Potassium Perchlorate: Past, Present, and Possibilities
Potassium Perchlorate Through the Ages
Anyone with a foot in the world of chemistry hears about potassium perchlorate early on, usually in stories about fireworks lighting up the night. Its roots stretch way back to the mid-19th century, cropping up in the race to find stronger oxidizers for explosives and colored displays. Originally, chemists hunted for oxidizers that gave more punch to mixtures without blowing up unpredictably. Potassium perchlorate delivered, packing a bigger oxidizing wallop than earlier salts like potassium chlorate. The historical push from chlorates to perchlorates was more than just about spectacle—the move cut down on instability and risk of accidental blasts, a real concern in ammunition factories and mining operations. Chemistry and real-world needs shaped its adoption, transforming it from a laboratory curiosity into a staple for industries looking for both power and a measure of safety in controlled reactions.
Today’s Potassium Perchlorate: What It Is and What It Does
Potassium perchlorate looks like many inorganic salts—white, crystalline, and unassuming until the right conditions make it spring to life. In my own lab, the jar never draws a crowd until someone mixes it with fuel, then all bets are off. Chemically, it carries the KClO4 formula, leaving little room for ambiguity. While students sometimes expect a wild scent or dramatic appearance, this compound disappoints the senses—no smell, no colored tint, no visible drama on the shelf. Its utility lies in its attitude under heat: stubbornly stable until things get hot, then it hands over oxygen faster than most other common salts. This stubbornness makes it especially useful for things that need controlled, reliable burns, like flares or propellants. Burn tests and historical data back up its performance, with reliable oxygen yield numbers and steady reaction rates.
A Close Look at the Structure and Behavior
At room temperature, potassium perchlorate stands firm against water, only barely dissolving—just 2 grams per 100 milliliters at 20°C. Many first-time users notice it takes a lot of effort and time to get it to budge in water, which puts it at odds with its cousin potassium chlorate. This poor solubility can be a blessing: less risk of runaway reactions in damp environments. The crystal structure, a regular array of potassium and perchlorate ions, explains both its stability and the sharp melting point around 400°C. Once it finally breaks down, the reaction releases oxygen and leaves behind potassium chloride, a benign byproduct for most uses. Its reluctance to react without provocation grants it a shelf life that pyrotechnic suppliers can rely on, strengthening its appeal in applications where accidents are not tolerated.
Labeling Realities and Technical Details to Get Right
Labeling potassium perchlorate always brings up the issue of hazard warnings. Its hazard code, warning icon, and handling instructions all have to underline the oxidizing risk. Regulations in many countries require a yellow “OXIDIZER” diamond on the container, which cuts through language barriers and makes its danger clear. Users in schools or pyrotechnic workshops need to respect those markings, no matter how commonplace the material starts to feel after years of handling. From a technical standpoint, purity matters—a few tenths of a percent in contaminants can turn a useful chemical into a risky one. Pyrotechnic-grade perchlorate runs at 99% or above, a level reached by careful purification and checks. Experience shows that shortcuts at this stage always come back to haunt downstream users, especially in high-stakes aerospace or medical applications. Keeping an eye on these details avoids unexpected failures that can sideline an entire production run.
Crafting Potassium Perchlorate: Tried-and-True Pathways
Commercial processes lean on electrolytic oxidation, using potassium chloride as the starting point. Running electricity through a brine of KCl, usually at a large plant, oxidizes the chloride ions to perchlorate step by step. Old textbooks describe setting up simple cells, but scaling up introduces headaches like controlling side-reactions and removing each stage’s intermediate. Eventually, this yields a solution of potassium perchlorate, which gets filtered and crystallized. In the past, some small labs tried direct chemical oxidation, but the yields and purity always fell short. Industry experience has shown that only modern electrolysis can consistently deliver the amount and purity that modern users demand, especially with environmental controls getting tighter each year.
Reacting Under Pressure: What Potassium Perchlorate Does in Practice
Potassium perchlorate sits on shelves for months, but light a match and it comes alive. Mixed with suitable fuels—powdered metals, sugars, or organic binders—it drives rocket propellants skyward and colors fireworks with intense whites, reds, and purples. Chemistry students love watching it break down, releasing oxygen and leaving behind the much-less-reactive potassium chloride. In my teaching, I’ve seen students’ surprise at how much energy comes out with just a small teaspoon. Its reactions don’t end in fireworks or rockets. In the laboratory, potassium perchlorate plays a role as a standard oxidizing agent for titrations and specialty syntheses, testing other compounds’ reducing power. Modifications, such as making double salts or adding stabilizers, expand its use into more sensitive roles—tailoring burn rates for airbags or cutting medical isotope production risks by offering a reliable oxygen source.
Names and Nicknames: Sorting Through the Synonyms
Potassium perchlorate appears under a handful of names, depending on where you look. Chemists stick to KClO4, perchloric acid potassium salt, or the slightly awkward potassium salt of perchloric acid. Pyrotechnicians and fireworks factories shorten it to simply “perchlorate,” but this can lead to confusion with sodium or ammonium versions, which have very different safety profiles. In global trade and shipping, you’ll see the UN number 1489 and references to “Class 5.1 oxidizer.” Knowing all these variations helps avoid sourcing the wrong grade or type—an expensive and sometimes dangerous mistake, especially with international procurement.
Staying Safe Without Killing Curiosity
Safety with potassium perchlorate deals less with toxicity and more with fire risk. A single spark can turn even a small amount mixed with fuel into a violent reaction. Experienced hands respect that danger—I shake out my sleeves and double-check scales before handling open containers. Newcomers often miss the silent threats: dust in the workspace, cross-contamination with reducing agents, or storing it near organic materials like paper or sawdust. Wearing gloves, goggles, and lab coats is not just some bureaucratic checkbox—it’s learned from cautionary tales when things went wrong. Regulatory bodies set strict limits on how much can be kept outside approved magazines to keep neighborhoods safe. Training and respect for the compound keep the worst-case scenarios at bay. Accidental ingestion or inhalation rarely happens outside the lab, but the Material Safety Data Sheet warns of kidney stress at high exposures and possible thyroid effects due to its role in disrupting iodine uptake. This calls for proper ventilation and clear policies for spills or accidental contact.
Lighting the Way: Where Potassium Perchlorate Shines
Applications pour in from all angles. Fireworks and rocket propellants make up the bulk, where its stability and high oxygen content guarantee a solid punch. Military flares, signal cartridges, and special effects in film rely on its bright burn and reliable shelf life. Safety matches—something most people use without a second thought—often contain a touch of potassium perchlorate to kick-start the action with the right amount of heat. Sitting at the intersection between lab and production, it also appears in chemical laboratories for analytical chemistry, serving as a pillar for calibrating equipment or preparing calibration standards. Dermatology and pharmaceuticals have eyed perchlorate as an experimental agent for hyperthyroidism, though these uses now come with caveats and less frequency. Environmental research now pays extra attention to residues from fireworks and military training, driving scientists to rethink perchlorate use in situations where pollution threatens groundwater.
R&D: The Science Marches On
Research teams around the world still look for ways to make potassium perchlorate safer, cleaner, and less environmentally tricky. With groundwater contamination drawing headlines, more studies eye methods for rapid perchlorate degradation—whether through bioremediation, chemical reduction, or advanced filtration. Full-scale substitution work continues, though so far, few alternatives match the reliability for certain propellant or flare formulas. Material scientists experiment with coating technologies to minimize dust and improve burn control. In universities, green chemistry programs challenge students to invent new preparation pathways that reduce waste, energy use, and resource consumption. Some researchers zero in on perchlorate’s lingering effects in soils long after fireworks shows, designing bacteria cultures or catalysts that eat it up without adding risk. This push for responsible use and disposal carries over to new product designs, so future generations inherit less chemical baggage.
Keeping an Eye on Toxicity and Human Health
The biggest toxicity concern links back to the thyroid. The perchlorate ion, once in the body, blocks iodine from hitching a ride into the thyroid gland. Regulatory agencies, like the EPA, juggle questions about what exposure level tips from safe to harmful, especially for pregnant women and children, who need reliable thyroid signaling for brain development. Most folks face perchlorate as a trace contaminant from fireworks or defense exercises, not from daily contact. Health surveys in regions with high groundwater pollution look for subtle effects—shifting hormone balances, delayed development in kids, or possible ties to immune suppression. Industry groups fund studies that aim to clarify dose and response, sometimes critiqued for bias, but the research base keeps growing. In daily lab life, I notice most labs choose extra caution: testing runoff, sealing waste, and adding special waste-handling instructions even if the science shows low direct risk.
What Comes Next for Potassium Perchlorate
Potassium perchlorate stands at a crossroads between tradition and innovation. Green chemistry ideals push both researchers and manufacturers to reinvent old processes for lower waste and less environmental harm, but market pressures still demand a substance that delivers where it counts. The rocket, fireworks, and signal industries—so deeply woven with this compound—face pressure to clean up not just what they send skyward, but what comes back to earth in the form of residue and runoff. Regulatory agencies in Europe and North America consider stricter rules on transportation, storage, and use, especially in regions where groundwater flows close to the surface. In response, industrial chemists develop closed-loop systems to recycle or neutralize perchlorate-laced waste. New analytical tools promise rapid field detection, empowering neighborhoods and regulators to spot pollution before it gets out of hand. While full substitutes remain rare, R&D inches forward, inspired and sometimes embarrassed by both the brilliance of fireworks and the hidden cost of a brighter night sky.
What is potassium perchlorate used for?
What Makes Potassium Perchlorate Stand Out
Potassium perchlorate grabs attention because of the oxygen it can deliver during rapid reactions. People often link it to fireworks, but its reach stretches much further. That strikes me as interesting since most folks never realize how much of the world’s rhythm flows from basic chemical ingredients.
Fireworks and Pyrotechnics: Creating Color and Lift
You can’t have those vivid purple fireworks seen at summer festivals without potassium perchlorate. It teams up with metal salts and fuels to send colors blazing across the sky. Years ago, I helped set up a Fourth of July show and saw how the tiniest change in mixing this chemical could shift the entire hue and height of a firework’s burst. It’s not just about spectacle, though. The stability of potassium perchlorate compared to other oxidizers like potassium chlorate makes handling it safer for both workers and crowds. Risks remain, of course, which means people in this industry must train hard to respect the materials in their hands.
Matches, Safety Flares, and Everyday Items
Think of those strike-anywhere matches most folks keep in the kitchen drawer. Often, potassium perchlorate helps make sure they light up with one smooth motion. Emergency flares guiding boats at night or warning oncoming traffic on the highway also depend on it to burn steadily in all kinds of tough conditions. Some friends of mine in construction carry road flares every day, trusting that single ingredient to work without fail if disaster strikes after dark.
Medical Uses That Surprise People
Here’s a surprise: potassium perchlorate played a role in medical treatments, especially before safer methods took over. Doctors sometimes gave it to patients with overactive thyroids, aiming to dial down the gland’s uptake of iodine. That use faded after researchers tied high doses and long-term exposure to possible side effects, especially in people with thyroid troubles or young children. Still, learning how quickly doctors could adapt their tools always struck me as both impressive and a bit sobering. Science never stands still, but neither do the risks.
Role in Laboratories and Industry
Lab techs use this chemical during tough analytical tests or to spark reactions that need a strong blast of oxygen. In manufacturing, it finds a home in processes ranging from ceramics to explosives, where consistency matters more than flash or drama. Potassium perchlorate’s properties allow for more predictable results compared to other, riskier oxidizers, and that consistency fuels a lot of industrial trust and long-term use.
The Question of Safety and the Environment
Potassium perchlorate’s widespread use raises tough questions about health and the environment. Traces sometimes slip into soil or water, and scientists worry about how much may linger, especially near sites with heavy fireworks or explosives use. The chemical affects thyroid function, so regular checks and strict safety procedures become essential. In my own work cleaning out an old pyrotechnics shed, protective gear and careful waste disposal turned out to be non-negotiable if we wanted to keep water sources clean and people healthy.
Moving Forward With Caution
Alternatives pop up as researchers and companies look to sidestep the drawbacks. Some places favor greener pyrotechnics or switch to other oxidizers that break down more easily in nature. Strong oversight and constant training help, but everyone handling these substances—from the backyard fireworks fan to the industrial chemist—bears some responsibility for safety.
Potassium perchlorate proves how one chemical can merge excitement, daily utility, and serious health questions. What’s clear is that the story never stays the same for long. Good judgment, clarity about the risks, and a push for safer solutions offer the best way through the challenges that potassium perchlorate brings.
Is potassium perchlorate dangerous or explosive?
Looking Beyond the Science Lab
Potassium perchlorate isn't a household name, but it lives quietly in a few unexpected places—fireworks, matches, even some lab experiments. Folks who work in chemistry or pyrotechnics usually see it as a routine part of the job, but the rest of us rarely hear about it unless something goes wrong. The big questions that come up: Is it as risky as people say, and what kind of trouble can it actually cause?
The Real Risks: What Experience Shows
Anyone who's cracked open a chemistry set knows chemicals get their power from how they react, not just what label sits on the jar. Potassium perchlorate lands in that tricky spot between useful and dangerous. It releases oxygen when mixed with fuel and heat. On its own, it's not going to leap off a shelf and explode, but it’s not a material for carelessness either. Damage comes when people ignore the basics—heat, friction, or careless mixing with other ingredients. Stories circulate about amateur rocket builders or people experimenting without the right safety gear. Even a small mistake sometimes brings a burst or an unexpected fire.
Mishandling this white, seemingly harmless powder can cause explosions, especially if it finds itself mixed with sugar or sulfur. Mixes like that burn with intense energy, which is great in fireworks, but a problem almost everywhere else. That's why professional handling makes the difference. At work in controlled labs and licensed factories, strict rules keep bad surprises in check. My time in college labs taught me to respect peri chemicals like this. The room felt thick with tension whenever we worked with it, even among careful hands. Experience told us not to cut corners.
Environmental and Health Concerns
Potassium perchlorate carries its own health questions. Old-school munitions plants sometimes left the compound in the ground or water. Perchlorate ions disrupt thyroid function because they interfere with iodine uptake—a real concern if water ends up near livestock or folks with thyroid conditions. A study by the U.S. Environmental Protection Agency found traces in drinking water that called for a closer look. This isn’t a substance for casual dumping or thoughtless disposal. Treating it casually meant risking the health of entire communities. Clean-up takes time and effort, and, as usual, the responsibility lands on more than just the people who used it in the first place.
How to Stay Safe
Anyone dealing with potassium perchlorate needs training, good ventilation, and a clear plan for storage. There’s no shortcut to safety. People working in educational settings, or any facility using this chemical, need regular reminders about the basics: keep it dry, keep it cool, and keep it away from anything that burns easily. Routine checks and clear labeling help too. Accidents and long-term health problems often start with a small oversight—a missing label, a poorly sealed container, a moment of daydreaming while measuring out powder. Culture plays as big a role as science here.
Where Solutions Can Start
Safer handling starts with better education, not just for chemists and engineers, but also for school science programs and anyone in charge of supplies. Laws only work if people know why they exist. Enforcement helps, but real safety comes from culture—one where people speak up, share what they see, and refuse to treat short cuts as normal. Some industries have experimented with safer alternatives for specific uses, which helps lower the risk altogether. Until those swaps become standard, taking potassium perchlorate seriously remains the surest way to avoid the dangers it brings. With the history it has—and the potential it holds—the material deserves nothing less.
How should potassium perchlorate be stored?
Understanding the Substance
Potassium perchlorate has provided essential value in pyrotechnics, laboratory research, and industrial applications. It powers fireworks, provides oxygen in flares, fuels rocket igniters, and supports various chemical syntheses. The flip side shows up in its strong oxidizing character, which brings real hazards into the story. Old-timers in the lab remember accidents traced back to careless storage — containers left open, forgotten bags absorbing humidity, and, sometimes, desk drawers caked in white powder after a spill.
Humidity and Contamination: The Everyday Enemies
Moisture triggers headaches with potassium perchlorate. The chemical happily draws water from the air, clumping together and becoming tough to handle. Water doesn't just spoil the texture — over time it can nudge potassium perchlorate into forming other chemicals. Plastic tubs with tight lids or double-sealed containers built from polyethylene or polypropylene stand up well to this hazard.
Cross-contamination causes bigger risks than most people expect. Mixing even tiny bits of organic material or reducing agents with potassium perchlorate can turn a safe shelf into a future accident scene. Something as innocent as spilled sugar or stray iron filings creates explosive mixtures inside one careless tub. Labels marked with bright ink make a difference; it keeps everyone honest and prevents kitchen-style mixing mistakes in the storage room.
Containers and Shelving: Getting the Basics Right
Glass jars might look sturdy, but most professionals shy away from them. A dropped glass container gives you a nightmare of broken shards dusted with oxidizer, each piece dangerous to clean. Instead, sturdy plastic bottles, sold through chemical supply houses, cost little but protect a lot. Screw tops with gaskets block moisture, and labeling stands up over time. It's a practice learned from handling powdered metals; stack jars too high and risk them tipping onto the floor. Shelves work best at eye level, deep enough to prevent anything from falling off, far enough apart to stop bottles from bumping into each other. Sparks from static electricity deserve respect, especially during the dry winter months.
Temperature and Location
Heat plays a silent role with oxidizers. Potassium perchlorate remains stable at room temperature, but exposure to direct sunlight or radiators gradually pushes up risk. Shadows and cool air keep things safer. Chemical storage rooms lined with brick or concrete do a solid job, but ventilation counts more than most realize. Anyone who has smelled a pungent chemical closet knows that airflow matters — not just for comfort, but for safety. Positioning storage far from acids, fuels, and other reactants builds an extra wall against trouble. Some operations mark out dedicated cabinets, painted with warning colors, away from doorways and main work areas.
Work Culture and Training
The best storage plan changes nothing if people ignore it. A shared respect for chemicals grows when everyone from new interns to senior staff gets hands-on safety training. Regular checks — lids tight, labels clear, no unexplained powders chipping away at the shelf — catch problems before they grow. Emergency sprinklers rarely help with oxidizers, sometimes making things worse, so fire extinguishers rated for chemical hazards sit close by instead. If a container starts leaking, most teams treat it as an immediate cleanup job, not a chore to pass to the next shift.
What safety precautions are needed when handling potassium perchlorate?
Understanding What’s on Your Bench
Potassium perchlorate gets its reputation for a reason. If you’ve ever seen someone light a model rocket or watched fireworks color a night sky, there’s a good chance potassium perchlorate played a part. At its core, this chemical moves oxygen around for reactions that don’t wait for anybody. In my own college chemistry labs, even the hint of perchlorate meant gloves on, nervous glances all around, and someone retelling the tale of when a classmate’s careless scoop nearly earned everyone a spooky story for life.
The Hazards: Not Just Theory
Science textbooks make clear potassium perchlorate can be a fierce oxidizer. It speeds up combustion in ways that feel a lot more like a threat than a feature. The real-life risks extend beyond an accident in a lab. Poor storage or a slipup with contaminated tools can set off serious fires and explosions. This isn't just a scare tactic. Factories have lost entire wings, and home pyrotechnics have sent too many to the ER.
Inhaling dust or letting it touch the skin isn’t harmless, either. It can get into your system, disrupt the way your thyroid works, and tinker with how your heart feels a beat. As much as people talk about “responsible use,” sometimes it just takes a powder-stained bench and someone in a hurry for things to go wrong. That makes respect for safety more than a box-ticking exercise.
Everyday Safety Steps: More than Just Rules
Locking chemicals away in cool, dry cabinets with clear labels does more than organize a workspace. This step guards against heat buildup, moisture, and the unlucky mix with strays like sulfur or metal powders. Good storage also stops the curious or the untrained from stumbling into trouble. As a habit, treating every perchlorate container as if it could go off is worth building into muscle memory.
During handling, a well-fitting face mask keeps the dust clear of your lungs, and goggles keep surprise splashes at bay. Non-sparking tools — made from plastic or wood — end up as your best friends, since metal-on-metal can toss a spark at the worst moment. Even a forgotten metal file from a previous experiment has started fires that demanded fire extinguishers and quick feet.
If you spill potassium perchlorate, dry sweeping or using compressed air is a fast way to lose your workspace and maybe your eyebrows. Sweep with a damp cloth and treat every cleanup like you’re handling live ammunition. Toss contaminated rags and waste into airtight, labeled hazardous bins — not in the nearest trash can. Colleges and chemistry teachers I've worked with keep this policy taped over every disposal point.
Training and Real Accountability
Telling someone once about the dangers isn't enough. Repeated drills, real-life scenario training, and visible logs of who used what make a difference. In places I’ve worked, nobody handled perchlorates alone. There was always someone double-checking. That may sound strict, but it kept bad days from becoming headlines.
Toward Safer Communities and Workspaces
Companies and schools shouldn’t cut corners. Clear rules, proper equipment, and regular spot checks matter more than any written policy. For home experimenters, it means pausing before mixing unknown powders or sharing recipes online. The best advice: treat potassium perchlorate like a fast-moving snake — handle with intention, knowledge, and never without a plan for what happens if something slips.
Is potassium perchlorate available for purchase by the general public?
Why Do People Care About Potassium Perchlorate?
Potassium perchlorate sparks a lot of debate, partly because it’s known as a strong oxidizer that powers fireworks, model rocket engines, and even explosives. Some folks remember it from chemistry class or high school rocketry clubs. It’s also no secret that pyrotechnics enthusiasts often look for ways to get their hands on it. But as someone who grew up in a town where the Fourth of July meant more than just sparklers, I can tell you that backyard use comes with a lot of headaches for regulators and worried neighbors.
Who Can Actually Buy It?
Retailers don’t just have potassium perchlorate in stock. Stores like hardware shops and garden centers avoid it because of strict rules and safety concerns. Back in the 1990s, ordering chemicals online felt like stumbling into a Wild West trading post, but things have changed. Now, reputable chemical suppliers require proof of a legitimate use, such as educational or professional needs. Even if you find a website claiming to ship to anyone, shipping carriers and government agencies keep a close watch. Online marketplaces block these sales, and border patrol isn’t shy about seizing suspicious packages.
Why Is It Restricted?
Safety drives these sales limits. Potassium perchlorate doesn’t catch fire by itself, but mix it with the wrong materials, and it turns into serious trouble. The U.S. Department of Homeland Security knows that even a small stash could be misused. In 2007, the Chemical Facility Anti-Terrorism Standards put it among “chemicals of interest.” Some states ban sales outright, or demand extra paperwork even for educational labs. This helps keep it away from anyone hoping to make homemade explosives. After incidents like the Boston Marathon bombing, lawmakers doubled down on tracking and limiting anything with explosive potential.
Health and Environmental Risks
Potassium perchlorate isn’t just risky in terms of explosions. If it seeps into drinking water or soil, it can mess with thyroid function. The Centers for Disease Control and the Environmental Protection Agency have studied these effects after sites near rocket fuel plants showed contamination. Children and pregnant women face higher risks. That’s enough to explain the outcry in areas with contaminated groundwater, where parents want answers faster than regulators usually respond.
Is There a Middle Ground?
Any hobbyist who wants to experiment with chemical reactions or model rockets can run into frustration, since chemistry as a hobby isn’t the danger — carelessness and secrecy create problems. Plenty of schools and clubs work through proper channels, sourcing from vetted suppliers in small quantities and following storage rules. Some groups push for permitting systems, but costs and red tape can shut out both troublemakers and responsible hobbyists. Community science programs offer one way to keep experiments safe, public, and educational. More transparency from both buyers and sellers builds trust, and that’s worth pursuing.
Finding Solutions
Ensuring public safety, while nurturing scientific curiosity, means updating laws without killing genuine interest. Real oversight, not just blanket bans, offers a path forward. Training for educators and clear communication with regulators go a long way. If people understand that potassium perchlorate is more than just an ingredient — it’s something that can change lives for better or worse — they treat it with the care it deserves. Putting safety first remains the best way to answer any question about who should have access to it.
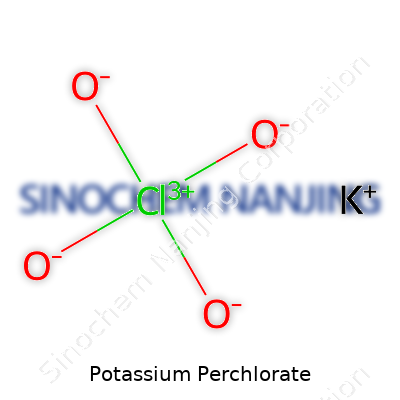
| Names | |
| Preferred IUPAC name | Potassium perchlorate |
| Other names |
Perchloric acid, potassium salt UN 1489 Potassium salt of perchloric acid KClO4 |
| Pronunciation | /pəˌtæsiəm pərˈklɔːreɪt/ |
| Identifiers | |
| CAS Number | 7778-74-7 |
| Beilstein Reference | 1207933 |
| ChEBI | CHEBI:83414 |
| ChEMBL | CHEMBL1087886 |
| ChemSpider | 12110 |
| DrugBank | DB11110 |
| ECHA InfoCard | 100.006.641 |
| EC Number | 231-912-9 |
| Gmelin Reference | Gmelin Reference: 2151 |
| KEGG | C18735 |
| MeSH | D011090 |
| PubChem CID | 24428 |
| RTECS number | SC4825000 |
| UNII | UJ8356C1VO |
| UN number | UN1489 |
| Properties | |
| Chemical formula | KClO4 |
| Molar mass | 138.55 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.52 g/cm³ |
| Solubility in water | 1.5 g/100 mL (25 °C) |
| log P | -7.3 |
| Vapor pressure | Negligible |
| Acidity (pKa) | -10.0 |
| Magnetic susceptibility (χ) | −32.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.338 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 149.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -431.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −295.4 kJ/mol |
| Pharmacology | |
| ATC code | V03AB17 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS01,GHS03 |
| Signal word | Danger |
| Hazard statements | H272, H319 |
| Precautionary statements | P210, P220, P221, P280, P370+P378, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 0, Instability: 3, Special: OX |
| Autoignition temperature | 400 °C |
| Lethal dose or concentration | LD50 oral rat 1870 mg/kg |
| LD50 (median dose) | 1,870 mg/kg (Rat, oral) |
| NIOSH | SN2875000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 50 mg/m³ |
| Related compounds | |
| Related compounds |
Potassium chlorate Potassium chlorite Potassium hypochlorite Sodium perchlorate Ammonium perchlorate Perchloric acid |

