Opinion: Potassium Silver Cyanide — The Substance That Shaped a Century of Surface Science
History Etched in Silver
Potassium silver cyanide tells a story that stretches back to the heyday of the industrial revolution. Chasing new finishes, industries banked on this salt for silver electroplating, craving both reliable conductivity and a polished look. Its breakthrough can be traced to the mid-nineteenth century, when inventors like John Wright pushed the science of gilding and plating past simple ornamentation and into the world of telegraph wires and tableware. The compound played a quiet but transformative role, not on stage but in the workrooms of manufacturing cities. Understanding its history sheds light on how industrial chemistry walks hand in hand with culture—silver-plated forks and cutlery became accessible, often changing how everyday people experienced the rituals of dining or technological progress.
What This Salt Brings to the Table
In a clear, white crystalline form, potassium silver cyanide reveals its purpose: a stable, highly soluble compound ready for precise processes. Unlike pure silver, it packs a chemical punch, offering a controlled route for depositing silver onto all sorts of metals. Its water solubility means baths mix easily, and the cyanide group unlocks the power to dissolve silver oxide, preparing it for an even plating layer. At room temperature, the salt won’t give off much of a smell, and the prismatic crystals powder when crushed, hinting at the ease with which it enters electroplating solutions. Anyone who’s handled it knows to respect its potency—one accidental gust, and there’s trouble.
Dealing with Details: Specs and Labels
Every vessel carrying potassium silver cyanide wears a clear, blunt label—a warning as much as an identifier. Purity levels loom large in technical sheets, often reaching well over 98 percent silver content to meet plating demands. Tiny shifts in impurity levels can ripple out to massive quality issues during manufacturing. Real-world commercial users trust their supplier to get these numbers right because anything less clogs up the process or contaminates entire baths, risking hardware and reputation.
From Lab Bench to Factory Floor
Making potassium silver cyanide isn’t a project for the casual hobbyist. The method pulls in two core ingredients: silver nitrate and potassium cyanide. In practice, a silver nitrate solution meets potassium cyanide, leading to the precipitation of potassium silver cyanide crystals. Chemistry drives this reaction hard in both industry and laboratory, and it’s fair to say that most who’ve prepared this substance keep windows open and protective equipment on at all times—cyanide risk makes mistakes unforgiving. Over the decades, procedural tweaks have improved yields and cut down on waste, but the fundamentals haven’t changed much; handling always demands full attention and a level of experience earned through long exposure, not quick study.
Chain Reactions: Chemistry in Motion
Potassium silver cyanide doesn’t just sit idly on the shelf. Plunged into a bath and connected to a current, the silver ions leap onto cathode surfaces in the process that built everything from jewelry to electronic connectors. The underlying reactions—decomposition under voltages, exchange reactions with acids, breakdown by bright sunlight—fascinate from both practical and theoretical standpoints. Mix it with strong acids, and deadly hydrogen cyanide gas escapes; introduce light, and the crystals slowly darken, a demonstration of chemistry’s persistence to shift and adapt. Over the years, researchers have prodded these reactions, looking for ways to boost efficiency, reduce waste, and redesign the process to sidestep environmental fallout.
Names Through the Ages
Whether it’s called argentic potassium cyanide, silver potassium dicyanide, or simply KSAg(CN)2 in shorthand, this compound collects synonyms as chemistry uncovers new quirks. The best-known title signals the silver at its core, but trade literature and older texts often use a jumble of names, a reminder of the long lineage and diverse users. Language keeps evolving, but across geographies and translations, most working chemists recognize the story behind the formula.
Rules for Safety and Working Standards
Anyone with years in laboratory work or a factory setting will tell you: potassium silver cyanide doesn’t tolerate shortcuts. Strong, consistent ventilation, sealed containers, and regular safety drills come with the territory. Cyanide poisoning can shut a site down, so teams set up spill kits and monitor the air for any signs of toxic gas. Gloves and goggles aren’t just recommendations—they’re basic tools, as essential as the chemicals themselves. The shift toward automation in plating lines and strict safety audits has cut down on accidents, but vigilance remains. In my experience, the teams who keep the best records and training logs avoid drama; those who cut corners pay the price in both human and financial terms.
Applications and Where It Leads
The real-world uses of this chemical haven’t faded, even as alternative technologies push forward. Telecommunications and electronics still rely on silver’s unmatched conductivity and resistance to corrosion. In jewelry and art, the materials haven’t changed: customers crave shine that lasts, and only silver plating—or, less often, gold—delivers on both price and performance. Microfabrication, once a far-off goal, now turns to potassium silver cyanide in processes for touch screens and connectors. A background in materials science shows how the compound became a quiet backbone—most folks never see it, but nearly everyone benefits from the devices and tools it creates.
R&D: Progress and Obstacles
Research teams keep hunting for greener, safer ways to manage cyanide. Decades of studies target process optimization, waste minimization, and recovery of spent baths through electrochemical treatment. Breathing room for innovation comes from collaboration; universities partner with businesses to redesign plating cells, monitor emissions more tightly, and build closed-loop systems that slash both cost and hazard. Some lines of inquiry focus on non-cyanide routes, but matching the old salt’s efficiency and quality hasn’t happened yet. In the end, the work requires a blend of academic theory and daily practice—lab breakthroughs only matter once they stand up to industrial demands.
What the Research Says About Toxicity
Cyanide remains one of the better-studied occupational hazards in chemistry. Chronic or acute exposure brings on headaches, muscle spasms, and at higher levels, life-threatening shutdowns of heart and lung function. Silver itself rarely reaches toxic levels, but in the presence of cyanide, all bets are off. Long-term monitoring of workers exposed to low levels keeps revealing subtle effects; careful epidemiological research and animal studies guide regulatory limits. Decent workplace routines—prompt cleaning, strict personal protective equipment rules, air monitoring systems—mark the line between a career and a crisis. Here, experience counts most: learning from incidents, sharing results, tweaking procedures, all keep future harm at bay.
The Road Ahead
There’s no escaping the tension between the usefulness of potassium silver cyanide and its risks. Green chemistry researchers press on, probing new complexes and plating processes that might strike a balance between performance and safety. Industry demand hasn’t slowed—connectors and high-end finishes still call for the same silvery layer. As technology advances, pressures for both environmental responsibility and production efficiency mount. Lessons stack up year after year, pushing incremental gains in handling, reclamation, and substitution. Looking back at the role this compound played in industry, one thing stands out: progress comes from those willing to acknowledge both the power and peril in chemistry, always striving for a safer, smarter tomorrow.
What is Potassium Silver Cyanide used for?
The Real Workhorse Behind Silver Plating
Potassium silver cyanide stays mostly out of the headlines, but in metal finishing shops, it’s a familiar chemical with a specific job. People in plating industries use it for one main purpose: coating other metals with a thin, even layer of silver. This makes forks shine, jewelry durable, circuit boards functional, and even components in medical devices reliable. In these trades, the go-to way of “silvering” an object means reaching for this chemical compound.
Most of us carry something silver-plated without realizing what makes it so bright and tough. The plating bath relies on potassium silver cyanide to do its job. The process uses electricity to move silver ions from the compound onto whatever needs that finish, whether it’s fancy cutlery, costume brooches, or high-tech connectors. You end up with the surface benefits of silver — corrosion resistance, conductivity, polish — while using only a fraction of the pure metal.
Why Factories Stick With It
This compound gets picked not just for its silver content but for how it behaves. It dissolves well, gives steady results, and keeps the metal finish consistent time after time. Over the years, shops have learned that switching from potassium silver cyanide to another method can cut corners. That risk isn’t welcome, especially in industries like electronics, where a tiny breakdown could affect a whole device.
If you ever visited a facility where metal finishing happens, you would notice how seriously the staff take safety. Potassium silver cyanide, like other cyanide compounds, is highly toxic. A mask, gloves, and tight procedures aren’t just “rules”—they’re lifelines. Years ago, I watched a supervisor double check everything herself during a training exercise. One careless step could cost someone their health. So every person working with this stuff understands what’s at stake.
The Double-Edged Sword of Progress
We often talk about progress as if it means moving forward, but for people working in plating, progress also means safety. Regulators keep an eye on potassium silver cyanide, and for good reason. Accidental releases can poison water, hurt wildlife, and make neighborhoods unsafe. Clean-up projects from past decades speak for themselves. Yet, pulling this chemical out of the plating world isn’t easy. From what I’ve seen, the main challenge comes from balancing business priorities with the health of people and ecosystems.
Are Safer Alternatives in Sight?
Researchers and environmentalists push for alternatives that can do the same job with less danger. Silver thiosulfate baths, proprietary non-cyanide mixes, or mechanical techniques all get attention. The problem comes down to performance. Many alternative baths struggle to match the results or reliability of potassium silver cyanide under everyday factory conditions. Some methods leave deposits too soft or patchy for sensitive electronics or heavy use.
Factories can improve beyond just swapping chemicals. Systems that reuse bath chemicals, monitor air and water, and lock up residues help limit risk. It takes investment and training to stay ahead of accidents. People who know the inside of a plating shop understand this isn’t a “set it and forget it” task. With more oversight and smarter technology, it’s possible to keep metal finishing strong while lowering hazards from chemicals like potassium silver cyanide.
Is Potassium Silver Cyanide hazardous to health?
Learning from Experience in the Metal Finishing Industry
Potassium silver cyanide often comes up in the world of electroplating and jewelry making. I’ve worked in workshops where this chemical was handled daily, sometimes by people who never gave its dangers much thought. Watching new workers pour a little powder without gloves or a mask made me realize how easily people ignore the risks until something goes wrong.
Understanding Why It’s a Real Hazard, Not Just a Technical Term
This chemical isn’t just another ingredient. Potassium silver cyanide contains cyanide ions, the same dangerous part found in many infamous poisons. When this compound gets absorbed through the skin or, worse, inhaled or ingested, it interrupts the body’s ability to use oxygen. Syptoms can show up fast: headache, dizziness, shortness of breath, and worse—loss of consciousness. These aren't just rare scenarios or warnings found in manuals. Hospitals have actually treated workers poisoned by accidentally inhaling dust or spilling a solution onto their hands.
Talking Safety Isn’t Enough—It’s About Habits
Reading chemical labels doesn’t protect anyone if habits don’t match the hazard. In workplaces I’ve seen, those who felt rushed or didn’t have the right training skipped basic steps like wearing gloves or making sure the room had good airflow. Respirators and sturdy gloves might sound like overkill to someone who’s handled chemicals for years, but accidents don’t give warnings. Even small spills and a little bit of powder in the air can cause real damage.
Veteran Advice: Physical and Mental Health Stakes
After a coworker suffered burns and lung irritation from a spill, I understood how far-reaching these accidents can be. It goes beyond poison—potassium silver cyanide can cause serious burns on skin and eyes. Exposure isn’t always an event with alarm bells; sometimes symptoms creep up and long-term health gets chipped away quietly. Chronic exposure links to nerve damage and persistent breathing problems. The mental toll of a poisoning scare changes how people view their jobs forever.
What Actually Works to Prevent Poisoning
I’ve seen improvements where supervisors pushed for better safety routines every day—not just during regulatory checks. Clear training and constant reminders kept safety steps fresh in our minds. Good ventilation made a difference. Dedicated storage bins, double-checked before closing up for the day, helped contain accidents. Making gloves, goggles, and proper masks as common as aprons or boots raised the chances people would use them.
Community and Policy Support
Protecting health isn’t just about better gear or quick warnings. Standards from groups like OSHA set limits on workplace exposure, but enforcement depends on regular inspections and speaking out when rules slip. Companies investing in safer alternatives, better tools for containment, and regular medical check-ups actually cut down on health problems over the years. Being open about near-misses and incidents means the message stays strong: handling potassium silver cyanide always carries risks, so respect for the rules has to be real and ongoing.
How should Potassium Silver Cyanide be stored?
Dealing with Potassium Silver Cyanide Is Not for the Inattentive
Potassium silver cyanide has a reputation, and not for making things simple. Anyone who's ever worked with it in a plating shop will admit that there’s no room for improvisation. The stuff earns respect fast: a single mistake can cause a mess that endangers health and, in tight operations, livelihoods. My first months on the floor, the supervisor hammered home the importance of not trusting memory or hope — only good habits and strict protocols stand between you and a very expensive, very dangerous mistake.
Why Respect the Danger?
Potassium silver cyanide contains cyanide ions—infamous for their lethality. A single whiff of cyanide gas can land you in the hospital. Silver is less of a problem by itself, but cyanide takes things up ten notches. Storing the compound asks for more than just keeping it on a high shelf. Moisture leads to hydrolysis, which can release hydrogen cyanide. Carelessness literally poisons the air.
Worldwide, deaths and poisonings linked to improper cyanide storage pop up every year, even in seasoned industries. Factory floors and jewelry workshops depend on keeping workspaces safe so the job doesn't become a gamble.
Keep Things Simple, Not Slack
Rule one from my old supervisor: never store potassium silver cyanide unless you have a sealed, airtight container. Forget jars, Tupperware, or anything less than a proper chemical storage container. Not every plastic works; many degrade or become brittle with time. Only containers rated for highly toxic and corrosive substances should ever be used.
Most newer storage containers feature locking mechanisms and gaskets. These simple tools keep humidity out and make sure a cap doesn't loosen. Every label must remain legible, with clear hazard symbols and emergency instructions. Sloppy handwriting gets called out right away—it's not about neatness, but about clear, fast action if someone gets exposed.
Keep Out Light, Heat, and Curiosity
The storage room needs to stay cool and dry. Direct sunlight or heat sources don’t belong here. Temperature swings only increase the risk of container rupture or vapor buildup. Workers who handle the chemical must know not to move it between rooms willy-nilly. Limit access to those who have the right training and understand both what’s at stake and what can go wrong.
I remember once finding a new worker poking around near the cyanide locker. Training happened the next day—not to punish, but to explain exactly why some materials can turn a small mistake into a disaster.
Keep Emergency Gear Close
Local regulations require spill kits, ammonia inhalants, and well-stocked first-aid supplies next to any cyanide stockpile. A locked storage cabinet gets checked every shift, along with ventilation and exhaust systems. Fire extinguishers stay close, since a fire can force the release of deadly gases. Ventilation should be mechanical, with airflow directed outside, never recirculating within the work area.
Teams always run regular drills on what to do if a spill or leak happens. This isn't paranoia—it's muscle memory. Nobody argues when someone insists on double-checking locks or temperature gauges.
Continual Training: Non-Negotiable
Every year, updated training on hazardous chemical storage keeps everyone sharp. I’ve never met anyone who regretted investing in safeguards or ongoing education. Local safety authorities update rules often enough that it pays to revisit safety data sheets before opening old stock or reordering supplies.
Potassium silver cyanide asks for diligence, not shortcuts. I’ve seen firsthand the difference between guesswork and following good advice—the first leads to disaster, the second to safe, focused work where everyone clocks out healthy at the end of the day.
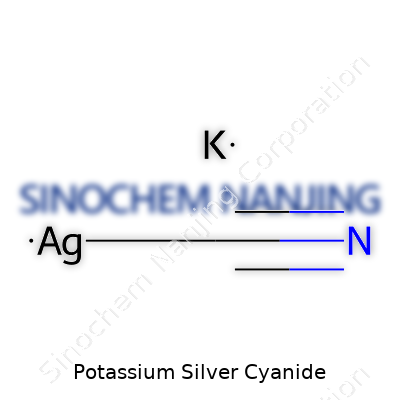
What is the chemical formula of Potassium Silver Cyanide?
Getting to the Formula
Potassium silver cyanide carries the chemical formula KAg(CN)2. You get this compound by mixing silver nitrate with potassium cyanide, ending up with a salt that finds a place among metal finishers and jewelers pretty fast. The reaction winds up as:
AgNO3 + 2KCN → KAg(CN)2 + KNO3
It’s strange how the things we see in everyday life—coated jewelry, reflective surfaces—trace back to a small, neat chemical equation. This compound isn’t some textbook oddity. Potassium silver cyanide shapes the looks of the watches, rings, and forks most of us have touched.
Why It Matters in Electroplating
Factories count on KAg(CN)2 for silver plating. In practice, this means drying racks crowded with items dipped in a bath, hooked up to electricity. Silver ions leave the solution, plate the workpiece, and create that familiar bright, durable finish. This matters because products last longer, corrode less, and look better—a triple win. For many jewelry makers, KAg(CN)2 beats other silver sources for its reliable, smooth deposits.
According to the Occupational Safety and Health Administration (OSHA), silver plating using this compound remains a big part of manufacturing’s backbone, supplying electronic and decorative industries worldwide. You don’t have to dig deep to see the economic impact. The plating sector supports thousands of jobs, directly and indirectly.
Safety Concerns Need Real Solutions
Chemistry class always warned about cyanides. Potassium silver cyanide doesn’t break out of that mold. It contains both silver and cyanide ions—both bring risks. Cyanide is notorious for its ability to disrupt the body’s use of oxygen, and anyone handling plating baths faces danger from splashes or fumes. I remember touring a metal finishing facility as a student. The chemical “smell” lingered on my clothes all day, and the workers never let their guard down. Safety came before anything else.
Poor handling caused newsworthy hospitalizations in the ‘80s in the US. Later, the Environmental Protection Agency (EPA) placed strict rules on disposal and air quality inside plating shops. Now, wastewater treatment, air capture systems, and regular safety training are non-negotiable.
Room for Safer Alternatives
More factories started using less-toxic silver complexes or techniques like physical vapor deposition. These lack the environmental punch of cyanide-based recipes but sometimes make trade-offs with cost or finish quality. The silver industry gradually introduces these alternatives, but old habits hold on tight in small shops because they know and trust the classic cyanide approach.
The way forward looks clear enough to me: tighter controls, real investments in safety, and practical support for small businesses ready to experiment with green chemistry. Nobody wants workers in harm’s way, just to get a shiny spoon on the store shelf.
The Bottom Line
Remembering the formula KAg(CN)2 means more than passing a chemistry quiz. It means recognizing a mix of science, industry, safety, and history behind your favorite silver piece. Pushing for better protective gear, cleaner water, and alternative processes keeps both workers and customers in good hands—without leaving tradition entirely behind.
How do you safely dispose of Potassium Silver Cyanide?
Potassium Silver Cyanide: More Than Just a Lab Chemical
Most folks never come across potassium silver cyanide unless they’re in the metal plating world, a chemistry lab, or taking on some specialized work. This stuff often turns up in silver electroplating, a process used to coat jewelry, electronics, and even medical equipment. The truth: it’s extremely toxic. Cyanide can kill in small amounts, and silver compounds bring their own risks. I remember the handful of times I handled cyanide compounds in college. The sense of caution never left my mind.
At its core, this chemical holds a triple threat—cyanide, potassium, and silver. Cyanide blocks the body’s cells from using oxygen. That means anyone exposed without protection faces trouble, starting with headaches and dizziness, moving quickly into life-threatening territory. On top of that, improper disposal doesn’t just endanger people; it poisons wildlife and groundwater.
Disposal in the Real World: No Room for DIY or Shortcuts
Think about flushing potassium silver cyanide down a drain, burying it in the backyard, or covering it up and hoping nobody finds it. Any of these risky moves could put pets, kids, neighbors, and the wider environment in danger. The law is strict for a reason. Improper disposal counts as a crime in a lot of regions, and environmental agencies track violators closely.
When I worked at a small plating shop, all our cyanide wastes went straight to specialized containers that locked tight. No open bottles, no improvisation. I watched my boss load those drums into a locked cage for the monthly pickup by a certified hazardous waste contractor. Folks who cut corners almost always wind up regretting it—often after some mishap or health scare.
Dealing with the Waste Properly: Steps and Precautions
You need solid training to handle this chemical safely. Before considering disposal, experts neutralize cyanide by converting it into a less harmful substance. The standard process uses oxidizing agents; sodium hypochlorite (household bleach) or hydrogen peroxide are common choices, but they require exact dosages, close monitoring, and strong ventilation. This converts deadly cyanide ions to nontoxic cyanate. Skip a step or miscalculate, and the risk returns fast. After neutralization, the waste still holds silver, so a facility recovers precious metals before sending the final waste stream to treatment.
No one should attempt these steps at home. Handling potassium silver cyanide without protective gear and training invites disaster. Labs and plating companies turn to licensed hazardous waste handlers who know how to test, treat, and transport chemicals like this. Most cities and states keep lists of authorized contractors. Call around—don’t let old supplies gather dust under a sink or in a garage.
What’s at Stake: Accountability and the Bigger Picture
Poisoning water, harming soil, or risking a child’s safety never justifies the cost or effort saved by ignoring proper disposal. The EPA reports thousands of chemical spills and contamination cases every year, often traced back to improper handling of substances like potassium silver cyanide. Cleanup can take decades and cost millions, hitting taxpayers and communities.
For anyone storing even a small batch, local health departments and environmental regulators give advice or require reporting. If you’re unsure, ask for help. Safe disposal really means protecting families, neighborhoods, and even future generations. I’ve seen close calls—trust me, nobody ever wishes they’d risked it. Stay safe, and choose the responsible path.
| Names | |
| Preferred IUPAC name | potassium dicyanoargentate(I) |
| Other names |
Argentous cyanide Silver potassium cyanide |
| Pronunciation | /pəˈtæsiəm ˈsɪlvər saɪˈænaɪd/ |
| Identifiers | |
| CAS Number | 506-61-6 |
| Beilstein Reference | 3924627 |
| ChEBI | CHEBI:86459 |
| ChEMBL | CHEMBL1201807 |
| ChemSpider | 79419 |
| DrugBank | DB14671 |
| ECHA InfoCard | ECHA InfoCard: 100.013.791 |
| EC Number | 208-734-8 |
| Gmelin Reference | 26244 |
| KEGG | C18768 |
| MeSH | D011104 |
| PubChem CID | 24658 |
| RTECS number | SZ7200000 |
| UNII | F52G7EENS7 |
| UN number | UN1684 |
| Properties | |
| Chemical formula | KAg(CN)2 |
| Molar mass | 165.1 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.556 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.5 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 11.71 |
| Magnetic susceptibility (χ) | +45.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.870 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 171.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -255.5 kJ/mol |
| Pharmacology | |
| ATC code | V03AB32 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled or in contact with skin; releases hydrogen cyanide gas upon contact with acids; very dangerous to aquatic life |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P260, P262, P264, P270, P273, P280, P302+P352, P304+P340, P305+P351+P338, P310, P320, P330, P361, P363, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-0-☠ |
| Lethal dose or concentration | LD50 oral rat 36 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 36 mg/kg |
| NIOSH | SN4550000 |
| PEL (Permissible) | PEL: 0.01 mg/m3 |
| REL (Recommended) | ALPHA |
| IDLH (Immediate danger) | **25 mg/m3** |
| Related compounds | |
| Related compounds |
Calcium cyanide Sodium cyanide Potassium cyanide Sodium argentocyanide |

