Potassium Nitrite: A Broad-Spectrum Commentary
Historical Development
Potassium nitrite didn’t arrive overnight. Early observations of its natural formation date back centuries, often traced to the decomposition of organic material in certain types of soil. Alchemists once mistook it for saltpeter’s close cousin, puzzled by the subtle differences between nitrites and nitrates. Real progress came with the rise of chemistry in the eighteenth century, as scientists identified key differences by isolating potassium nitrite as a distinct substance, which led to its adoption in both the laboratory and the manufacturing floor. Historical records show its use in metallurgy and glassmaking, long before food technology begun applying it in preservation. This history shaped how industries handle nitrites today, tracing a line through generations of discovery, regulation, and scrutiny.
Product Overview
Potassium nitrite stands as an inorganic compound with the formula KNO₂. It appears as a white or slightly yellow crystalline powder—odorless and moderately hygroscopic. Industrial-grade potassium nitrite shows up graded in purity, with quality controls addressing specific contaminants to keep the risk profile in check, whether destined for industrial use or research labs. Chemists value its strong oxidizing power and reliable solubility in water, making it useful in chemical synthesis, textile dyeing, corrosion inhibition, and sometimes even as a food preservative in rigorously controlled settings. Each sector has its own story, but across the board, potassium nitrite plays a supporting role in processes demanding exacting standards.
Physical & Chemical Properties
Potassium nitrite melts at about 440°C and decomposes just above that temperature. It dissolves swiftly in water, creating mildly alkaline solutions. The compound offers high thermal stability as long as it’s kept away from strong acids and reducing agents, which can lead to unwanted chemical events. Young chemists and seasoned professionals alike have learned—often through trial and error—how its reactivity can go from dull to dangerous if mixed incorrectly. Storing it away from heat, sparks, or incompatible chemicals prevents unpleasant surprises and protects workers, equipment, and the broader environment.
Technical Specifications & Labeling
Any bottle of potassium nitrite—from lab reagent to wholesale drum—carries a detailed label spelling out concentration, impurities, lot number, manufacturer, hazard pictograms, and safety instructions. Regulations force manufacturers to lay all this out in the open, based on region and end use. Products for sensitive applications, such as pharmaceuticals or research, carry even tighter tolerances, with data sheets carefully audited for accuracy. I’ve learned to check not just the label but the supporting certificates—overlooking a minor detail in labeling or paperwork can cost more than just money if it means cross-contamination or misuse of a reactive chemical.
Preparation Method
Industrial production methods typically rely on reducing potassium nitrate with lead, copper, or other agents at elevated temperatures, although greener options are gaining ground, such as catalytic reduction in water-based systems. This process calls for careful monitoring to control both yield and purity of the finished product, since leftover traces of heavy metals or other byproducts pose health and environmental risks. Smaller-scale labs often prepare potassium nitrite by carefully controlling the partial reduction of nitrates—a balancing act involving precise heating, stirring, and purification. Each method echoes the tug-of-war between efficiency, cost, and safety, a challenge faced each day by those who produce, handle, and use it.
Chemical Reactions & Modifications
Potassium nitrite behaves as a powerful oxidizer and reducing agent in specific reactions. It takes part in diazotization, nitrosation, and formation of azo dyes. It reacts briskly with ammonium salts to yield nitrogen gas, while acids convert it quickly to nitrous acid—an unstable species notorious for breaking down and brewing toxic fumes such as nitrogen oxides. This ability to switch roles in redox chemistry gives potassium nitrite broad utility but demands tight control in manufacturing, as one wrong move can set off violent decompositions or release hazardous gases. Over the years, advances in process engineering and automation have trimmed these risks, yet every handling incident is a reminder that the chemistry’s potential for harm matches its promise for good.
Synonyms & Product Names
Ask for potassium nitrite in a hardware store and you may get blank stares, but ask a chemist—or check regulatory records—and you’ll spot references like “Nitrous acid, potassium salt” or “E249” in the context of food preservation. Some catalogs call it potassiumnitrit or potassii nitris, borrowing from old Latin nomenclature. Food industry paperwork still refers to it mostly by the E-number, an artifact from decades of regulatory negotiation. These alternate names reflect a tapestry of applications, languages, and histories, and I’ve learned that knowing them all helps avoid confusion in communication—especially across global supply chains.
Safety & Operational Standards
No one should overlook the risks tied to potassium nitrite. Strict handling measures include gloves, eye protection, tight ventilation, and emergency shower access. OSHA, EU-REACH, and other authorities list it as hazardous due to its toxicity and potential as an environmental contaminant. Workplaces implement exposure monitoring and compulsory training, based on both accident records and the chemical’s toxic reputation. In my experience, lapses in procedure often trace back to skipped steps or misplaced assumptions about “routine” tasks—yet incidents involving potassium nitrite have led to updated protocols and better enforcement. For specialized uses, such as injection in food preservation, only certified personnel armed with detailed training can handle and dose it, to prevent accidental overexposure.
Application Area
Industry leans on potassium nitrite for its oxidizing punch, using it to bleach fibers, develop dyes, and flash-finish some metals to achieve certain lusters or corrosion resistance. It shows up in heat transfer mixtures and specialty lubricants as well. Historically, food manufacturers depended on it for curing certain preserved meats, but rising evidence about health risks has pushed for a sharp drop in use within this sector—especially in places with tight regulation. Instead, its applications have shifted toward technical roles in industrial processes and analytical laboratories where alternatives just don’t work as well. Customers outside these niches often look elsewhere, as health and sustainability pressures reshape the landscape.
Research & Development
Research teams continue to hunt for safer nitrite alternatives for food and pharmaceuticals, but advanced applications in nanotechnology and catalysis see new life breathed into old chemistry. Green chemistry labs are retooling production methods to limit byproduct contamination and energy use. Regulatory updates—sometimes inspired by new toxicity data—force researchers to adjust product design and documentation to match evolving safety standards. Academics keep exploring the redox behavior of nitrites for energy storage, sensor design, and water treatment, betting that either new uses or cleaner processes will trump old criticisms. This ongoing work, driven by collaboration between academia and private industry, aims to maximize potassium nitrite’s utility without repeating past mistakes.
Toxicity Research
Modern studies highlight how potassium nitrite can interfere with hemoglobin, turning it into methemoglobin and choking off oxygen delivery in the blood—a risk recognized even among seasoned chemists. Low doses, especially through food, seem manageable under strict guidelines, but both acute and chronic exposure can trigger a cascade of health effects, from headache and nausea to severe respiratory distress or, rarely, death. Scientific attention has focused on occupational safety, residue analysis, and interaction with other food components, which can generate dangerous nitrosamines. Regulatory agencies have responded by tightening exposure limits. Looking back, these health scares and poisonings have prompted the chemically literate to tread carefully and regulators to rethink the wisdom of wide application outside narrowly defined circumstances.
Future Prospects
Regulatory constraints continue to tighten, shrinking potassium nitrite’s food-sector role but keeping it alive in technical and scientific fields that demand its properties. Ongoing efforts to develop “greener” alternatives may eventually dethrone it, yet its reliability in chemical syntheses and research labs suggests it will stick around in some form for years. Advanced manufacturing, clean energy storage, and specialty catalysis stand out as likely growth spots, provided developers square safety with performance. Lessons from history, including mishaps and misapplications, serve as a guide in shaping modern policy and technology. More robust monitoring, rapid toxicity screening, and digital inventory controls could drive safer and more responsible use in the next chapter of potassium nitrite’s long journey.
What is Potassium Nitrite used for?
What Happens with Potassium Nitrite?
Potassium nitrite has found plenty of practical uses in fields like chemistry and food processing. Many people wonder why this material keeps showing up in labs, factories, and sometimes even in products that land on dinner plates. It’s not a household name, but its impact runs deep.
Keeping Meat Safe and Appealing
Food producers often lean on potassium nitrite for curing meats like ham and bacon. It keeps meat from turning gray and helps prevent the growth of harmful bacteria, especially the one that causes botulism. Nobody wants to risk foodborne illness at the family table, and the reliability of this chemical plays a part in making processed meats last longer on store shelves.
Health experts stay interested in how nitrites react in the body. Too much of these preservatives can raise concerns, since nitrites may combine with amines in food under high heat and form nitrosamines. Some studies link these byproducts to certain cancers. Regulatory agencies keep close tabs and set limits on how much potassium nitrite producers can add to meat, which helps protect public health. Conversations around nitrites led to products with lower concentrations and the rise of alternatives like celery powder, which also produces nitrites naturally.
Lab Work and Industrial Chemistry
It’s not all about bacon and sausage. Potassium nitrite does serious duty in the lab. Chemists rely on it for organic synthesis and as an oxidizing agent. Used the right way, it helps create dyes, medicine, and photography chemistries that show up in everything from hospital labs to art studios. My own experience in a university chemistry lab came with strict training on how to handle chemicals—potassium nitrite included—because of its potential hazard when mixed with common substances.
Factories who use potassium nitrite don’t ignore the dangers. This compound reacts fast with reducing agents, which means untrained workers could risk fires or toxic gas leaks. The focus here needs to be on safety. Employers have set up strict safety protocols, ventilation, and storage guidelines. It’s clear that while chemistry can open doors, it’ll close them just as quickly if corners are cut.
Looking Forward: Finding Balance
Progress in technology and food science points towards safer, more transparent use of compounds like potassium nitrite. Food makers invest in new preservation techniques that look to keep food safe while lowering chemical intake. As someone who cares about honest labels and healthy lunches, I want to see clearer food labeling and better consumer education, not scare tactics. Giving people the information to choose lower-nitrite foods or nitrite-free meats empowers them without causing confusion in the aisle.
In industry, training continues to matter. Regular equipment checks, up-to-date safety gear, and detailed protocols mean accidents are less likely. Teaching the next generation of chemists to respect but not fear compounds like potassium nitrite prepares them to handle challenges, whether in a research lab or a food production facility.
Solutions Are Within Reach
Safer alternatives and improved safety standards shape the future of potassium nitrite’s use. Instead of trying to wipe it away, balancing its benefits and risks looks like the smarter move. Encouraging collaboration between regulators, industry leaders, and consumers helps maintain safety without sacrificing progress or tradition.
Is Potassium Nitrite safe to handle?
Safety Risks, Straightforward Facts
Potassium nitrite shows up in more places than you’d expect—labs, classrooms, industrial settings, sometimes even in food research. Bright yellow and water-soluble, this chemical doesn’t hide its presence. At first glance, a bottle of potassium nitrite looks harmless compared to some lab staples, but appearances don’t tell the real story.
Potassium nitrite doesn’t usually get top billing on the list of dangerous substances, but it has its share of risks. If someone isn't paying attention, it slips into the category of substances that disrupt health fast. Even small doses absorbed by the body interfere with how blood carries oxygen. This happens because nitrite ions convert hemoglobin into methemoglobin, which can’t shuttle oxygen where it needs to go. Higher doses bring on dizziness, headaches, and even convulsions. A curious child or an inexperienced lab worker can get into real trouble fast.
Industrial and Everyday Exposure
Nobody needs to panic just from standing near a container, but direct contact or inhalation takes the danger up a notch. Potassium nitrite can irritate skin and eyes, and accidental ingestion or inhalation in a poorly ventilated space can cause real harm. For many years, industries requiring potassium nitrite have stressed protective gear: gloves, goggles, and solid ventilation. Those aren’t just for show. The health risks follow a clear line from unsafe practices.
In the food sector, the bigger question hits closer to home. Some people still recall when nitrites popped up in processed meat products. Scientists flagged the potential for nitrosamine formation—a group of compounds connected to cancer risks. Recognizing this connection pushed governments and food safety experts to monitor how nitrite salts get used. Oversight and limiting intake, based on toxicological studies, makes sense. Potassium nitrite is not allowed in food in the same casual way as table salt.
Supporting Safe Practices
Reliable sources like the Centers for Disease Control and Prevention and the European Food Safety Authority offer clear guidelines for potassium nitrite exposure. They don’t leave the dangers open for debate: be vigilant, store away from acids and organic materials (because it reacts fast), and use personal protective equipment at all times. Forgetting about these rules or cutting corners almost always leads to a close call.
It helps to give workers straightforward training and keep material safety data sheets close at hand. This transparency lets people understand what they’re handling, instead of just going through the motions. Anyone who’s done chemical inventory in a crowded storage locker knows the value of strong labels and protocols you can trust.
Solutions for Safer Handling
Real safety comes with more than just rules on paper. Regular refresher training turns good advice into habits. Setting up spill kits and neutralizing agents nearby adds a final line of defense if something goes wrong. In research labs, tracking chemicals with up-to-date logs avoids forgotten containers and surprise exposures.
Looking out for each other makes a difference, too. Open communication, where workers compare notes on best practices or speak up when something feels off, lowers the risks. A teachable moment from a small mishap often sticks longer than any printed reminder. Potassium nitrite is less mysterious and less risky when everyone shares knowledge and respects the science behind its effects.
What are the storage requirements for Potassium Nitrite?
Understanding the Chemical
Potassium nitrite isn’t something to leave lying around the garage or ignore in a storage room. This compound, often a white or light yellow powder, acts as a strong oxidizer and shows toxic effects if people aren't careful. When it meets the wrong conditions, it reacts quickly and has the potential to release toxic gases. For anyone who handles chemicals—even in research or food industries—taking shortcuts on storage can lead straight to trouble.
Storage Conditions: What Works and What Doesn’t
Potassium nitrite reacts to moisture. Humidity in the air can turn a shelf-stable powder into a sticky, unusable mess, which can also eat through container linings. A dry, cool, and well-ventilated space is non-negotiable. Many laboratories and manufacturers keep this chemical locked up in climate-controlled rooms, avoiding basements, storerooms near wash stations, or areas exposed to open windows.
Direct sunlight and strong sources of heat also make poor neighbors for chemical storage. The powder should go into containers with tight lids, not just any plastic jar or paper bag. Glass, high-quality plastic, or specific metal canisters resist both corrosion and leaks. Checking container integrity after every use stops small problems before they add up. If the package looks compromised—maybe from a drop in temperature that caused condensation or a sudden spike in ambient heat—it’s smarter to replace it than to risk contamination spreading.
Segregation and Safety Measures
Mixing potassium nitrite with incompatible materials ranks among the biggest safety mistakes anyone can make in a workplace. Flammable substances, strong acids, and reducing agents should all stay far away. If stored nearby, even a single spill or leakage could trigger an explosive or poisonous reaction. In shared storage rooms, signage and clear labeling help keep everyone on the same page, especially in busy environments where several people access the area.
From personal experience in laboratory settings, storing reactive chemicals close to the eye wash station may sound convenient but makes accidents more likely. Chemicals like potassium nitrite find their safe space behind marked doors, preferably inside secondary containment bins. These bins catch leaks and stop dust from spreading. Some facilities even use locked cabinets with restricted access to limit who can handle or disturb these chemicals.
Training and Emergency Actions
Mistakes happen most often when people cut corners or skip steps out of routine. In workplaces, regular training refreshers keep both new and experienced staff alert. Even a small amount of powder spilled on a bench can pose a real threat, especially if someone sweeps it away or rinses it without a proper neutralizer. Clear instructions for managing small spills and exposure can reduce panic. Most places keep spill kits, gloves, and chemical-resistant aprons nearby for this reason.
Keeping a chemical inventory up to date helps spot expired or poor-quality materials before they cause bigger problems. Many organizations use digital logs to monitor quantities and expiration dates, making audits quicker and more accurate.
Avoiding Common Pitfalls
Secure storage isn’t just about following rules—it’s about protecting real people from unnecessary risk. In my experience, a culture of checking twice and replacing damaged gear beats wishful thinking every time. Simple, consistent habits—like labeling, inspecting, and never stacking incompatible containers—maintain safety for everyone in the building. Potassium nitrite has value in the right setting, but it asks for respect and attention at every stage from delivery to disposal.
How should Potassium Nitrite be disposed of?
The Real Risks Behind Potassium Nitrite
Potassium nitrite carries a reputation in the lab for a reason. This chemical brings both benefits and hazards. From my days working in an environmental testing lab, we treated even a pinch with respect. Direct exposure puts health and the local environment at risk. Unlike organic kitchen scraps or old batteries, just tossing potassium nitrite in the trash or down the drain can harm water sources. Nitrates and nitrites get into rivers, lakes, or soil and lead to fish kills, less oxygen for aquatic life, and dangerous changes in water chemistry over time. These problems don’t disappear on their own. Towns struggle to purify water affected by chemicals like this one, so avoiding carelessness matters.
Where the Buck Stops: Responsibility and Rules
State and federal guidelines demand responsible action. The EPA and local authorities keep a close eye on nitrite compounds because of the way they interact with both ecosystems and people. OSHA and DOT rules cover workplace safety and shipping. Both chemistry and common sense suggest treating even small leftover amounts with seriousness. A quick Internet search reveals real stories: one mishap at a college lab led to sick staff after improper disposal; another case involved contamination of a creek, setting off expensive cleanup efforts. Chemical safety officers evidence the consequences of ignoring disposal rules, so cutting corners poses a huge risk to both health and wallets.
Disposal Starts with Preparation
At work or at school, the first step always starts with a careful look at that container. Check the label, make sure it hasn’t mixed with anything else, and keep it far from heat, acids, or organic materials. I once saw a student mix waste streams by accident – the resulting fumes cleared half the building. Segregating potassium nitrite waste stands as a basic rule.
Options That Actually Work
Licensed hazardous waste disposal companies handle potassium nitrite safely by collecting it in sealed, labeled containers and transporting it under permits. They neutralize or stabilize the chemical before disposal, tracking every step. Some university labs have special programs or satellite collection stations that manage smaller amounts, keeping big shipments out of public roads. If something like this piles up at home—say, from an old chemistry set—calling a local household hazardous waste drop-off center offers a straightforward fix. Most cities run collection days for the public, keeping dangerous substances out of the regular trash system.
Reducing the Need for Disposal
Shaving off the source of the problem makes the whole process easier. Ordering only the amount needed, sharing excess with certified labs, and using proper storage methods all help to avoid buildup. Teachers and hobbyists stick with “green” classroom chemistry projects now, steering away from unnecessary risk. These efforts keep disposal headaches smaller for everyone.
The Bottom Line for Communities
Every time potassium nitrite gets disposed of safely, a community avoids a potential environmental mess. Clean water matters to everyone, whether you’re fishing, swimming, or just turning on the tap in your kitchen. Real solutions come from handling chemicals with clarity, using local resources, and following established rules. By staying alert and educated, anyone with a leftover bottle of potassium nitrite can do their part to keep harm at bay.
What precautions should be taken while using Potassium Nitrite?
Respect the Risks
Potassium nitrite is no ordinary white powder. Plenty of industries use it for chemical synthesis and laboratory work. Sounds helpful, but its risks should never blend into the background. Even though chemistry teachers and technicians handle it regularly, each fresh scoop carries real responsibility. Breathing small amounts can mess with your blood’s ability to carry oxygen. Swallowing a small amount poses bigger health dangers. Some people don’t realize how fast skin can absorb hazardous chemicals. That’s why gloves and protective clothing always go on before you open the jar.
Handling in the Lab or Workshop
The best labs operate on strict routines. Double-check the label before scooping anything, even if you’re in a hurry. Potassium nitrite looks a lot like salt or sugar under old fluorescent lights. Labels rubbed off or taped over make accidents more likely. Keep the container tightly sealed and reach for tools, not bare hands, to transfer it. You don’t want to introduce any extra moisture either—getting it wet can turn a minor spill into corrosive vapors, which nobody wants in their eyes or on their lungs.
Personal Protective Equipment: No Cutting Corners
Too many accidents start with skipped safety gear. Lab coats stop powder landing on your arms. Gloves keep absorption and allergic reactions at bay. Closed-toe shoes give a last defense against spills hitting your feet. Eye protection needs to rest snugly on your face, not on top of your head. Everyday glasses don’t cut it; you need goggles designed for splashes. I’ve seen folks turn red and dizzy just from breathing the wrong fumes for a few minutes.
Air, Clean Surfaces, and Waste
Chemicals like potassium nitrite demand good airflow. Fume hoods belong switched on and used properly. If all you’ve got is a fan or open window, you’re not set up right. Never snack, drink, or chew in spaces where chemicals live—it’s tempting, but not worth it. Any surfaces need regular cleaning, and spill kits should sit nearby, not locked away. Disposal isn’t just dumping leftovers in the trash. Local hazardous waste rules spell out safe disposal steps, and anything flushed down the drain can cause real harm to water supplies and wildlife.
Emergency Know-How
Memorizing emergency protocol saves lives. I’ve witnessed folks freeze when things go wrong, just because they never practiced a drill. Know the eyewash station’s location. Make sure the emergency shower actually works. Have the right spill kit for nitrites—those universal absorbent pads might not neutralize nitrite’s dangers. Store an SDS (Safety Data Sheet) nearby so you can grab the instructions in an instant.
Final Thoughts: Take the Small Stuff Seriously
Potassium nitrite looks innocent but behaves like a ticking clock if mishandled. Trust in routines and personal responsibility more than luck. Start every use by double-checking, and finish every work session with a careful cleanup. Small steps turn dangerous substances into useful tools instead of disaster stories.
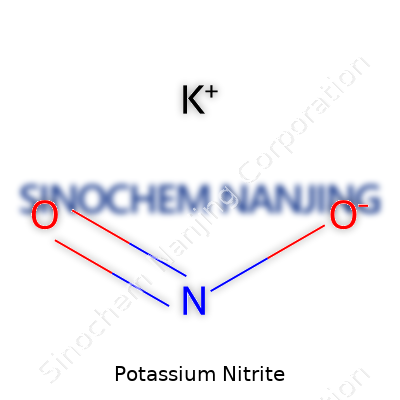
| Names | |
| Preferred IUPAC name | Potassium nitrite |
| Other names |
Nitrous acid, potassium salt Nitrite of potash Potassium salt nitrous acid |
| Pronunciation | /pəˈtæsiəm ˈnaɪtraɪt/ |
| Identifiers | |
| CAS Number | 7758-09-0 |
| Beilstein Reference | 3539826 |
| ChEBI | CHEBI:77658 |
| ChEMBL | CHEMBL1200972 |
| ChemSpider | 10212 |
| DrugBank | DB01427 |
| ECHA InfoCard | 100.029.807 |
| EC Number | 231-555-9 |
| Gmelin Reference | 653 |
| KEGG | C14355 |
| MeSH | D011074 |
| PubChem CID | 238681 |
| RTECS number | QR6300000 |
| UNII | 7Z403HQ5ZB |
| UN number | UN1488 |
| Properties | |
| Chemical formula | KNO2 |
| Molar mass | 85.103 g/mol |
| Appearance | white to slightly yellow crystalline solid |
| Odor | odorless |
| Density | 1.915 g/cm³ |
| Solubility in water | 81.3 g/100 mL (20 °C) |
| log P | -3.4 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 3.4 |
| Basicity (pKb) | 14.5 |
| Magnetic susceptibility (χ) | Paramagnetic |
| Refractive index (nD) | 1.997 |
| Dipole moment | 7.12 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 114.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -363.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -391 kJ/mol |
| Pharmacology | |
| ATC code | V03AB10 |
| Hazards | |
| GHS labelling | **GHS02, GHS03, GHS06** |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H272, H301, H319, H335 |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P304+P340, P308+P313, P312, P330, P405, P501 |
| NFPA 704 (fire diamond) | 3 0 2 OX |
| Autoignition temperature | 480 °C (896 °F; 753 K) |
| Lethal dose or concentration | LD50 oral rat: 180 mg/kg |
| LD50 (median dose) | 85 mg/kg (rat, oral) |
| NIOSH | WS3650000 |
| PEL (Permissible) | 0.2 mg/m³ |
| REL (Recommended) | 0.2 mg/m3 |
| IDLH (Immediate danger) | IDLH: 75 mg/m3 |
| Related compounds | |
| Related compounds |
Sodium nitrite Potassium nitrate Potassium chlorate |

