Potassium Periodate: A Deep Dive Beyond the Laboratory Bench
Historical Pull and Scientific Shift
Potassium periodate’s story weaves through more than a century of chemical curiosity and discovery. Chemists in Europe first noticed the peculiar oxidative properties of periodates back in the 1800s, drawn by their role in breaking down organic matter. The industrial age drove demand for reliable oxidizers, and potassium periodate earned its badge for precision and strength. In my own work in undergraduate labs, periodates always carried a sense of respect—one misstep around these crystals meant stained benches and sometimes ruined equipment. Despite this, the older generations of researchers often leaned on periodates for classic reactions, knowing that their clean, forceful oxidation could outmatch softer tricks found in many organic labs.
Looking at the Material—Up Close
Potassium periodate usually arrives as a milky white, crystalline powder—no real odor, yet a certain weight that tells you it means business. Handling a handful at the start of a shift, grains almost sparkle, reflecting the strict lattice of its crystal structure. The chemical formula, KIO4, reflects a strong oxidizer, with iodine in a high oxidation state. Melting point sits quite high, out of reach for any casual flame, and it dissolves in water but leaves traces in ethanol. These basic details become more than trivia in the lab—knowing the powder can drift into skin or lungs changes how people respect it. A strong oxidizer sitting near anything combustible practically throws warnings into the air, echoing every time someone in a lab coat shuffles by with a careless sleeve.
Technical Details and Labeling—More Than Just a Checklist
The labels on chemical drums hide stories behind brittle standards. Potassium periodate demands precise attention: purity above 99 percent, crystal size that can swing the outcome of a reaction, even the tightness of the seal and the warning symbols. In an age wary of chemical mishaps, regulators force explicit signage—oxidizer diamonds, skulls if warranted, and a firm reminder that this isn’t a backyard chemistry set. That level of detail speaks volumes, especially to newer students or factory crews who don’t get lulled into routine. I’ve watched colleagues almost skip reading a new batch’s spec sheet, nearly dumping a high-purity material into a reaction meant for a rougher grade. Mistakes like that cost money and safety.
Synthesizing—Not for the Untrained
Cook up potassium periodate and you face a process built on careful, clean reactions. Common practice involves oxidizing potassium iodate with chlorine or other powerful agents, under strict temperature control. Yields change with the patience and skill of the chemist. Get it wrong, and you at best get a sludgy, contaminated mess—at worst, you risk a runaway reaction. Stories float around of early academic labs clouded with choking gas after someone misjudged stoichiometry. Modern routines don’t forgive slackness; ventilation, steady stirring, and clean glassware set apart a professional prep from a dangerous gamble.
In the Reaction—Potassium Periodate at Work
In the hands of creative researchers, potassium periodate’s oxidative punch triggers transformations few other chemicals match. Slice through vicinal diols to yield crisp cleavage products, a move that enchanted organic chemists aiming to decipher complex structures. Tie this into the periodic table’s deeper wisdom: periodate’s large, reactive iodine center draws in electrons, making it perfect for unraveling stubborn bonds. In analytical labs, mixing periodate with colorless organics often generates visible, measurable color shifts, helping forensic teams trace contaminants or break down unknown substances. My time spent in these tests often felt like chemistry’s version of detective work.
What People Call It—Names that Travel
Ask around and you’ll hear potassium periodate called by synonyms: orthoperiodic acid, potassium salt; or sometimes just KIO4. Names multiply in trade and different countries, but each points to the same crystalline punch packed in every bottle.
On Safety—Where No Corners Tolerate Cutting
Few chemicals invite more caution than potassium periodate sprinkled on an open bench. As a seasoned chemist, I’ve watched hands tremble the first time someone realizes a stray crystal could set off a chain reaction, especially near flammable dust or paper. The rules command gloves, goggles, and a thick layer of wariness—nobody trusts a naked eye or bare hand with oxidizers of this pedigree. Regulations push for sealed secondary containment, grounded containers, and ventilation systems built with redundancy. I’ve seen more than one old-timer dismiss protocol, only to snipe an eyebrow when a new recruit brought out a spill kit: both sides recognize that good habits prevent disaster.
Where It’s Used—Industry to Academia
Potassium periodate stretches its reach far past ivy-covered walls. In analytical chemistry, it sorts sugars or spots trace metals with almost showy accuracy. Water treatment operators sometimes spike stubborn contaminants with periodate to trigger breakdowns no other agent could finish; the clear, sharp oxidizing power bleaches out toxins and opens pathways for safer molecules. In specialized manufacturing, smaller companies rely on periodate for surface treatments, cleaning metals, or prepping thin films for electronics. Academic research never runs out of uses—from illuminating reaction mechanisms in protein science to driving method development in forensic labs. Over time, the chemical shifted from an academic curiosity to a workhorse behind solutions that matter in public health, ecology, and technology.
Research Keeps Digging Deeper
Nobody truly leaves potassium periodate behind—not with current demands for better oxidizers and safer processes. Young researchers invest thousands of hours tracking new periodate-based catalysts, trying to lower the risks or improve waste handling. Newer environmental research targets smaller, more selective uses, looking to reduce the environmental toll and improve recoverability. Industrial teams focus on reusing periodate or breaking down reaction byproducts before they drift into the waste stream. The press for “greener” chemistry means potassium periodate’s future rests on clever engineering and inventive tweaks, not just brute force oxidation. Conferences now buzz with ideas about immobilized periodate or hybrid catalysts, and some labs even push toward applications in energy materials.
Understanding Toxicity—No Blind Spots Allowed
Toxicology data earned through hard lessons and animal studies underscore potassium periodate’s potential harm. Even a whiff of airborne dust, if enough gets into the lungs, sparks worry about irritation and longer-term lung damage. Ingestion risks iodine overload and burns, with the body rebelling against this level of exposure. I remember the urgency in training room voices spelling out spill cleanup procedures: never improvise, never underestimate the sting. Regulators enforce strict exposure limits, and disposal seldom gets left to junior staff—trained professionals handle every spent batch.
Where It Could Go Next
Innovators keep testing the boundaries, tying potassium periodate’s unique reactivity into sustainable chemistry and niche manufacturing. Ideas float about its use in precision nanomaterials, microelectronics, and even biomedical diagnostics where safety and performance mesh in tight tolerances. As demands grow for cleaner oxidants and more efficient syntheses, periodate’s crystal structure seems ready for a second act—Tinkerers pitch safer blends, stabilized powders, and even “closed loop” lab systems reducing waste. Environmental groups demand full life-cycle reviews, so new research tunes the molecule’s legacy for the next era—maybe greener, maybe smarter, but never forgotten.
What is Potassium Periodate used for?
A Closer Look at Potassium Periodate
Potassium periodate never shows up in everyday conversation, but anyone who’s spent time in a chemistry lab will recognize its name. It’s a white crystalline powder—really not much to look at—yet labs and industries put it to work in important ways. The thing that stands out about this compound lies in its oxidizing power. Chemists count on it to promote reactions that take other compounds to places they can’t reach alone.
Everyday Uses in Laboratories
Think about the number of tests out there that look for sugars, proteins, or specific metals—potassium periodate plays a role in more of these than most people realize. Analytical labs use it for oxidation in qualitative and quantitative testing. For instance, in old-school analytical chemistry, chemists use it to break down certain organic molecules to figure out what’s really present in a mixture. It acts almost like a traffic cop for electrons, guiding these tiny particles so that chemical changes can happen in a controlled way.
Potassium periodate also helps in prepping samples for detection of some metals—like manganese—and in biological research for cutting carbohydrates at certain points in a molecule. The selectivity in these reactions means researchers can dig into the specific structure or function of compounds they’re studying.
How Industry Depends on It
The reach of potassium periodate goes past the lab bench. It finds its place in the electronics world. Makers of printed circuit boards sometimes use it for etching copper patterns. Without this chemical’s help, building complex electronics would get even trickier. There’s also a spot for it in photography, though digital imagery has crowded out its use in the darkroom. For years, potassium periodate contributed to refining photographic chemicals and adjusting light sensitivity for better results.
Water treatment plants may tap into its oxidizing strength as well. By breaking down unwanted organic substances, potassium periodate can help produce cleaner water. Still, broader adoption comes with a big caveat: its cost and reactivity limit where it works best. Facilities aren’t going to pour this compound down the drain without a good reason.
Safety and Environmental Concerns
With every strong chemical, safety comes first. Potassium periodate doesn’t explode at a sneeze, but it brings risks—especially since it liberates oxygen and can accelerate fires if it contacts flammable materials. Anyone handling it should respect the lab’s rules: gloves, goggles, and keeping incompatible materials far apart.
Disposal raises questions too. Pouring strong oxidizers into the environment could mean trouble, including harm to aquatic life or water systems. Companies and universities track best practices: neutralize leftovers, collect waste properly, and never treat any unused chemical as trash.
Room for Better Practices
The chemistry world keeps evolving. Cleaner, safer, and less costly alternatives get attention with green chemistry gaining popularity. Some research groups explore other oxidizing agents that do similar work with fewer dangers. Meanwhile, for jobs only potassium periodate can handle, training and vigilance cut risks for workers and the planet.
Why Potassium Periodate Still Counts
There are hundreds of chemicals out there, yet only a few offer the combination of strength and specificity that potassium periodate provides. Its high performance in key reactions keeps it relevant in science and technology. Every time an old chemical like this remains useful, it’s a reminder that progress builds on lessons learned in dusty classrooms, busy labs, and tough factories. Using it wisely means paying attention to both the science and the consequences—something every chemist and industry professional learns, sometimes the hard way.
Is Potassium Periodate hazardous to handle?
Understanding Potassium Periodate in Daily Use
Potassium periodate shows up in science classrooms, some research labs, and even a few niche industrial spots. Never heard of it? Most folks outside chemistry circles haven't. But ask anyone who's worked with strong oxidizers and they’ll tell you this isn’t something to scoop out with bare hands.
Getting Familiar with the Risks
Staring at potassium periodate, you’d see a fine, white powder. It doesn’t look scary, doesn't smell odd. Still, its chemical properties spell trouble if you aren’t careful. Periodate compounds love to donate oxygen and trigger fast, sometimes dangerous, reactions. Accidental mixing with organic material—paper, clothing fibers, even a droplet of ethanol from a cleaning pad—can result in sparks, burns, or a sudden burst of heat.
The risks aren’t just about fire or explosions. Potassium periodate irritates skin, eyes, and lungs. A tiny bit in the eye can set off burning pain and lasting damage. Inhaling the dust might remind you of that time you caught a lungful of chalk—only much worse—leading to coughing fits and possibly longer-term trouble.
Learning from Experience
I worked in a university lab fresh out of college, eager to impress and shrugging off warnings about PPE. One semester, a classmate dropped a small scoop of periodate near a bench edge. We swept it up like sugar, until a TA spotted us and hit the panic button—literal evacuation, questions from the safety officer, new respect for those bold hazard diamonds. No one got hurt, but the lesson stuck: casual attitudes turn labs into accident magnets, especially with strong oxidizers around.
Why the Stakes Matter
Potassium periodate hasn’t popped up in major headlines, but reports surface in chemical safety bulletins and industrial incident logs. OSHA, NIOSH, and the CDC publish guidelines keeping people out of danger. Hazard statements use direct words: harmful if swallowed, causes severe skin burns and eye damage, may catch fire in contact with organic material. For anyone in charge of a workplace or school, lax training or out-of-date safety data sheets spell legal trouble and risk for everyone nearby.
Protecting Yourself and Others
Handling potassium periodate safely isn’t rocket science but skipping steps brings regret. Goggles, lab coats, gloves, and good ventilation matter. Fume hoods keep dust away from faces. Spilled powder goes into special waste bins, not regular trash—otherwise, trash fires or worse surprise you later. Proper labeling on all containers makes quick work of spotting a risk in an emergency.
Before using it, staff need regular training refreshers—not just a once-a-year safety slideshow. No one remembers every rule forever, especially in busy labs or classrooms juggling a dozen projects. Spreading awareness with hands-on demos and real-life case stories helps keep everyone sharp and ready.
Moving Forward: Solutions That Stick
Regular audits and surprise inspections find weak spots in storage or handling routines. Supervisors who walk the walk—using PPE and modeling good practice—set the tone. Emergency stations for eye and skin wash should sit close, not stashed behind stacks of old boxes. If the job allows for a safer substitute, switching away from potassium periodate makes sense.
People rely on trust in their teams and confidence in clear instructions. Mistakes happen, but clear communication, strong habits, and a little healthy caution keep everyone in one piece. Potassium periodate deserves respect; treating it like just another powder leads to preventable disasters.
What is the chemical formula of Potassium Periodate?
Looking at the Formula: KIO4
Potassium periodate, with the formula KIO4, doesn’t tend to grab headlines. Over the years in the lab, this compound stood out thanks to its strong oxidizing power. Potassium, iodine, and oxygen come together in a single, unmistakable molecule. Some might look at that collection of elements and move along, but it’s worth stopping for a second. That formula shows a direct bond to safety, education, and even industry.
Why Knowledge of KIO4 Shapes Decisions
I once worked on a project that involved testing water for certain contaminants. We used potassium periodate because its high oxidation state—a hallmark spelled out in the KIO4 formula—made detection easier and more reliable. Periodates like this one work at a level above other oxidizers, giving chemists a tool when others fall short. Ignoring the formula can mean missing out on crucial reactions, or worse, ending up with unexpected results.
Mistakes can happen in the lab any time someone grabs the wrong bottle. Strong oxidizers aren’t things to mix up. One year, a student mixed a periodate with something it shouldn’t have contacted. Fast thinking and good labeling—no thanks to any accidental uncertainty—kept the reaction under control. Clear knowledge of KIO4 prevents these issues. Its formula links directly to its properties: potassium for solubility, iodine at a high oxidation state for reactivity, four oxygen atoms shaping chemical possibilities that aren’t always obvious with less oxygen packed in.
Education and Outreach
Teaching high school and first-year college students about chemical formulas, simple as it might seem, builds a foundation. There’s a real difference between memorizing and understanding. Those four oxygen atoms in KIO4 don’t just sit there—they contribute to why potassium periodate is strong enough to oxidize manganese(II) ions to permanganate. In classrooms, showing the formula side by side with the reaction turns an intimidating equation into an understandable change. Those “aha” moments matter.
Plenty of folks ask why it isn’t just KIO3. Explaining the difference between periodate and iodate means walking students through the role of oxidation numbers, which pop up everywhere from analytical chemistry to environmental tests. KIO4 isn’t there just for show; it has everyday impact on what gets discovered, cleaned, and measured.
Application and Safety
Having worked with dozens of oxidizers, I’ve seen how correct formulas drive safety protocols. Potassium periodate reacts fast, sometimes even explosively, when paired with organic material. This isn’t theory—it’s hands-on chemical safety driven by the number of oxygens and the strength of that oxidizing iodine. Accurate identification through formula knowledge means better storage, smarter reactions, and more predictable outcomes. Every time someone double-checks the KIO4, hazards get cut down.
Plenty of modern sensors and color tests for metals and organics depend on periodates’ signature properties. Labs move forward on the back of compounds like this, so knowing what KIO4 actually means isn’t trivia; it’s foundation.
Paths Toward Safer and Smarter Chemistry
Some simple steps keep things running well. Clear labeling, tighter controls on who handles KIO4, and better training on what those oxygen atoms change in a reaction. Scientists who learn more than just the name—and truly grasp the chemical formula—carry forward safer habits and stronger experiments. In a world overflowing with new chemicals and combinations, getting the small details like KIO4 right goes a long way.
How should Potassium Periodate be stored?
Why Potassium Periodate Demands Respect
Every lab tech or chemistry student knows some materials rank high on the “handle with care” scale. Potassium periodate lands right in that group. With a keen knack for releasing oxygen and reacting fast with the wrong partners, this oxidizer commands attention the minute a new bottle enters the storeroom. Most accidents involving strong oxidizers happen because someone shrugged off the basics or packed chemicals too close together. No one wants an unexpected cleanup, so getting storage right saves headaches down the road.
Simple Rules: Storage Makes All the Difference
Start by finding a cool, dry shelf that skips out on direct sunlight. Heat turns potassium periodate from a tidy white powder into a conversation piece for all the wrong reasons. Humidity isn’t a friend either, since moisture speeds up unwanted reactions and shortens shelf life. My advice—skip storing anything in cellars or next to sinks where drips or condensation collect.
One experience stands out. At my old university lab, a careless placement next to a radiator taught us how fast the stuff degrades. The unmistakable salty smell and odd purple tinge were dead giveaways. That batch never even made it to the next experiment. The lesson spread fast: treat potassium periodate like an egg—one shake, drop, or steam blast, and the situation could get messy.
Stay Away from Trouble: Avoiding the Wrong Neighbors
Chemicals don’t like neighbors who get them riled up. Potassium periodate prefers distance from flammables, organic materials, acids, or anything sulfur-based. One old story from a high-school chemistry teacher showed why this matters. She stored periodate next to paper towels. The janitor’s mop bucket tipped over, soaked the stack, and sparks followed. Even something as harmless as a cleaning spill spells disaster if storage gets sloppy.
On a shelf, keep potassium periodate in sturdy containers with tight lids, made of glass or high-grade plastic. No rubber stoppers—they break down and let air seep in. Every container should show a clear label with storage dates. If you notice any crust, clumping, or color shift, call it time to dispose of the old stock. Plenty of labs hang onto chemicals too long, but saving a few bucks by using dated material doesn’t stack up against the risk.
Stepping Up: Staff, Signage, and Expecting the Unexpected
Short, clear instructions on storage go a long way. Print them, post them, talk about them. Have spill kits and basic personal protective gear within arm’s reach. Goggle up before poking around near the oxidizer shelf, and wear gloves that won’t dissolve if a bit of dust lands on them. I always kept baking soda close—it neutralizes acids, helping tamp down risk if the periodate ever meets the wrong liquid.
Double-check the area for liquids stored below or above. One slow drip spells disaster. Hydrostatic lockboxes or separate cabinets for strong oxidizers offer peace of mind. Fire marshals love custom cabinets with warning stickers and will point out poor storage during checks. Listen and adjust before anyone else needs to remind you.
Small Steps, Big Payoff
Taking these precautions gives you peace, keeps labs running, and stops accidents before they start. Storing potassium periodate isn’t about paranoia—it’s just good sense. My years in shared research spaces showed me that small lapses lead to big issues, and smart storage sidesteps trouble every time.
What is the purity or grade of Potassium Periodate available?
Understanding What’s On Offer
Potassium periodate, with its chemical identity KIO4, usually lands on the market in a couple of purity levels. Chemists see it billed as “reagent grade” or “analytical grade.” Sometimes a “puriss” or “ultra pure” option joins the menu. Most offer a minimum purity of 99%, and the difference between them boils down to trace contaminants more than the molecule itself. That decimal or two feels trivial to outsiders, but for anyone needing reliable results, it isn’t small beans.
Lab Work Demands Trust
I’ve spent enough hours at the bench to lose count of how often one rogue impurity has crashed a day’s work. Trace metals, leftover contaminants from the production line, or even moisture trapped during bottling—these ghost guests can ruin chemical reactions, skew instrument readings, or leave mysterious shadows on a chromatogram. Analytical grade potassium periodate wins trust for most research because it comes with a certificate outlining what’s buried in those last few decimal points. The document tells you the levels of sodium, chloride, or even heavy metals. Using chemical feedstock from a random supplier can give “surprises” that are hard to uncover until something goes wrong.
Why Sticking to Grade Pays Off
Potassium periodate gets used for oxidation reactions, mostly in organic chemistry. Some years ago, I learned this lesson the ugly way. We bought lower-cost industrial grade oxidizer to stretch a shrinking grant. Yields in our synthesis tanked for weeks—tough to pin down until we caught wind of barium sneaking in with the potassium. Swapping to a bottle with an honest assay certificate changed everything overnight. For schools, hobbyists, and research labs where cost pressures weigh heavy, the temptation to save a dime knocks at the door. Cutting corners on purity practically guarantees more headaches and false leads in delicate processes.
Production and Supply Chain Pressures
Global demand ties into the purity grades available. The big players in the chemicals trade—Sigma-Aldrich, Alfa Aesar and others—stick to tried-and-tested purification routines. Smaller dealers may promise identical grade labels but lack the same batch-to-batch traceability or robust testing. Laboratory staff notices the difference over time. Price isn’t always a reflection of purity, and regulatory paperwork sometimes clouds real quality. These wrinkles matter when you’re doing work where impurity profiles count: analytical chemistry, material development, or pharmacological applications. Those traces can cause false positives, batch failures, or regulatory headaches when scaling up production.
How Buyers Can Get Real Assurance
Not all certificates are created equal. Ask for the complete assay, not just a generic grade letter. Look for references to impurity limits—sodium, calcium, heavy metals—down to parts per million or better. Contacting the supplier for a full breakdown often reveals more than what’s printed on the datasheet. If the project is high stakes, third-party testing of a random sample can flag hidden risks before they turn up in critical experiments or production lines. In the end, documentation and open dialogue with suppliers pave the way for smarter buying—no one wins by “finding out later” that an unwanted trace ruined their results.
| Names | |
| Preferred IUPAC name | potassium periodate |
| Other names |
Potassium metaperiodate Potassium salt of periodic acid Periodic acid, potassium salt |
| Pronunciation | /pəˈtæsiəm pɪˈrɪəˌdeɪt/ |
| Identifiers | |
| CAS Number | 7790-21-8 |
| Beilstein Reference | 4039359 |
| ChEBI | CHEBI:48641 |
| ChEMBL | CHEMBL1200671 |
| ChemSpider | 21431 |
| DrugBank | DB16555 |
| ECHA InfoCard | 100.028.817 |
| EC Number | 231-715-2 |
| Gmelin Reference | Gm. 1498 |
| KEGG | C18636 |
| MeSH | D011099 |
| PubChem CID | 24858 |
| RTECS number | SD7520000 |
| UNII | 80KOU88O9D |
| UN number | UN1479 |
| Properties | |
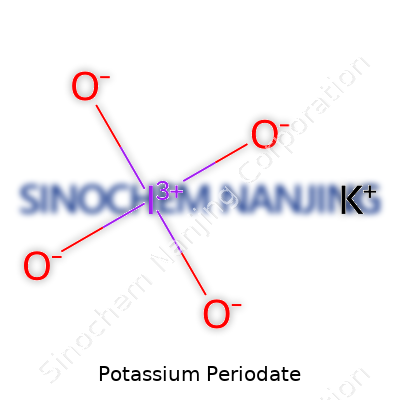
| Chemical formula | KIO4 |
| Molar mass | KIO4: 230.00 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 3.618 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | -4.0 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 10.6 |
| Basicity (pKb) | 11.4 |
| Magnetic susceptibility (χ) | -46.0e-6 cm³/mol |
| Refractive index (nD) | 1.826 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 214.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -492.6 kJ/mol |
| Pharmacology | |
| ATC code | V03AB15 |
| Hazards | |
| Main hazards | Oxidizer, may intensify fire; harmful if swallowed or inhaled; causes eye, skin, and respiratory irritation |
| GHS labelling | GHS05, GHS07, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H272, H319 |
| Precautionary statements | P210, P220, P221, P264, P280, P370+P378, P306+P360, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-0-3-OX |
| Lethal dose or concentration | LD50 oral rat 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 752 mg/kg |
| NIOSH | KWG181 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 mg/m³ |
| Related compounds | |
| Related compounds |
Potassium perchlorate Potassium iodate Sodium periodate |

