Potassium Persulfate: Progress, Promise, and the Push for Safer Chemistry
Historical Development
The roots of potassium persulfate stretch back to the late 1800s, a time when the chemical industry was hungry for strong oxidizers. As industrial chemistry branched beyond acids and bases, researchers chased compounds that could open new routes for oxidation. Potassium persulfate, with its punchy formula K2S2O8, drew attention for its ability to boost polymerization and etching technologies. Through the decades, its role expanded, tagging along with the boom in plastics, printed circuits, and water treatment technology. Anyone who’s ever worked in a lab with acrylics or watched a circuit board take shape has a practical stake in the legacy of this white, crystalline powder.
Product Overview
Potassium persulfate stands out in the crowd of oxidizers. In a world stacked with bleaching powder and hydrogen peroxide, this salt quietly amplifies etching and starts polymer reactions that turn runny liquids into tough, solid plastics. Folks in the textile industry still reach for it to make dyes take hold, while those in electronics rely on its ability to bite into metals—delicately enough to craft precise circuits. The food and pharmaceutical sectors use it with care, aware of its power to clean and purify. Because of this, demand rarely seems to stall, and its shelf presence reflects a reputation for reliability in both industry and research.
Physical & Chemical Properties
Potassium persulfate carries a simple appearance—a white, odorless powder—but its chemistry is anything but dull. With a molecular weight around 270 grams per mole and a composition that brims with available oxygen, it brings high reactivity to every table. It dissolves easily in water and boasts a strong oxidizing edge, which means it can kick-start reactions you wouldn’t dare attempt with gentler chemicals. While it won’t explode under typical storage conditions, exposure to heat, moisture, or organic materials can start a race of decomposition or release of gases. Airborne dust from careless handling can irritate skin and lungs, so most people treat it with as much respect as they do a power tool. The chemistry makes it uniquely suited for radical generation and redox applications, finding favor among practical chemists for its dependability.
Technical Specifications & Labeling
Technical data for potassium persulfate circles purity (often pegged above 99%), moisture content, and maximum allowable impurities like heavy metals. Labels aren’t just stickers—regulations demand clear signals, hazard statements, and first-aid basics. This direct communication helps minimize the kind of confusion that leads to chemical mishaps. Batch numbers and preparation methods often sit underneath the main chemical name, making life easier for traceability in large-scale labs or in the case of recalls. Good labeling guarantees each pouch or drum tells its own story, long before anyone thinks about opening it.
Preparation Method
Chemical plants rely on the electrolysis of potassium sulfate solutions to churn out potassium persulfate. Placing the solution between platinum electrodes, manufacturers coax out the peroxydisulfate ion through a precise electrochemical reaction. As with any industrial synthesis, small changes in current, pH, or temperature ripple down the line and affect both yield and quality. This method packs less environmental baggage than older, dirtier oxidation techniques, nodding to the growing demand for safe, predictable supplies. Smaller labs occasionally make their own, but most professional users value the quality controls that come with large batches.
Chemical Reactions & Modifications
Potassium persulfate launches into redox mode as soon as it enters water, paving the way for radical reactions. It splits into sulfate radicals, eager to tear electrons from nearby molecules and generate chains that lead to polymers or decompose organic pollutants. Adding different transition metals transforms its reactivity, giving chemists extra dials to fine-tune selectivity or speed. For those in synthesis, the ability to direct outcomes with catalysts or solvents has turned potassium persulfate into a versatile asset. As a student, building acrylic polymers seemed almost magical with this single ingredient as the engine behind chain initiation. Watching it in action impresses even the most jaded undergraduate.
Synonyms & Product Names
Chemists bounce around several names for potassium persulfate: potassium peroxydisulfate and potassium oxodisulfate crop up often, alongside various translations and trade names. The chemical shorthand KPS appears in research and product brochures, often shortened for quick reference in synthetic recipes. Academic researchers and industry veterans both know to cross-check synonyms to avoid misorders or confusion, especially when dealing with international suppliers. Global harmonization in labeling and safety data sheets continues to iron out past confusion, helping lab managers and procurement teams avoid costly mix-ups.
Safety & Operational Standards
Working with potassium persulfate means following strict ground rules. Industrial sites set up fume hoods, dust control, and spill kits to keep accidental exposure out of the news. The main threats come from its oxidizing punch: dust can hurt skin and eyes, and mixing carelessly with organics or combustibles risks swift reactions. Familiarity with the material safety data sheet and regular drill refreshers set the foundation for responsible work. Regulatory agencies keep updating best practices, asking companies and labs to prove competence through audits and staff training. In my own experience, a simple misstep—like ignoring a puff of dust—is enough to send folks scrambling for eyewash and medical help.
Application Area
Potassium persulfate thrives in multiple fields: plastics, water treatment, textiles, electronics, even photofinishing. Its real superpower is in launching free radical reactions—essential in building tough polymers and etching delicate patterns on electronic circuits. Textile companies use it for bleaching, where high-purity requirements push manufacturers to refine their processes constantly. Wastewater plants use it to break down pollutants that resist biological treatment, giving communities the peace of mind that comes when old chemicals vanish from the water supply. Hobbyists recognize it as a staple kit item for etching copper in home circuit projects, though big factories leave nothing to chance, building automated systems dedicated to safety and quality assurance. Some researchers push its limits further, tuning reactivity to hunt down traces of chemical contaminants in food or measuring oxidative stress in biological samples. In each of these areas, its value centers on reliability and controlled power.
Research & Development
R&D teams invest time searching for ways to squeeze even more utility or safety from potassium persulfate. Some projects look at alternative synthesis that lowers energy use or makes use of recyclable materials. Others hunt for new catalysts or solvents that notch up reaction speed or selectivity without raising costs. There’s a push for variants that produce fewer environmental byproducts, especially for applications in food or agriculture. Open-access publishing and global partnerships mean college labs and commercial giants both have a chance to join the conversation. My own foray into R&D taught me that tiny improvements—say, in how potassium persulfate stores or blends—can ripple out to reshape entire supply chains or open doors to new end-products. The fast-growing circles around green chemistry keep potassium persulfate in the spotlight as a subject ripe for both practical improvement and scientific discovery.
Toxicity Research
While potassium persulfate performs with controlled aggression in a beaker or reactor, its effects on health came under scrutiny as industry use expanded. Exposure can spark skin, eye, or respiratory irritation. Asthmatics face special risk from dust, prompting manufacturing plants and schools to crack down on airborne material. Animal studies set safe limits, but regulators keep gathering human data to drive guidelines that mirror both occupational risks and environmental exposure. Scientists probe long-term toxicity and ecosystem impact, especially with increased scrutiny of micro-contaminants making their way from industrial outflows into rivers and soil. Workers and the wider public call for full transparency, pushing companies to fund independent toxicity tests and invest in remediation where necessary. The days of hand-waving away concerns no longer pass muster, not only because of legal pressure but a real ethical responsibility to everyone who comes in contact with modern chemicals.
Future Prospects
Looking at the future, potassium persulfate stands at a curious intersection of legacy and innovation. As industries grow more conscious of environmental footprints, demand rises for oxidizers that can do the job cleanly or can be recovered and reused. Its strong, predictable chemistry leaves room for upgrading both green credentials and workplace safety. Research into nano-scale or hybrid applications hints at untapped potential, including medical diagnostics and emerging fields like environmental sensing. Some start-ups pair it with renewable energy sources for decentralized synthesis, appealing to lower-carbon supply chains. Widespread digitization allows real-time monitoring and smart dosing, shrinking waste while raising process safety. Potassium persulfate’s continued relevance reflects the ever-changing balance between what’s good for business and what’s healthy for people and the planet. New uses and tougher standards will likely keep this chemical both under the microscope and on the shop floor for years to come.
What are the main uses of Potassium Persulfate?
Essential Chemical, Everyday Impact
Potassium persulfate pops up in places where many people don’t expect it. In my years talking with folks in manufacturing, hair salons, even farmers, the same compound kept coming up. That white, crystalline powder—simple to look at—ends up doing a surprising amount of work behind the scenes.
Bleaching and Coloring: More Than Just a Bright Look
Anyone who’s worked in a salon knows potassium persulfate’s role in hair lightening. It acts as an oxidizing agent, breaking down natural pigments and prepping strands for bold colors. Consumers don’t always see this side, but it’s what helps stylists turn jet-black locks platinum blonde. The tech comes with some risk. Overuse can damage hair; salon pros use strict handling protocols because inhaling the dust or letting it linger on skin causes irritation. It pays to know exactly what you’re working with and why those gloves matter.
Printed Circuits and Electronics
Electronic tinkering has become more than just a hobby—some folks now build small runs of circuit boards in home workshops. Potassium persulfate works as an etchant in this world. It removes copper from printed circuit board blanks with precision, letting only the wanted traces remain. Large-scale manufacturers and garage inventors both benefit. People I’ve met who make custom electronics choose this chemical because it works fast and clean, compared to older ferric chloride methods. I always suggest good ventilation and careful storage, because its strength brings safety challenges.
A Touch in the Textile Industry
Textile work sometimes gets overlooked, but potassium persulfate plays a subtle yet essential part. In the past, I visited a dye house that used it as a bleaching agent to clean up wool and cotton before coloring. With fabric demands rising, consistency counts for everything—chemical oxidizers like this keep colors sharp and fabric blemishes down. Manufacturers pick it for speed and clarity, always balancing the need for high performance with tough labor safety rules.
Polymerization and More: Industrial Grit
If you’ve ever noticed the snap of a new acrylic nail or the stretch of modern rubber, there’s a strong chance potassium persulfate played a role. The chemical acts as a free-radical initiator in making plastics, resins, adhesives, and latex products. An old friend of mine in plastics manufacturing said they use it for kickstarting powerful reactions at steady temperatures, speeding up output without sacrificing product properties. Missteps in measurement or process control waste materials or worse, leave behind dangerous residues, so training on chemical handling never gets skipped.
Soil and Water Treatment
Environmental cleanup isn’t flashy. Plenty of remediation projects call for potassium persulfate to break down organic contaminants in soil and groundwater. I watched a cleanup crew in action as they treated polluted sites with it, often in combination with iron catalysts. That helped transform stubborn toxins into compounds easier to remove. Regulatory oversight remains strict here, and rightly so, since misuse brings its own risks. Qualified handling, environmental monitoring, and follow-up water testing are essential steps.
Looking Ahead
Every chemical comes with trade-offs: potassium persulfate’s value balances against its potential hazards. Regular hands-on safety training, better packaging, clearer labeling, and public education can fill knowledge gaps. Chemists, techs, hobbyists, even artists who use this material serve as reminders that science never really sits behind closed doors.
Is Potassium Persulfate hazardous or toxic?
What is Potassium Persulfate?
Potassium persulfate has found a permanent place on the shelves of hair salons, chemistry classrooms, textile plants, and industrial facilities. The white, crystalline powder acts as a powerful oxidizer, which means it helps kick off chemical reactions by breaking things apart — helpful for bleaching hair, treating water, even etching electronics. My first moments learning about this stuff came from chemistry class in high school, watching a teacher pour it into water and trigger a fizzy, unmistakable reaction.
Despite its usefulness, potassium persulfate comes with health and safety issues that easily get pushed aside for convenience. That is a mistake. Plenty of people use this chemical without realizing how risky it can be, especially once airborne powders are involved.
Does Exposure Lead to Health Problems?
The short answer: yes. Breathing in potassium persulfate causes nose and throat irritation, coughing, and—sometimes—serious allergic reactions. Pulmonary issues, such as asthma and chronic bronchitis, have been linked to repeated exposure, especially among hairdressers who regularly work with hair bleach containing this substance.
Some studies, including one from the British Journal of Dermatology, document increased asthma rates among people handling these powders at work. It’s not just respiratory: skin contact leads to rashes, itching, and blisters. Eye contact makes things even worse, causing redness or damage. In some cases, longer-term, repeated exposure can make people extra sensitive, so that even a little contact will trigger symptoms. For anyone who already deals with allergies or respiratory issues like asthma, using this chemical at work means taking extra risks.
Safety in Everyday Use
Learning how to handle potassium persulfate safely took trial and error on my part. Gloves, goggles, and good ventilation are non-negotiable. Even small amounts of airborne powder hang in the air for hours, so just mixing up a bleach solution in a closed room means trouble. The Occupational Safety and Health Administration (OSHA) has guidelines, but I’ve seen more than one business ignore them to save time. Hair salons especially cut corners because appointments stack up, with workers mixing bleach by hand.
Accidents involving potassium persulfate tend to be sudden. Water exposure can set off a reaction that causes burns. Dust finds its way onto surfaces and hands, traveling from the back-room sink to the lunch table without anyone noticing. Many cases of allergic skin reactions happen because people don’t realize the importance of hygiene after handling the chemical.
Why Regulations Matter
Government standards limit how much dust workers should breathe, but those standards mean little if ignored. OSHA and the National Institute for Occupational Safety and Health (NIOSH) both warn about the risks, placing focus on personal protective equipment and proper workplace ventilation. Enforcement often falls short, especially in small shops or locations outside big cities.
Safe alternatives for bleaching and water treatment are slowly gaining ground, but they haven’t replaced potassium persulfate yet. Until then, better education seems sensible. Employers need to provide training, not just a safety poster. Regular air checks, strong ventilation, and strict glove policies go a long way.
After seeing co-workers struggle with chemical-induced asthma, I decided that a few extra minutes spent cleaning and suiting up isn’t wasted. Potassium persulfate will keep showing up across industries, so staying informed and insisting on safe practices means longer, healthier careers for workers and fewer surprise visits to the doctor.
How should Potassium Persulfate be stored?
Why Storage Matters for Potassium Persulfate
Potassium persulfate often lands on the shelves of labs, hair salons, and even some art studios. Anyone who’s handled it knows how easily it can react with just about anything. The wrong storage can turn this dependable chemical into a serious hazard, turning a routine day into a scramble for the emergency kit. Potassium persulfate brings a strong oxidizing punch, raising the risk for fires or dangerous reactions, especially if it runs into the wrong chemicals or catches a bit of extra moisture. Problems rarely start with dramatic incidents; they begin with small lapses, like a box sitting on a damp shelf or a jar left just a bit too close to cleaning solvents.
Recognizing the Risks
Unopened containers can seem safe, but this stuff doesn’t tolerate carelessness. Moisture triggers slow decomposition, which releases oxygen. This means a bottle stored in a damp spot becomes a tiny time bomb over time. Unlabeled containers or casual handling can lead to mixing with organic material, such as paper towels or leftover cleaning rags. Fires that start this way don’t wait around. Lab workers and people handling industrial chemicals have learned this the hard way.
Incidents reported by the CDC and chemical safety agencies show most accidents happen due to storage mistakes. For instance, storing potassium persulfate near flammable materials or acids starts a chain reaction. In small businesses, staff sometimes stash containers under the sink, thinking it’s out of the way. These shortcuts add up to real danger. It only takes one spilled drink or leaky pipe to turn a routine storage area into a hazard zone.
My Experience: What Works and What Doesn’t
Years spent working in a community makerspace meant running into potassium persulfate more often than most. I saw issues crop up due to relaxed attitudes toward chemical storage. One year, condensation from a poorly insulated pipe dripped into an open tub. The distinct odor tipped us off just in time, but the near-miss stuck with me. Clear labels, secondary containment, and dry cabinets really make a difference.
Smart storage routines set the tone. Keeping potassium persulfate in tightly sealed, moisture-proof containers cuts down on the risk dramatically. Glass or plastic works, as long as it completely blocks out air and water. Dedicated shelves lined with chemical-resistant materials stay clean and dry. No cardboard or porous surfaces allowed, since these materials tend to pull in humidity from the air. Some workplaces overdo things by locking everything down, but most problems vanish when rules get followed day to day: lids go on tight, labels stay visible, and containers stay upright.
Best Practices Backed By Science
Industry guidelines, like those from OSHA, echo what many careful labs already do. They recommend cool, dry, well-ventilated locations far from direct sunlight or any heat sources. Experienced techs avoid stacking heavy items on top of persulfate containers. Remote storage rooms, with humidity sensors and regular inspections, lower the chance of surprise leaks or condensation. Secondary containment pans add another level of defense. Storing oxidizers like potassium persulfate far from fuel sources, acids, and organic materials helps prevent dangerous mix-ups. Routine housekeeping keeps surprise spills at bay.
If the work area doubles as a break room, chemical storage belongs elsewhere. Food and chemical containers easily get swapped, which leads straight to trouble. Used containers get rinsed and disposed of right away. Training everyone—down to the newest hire—keeps everyone safer.
Choosing Safety and Accountability
Storing potassium persulfate safely doesn’t require over-the-top measures. It does take discipline, consistency, and some respect for how quickly things can go wrong. Stories about close calls, supported by research and agency guidance, point to real-world benefits from these basic, practical steps. Investing in proper storage keeps people healthy, businesses running, and reputations intact.
What is the chemical formula of Potassium Persulfate?
Understanding the Basics
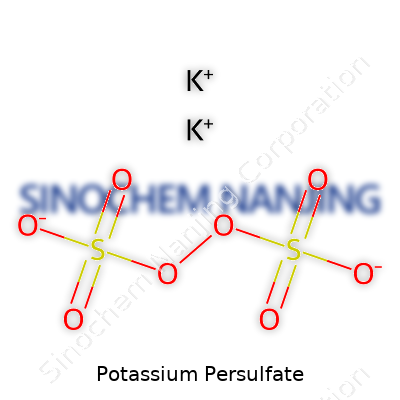
Potassium persulfate is a name I’ve come across plenty of times during both my college chemistry days and in real-world applications, especially related to cleaning and industry. Its chemical formula is K2S2O8. This equation isn’t random—it tells a story about the atoms making up each molecule. You’ve got two potassium atoms, two sulfur atoms, and eight oxygen atoms bonded together. That combination turns this compound into a strong oxidizing agent, meaning it’s great for sparking chemical reactions by grabbing onto electrons and helping things break down.
Where Potassium Persulfate Shows Up
This compound isn’t just for scientists in lab coats. It’s part of the toolkit in several areas you might not expect. In water treatment, potassium persulfate helps break down tough organic contaminants. Salons rely on it for hair bleaching because it ramps up the process, making lightening quicker and more reliable. Even printed circuit boards for electronics owe their intricate designs to the etching power of this persulfate. These uses put it right at the crossroads of public health, style, and technology advancement.
Why People Should Care
Most folks aren’t thinking about chemical reactions in their day-to-day lives, yet chemicals like potassium persulfate shape much of what’s around us. The water running from a tap, the hair color options at the drugstore, the reliable electronics at home—all rely on technical solutions that started in the chemistry classroom. Potassium persulfate’s action as an oxidizer makes all this possible. Industry counts on its reliability. The potential for it to cause skin irritation or respiratory problems means using the right protective measures matters, too.
Challenges and Solutions
It’s not only upside with this compound. Overexposure in industrial environments has led to allergic reactions and asthma, especially among workers in close contact with persulfate powders. During my time interning at a materials processing plant, safety goggles, gloves, and good ventilation weren’t negotiable—they mattered for getting through a shift without health issues. Training and awareness could be deeper in many workplaces. Manufacturers can improve packaging to cut down dust spread and use warning labels that get straight to the point about risks.
On the environmental front, heavy use in some settings means leftover chemicals could enter local water systems if not disposed of properly. That’s not a far-off concern—it’s a reality for regions with lots of manufacturing. Building clearer rules for disposal, following those rules, and developing greener alternatives for industrial uses would lessen the load on waterways.
Looking Forward
Teaching the next generation of chemists and technicians to balance chemical benefits with human safety and environmental health sets a better standard. Tools and technologies will keep evolving, but the basic knowledge about a formula like K₂S₂O₈ stands as a foundation. Practical steps—strong training, improved safety gear, responsible manufacturer practices—help everyone benefit from what chemistry has to offer without falling into the same health or pollution traps. Watching how we use even the most familiar chemicals pushes industries toward better habits and keeps both people and the planet in mind.
What safety precautions are needed when handling Potassium Persulfate?
Understanding the Risks
Potassium persulfate is no stranger in labs and industrial settings. It finds a spot in etching, polymerization, even some hair bleaching products. I remember working my first college chemistry project and reading up on it—the warning labels kept my attention more than any textbook paragraph ever did. The risks are clear: strong oxidizer, capable of causing burns to skin and eyes, can trigger fires if mixed with organic matter or combustibles, and its dust plays nasty tricks on your lungs if you don’t respect it.
Personal Protective Gear Is Non-Negotiable
I’ve learned the hard way that goggles should always fit snug and create a proper seal. Splashing potassium persulfate, even in small amounts, can burn right through comfort zones. Full-length gloves that cover sleeves block unexpected dribbles, and a lab coat will spare shirts, arms, and the regret of careless exposure. A sturdy, chemical-rated mask or respirator matters when you’re opening containers or weighing out the powder. Potassium persulfate throws off fine dust—skip the mask, and you’ll feel it scratch at your throat before long.
Engineering Controls Can't Be Skipped
The simple act of measuring chemicals brings exposure front and center. A fume hood draws vapors and dust away from faces and communal air. Any container holding potassium persulfate belongs in a cool, dry, and tightly closed space, away from even the hint of flame or spark. Storing acids or reducing agents close by can lead to runaway reactions if things go sideways. I always label containers clearly, so nobody grabs the wrong bottle after a long, late shift.
Spill Response Must Be Quick and Smart
I’ve found spill drills to be about as thrilling as fire drills—until the first time you see crystals scattered across a bench. Scooping up potassium persulfate with bare hands, or letting it hit moist surfaces, creates more problems. Grab a dustpan and broom for dry spills, and never use water unless advised by your site’s safety team. Ventilation is your best friend if dust fills the air. I keep an emergency eyewash and shower station mapped out in my head because every second counts in a crisis.
Health and Training Should Stay on the Radar
Some people, myself included, get skin rashes or worsened asthma from persulfate exposure. It’s not rare. If anyone feels irritated throats, stinging eyes, or trouble breathing, they ought to speak up quickly and head for fresh air. Regular training on chemical hazards, spill response, and first aid helps keep habits sharp and accidents down. I encourage asking questions if you’re not sure about PPE or procedure—nobody ever regretted being extra safe in my circles.
Thinking Ahead: Disposal And Environmental Impact
Leftover potassium persulfate does not just get dumped down the drain. It can tangle up wastewater systems, damage pipes, and react downstream. Designated chemical waste bins with proper labeling stop problems before they leave the lab. Environmental and lab regulations both set the tone for safe disposal, but I believe in going beyond the rulebooks—reputation and conscience matter more than paperwork.
Finding Real Solutions
Substitution works best. If a project or process means you can sidestep potassium persulfate for a safer option, it saves headaches. Digital resources, peer groups, and EHS (environment, health, safety) staff can point out less hazardous choices and safer handling routines. I’ve watched entire teams improve their culture around chemical use with clear protocols, reliable training, and the right safety tools on hand—turns out, good habits and respect for risk leave room for smoother work and fewer close calls.
| Names | |
| Preferred IUPAC name | Dipotassium peroxodisulfate |
| Other names |
Peroxydisulfuric acid potassium salt Potassium peroxydisulfate Dipotassium peroxodisulfate Potassium persulphate Potassium peroxodisulphate |
| Pronunciation | /pəˌtæsiəm pəˈsɜːlfeɪt/ |
| Identifiers | |
| CAS Number | 7727-21-1 |
| Beilstein Reference | 4031146 |
| ChEBI | CHEBI:48607 |
| ChEMBL | CHEMBL1083088 |
| ChemSpider | 14118 |
| DrugBank | DB11090 |
| ECHA InfoCard | 100.221.894 |
| EC Number | 231-781-8 |
| Gmelin Reference | 1157 |
| KEGG | C18683 |
| MeSH | D011188 |
| PubChem CID | 24412 |
| RTECS number | SE0350000 |
| UNII | G423D7V79L |
| UN number | '1492' |
| CompTox Dashboard (EPA) | CompTox Dashboard (EPA) of product 'Potassium Persulfate' is **DTXSID2020437** |
| Properties | |
| Chemical formula | K2S2O8 |
| Molar mass | 270.320 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.477 g/cm³ |
| Solubility in water | 7.4 g/100 mL (20 °C) |
| log P | -4.7 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 2.0 (H₂SO₄, related acid) |
| Basicity (pKb) | 12.0 |
| Magnetic susceptibility (χ) | +51.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.495 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 151.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1247 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1094 kJ/mol |
| Pharmacology | |
| ATC code | V03AB17 |
| Hazards | |
| Main hazards | Oxidizer, may intensify fire; harmful if swallowed, causes skin and serious eye irritation, may cause allergic skin reaction, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS03,GHS07 |
| Signal word | Warning |
| Hazard statements | H272, H302, H318, H335 |
| Precautionary statements | P210, P220, P221, P234, P264, P280, P301+P312, P302+P352, P305+P351+P338, P306+P360, P321, P330, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-0-3-OX |
| Autoignition temperature | > 250°C (482°F) |
| Lethal dose or concentration | LD50 (oral, rat): 802 mg/kg |
| LD50 (median dose) | Oral rat LD50: 802 mg/kg |
| NIOSH | NIOSH: SE0350000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Potassium Persulfate: "5 mg/m³ (as respirable dust, OSHA PEL) |
| REL (Recommended) | > 0.1 mg/m³ |
| IDLH (Immediate danger) | > 1,500 mg/m3 |
| Related compounds | |
| Related compounds |
Ammonium persulfate Sodium persulfate Potassium peroxymonosulfate Potassium sulfate |

