Potassium Selenate: A Closer Look at Its Journey, Uses, and Future
Historical Development
The story of potassium selenate started in the nineteenth century, tied closely to research into selenium chemistry and the broader boom in inorganic salt discoveries. Chemists in Europe, particularly in Germany and Sweden, began isolating selenium compounds after Berzelius identified selenium in 1817. As industry spun up glassmaking and metallurgy, applying selenium to color glass or alter alloys, the need for reliable selenium sources grew. Potassium selenate appeared soon after, as researchers experimented with alkali metal salts and saw the unique results that selenium brought to both industry and laboratory studies. In the twentieth century, potassium selenate became a staple for testing in analytical labs, fertilizer research, and specialized manufacturing.
Product Overview
Potassium selenate stands out as a white, water-soluble salt, valued in specialized applications where selenium supply needs a clean, measurable form. Its most direct use shows up in agriculture, particularly rural regions where soil naturally lacks selenium. The salt ends up added to fertilizers to address crop deficiencies, so livestock and people downstream don’t fall short of this trace nutrient. Chemists rely on potassium selenate in research settings, especially when they demand consistency across experiment batches. In industry, it plays a role in certain glass colorants, handling glass redox reactions, or serving niche electronics applications.
Physical & Chemical Properties
Potassium selenate arrives as colorless, odorless crystals, dissolving quickly in water and forming a neutral solution. Its empirical formula is K2SeO4, with a molar mass landing at 228.07 g/mol. It won’t melt down in a home oven—decomposition happens at about 930°C, where it turns to selenium trioxide and potassium oxide instead of a true liquid. One major factor chemists lean on lies in its high solubility, over 160 grams per liter at room temperature. This trait gives it an advantage over potassium selenite or elemental selenium in applications needing measured selenium doses. The compound stands relatively stable in storage, so one doesn’t need constant worry over unwanted reactivity at room temperature. It won’t act as a major oxidizer, yet it reacts steadily with strong acids or with reducing agents, producing products useful or risky, depending on the context.
Technical Specifications & Labeling
Commercial suppliers label jars of potassium selenate with purity levels—usually above 98%—and a clear warning about toxicity. Labels cite both the United Nations (UN 2630 for transport) and the European Union’s CLP/GHS hazard statements: H301 (toxic if swallowed) and H373 (risk of longer-term health effects). Quality control relies on confirming metallic impurities remain below certain levels, as iron, lead, or cadmium could turn a safe-agriculture product into a dangerous one. The packaging often comes in HDPE bottles with secure caps to fight moisture and accidental spills. Safe transport requires paperwork that helps handlers recognize risks if containers break open in a shipment disaster.
Preparation Method
Potassium selenate production starts in chemical plants by oxidizing elemental selenium or treating selenite with potassium hydroxide under oxygen-rich conditions. If producing from selenium metal, workers dissolve it first in hot concentrated nitric acid, creating selenic acid, followed by neutralization with potassium carbonate or potassium hydroxide. This reaction generates a solution of potassium selenate, which then gets cooled and crystallized out. Alternatively, potassium selenite reacts with additional oxidizers—think hydrogen peroxide or chlorine—in alkaline water, coaxing it into the selenate form. As someone who’s watched these processes in university teaching labs, I know how even faint air currents carry dust, so handling involves fume hoods and disposable gloves to avoid contamination or toxic exposure. Plants recover pure phosphate-like crystals through careful filtering and slow evaporation.
Chemical Reactions & Modifications
In the lab, potassium selenate works best when mixed into aqueous solutions. It acts as a selenium donor in redox reactions, strongly favoring conversion to selenite or elemental selenium with modest heating and reducing agents. Reactions with acids produce selenic acid, a powerful oxidizer in its own right. With enough heat, potassium selenate gives up oxygen, decomposing to potassium selenite and, ultimately, volatile selenium oxides. For anyone making selenium nanoparticles or catalysts, this property makes potassium selenate an approachable precursor: dissolve it, add a mild reducing sugar or ascorbate, and out come zero-valent selenium or modified compounds with precisely tuned properties. Such reactions speed up both in hot solutions and under acidic conditions, so experimentation with temperature control or graduated acid titration leads to finely controlled results.
Synonyms & Product Names
Chemical supply catalogs might list potassium selenate under several names—potassium(VI) selenate, potassium selenate(VI), or by its registry number, CAS 10102-18-8. Some companies sell it labeled as “selenium supplement (fertilizer grade)” in rural outlets, while lab-grade material takes the name “K2SeO4, analytical reagent.” In older chemistry texts, one might spot “selenium acid, dipotassium salt,” tying it back to the selenic acid production route that dominates modern processes.
Safety & Operational Standards
All lab workers and agricultural suppliers take potassium selenate safety procedures seriously. Inhalation or swallowing risks acute illness—toxicity can present as gastrointestinal distress, tremors, or, at high enough doses, chronic heart and nerve problems. Chronic exposures raise questions of carcinogenicity and animal toxicity, as selenium’s “nutrient window” stays narrow. Procedures require gloves, goggles, and close attention to ventilation. Spills need fast clean-up with damp cloths or HEPA vacuums, never brooms. Farms using this compound mix it in liquid storage tanks with backflow prevention and strict labeling, hoping to avoid drift into water tables or onto non-target fields. Material safety data sheets detail rescue steps if exposure occurs and instruct on rapid first aid for poisoning.
Application Area
The most visible role comes from fertilizer enrichment in vast grain-growing regions—Brazil, Finland, and China all supplement selenium-poor soils to prevent animal and human deficiency diseases. Feed additive producers add potassium selenate to premixes, keeping doses below national regulatory limits to avoid toxic buildup. In the glass industry, potassium selenate finds use adjusting color and oxidation states. Certain specialty glassmakers add it deliberately, crafting red and pink hues prized by artists and architects. Researchers put the compound to use as a starting point for synthesizing selenate nanoparticles, bioactive complexes, and catalysts. In environmental studies, potassium selenate appears in water treatment pilots for selective selenium removal, testing biological or chemical traps in rivers contaminated by mining runoff.
Research & Development
Academic groups continue finding new angles for potassium selenate, from medical biochemistry to green chemistry. Its role as a precursor for cancer research compounds, selenium-labeled proteins, and antioxidant enzyme mimics has gained momentum over the past decade. Biomedical teams look at how controlled-release selenate nanoparticles can deliver therapeutic doses, reducing risks of overdose that haunt more traditional selenium supplements. Chemical engineers work to realize cleaner synthesis processes, experimenting with ultrasound or microwave-initiated crystallization that trims waste and energy use. Studies appear on using potassium selenate in low-temperature materials synthesis, where its high reactivity produces novel materials quickly.
Toxicity Research
Potassium selenate’s toxicology profile earns close attention. At trace levels, selenium counts as a vital dietary nutrient, but the difference between need and harm remains slight. High doses produce “selenosis,” a sometimes fatal syndrome marked by hair loss, cracked nails, and neurological damage. Animal trials over the years tracked dose-response curves, finding that different livestock species process selenium at varying rates. Regulatory agencies—U.S. EPA, EFSA in the EU, China’s Ministry of Agriculture—set daily intake maximums (roughly 55 micrograms for humans, depending on age and health). Environmental impact research expanded rapidly in the 1980s and 1990s following catastrophic wildlife poisonings downstream of agricultural runoff. Today’s research covers chronic, low-level exposure effects, potential accumulation in crops or aquatic food chains, and interaction with other soil minerals. Topics under study include the ability of organic-binding processes to limit selenate mobility and methods for in situ monitoring of bioavailable selenium.
Future Prospects
Looking forward, potassium selenate has plenty of life left as an agricultural solution and research tool. Countries suffering widespread selenium deficiency treat this compound almost as a health supplement for crops and herds. Industry trends focus on better application strategies—slow-release fertilizers, precision dosing, and in-field monitoring—to prevent waste and minimize risks of waterway contamination. Scientists explore prebiotic and cancer-preventive roles for selenium compounds, and potassium selenate remains an easy entry point for pilot manufacturing or preclinical tests. If green chemistry advances make production cleaner and new analytical tools further map out safe environmental windows, those relying on this compound could cut risks without sacrificing benefit. In my own experience working on a selenium soil remediation project, I witnessed both the hazards and the profound impacts on public health when the balance is struck right. For now, potassium selenate holds ground as an essential but tightly regulated link in the expanding story of human and environmental health.
What is Potassium Selenate used for?
Potassium Selenate: Why It Matters
Most people hear about selenium in the context of dietary supplements or food fortification. Fewer know that potassium selenate, a simple salt with a long name, plays an important part in our food chain before it even reaches the grocery store. I grew up in a farming community in the Midwest, and I’ve seen how some soils struggle to provide enough micronutrients for healthy crops. That’s where potassium selenate comes in — it’s not a headline-grabber, but it quietly supports healthier ecosystems and, eventually, healthier people.
Supporting Crop Health and Human Nutrition
In my experience, farmers pay close attention to the trace minerals in their fields. Selenium often shows up on those soil tests as “low” or “deficient.” Without enough selenium in the ground, grains and vegetables end up lacking this vital mineral. Potassium selenate acts as an efficient way to boost selenium content in crops. Applied at the right time, it works its way from the soil into plant tissue, increasing the selenium content of wheat, barley, and other staples. Research from agricultural universities backs this up — even a modest application can make a noticeable difference in selenium levels in food.
This form of selenium also stands out because of its solubility. Unlike other compounds that take forever to break down, potassium selenate dissolves quickly and gets into the root zone fast. Farmers and agricultural advisors use it strategically, not just tossing it on every field, but targeting areas most at risk of selenium deficiency. Getting this step right means the food produced contains enough selenium to help meet human dietary requirements.
Animal Feed and Food Security
Beyond plant nutrition, potassium selenate finds its way into animal feed. Mineral premixes for cattle, sheep, and poultry often rely on this compound to keep livestock healthy. Selenium supports immune function and reproductive health in animals, much like it does for people. I’ve spoken to ranchers who remember the days before selenium supplementation, when “white muscle disease” in calves caused real losses.
For regions where the soil naturally contains little selenium, supplementing with potassium selenate has helped reduce these problems, improving the welfare of animals and also ensuring a more secure food supply. One major benefit: by supporting animal health, potassium selenate reduces the need for other interventions, which saves time and money for producers and, ultimately, for consumers.
Food Safety, Dose, and Environmental Concerns
The flip side to all the benefits is the need for careful management. Too much selenium can be toxic. This isn’t just a theoretical problem — there have been well-publicized incidents, especially in large herds, where over-supplementation led to health issues. Regulatory agencies in the US and Europe set clear limits for selenium additives, including potassium selenate, in both crops and feed. Following these guidelines protects both public health and the environment.
Managing this balance requires experience, good testing, and up-to-date advice. Soil labs, veterinarians, and crop specialists all play a role. The push for precision agriculture offers a path forward: better data, smarter application. New sensors and monitoring tools make it easier to apply only as much potassium selenate as is truly needed.
Looking Forward
Potassium selenate may sound technical, yet its value plays out in everyday health, from the bread on our tables to the wellbeing of animals in the field. It deserves attention from policymakers, farmers, and even consumers who want to know more about what goes into their food. With continued care and scientific input, this humble salt keeps working behind the scenes for healthier land and people.
Is Potassium Selenate safe for human consumption?
Looking Closely at What Potassium Selenate Brings
Potassium selenate shows up in chemical catalogs and some agricultural products. Not many people talk about it unless they spend time reading through food science papers or labels on plant fertilizers. As someone who keeps an eye on nutrition research, I believe it’s important to dig into what this chemical means for the food chain and consumer health.
Selenium: Small Dose, Big Effect
Selenium doesn’t work like common minerals such as calcium or magnesium. The body uses it in tiny amounts. Potassium selenate delivers selenium, which acts in metabolic processes—helping to build proteins that defend against cellular damage. People get selenium from foods like nuts, fish, and grains. In some parts of the world, soil runs low on selenium, which brings up the topic of fortification.
Both the U.S. Department of Agriculture and the National Institutes of Health point out that most people in North America and Europe meet their selenium needs through diet. Issues surface in regions with poor soil or diets lacking variety. In these places, farmers and governments sometimes consider adding selenium to crops or animal feed, and potassium selenate has made its way into that conversation.
The Fine Line Between Nourishing and Toxic
Potassium selenate has a narrow window between enough and too much. The World Health Organization and the European Food Safety Authority both confirm that low selenium can weaken the immune system and slow growth. Too much leads to selenium poisoning—hair loss, brittle nails, skin rashes, and in extreme cases, nervous system problems or death.
Potassium selenate dissolves easily, and the body absorbs it well. This makes it effective for quick correction of selenium deficiency, yet also increases the chance of overdose. An adult only needs about 55 micrograms of selenium daily. Anything over 400 micrograms, over time, sends health risks climbing.
Food Additives and Supplement Safety
In agriculture, controlled selenium supplementation can help produce healthier crops and livestock. Food manufacturers sometimes fortify products with selenium, though most commonly in the form of selenomethionine or sodium selenite. Potassium selenate rarely appears on supplement labels in the U.S. due to its higher toxicity compared to organic forms.
Countries that permit potassium selenate ask for strict controls on how much goes into foods or animal feeds. The European Commission, for example, limits selenium supplementation and asks manufacturers to show that their products do not exceed those levels. The U.S. Food and Drug Administration also enforces maximum limits, and recalls happen when companies miscalculate dosages.
Real-World Practices and Next Steps
In a household kitchen, you won’t spot potassium selenate among the spices or pantry items. Even so, the chain of production—farms, animal feed, processed foods—brings it into indirect contact with consumers. Quality assurance testing and transparent food labeling can offer the best confidence for people choosing packaged foods.
For anyone thinking about dietary supplements, I recommend sticking to products that clearly state their selenium source. Natural sources or those backed by studies with established human safety records present much less risk. Limiting intake to recommended dietary allowances protects against both deficiency and toxicity risks—and keeps nutrition from turning into a guessing game.
How should Potassium Selenate be stored?
Why Proper Storage Shapes Safety
Anyone who’s worked in a lab or managed a storeroom full of reagents knows that how you store something like potassium selenate isn’t just a box-ticking exercise. This compound, like many selenium salts, ranks high on the safety meter—not just for its toxicity, but because it tends to absorb moisture from the air and doesn’t play nice with everything around it. Mistakes in storage can ruin samples, wreck experiments, or worse, hurt people. So, taking storage shortcuts isn't about saving time or space—it’s about avoiding health scares and wasted resources.
Dry, Cool, and Sealed: The Basics That Keep on Giving
After years working with small chemical inventories, I learned that cool and dry doesn’t just mean “not in the sun.” Humidity will turn potassium selenate into a lumpy mess, and heat can speed up its degradation or increase the risk of accidental release. Best results come from storage in a dedicated cabinet away from strong oxidizing agents, acids, and combustible materials. A standard chemicals cabinet, kept closed and clean, does the trick. Don’t stack it next to the open window or a constantly rattling fridge—both can lead to unpredictable messes.
Airtight containers are a must. Glass bottles with tight-fitting lids or high-quality plastic jars cut down on moisture intrusion. Silica gel packs tossed in for good measure pull out residual humidity, especially in old buildings where AC never quite cuts it. A clear label, with the date received and opened, helps track freshness—selenium compounds never last forever.
Training and Visibility: People Matter
Even the best storage protocols fall flat without staff who take them seriously. During my first internship, a senior tech drilled into everyone that hazard training is as important as the lab manual. Potassium selenate may sound obscure, but symptoms of selenium poisoning aren’t. Regular review of Safety Data Sheets (SDS), signage on the storage cabinet, and quick training refreshers ensure new folks know what they’re dealing with—and remind old hands how easy it is to overlook the basics. It’s better to be the person who double-checks containers than the person running for the emergency shower.
Disposal Plans from the Start
Responsible storage includes thinking about the endgame. Labs that treat disposal as an afterthought wind up with leaky bottles and mystery goo in the back corner. Potassium selenate qualifies as hazardous waste in most regions. Planning its exit when it arrives prevents “forgotten bottle” syndrome. A logbook noting quantities and a relationship with a certified disposal company smooths the whole process. Regulations change—especially around selenium compounds—so updates to protocol need to happen whenever new laws come down the pipe.
Finding Room for Improvement
New storage tech—better containers, humidity monitors, and digital inventory systems—pops up every year. Adoption can feel costly or annoying, but the labs that invest in airtight storage or even basic dataloggers tend to find their cleanup bills and safety incidents dropping. Simpler ideas help, too: grouping all selenium compounds on a single shelf with a bright label puts risk where everyone can see it.
Practical Steps Build Better Habits
Potassium selenate demands respect, whether you're working in a research lab, a fertilizer plant, or a teaching classroom. It’s diligence in the details—securing lids, logging inventory, placing reminders—that keeps accidents away. Anyone can memorize guidelines, but it’s the practical, everyday habits that hold the line between routine and disaster.
What is the recommended dosage of Potassium Selenate?
Understanding Potassium Selenate
Potassium selenate is a potent source of the trace mineral selenium. In agriculture, medicine, and dietary supplements, it often appears as a solution for selenium deficiency, but the fine line between helpful and harmful doses makes it worth a closer look.
Why Selenium Matters
Selenium supports the immune system, thyroid health, and protects cells from oxidative damage. In my own experience working on sustainable nutrition programs, I’ve seen how a lack of selenium can weaken livestock and reduce crop quality. On the other hand, selenium overload creates toxicity risks, which can cause symptoms like stomach upset, hair loss, nerve damage, and even pose a threat to life.
Dosage Recommendations
Facts from organizations like the World Health Organization and the National Institutes of Health say most adults need around 55 micrograms of selenium every day. Potassium selenate is extremely concentrated — one must measure doses carefully. For crops, soil analysis drives application rates; too much can poison both plants and the land. In supplements and fortified foods, selenium rarely exceeds 100 micrograms per serving, keeping intake below the average safe upper limit of 400 micrograms per day for adults.
Toxicity happens when intake goes above those boundaries over time. The soil in some parts of the world, like certain regions in China or the United States, already holds enough selenium. Blindly adding more raises serious health and environmental concerns. From what researchers tracked, animals can show hoof problems and neurological symptoms if they graze on pasture with high selenium from fertilizers or supplements.
Practical Approaches
Instead of defaulting to supplementation, testing soil, water, or blood levels provides data for tailored solutions. In agriculture, farmers seek advice from agronomists with experience reading lab reports. Home gardeners check for local selenium data before looking for additives. Doctors rely on lab tests and established medical guidelines before prescribing selenium compounds.
I have met families in rural areas still using basic multivitamins because of marketing campaigns pushing minerals as cure-alls. They may not realize their diet already brings in enough selenium from nuts, grains, eggs, or fish. Without good guidance, they risk crossing into excess.
Possible Solutions for Safe Usage
Education can fill the gap. Clear labels on supplements put in perspective how much selenium comes from each pill compared to a day’s worth of food. Doctors and pharmacists continue to remind patients about the narrow safety window of nutrients like selenium. Manufacturers use technology to better control the amount in fertilizers and feeds.
Open access to simple testing for patients and growers makes the biggest impact. Where selenium deficiency threatens, targeted programs prove useful. Where natural levels run high, new fertilizer rules and crop selections keep both people and the planet safe. After all, too much of anything, even an essential mineral, rarely brings good results.
Are there any side effects or risks associated with Potassium Selenate?
Getting the Facts Straight on Potassium Selenate
Potassium selenate pops up in certain supplements and agricultural products, often marketed for its selenium. Selenium counts as an essential nutrient; your body relies on it in trace amounts for a healthy immune system and thyroid function. Food doesn’t always supply enough for everyone, especially in some regions. This is why some reach for supplements. Still, more does not mean better, and the risks of too much selenium can get serious.
How Potassium Selenate Works in the Body
Selenium changes an enzyme in the thyroid and helps protect cells from damage. Potassium selenate acts as a delivery system for this element, making sure selenium gets absorbed once it’s inside you. The downside: just a little extra selenium above your body’s need tips the scale from helpful to harmful surprisingly quickly. Adults require about 55 micrograms per day. Anything much higher for a stretch risks trouble.
Short-Term and Long-Term Effects
Taking potassium selenate without proper guidance puts you at risk of selenium toxicity, or selenosis. Too much selenium at once can cause nausea, vomiting, and headaches. I remember hearing stories of multivitamin overdoses in health forums. Most people want to be healthy and feel in control, but it only takes a few reckless doses—hundreds of micrograms per day—to start noticing hair loss, brittle nails, garlic breath, or even fatigue and irritability. At higher levels, selenium toxicity can hit the nervous system and may lead to tremors, confusion, or more serious symptoms. The National Institutes of Health points out a tolerable upper limit of 400 micrograms per day for adults.
Environmental and Agricultural Exposure
Potassium selenate doesn’t only come in pills. It’s used in fertilizers, especially in countries with selenium-deficient soil, to boost the nutrient content in crops. Workers and farmers face the risk of overexposure if they don’t wear protective equipment or use it irresponsibly. Research shows inconsistent selenium use in agriculture leaches it into water supplies, creating a possible hazard both for humans and for wildlife. Animals, especially fish, don’t handle selenium buildup well. Even small increases in their diets can harm reproductive health and impact the food chain.
Reducing the Risk
Checking supplement labels and watching for selenium in multivitamins makes a difference. Anyone thinking about using potassium selenate supplements should talk with a healthcare provider. Doctors can run a simple blood test to measure selenium status and spot any early signs of trouble. People already eating a balanced diet with Brazil nuts, seafood, eggs, and whole grains usually get enough without trying hard. For those working around fertilizers or in agriculture, wearing gloves, masks, and washing up after handling selenium compounds lowers exposure. Regulatory agencies keep an eye on agricultural use, but individual choices at home and on the job matter most day to day.
Personal Experience: The Slow Creep of Selenium Overdose
I’ve seen the fallout when someone chases a health trend without understanding the risks. Years back, a friend popped a mix of supplements with the right intentions but lost hair and energy, puzzled until a doctor flagged high selenium in routine labs. Cutting nonessential supplements made all the difference, but it’s a lesson worth sharing. Too much potassium selenate doesn’t make anyone bulletproof. Staying informed, asking questions, and favoring whole foods over pills kept my own health on track.
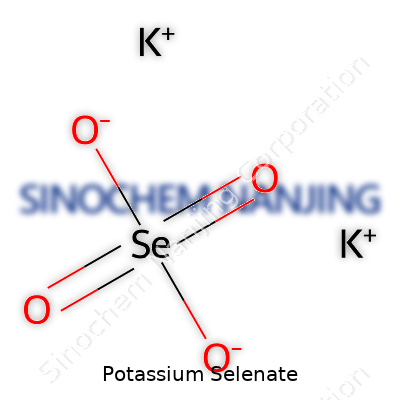
| Names | |
| Preferred IUPAC name | potassium selenate |
| Other names |
Dipotassium selenate Potassium selenate(VI) |
| Pronunciation | /pəˈtæsiəm ˈsɛlɪneɪt/ |
| Identifiers | |
| CAS Number | 10102-19-9 |
| Beilstein Reference | 1906088 |
| ChEBI | CHEBI:77637 |
| ChEMBL | CHEMBL1201596 |
| ChemSpider | 23821 |
| DrugBank | DB11092 |
| ECHA InfoCard | 03d7a6ef-c40d-4480-9f5c-84e1a67a79b2 |
| EC Number | 231-907-1 |
| Gmelin Reference | 8112 |
| KEGG | C18645 |
| MeSH | D011402 |
| PubChem CID | 24508 |
| RTECS number | VS8025000 |
| UNII | 8QQV152D4W |
| UN number | UN2630 |
| Properties | |
| Chemical formula | K2SeO4 |
| Molar mass | K2SeO4: 221.13 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 3.049 g/cm³ |
| Solubility in water | Soluble |
| log P | -4.09 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 2.0 |
| Basicity (pKb) | pKb ≈ 6.9 |
| Magnetic susceptibility (χ) | -57.0e-6 cm³/mol |
| Refractive index (nD) | 1.445 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 164.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1387.2 kJ/mol |
| Pharmacology | |
| ATC code | A12CE02 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; may cause damage to organs; environmental hazard. |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H332: Toxic if swallowed or if inhaled. |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P330, P391, P501 |
| NFPA 704 (fire diamond) | 2-0-0-OX |
| Explosive limits | Non-explosive |
| Lethal dose or concentration | LD₅₀ oral rat: 16 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 5 mg/kg |
| NIOSH | WN2950000 |
| PEL (Permissible) | 0.2 mg(Se)/m³ |
| REL (Recommended) | 0.02 mg Se/m³ |
| IDLH (Immediate danger) | 1 mg/m3 |
| Related compounds | |
| Related compounds |
Potassium sulfate Sodium selenate Potassium selenite |

