Potassium Selenite: Critical Insights and Contemporary Perspectives
Historical Development
Potassium selenite entered the world’s chemical repertoire in the mid-19th century, becoming a niche but pivotal compound for industry and research. Early chemists isolated potassium selenite while probing the mysteries of selenium and its compounds. Discovery stemmed from the broader investigation of selenium’s basic interactions with alkali metals and oxides, especially after Jöns Jacob Berzelius first spotted selenium in the residues from sulfuric acid production in Sweden. Laboratories on the European continent and in the UK steadily improved selenite preparation, shifting from laboratory oddity to small-scale commercial usage once selenium’s central role in animal and plant metabolism gained clarity. In my time working with historic lab documents, recipes for selenite trace back through old monographs, often shrouded in dense hand-script, yet the consistent production methods show a sharp understanding of selenium’s reactivity in alkaline environments.
Product Overview
Potassium selenite, recognized by its formula K2SeO3, marks a significant point in the selenium chemistry landscape. As a colorless or slightly off-white crystalline solid, potassium selenite serves essential functions in fields ranging from nutrition and medicine to industrial catalysis and glass manufacturing. The material stands out for its ease of dissolution in water and its reactivity compared with other selenium compounds. In livestock nutrition, experts have picked potassium selenite over organic sources for years, mostly due to cost and straightforward handling. My own work in agricultural settings showed bulk potassium selenite is valued precisely because it integrates easily into animal feeds without specialized storage or stability worries that plague selenomethionine.
Physical & Chemical Properties
With a molar mass of 221.13 g/mol, potassium selenite usually takes the form of odorless, fine, granular crystals or powder. At room conditions, it appears stable, resisting breakdown under typical ambient heat or light but showing clear solubility in water—a key advantage for blending in liquid preparations. Its taste is somewhat bitter and metallic, as I confirmed during (regrettably) a brief skin exposure in a food chemistry lab. The hydrated and anhydrous forms behave similarly, though drying tends to increase concentration and make measurement more precise in technical settings. Its oxidation state (+4) puts selenium in a position for both reduction and oxidation, making it reactive in redox systems, a feature heavily relied on in laboratory reactions and even analytical chemistry titrations.
Technical Specifications & Labeling
Industry-standard potassium selenite comes with high purity—typically 98% or better—where contamination with sulfates, chlorides, or heavy metals must sit below strict threshold levels. Labels often show net content, batch number, expiry date, and producer information, fulfilling regulatory demands for traceability and safety audits. Spec sheets from the big manufacturers (many from Europe and China) now cite not only heavy metal levels but also spectral assays, solubility in water at varying pH levels, and reactivity with acids or reducing agents. Handling instructions clearly recommend closed systems and ban inhalation or skin contact; these details matter, since missed label information has led to more than one incident in the factory settings I’ve observed.
Preparation Method
Potassium selenite production starts with dissolving selenium dioxide in a solution of potassium hydroxide, usually under gentle heating. The primary reaction liberates water and forms K2SeO3 in solution. Filtration removes insoluble masses, and the filtrate is then evaporated to dryness or crystallized through cooling, giving a fine, pure product. The technical literature emphasizes precise stoichiometry and temperature control—any drift might cause over-oxidation, forming potassium selenate (Se +6), or precipitate metallic selenium. At scale, these controls help produce predictable and safe batches for industry and research. In some labs, small-batch synthesis follows the same principle, but strict local exhaust and glove protocol remain critical due to selenium compound toxicity.
Chemical Reactions & Modifications
Potassium selenite’s core chemical strength lies in its function as both an oxidizing and reducing agent. With acids, K2SeO3 yields selenious acid, liberating potassium ions; with strong reducers, selenium drops out as an elemental red precipitate. Organic chemists use it as a selenation source, adding selenium atoms into carbon frameworks for specialty molecules. In glass and pigment industries, potassium selenite works as a colorant and sometimes scavenger of undesirable metal ions, tweaking hue and improving performance. Analytical labs regularly exploit selenite’s reactivity to test for trace metals or assess redox potential in samples. As a modifier, mixing with transition metal ions can form mixed selenites, unlocking uses in catalysis, ion-selective electrodes, and even in the development of new materials.
Synonyms & Product Names
Industry and academia call potassium selenite by several names, depending on context. Potassium selenous acid salt and dipotassium selenite show up in older and European literature, though most regulatory documents stick to potassium selenite. Registrations under EC and CAS list numbers such as 10102-18-8. Feed-grade and pharma-grade forms frequently pick up “food supplement” or “nutritional additive” tags, depending on end-use. I’ve come across brands using trade-friendly names like Selenex-K or Selifeed, though those mark private formulations blended with other micronutrients. Recognizing these alternatives is practical for buyers juggling global suppliers, as a mismatched trade name too often delays procurement and quality checks.
Safety & Operational Standards
Safety protocols require strong attention with potassium selenite. Inhalation, ingestion, or skin contact with dust or solutions can cause acute selenium poisoning, leading to gastrointestinal distress, neurological symptoms, and, at high doses, lethal toxicity. Storage aligns with the rest of the oxidizer category: tight, clearly labeled containers, dry and segregated from acids and combustibles. Precautions include gloves, protective eyewear, and designated spill management kits. Regulatory requirements in Europe, North America, and Asia list potassium selenite under hazardous materials for shipping, mandating training for anyone handling or moving commercial quantities. Regular staff safety audits and real-world spill drills, like those seen in chemical depots I’ve visited, sharpen awareness and reduce accident rates, showing safety isn’t just paperwork but daily discipline.
Application Area
Most potassium selenite ends up in micronutrient supplements for livestock. Its bioavailable selenium content helps prevent disorders like white muscle disease, which cost billions in agriculture losses every year. In pharmaceuticals, some fortified intravenous nutrition bags include potassium selenite to deliver selenium directly to patients lacking dietary sources. Industrial settings use the compound to adjust glass and pigment color, giving precise shades otherwise hard to reach, due to selenium’s interaction with iron and other ions. Analytical labs reach for potassium selenite during spectroscopic metal testing or for prepping test solutions. In technology research, selenite-based compounds turn up as precursors for new sensors and semi-conducting materials, stretching its impact from farmyard to the electronics bench.
Research & Development
The story of potassium selenite in research circles reveals a steady shift toward fine-tuned nutrition and advanced materials. Scientists continue probing selenium’s role in oxidative stress response and immune modulation, which starts with clarifying which selenium compound works best in what matrix. Experiments in my former university labs tested potassium selenite’s absorption versus organo-selenium compounds in small animal models, aiming to set new feed standards. In material science, exploration pushes into mixed selenite crystal growth and transparent conductive films, riding on potassium selenite’s flexibility and clean reaction profile. Nanotechnology gains momentum as researchers seek to shrink selenium carriers down to boost absorption and cut toxicity, keeping potassium selenite in the mix as a trusted starting point.
Toxicity Research
Potassium selenite presents a double-edged sword for toxicology teams. At trace levels, selenium—whether as selenite or selenate—proves essential for mammalian and avian life, but toxicity emerges fast as dose rises. Animal studies repeatedly confirm chronic high exposure brings on symptoms from brittle hair and nails to severe neurological damage. Acute exposure, even accidentally, needs medicine capable of handling heavy metal toxidromes. Ongoing studies break down absorption, tissue retention, and excretion rates, aiming to adjust safe upper limits and improve emergency response protocols. Research into long-term environmental release warns of selenium building up in water systems, entering food chains, and harming fish and birds. Authorities like EFSA and EPA now use detailed toxicology data to refine dietary maximums, packaging requirements, and disposal protocols. From my own reading and practice, over-reliance on selenite supplementation, particularly in poorly regulated livestock operations, drives home the cost of ignoring cumulative toxicity risks.
Future Prospects
Looking out over the next decade, potassium selenite faces shifting ground in both opportunity and regulation. Nutritional scientists hope for more selective delivery systems, building on the ease of dosing selenite in feeds, yet searching for alternatives with less toxicity risk. Regulatory tightening—fueled by public sensitivity to trace metal contamination and environmental selenium buildup—pushes manufacturers to prove safety both in the field and after product disposal. Technical teams in optoelectronics and specialty glass remain keen to customize selenite compounds, crafting smart coatings or next-generation catalysts. Advances in nutrigenomics may unlock new insights into how selenite, in contrast with organic forms, shapes cell and tissue responses. I see potential for potassium selenite to stay relevant, so long as producers and users balance performance with responsibility, respecting both human health and the ecological web that can so easily tip under a wafer-thin line of excess.
What is Potassium Selenite used for?
Not Just Another Compound
Potassium selenite might not pop up in everyday conversations, but inside certain industries and scientific circles, it matters a lot. This chemical brings selenium into the spotlight, an element that plays a key role in human and animal health. From animal feed to dietary supplements, potassium selenite delivers selenium in a way that's both usable and easy to control. For anyone who has worked around livestock nutrition or food science, selenium isn't some abstract concept; it's a real-world nutrient with tangible effects on living things.
Selenium’s Place in Nutrition
Selenium itself is a trace mineral. The body doesn’t need much, but lack of it can open the door to a range of problems. Heart muscle issues, higher risk of infection, even fertility problems have been tied to low selenium. People who study animal health have wrestled with these challenges for decades. Potassium selenite steps in as a fix. Its chemical form dissolves easily and lets farmers or nutritionists add precise amounts straight into grain, supplements, or mineral mixes for livestock. Animals get what their bodies require, nothing less and nothing more.
Human Health and Dietary Supplements
For humans, potassium selenite appears on the label of some dietary supplements. It’s chosen because it’s a reliable, cost-effective way to help folks meet daily needs for selenium, especially where diets fall short. Populations in certain regions experience soil that simply doesn’t provide enough selenium for crops, so what ends up on the dinner table can lack this essential mineral. Supplements that use this ingredient give those populations a chance to sidestep deficiency.
Food Fortification and Safety Measures
Potassium selenite doesn’t only show up in animals and supplements; it finds its way into food fortification, too. Wheat, corn, and even infant formulas sometimes get boosted with selenium to make sure even the pickiest eaters get what they need growing up. Fortification projects don’t take selenium lightly. It’s powerful, even at low doses, and the gap between a helpful amount and a dangerous one isn’t huge. Regulatory bodies closely monitor how much potassium selenite can be added. Walk into any well-run feed mill or supplement factory, and you’ll see careful measuring, strict records, and plenty of safety warnings posted on the wall.
Possible Risks and What Needs Attention
Too much selenium leads to selenosis. Symptoms can range from upset stomach and brittle hair to far graver problems. In rural areas where selenium-rich soil naturally shows up, there have been incidents of livestock poisoning due to excessive levels. In my time consulting for feed operations, I heard stories of ranchers losing cattle when they didn’t pay close attention to total selenium intake from all sources. That’s why ongoing research, regular nutritional audits, and clear labeling matter so much in both farming and food production.
Looking Ahead: Smarter Use of Potassium Selenite
Better education around trace minerals and closer collaboration between scientists, veterinarians, and regulatory agencies promise a safer, more effective use of potassium selenite. Companies now invest in precision dosing systems that cut down on human error and give every animal or person the benefit of selenium—no more, no less. With a focus on scientific evidence and public health, potassium selenite stands as a bridge between what nature provides and what living bodies need to thrive.
Is Potassium Selenite safe for human consumption?
What Is Potassium Selenite?
Potassium selenite pops up in ingredient lists of vitamin supplements and some fortified foods. It’s a compound that delivers selenium, a trace mineral the body needs for thyroid hormone production and antioxidant function. Most folks get selenium from foods like Brazil nuts, tuna, eggs, and cereals. Potassium selenite enters the picture in places where dietary intake falls short or foodmakers want to add a controlled trace of selenium.
Why Selenium Matters
Selenium punches above its weight in human health. Not enough can bring real problems—weak immunity, slow thinking, even heart issues in some cases. But there’s a catch. The difference between an optimal dose and a dose that causes harm is not huge. The National Institutes of Health reports that adults need just 55 micrograms of selenium per day. That’s a tiny pinch. Cross 400 micrograms daily for extended periods and you’re courting toxicity.
What Does Potassium Selenite Do in Supplements?
Supplement makers use potassium selenite because it dissolves easily and delivers a predictable, measurable amount of selenium. The U.S. Food and Drug Administration and its European counterparts allow its use, but with strict limits. Most multivitamins with selenium contain microgram-level amounts—far below what triggers risk. Medical professionals sometimes use potassium selenite for patients with proven selenium deficiency, typically under close supervision, because too much damages cells and brings nausea, hair loss, and nerve pain.
What Do Studies Say About Safety?
Animal research shows clear warning signs: potassium selenite can harm cells at high levels by generating reactive oxygen species. Large doses over weeks might damage kidneys, liver, and nervous tissue. Doctors and toxicologists have a name for selenium poisoning: selenosis. Signs include loss of hair and nails, fatigue, a telltale garlic-like breath odor, and neurological effects. Though most cases come from bulk supplementation or industrial exposure, food safety authorities keep upper intake threshold well below these danger levels.
Concerns About Potassium Selenite in Food
Calls to swap potassium selenite for organic selenium compounds like selenomethionine are getting louder in some circles. Organic forms show higher bioavailability and a gentler absorption curve in most research, meaning fewer people experience temporary gastrointestinal upset or spikes in selenium levels. Still, potassium selenite remains cheaper and reliable. Stringent labeling laws help keep accidental overconsumption rare in places like the U.S. or E.U., but folks who take handfuls of supplements without tracking totals risk stacking up too much.
The Path to Responsible Use
Trust builds on transparency. Reputable supplement makers publish how much selenium each serving contains, and most foods fortified with potassium selenite undergo government review before hitting store shelves. Consumers who read the fine print, track their daily intake, and consult healthcare providers make the best use of potassium selenite. Nutritionists frequently remind people that a varied diet full of whole foods covers selenium needs for most adults. Supplementing usually serves those with malabsorption, low dietary diversity, or specific health needs under medical care.
Potential Solutions
Clear labeling stands at the front line. Health advocates want warnings on supplement bottles about the risks of overuse. Doctors suggest labs offer affordable blood selenium tests for those concerned about deficiency. Schools and clinics can help spread info about safe supplement use. Big brands might shift toward organic selenium forms and away from potassium selenite, but doing so would raise costs and not necessarily improve safety for most people following label directions.
At the end of the day, potassium selenite remains a safe ingredient for human consumption at the levels found in legally sold food and supplements. Problems usually rise from high-dose misuse and lack of oversight. Familiarity with serving sizes and a real look at total trace mineral intake can help anyone stay on the safe side.
What is the recommended dosage of Potassium Selenite?
How Much Potassium Selenite Is Right?
Potassium selenite stands out as a source of selenium—a trace mineral our bodies depend on in small amounts. Selenium fuels important enzymes, strengthens the immune system, and protects cells from free radicals. You often see potassium selenite listed in dietary supplements or animal feed. When it comes to proper use, exactly how much is too much or too little stirs plenty of debate.
Recommended Amounts for People
For healthy adults, the U.S. National Institutes of Health recommends 55 micrograms (mcg) of selenium daily. Pregnant people might go up to 60 mcg; lactating folks, 70 mcg. Most folks meet these levels through diet, mainly eating foods like nuts, eggs, seafood, and meat.
Potassium selenite sometimes gets used when someone can't get enough selenium from diet alone, or for folks with rare malabsorption illnesses. Doctors usually prescribe selenium in the range of 50-200 mcg per day overall—not just the selenite form.
Using potassium selenite for minerals means a tiny amount. Potassium selenite isn’t a multivitamin ingredient you can just toss in without thinking. For every 1 milligram of potassium selenite, you get about 0.46 milligrams of elemental selenium. Even slight miscalculations can cause trouble. Selenium in high doses brings on symptoms like garlic breath, brittle hair, or even nerve problems. Doses over 400 micrograms daily year after year set up risk for toxicity, so nobody should self-prescribe or double up on supplements, even when feeling sluggish or run down.
Why Dose Matters For Animals
In farming, potassium selenite gets added to animal feed to combat selenium deficiency, which can slow growth or cause disease in livestock. The National Research Council puts average dietary selenium needs at about 0.3 parts per million for cows, pigs, and chickens. Feed mills need close oversight to hit that target. Animals get sick if they eat either too little or too much. Ranchers adding loose supplements on their own sometimes overdo it, so trained nutritionists or veterinarians oversee this job.
Best Practices And Long-Term Health
Every supplement label should give a clear dosage and a warning about not exceeding recommended daily intake. Skipping label details or following internet tips instead of medical advice can end up hurting those using potassium selenite. In my own family, when my uncle decided to take selenium supplements without talking to his doctor, he wound up with headaches and fatigue until he quit. Only after checking his bloodwork did his doctor explain he’d quietly doubled what his body needed, thinking more would benefit his immune system.
Professional groups like the Food and Nutrition Board and health authorities all agree—selenium has a narrow safe window. Manufactured supplements have to undergo regular testing. The FDA doesn’t review dietary supplements before they hit shelves, so consumers have to trust either the manufacturer’s reputation or third-party testing. Researchers even suggest checking with healthcare professionals before adding new selenium pills to your routine, especially for those dealing with thyroid issues, autoimmune conditions, or pregnancy.
Making Sense Of The Choice
Getting enough selenium matters, but more is rarely better. Potassium selenite works best under guidance, as part of a gentle, supervised balance. A doctor or licensed nutritionist can confirm if there’s a real gap and point to the safest route. Sometimes, a well-tuned diet gives all the selenium most folks ever need.
Are there any side effects or risks associated with Potassium Selenite?
Why It Matters
Potassium selenite turns up in supplements, animal feed, and, at times, in medical settings. Selenium, which sits at the core of this compound, ranks as a trace element that bodies demand in small amounts. Too much—or too little—can tip the balance and spark real problems. Working in pharmacy, I've seen plenty of confusion over minerals like these. Folks think because it’s “just a mineral,” higher doses do more good. That’s far from true—particularly with selenium salts.
Known Side Effects: What Evidence Shows
Taking potassium selenite above recommended limits can lead to noticeable side effects. Early signs often show up as nausea, stomach pain, or a metallic taste. People sometimes notice hair loss or brittle nails after several weeks if they push dosing past safe levels. A body overloaded with selenium might react with skin rashes, irritability, or even tingling and numbness. These aren’t just mild annoyances—these are warnings to stop and get checked.
Larger doses increase the danger. Over time, selenium toxicity—also called selenosis—has made the rounds in several reports. Symptoms can escalate into trouble walking, muscle tenderness, or even breathing issues. In rare cases, sensitive people have faced kidney or liver troubles. What’s critical to know: the human body draws a fine line between what’s helpful and what’s harmful with selenium. Most adults need only a tiny amount—55 micrograms per day. Crossing above 400 micrograms daily gets risky.
At-Risk Groups and Interactions
Children, pregnant women, and people with chronic illness face higher risk from excess potassium selenite. These groups process minerals differently, and mistakes in dosing can pack bigger consequences. Kidney problems magnify the threat since waste products clear slower. People who pair selenium with vitamin C, certain antibiotics, or cancer drugs sometimes run into unexpected reactions since selenium can shape how these medicines behave.
Food and supplement interactions also play a role. Brazil nuts, fish, eggs, and whole grains provide plenty for most diets. Mixing potassium selenite into a regimen without screening for diet and other supplements raises the odds for accidental overdose.
Prevention and Smarter Use
The best path stays clear: measure and monitor. Blood tests show actual selenium levels and prevent guessing games. Trustworthy supplement makers list precise milligrams and micrograms—check labels closely. Doctors sometimes prescribe potassium selenite for deficiencies, but every case starts with lab testing, not self-diagnosis. The web brims with “immune-boosting” products. Most healthy people covering basic dietary ground won’t see extra benefit from piling on selenium.
Education forms the backbone of safer use. Health providers should speak plainly about risks and review all medications with their patients. Any new supplement goes through the same checklist. For those with kidney or digestive conditions, regular check-ups and bloodwork can stop problems before they start. Parents should keep all forms of selenium out of kids’ reach, since dosing errors in children can turn dangerous fast.
Final Thoughts
Potassium selenite, like any potent mineral, serves a purpose but doesn’t come risk-free. Following recommended allowances, reading product labels, and asking your doctor before making nutrition changes helps sidestep the biggest hazards. Paying attention to changes in how you feel matters just as much as any number printed on a bottle. Health comes down to balance, not more for more’s sake.
How should Potassium Selenite be stored?
Understanding Potassium Selenite’s Characteristics
Potassium selenite plays an important part in agriculture, nutrition, and laboratory work. It has the chemical formula K2SeO3. Its ability to supplement essential selenium adds value, yet safe handling becomes just as crucial as its function. This compound brings risks if storage slips off the radar. Potassium selenite is known to react with strong acids and can become unstable in moist or hot environments. Keeping these facts in mind, practical storage matters a great deal.
Why Storage Conditions Matter for Health and Safety
Anyone who’s worked in research or ran a farm supplementing feed knows that improper storage is a shortcut to trouble. Potassium selenite is toxic in concentrated amounts, especially when inhaled or ingested. Misplaced storage can lead to chemical reactions, container leaks, or accidental mixing with incompatible substances. For anyone with family, colleagues, or livestock nearby, the potential harm isn’t just a distant idea — real damage can happen.
Inhalation causes respiratory issues and skin contact may irritate. Mistakes in storage can result in costly spills or unexpected exposure. According to the United States Occupational Safety and Health Administration (OSHA), containers should be sealed and labeled clearly to prevent accidental handling.
Recommended Storage Guidelines
Potassium selenite stays most stable in a cool, dry place. Direct sunlight speeds up decomposition or causes heat build-up, so shelves near windows or heat sources should always stay empty. Humidity also poses a real threat; moisture can degrade the chemical and encourage clumping, making it harder to measure out exact quantities for lab protocols or animal feed. I keep a silica gel pack in chemical drawers for this very reason.
Contamination often sneaks up when storage space hosts multiple chemicals. Never store potassium selenite near acids, reducers, or organic matter. Cross-contamination doesn’t just spoil the product — it sends safety down the drain, sometimes literally.
Always use original containers made from compatible materials, usually plastic or glass, with tight-fitting lids. Manufacturers use these for a reason: they resist corrosion and keep out environmental elements. Secondary containment trays below the bottles catch leaks caused by worn-out seals or accidental drops. Regular inspection means you spot any problems before they turn into emergencies.
Emergency Planning and Everyday Responsibility
Like many chemicals, potassium selenite demands respect. Lab managers and warehouse crews should have safety data sheets handy in each chemical area. Quick access to eyewash stations and spill clean-up kits prevents small mistakes from ballooning. Training doesn’t just tick boxes; it builds habits that keep people safe.
I’ve learned it’s smart to rotate stock and check expiry dates regularly. Fresh product means fewer risks. Set up regular checks in your routine — schedule reminders if you have to. Don’t rely on memory alone, especially in busy, shared spaces.
Solutions for Safer Chemical Management
Lockable cabinets restrict access to those who understand the risks. Color-coded labels can signal hazardous content at a glance. Digital inventory systems help staff track what’s on-site and flag when supplies go low or containers pass their expiration window. These steps may sound basic, but they consistently prevent accidents both large and small.
Clear protocols and regular oversight maintain a safe work environment. With potassium selenite, small preventive steps keep harm far away and ensure this useful compound serves its intended purpose, not public health headlines.
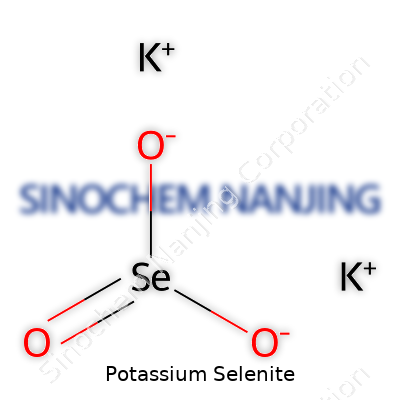
| Names | |
| Preferred IUPAC name | Potassium trioxidoselenate(2-) |
| Other names |
Dipotassium selenite Potassium selenious oxide Selenious acid, dipotassium salt |
| Pronunciation | /pəˈtæsiəm ˈsɛlɪnaɪt/ |
| Identifiers | |
| CAS Number | 10102-18-8 |
| Beilstein Reference | 1840789 |
| ChEBI | CHEBI:77637 |
| ChEMBL | CHEMBL1201640 |
| ChemSpider | 54816 |
| DrugBank | DB11129 |
| ECHA InfoCard | 03b85e65-3e53-4e3d-aaa5-24a9c0883f9c |
| EC Number | 231-555-9 |
| Gmelin Reference | 54612 |
| KEGG | C13956 |
| MeSH | D011400 |
| PubChem CID | 24828 |
| RTECS number | VV7325000 |
| UNII | 1K5696D41Y |
| UN number | UN2630 |
| Properties | |
| Chemical formula | K2SeO3 |
| Molar mass | 174.07 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.39 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -4.14 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 2.62 |
| Basicity (pKb) | 3.17 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.450 |
| Viscosity | Viscous liquid |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 120.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -675.6 kJ/mol |
| Pharmacology | |
| ATC code | A12CE02 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or absorbed through skin; may cause irritation to skin, eyes, and respiratory tract. |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P330, P391, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Autoignition temperature | Approximately 250°C (482°F) |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 1.6 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 7 mg/kg |
| NIOSH | WC5600000 |
| PEL (Permissible) | PEL: 0.2 mg Se/m³ |
| REL (Recommended) | 0.02 mg Se/m³ |
| IDLH (Immediate danger) | 1 mg/m3 |
| Related compounds | |
| Related compounds |
Sodium selenite Potassium selenate Sodium selenate Selenous acid Potassium tellurite |

