reach of sandwich lunches or milk cartons.
Real-World Solutions
Not every lab budget stretches far, but cutting corners with hazardous acids can turn penny-pinching into disaster. I recommend keeping small quantities on hand, only ordering what gets used in a month or two. In my experience, double containment works: place the original sealed bottle in a plastic secondary tray, which catches drips or breaks. Acid cabinets lined with plastic or corrosion-resistant metal give another layer of protection. Label everything clearly, using both chemical names and hazard codes, because a rushed junior chemist grabbing “the bottle with the blue cap” can bring confusion or worse.
Spill kits with acid neutralizers need to stay within arm’s reach, not buried behind stacks of printouts and solvent cans. Proper personal protective gear—face shield, gloves, splash apron—comes out every time, no exceptions. It takes only seconds for liquid acid to scorch skin or ruin favorite jeans.
Ventilation gets ignored too often. Fume hoods don’t just suck away funny smells, they pull harmful vapors out of general breathing air. At a local university, one fume hood malfunctioned, and after two hours even bystanders outside the lab started coughing—maintenance checks for airflow and duct integrity keep bad surprises at bay.
Building a Safer Workplace
Respecting chemicals like propiolic acid boils down to making storage habits a matter of pride, not paperwork. Training new folks hands-on, not just through slideshows, builds confidence. Taking shortcuts grows risk, not just for the individual, but everyone nearby. Learning from small mistakes, and sharing stories about them, keeps everyone sharper.
Can Propiolic Acid be used in pharmaceutical synthesis?
The Appeal and Challenges of Propiolic Acid
Propiolic acid, with its triple-bonded backbone, draws attention from chemists looking to spark new reactions in drug chemistry. The appeal comes from its simplicity and reactivity. As a molecule, it sits at a crossroad: basic in structure, but rich in potential. Many drug developers look for starting materials that can do more with less, and propiolic acid almost teases with its mix of ease and possibility.
Chemists have known it for decades. Its triple-bonded carbon makes reactions go places that saturated acids can’t reach. Whether adding to aromatic rings, creating new carbon-carbon bonds, or yielding functional groups, this acid offers unique shortcuts in complex syntheses. One breakdown product, propargyl alcohol, turns up often in painkillers and cholesterol-lowering drugs, showing how precursors like propiolic acid support certain big-name therapies.
Safety, Handling, and Real-World Lab Experience
Anyone who’s ever held a flask of propiolic acid knows its reputation. It’s sharp, volatile, and leaves a sting in the air. Skin contact or accidental inhalation means trouble—a fact no lab should ignore. Handling this chemical means gloves, goggles, and respect. In many university and pharmaceutical research labs, senior staff limit its use to fume hoods or even gloveboxes. I recall nervous first-year graduate students eyeing the bottle warily before any procedure. This tells us: Propiolic acid always demands attention to safety.
What Makes It Useful in Drug Synthesis?
The versatility shines in reactions like “click chemistry” and alkynylations. These broad synthetic tools build the scaffold of modern medicines, from antiviral drugs to new cancer agents. Alkynes, the class of molecules where propiolic acid belongs, offer easy routes to fine-tuning molecular shape and polarity—properties central to how a drug moves in the body. During a sabbatical at a drug company, I watched chemists use propiolic acid to stitch together drug fragments that wouldn’t connect using classic routes. This open-minded approach matters most in discovery.
Regulatory and Environmental Considerations
No one can avoid the regulatory hoops in pharmaceuticals. Propiolic acid, thanks to its volatility and health warnings, brings stricter storage rules and more paperwork. Regulators want proof that chemists can keep it contained, monitor exposure, and dispose of leftovers safely. Environmental teams pay attention to waste and release—its triple bond isn’t friendly to waterways or soil after use. Safe processes demand reliable ventilation and solvent management plans. Those extra steps slow routine projects, driving some companies to seek safer alternatives.
Potential Solutions and Industry Outlook
Many labs now look for ways around the hazards by seeking milder surrogates or sealed reaction systems that reduce risk. Flow chemistry setups, where proportions run small and reactions go on behind glass or steel, make a real difference. I watched a team in Boston scale reactions with only a few milliliters in play, keeping waste and risk much lower than a full beaker approach. Transparent reporting, purpose-designed training, and routine audits—these common-sense steps protect workers while letting innovation continue.
As drug discovery evolves, chemists will keep asking whether the ends justify the means. Propiolic acid offers unique shortcuts and creative solutions, but always as part of a broader balance between innovation, safety, and environmental responsibility. Its future use will depend on continued vigilance and a willingness to rethink how we build the molecules that matter for health.
| Names | |
| Preferred IUPAC name | prop-2-ynoic acid |
| Other names |
Propynoic acid Prop-2-ynoic acid 2-Propynoic acid Ethynylformic acid |
| Pronunciation | /proʊˌpaɪˈɑːlɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 471-25-0 |
| Beilstein Reference | Beilstein Reference: **1208416** |
| ChEBI | CHEBI:50199 |
| ChEMBL | CHEMBL11604 |
| ChemSpider | 5329 |
| DrugBank | DB04248 |
| ECHA InfoCard | 100.003.875 |
| EC Number | 204-107-1 |
| Gmelin Reference | 833 |
| KEGG | C02228 |
| MeSH | D011367 |
| PubChem CID | 7902 |
| RTECS number | UK4375000 |
| UNII | 2E42ONI5Y8 |
| UN number | UN2929 |
| Properties | |
| Chemical formula | C3H2O2 |
| Molar mass | 84.04 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | pungent |
| Density | 1.046 g/mL at 25 °C (lit.) |
| Solubility in water | soluble |
| log P | 0.09 |
| Vapor pressure | 0.32 mmHg (at 25 °C) |
| Acidity (pKa) | 1.90 |
| Basicity (pKb) | pKb = 24.23 |
| Magnetic susceptibility (χ) | -27.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.401 |
| Viscosity | 1.552 mPa·s (25 °C) |
| Dipole moment | 1.63 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 215.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –73.63 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -492 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS05 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331, H314, H373 |
| Precautionary statements | P210, P261, P280, P304+P340, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Flash point | 109 °F (43 °C) – closed cup |
| Autoignition temperature | 445 °C |
| Explosive limits | 2.4–12% (in air) |
| Lethal dose or concentration | LD₅₀ (oral, rat): 92 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 80 mg/kg |
| NIOSH | UC9100000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | REL: 3 mg/m³ |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Acetylenediol Acetylenedicarboxylic acid Propanoic acid |

