Potassium Nitrate: A Grounded Exploration
Historical Development
People have worked with potassium nitrate for centuries, long before chemical purity meant much in laboratories. In the Middle Ages, this salt sparked a drastic turn in how humanity waged war, lighting up the world of gunpowder. Simple nitraries—open-air pits collecting bird droppings and decaying matter—offered early sources before industrial thinkers began refining processes. Its reach soon spread beyond battlefields, fueling not just weaponry, but also the curiosity of agriculturalists, glassmakers, and even the folk who first understood mineral fertilizers. Today, potassium nitrate production draws from deep reserves and chemical plants, but its core value traces back to the alchemists and pioneers who shaped both practical use and chemical understanding out of raw, natural beds of saltpeter.
Product Overview
Potassium nitrate, known to many as saltpeter, stands out for its clear appearance and high solubility. The crystals don't carry much weight on their own, but when mixed or melted, they unlock energy for fertilizers, explosives, and heat transfer applications. It's one of the few salts serving so many distinct industries, from garden centers offering it in fertilizer blends to laboratories evaluating its oxidizing power. Over time, commercial potassium nitrate has taken several forms: prills, crystals, flakes—each one meant for its own set of uses. What often matters most is not how it looks on the shelf, but how readily it dissolves and releases its nitrogen and potassium to whatever task is at hand.
Physical & Chemical Properties
At room temperature, potassium nitrate appears as a white, crystalline compound. It melts at about 334°C, releasing no dangerous fumes until heated much higher, though it will decompose with sufficient heat or in the presence of strong reducing agents. Water pulls it apart eagerly, giving rise to high solubility that gardeners and chemists appreciate alike. Chemically, it brings together potassium and nitrate ions in a 1:1 relationship, with a formula of KNO3. Its role as a strong oxidizer sets it apart from less reactive salts—giving up oxygen atoms in chemical reactions and, by doing so, speeding burning or enabling explosive performance in pyrotechnics or propellants. In storage, it resists clumping and keeps potency if sealed dry, but humidity will pull it from air, making lumps unless handled with care.
Technical Specifications & Labeling
Most industrial buyers require at least 99% purity in potassium nitrate—agricultural blends may relax that standard to allow traces of sodium, calcium, or magnesium. Product labels give a full rundown of content, size of particles, and packaging method. Regulatory compliance remains key, especially since nitrate compounds may fall under explosives regulations, and buyers need certification for both quality and handling. Data sheets lay out each lot’s batch number, test results, country of origin, and recommended safety instructions. Even as a teenager reading over bags in the garage, I saw the rows of technical detail that come by law and by best practice. These numbers matter in every context from farm supply stores to chemical labs.
Preparation Method
Manufacturers tend to make potassium nitrate either by reacting potassium chloride with sodium nitrate in solution, followed by cooling and crystallization, or by converting potassium carbonate with ammonium nitrate. The final product gets washed, recrystallized, and dried. In the past, natural deposits and leaching processes met most needs, but chemical processing now runs the show, allowing tighter controls on purity and particle size. Through every stage—dissolving, mixing, cooling, separating—workers must manage risk of contamination because even small shifts in composition can change reactivity and suitability for use. Some batch facilities still draw on traditional methods, but global demand requires plants to scale up, monitor waste streams, and check byproducts so environmental releases stay below regulatory limits.
Chemical Reactions & Modifications
The oxidizing strength of potassium nitrate pops up in many reactions: it turns fuel into flames, helps split organic matter cleanly, and breaks down in the right setup to form potassium nitrite. Strong acids knock out nitric acid from it, while reducing agents trigger shifts that release nitrogen gases—sometimes dangerously so. Pyrotechnic crafters blend it with sulfur and charcoal for black powder, counting on its speed and thoroughness as an oxygen donor. Researchers sometimes adjust its properties by doping with other ions or changing physical structure—say, altering particle size with spray-drying—allowing fine-tuned application in rocket propulsion or slow-release fertilizers. Yet no modification truly changes the essential reality: potassium nitrate brings oxygen to reactions efficiently, swiftly, and with a predictability that engineers bank on through the years.
Synonyms & Product Names
Across regions and over time, potassium nitrate picked up a handful of names: saltpeter or Indian saltpeter being the best known. Scientific circles stick to KNO3 or potassium nitrate, but trade names abound in commercial catalogs and fertilizer warehouses. Some suppliers brand it as Niter or Chile saltpeter (though the latter sometimes also speaks to sodium nitrate, so confusion crops up). In older texts, the term “nitrum” may turn up, though that word ranged widely in historical alchemy. Wading through labels demands attention, as mismatched synonyms lead sometimes to costly mistakes, especially in cross-border procurement or when ordering in bulk.
Safety & Operational Standards
Potassium nitrate needs careful handling, not only because of its oxidizing nature but also due to its regulatory baggage. In the workplace, ordinary gloves and dust masks typically handle accidental exposure, but fire risk rises if the salt mixes with fuel or other combustibles. Standard practice keeps potassium nitrate separated from acids, oils, and reducing agents, locked in dry storage away from heat. Safety data sheets warn of eye and skin irritation, rare but serious allergic reactions, and the need to seek medical attention if large quantities get ingested or dust is inhaled. Strict documentation follows every shipment, especially in regions sensitive to explosive precursor controls, and training keeps workers aware of incompatibilities.
Application Area
No other single salt touches as many corners of life as potassium nitrate, especially for those of us who garden, shoot, or build. Farmers count on it as a fast source of both potassium and nitrogen, feeding field and orchard. Industrial operators feed it into heat transfer systems—its high melting point and heat-carrying capacity find use in solar thermal power plants. In the world of food, it’s a curing agent for meat, holding off bacteria like Clostridium botulinum in cured hams and sausages, though food safety standards now favor rigorous monitoring to keep dietary intake below risk thresholds. Chemists turn to its reactivity in teaching labs, propellant makers count on its dryness and purity, and pyrotechnicians trust it to give color and shape to fireworks. Even modern pharmaceuticals glance toward potassium nitrate in certain dental applications, using its moderate solubility for slow-release effects in sensitive toothpaste.
Research & Development
Researchers continue chasing ways to improve potassium nitrate’s usability and environmental footprint, from turning crop waste into sustainable production streams to tweaking crystal morphology for better absorption. Some labs look at combining it with slow-release coatings to deliver nutrients evenly through a growing season. Recent university research digs into blending nitrate salts for custom fertilizer blends, balancing yield with reduced runoff risk. Substance tracking gets smarter, too, as companies roll out digital systems to watch every sack from factory to field. One promising path involves capturing atmospheric nitrogen in new processes, reducing reliance on mining and classical chemical synthesis. Technologists test new thermal storage setups, pushing potassium nitrate to help balance grid power by holding solar heat for night release—a challenge bred from modern energy demand rather than an old alchemist's dream.
Toxicity Research
Potassium nitrate draws attention in toxicology because, beyond moderate nutritional applications, excess intake alters blood chemistry. Nitrates reduce to nitrites, then interact with hemoglobin to form methemoglobin, which can’t carry oxygen—a risk mostly to infants, certain patients, or livestock drinking from contaminated wells. High doses can also trigger mild gastrointestinal upset and headaches; workers exposed regularly must watch aggregate exposure closely. Governments put strict limits on nitrate residues in foods, particularly cured meats, and labs routinely test water—especially near run-off from fields or manufacturing sites. Animal studies hint at effects from long-term exposure, pushing regulators to call not just for dose limits but for strict labeling and periodic monitoring along the whole supply chain. Good hygiene, responsible dosing, and clear documentation curb most risk, but public health requires vigilance.
Future Prospects
Demand for potassium nitrate will keep shifting as people develop smarter crops, cleaner fuels, and safer energy storage. Efforts to cut agricultural runoff point toward new formulations that reduce leaching and match plant uptake. Synthetic and biological methods for nitrate production keep getting greener, and pressure mounts to reclaim the salt from waste streams—closing the loop between farm, factory, and natural water systems. Technology companies search for materials to hold more energy at higher reliability, and potassium nitrate looks ready for expanded roles in grid storage and high-temperature batteries. Food scientists dig deeper for alternatives but rarely stray far, since few substitutes bring such clean antimicrobial action with modest taste impact. Potassium nitrate reminds us that a well-known chemical can still play a role in the future, if its risks, roles, and chemistry stay open to innovation and careful management.
What are the main uses of Potassium Nitrate?
Feeding the World’s Fields
Growing healthy crops relies on more than rain and sunshine. Potassium nitrate stands out as a reliable fertilizer, delivering nitrogen and potassium in a form that plants soak up right away. These two nutrients make stronger stems and brighter leaves possible, especially in areas where soil naturally lacks them. Farmers show a clear preference for blends that prevent salt build-up and let plants take up nutrients at their own pace. I’ve seen how, in drought years, balanced fertilizer can mean the difference between a paid mortgage and a lost season.
Fireworks and a Burst of Color
Fourth of July crowds don't often stop to think about chemistry during the fireworks shows. Behind those bursts of red and green, you’ll find potassium nitrate hard at work as an oxidizer. It kicks off the chemical reactions that shape each color and sound, letting pyrotechnic artists set the night sky on fire. Sometimes, the same chemical finds a place in smoke bombs or safety flares. As a young kid on camping trips, I marveled at road flares giving stranded vehicles a fighting chance in the dark — potassium nitrate drives that reliable, bright burn.
Preserving Food the Old Way
Before refrigerators filled every kitchen, people turned to various ways to keep food safe. Potassium nitrate, known as saltpeter in these circles, brought a steady edge to meat preservation. It didn’t just keep bacteria away. It helped create the deep color of cured ham and sausages. In rural communities where electricity comes and goes, folks still value these old methods. Using nitrates in moderation and staying up-to-date on new food safety research keeps this tradition both alive and safe.
Making Gunpowder and Blasting Rock
Potassium nitrate once tipped the balance in battles centuries ago. It makes up a big part of traditional black powder used in firearms, cannons, and blasting for mines. That same chemistry powers some model rocketry and educational kits today. Scientific work continues to look at how to keep these uses as safe as possible and out of the wrong hands. Real-world handling means following regulations and learning from tragic mistakes. The importance of careful storage and secure transportation can’t be overstated.
Everyday Products You Don’t Notice
People spot potassium nitrate in things like toothpaste for sensitive teeth, where its calming effect makes hot and cold foods less painful. Dentists often steer patients toward these products because potassium nitrate works directly on nerves inside the tooth. Though regulations strictly manage the amount used, most people benefit from the relief it brings — especially in communities with limited access to advanced dental care.
Moving Toward Safer and Smarter Use
Potassium nitrate’s reach spans from agriculture all the way to fireworks. With any useful tool, harm can follow if people ignore how improperly handled chemicals affect health and safety. Education stands front and center. Farmers, hobbyists, and families need trustworthy information on safe storage and responsible use, not scare tactics. Environment-friendly policies and good labeling also play their part in keeping people informed. Meeting food needs, lighting the sky, and protecting health get a bit easier with chemicals like potassium nitrate put to wise work.
Is Potassium Nitrate safe to handle and store?
What Potassium Nitrate Is Used For
Many people know potassium nitrate as a component in fertilizer, sometimes fireworks, or even old-school curing salts for meats. It’s a compound with decades of agricultural and industrial history, not just a chemical tucked away in a lab cabinet.
Handling potassium nitrate isn’t quite like dealing with table salt or sugar. Once I spent a summer working in a small rural supply store. Bags of this stuff arrived for farmers tending to their crops, and every shipment came with clear warnings. It wasn’t just legalese—years of accidents had shaped those safety standards. At home, some folks might remember stories about gunpowder and even home experiments gone wrong.
Understanding the Real Risks
Potassium nitrate catches attention because of its oxidizing nature. Unlike many other white powders, if exposed to heat or mixed with certain fuels, it can speed up fires. Fires in storage areas have caused serious damage, so authorities started to require regular checks. In the United States, the Occupational Safety and Health Administration classifies it as a hazardous substance.
Health wise, it irritates skin and, more seriously, can damage lungs if the dust gets airborne and breathed in. Warehouse workers learn to wear masks and gloves—not out of paranoia, but from hearing stories where people skipped the basics and regretted it later. Swallowing it directly isn’t common sense, but accidents happen, especially with kids or pets around. The U.S. National Institute for Occupational Safety and Health has found that exposure at high levels can even cause anemia or kidney issues.
Safe Storage Means Thinking Ahead
Storage depends on keeping potassium nitrate dry and separate from organic materials or flammable items. In rural shops, we never stacked those bags near fuels, paints, or even basic gardening mulch. From a personal experience, we learned that storing any oxidizer next to paint thinner or sawdust asks for trouble—insurance companies rarely pay out if rules get ignored.
Keeping humidity low tops the list, too. Dampness turns the powder clumpy and can change how it reacts with other materials. More than one warehouse anecdote tells of leaks or water getting into a shipment, leading to ruined products or, in rare cases, self-heating piles that smoldered for hours. So, tight lids or sealed bins do more than just keep out rodents.
Practical Safety Steps That Work
Respirator masks may sound extreme, but in facilities where dust can spread, those masks prevent real issues. Simple gloves stop irritation, and splash goggles keep the powder from getting in eyes. Out in the field, keeping bags tied and off the ground saves more headache than chasing loose powder later.
Large-scale users—like commercial growers—often go for locked storage rooms, regular checks for leaks, and clear signs. These aren’t just government rules; they build habits that help avoid lawsuits, lost crop cycles, or tragic fires. For folks handling small amounts, think pantry safety: label everything, keep out of reach of little hands, and double check lids.
Trusted Information Wins
Sticking with information from university extension offices, seasoned chemists, or experienced agricultural suppliers pays off. Online sources vary wildly, but experts agree that keeping potassium nitrate in its own space, using it as intended, and taking basic precautions goes a long way. Mistakes rarely happen when people take advice seriously and stay away from mixing it with the unknown.
How should Potassium Nitrate be stored?
Why Storage Makes All the Difference
Potassium nitrate may seem ordinary on a chemistry shelf, but as someone who’s worked with labs, gardens, and a few makeshift fireworks on holiday nights, I know this chemical demands respect. Whether it’s feeding plants, preserving meats, or fueling science experiments, people reach for this white powder for all sorts of reasons. It’s what happens after the bag is opened that raises real safety questions.
Humidity, Heat, and the Trouble They Bring
In places where the air hangs heavy or where summer turns sheds into ovens, potassium nitrate turns clumpy or cakes up pretty fast. Left unsealed, even a small bag left in a drippy garage can harden so badly you’ll need a hammer and chisel to break it loose. More important, damp powder raises the risk of unwanted chemical reactions. Lessons from labs and classrooms tell the same story—keep the powder dry or invite headaches later. Storing it in airtight containers, away from open windows and water pipes, keeps things safe and frustration low.
Fire Risks: No Joke, No Corners Cut
Plenty of folks use potassium nitrate for pyrotechnics or stump removal, probably thinking only big operations run into trouble. The facts tell a different story. Left near oily rags, paint cans, or garden fertilizers, this salt can find trouble. Dropped cigarettes or an old extension cord can set off a race nobody wants to run. A shed packed with flammable gear or yard equipment turns dangerous quickly if a container spills or breaks. That’s why choosing a separate shelf—metal if possible, away from flame sources—makes sense every time.
Children, Curiosity, and the Importance of Labels
Every parent or caretaker knows a bored kid will reach for whatever looks interesting—bags, jars, bottles. One family friend left a half-used bag in his kitchen one summer, thinking nobody would mess with it. Curiosity trumped good sense, and only quick thinking kept things from ending badly. Labels with large, clear writing and danger symbols work a lot better than scribbled notes. Locking up supplies matters as much at home as in the school chemistry closet.
Storage Practices that Work
Good storage starts with the simple stuff. Glass jars with screw tops keep moisture out. A metal cabinet with a padlock keeps hands (and pests) away. Big, bold labels alert anyone who opens a drawer. If dusty bags collect at the bottom of a shed, it’s time to check for tears and toss what no longer looks right. Local regulations might call for special disposal—nobody wants a chemical spill trickling into a garden, creek, or trash bin.
The Role of Knowledge and Routine
Handling potassium nitrate safely isn’t about following rules out of fear; it’s about building habits you don’t have to think about. I learned early on after ruining a small garden crop with a damp batch of fertilizer that saving a few minutes upfront only causes bigger headaches later. Getting safety right pays off with less waste and fewer scares. Regular checks, locked storage, and dry ceilings all add up to safer homes, sheds, and workbenches.
The Bottom Line
Potassium nitrate serves many uses, but the same rules apply everywhere—keep it dry, keep it separate, keep it labeled, and keep it out of reach. Safe storage isn’t complicated. It’s just practice, built up over time, and reinforced by the lessons that come from other people’s close calls and personal mistakes.
What is the chemical formula and properties of Potassium Nitrate?
Potassium Nitrate’s Formula and Its Simple Structure
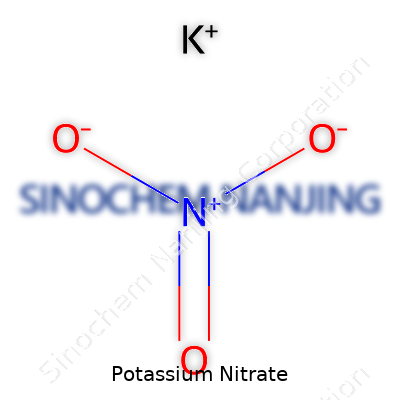
Potassium nitrate carries the chemical formula KNO3. The “K” stands for potassium, “N” for nitrogen, and “O” for oxygen. Looking at a handful of these crystals tells a lot about the compound. Potassium ions hold a single positive charge, and nitrate ions bring along their three-oxygen triad, each oxygen with a small negative charge. Everything balances neatly, which keeps the salt stable. People sometimes call it “saltpeter.”
Physical Properties in Everyday Life
Potassium nitrate forms colorless crystals or white powder. They dissolve pretty well in water. On a summer day, adding some of these crystals to a cup of water shows just how eager they are to disappear into clear solution. That solubility puts potassium nitrate in a different league from salts like calcium sulfate, which fights to stay solid. In science class, touching a piece of the crystal feels cool—there’s no greasy or sticky texture. The powder barely smells, which makes it much easier to work with than ammonium salts.
Potassium nitrate doesn’t burn by itself, but it feeds flames very well. Drop a pinch on burning charcoal; the sparks leap higher. This comes from nitrate’s deep supply of oxygen—each molecule carries three oxygen atoms, just waiting to let fuels catch fire and burn hotter. Potassium does not give the harsh bitterness of sodium; it’s also less likely to bring unwanted side-products to the burn. This is the main reason it shows up in fireworks, stump removers, and black powder recipes handed down over centuries.
Chemical Properties That Matter
This chemical barely reacts at room temperature. Mix it in with other solids like sugar or charcoal, and it mostly keeps to itself. But bring in heat, and potassium nitrate starts to release oxygen gas, leaving behind potassium nitrite and then potassium oxide if you keep cranking the heat higher. That makes it really useful whenever oxygen is hard to come by but fire needs fuel, like in matches and fuse cords.
Around the garden, potassium nitrate has fans who grow tomatoes and peppers. Plants crave potassium for healthy fruit and strong stems, and the nitrate ions bring nitrogen—a must for lush, deep-green leaves. Toss too much of it into soil, and plants might soak up too much salt and dry out. Growing up in a rural area, I saw old-timers weigh out the powder carefully, not dumping it by the handful. They’d sprinkle it sparingly, aware of both the yield it promises and the harm it can do if handled carelessly.
Responsible Use and Looking Ahead
This chemical has a tangled past, from gunpowder in the early days to fertilizer in modern agriculture. Unregulated storage and careless handling have led to accidents, and authorities in many countries watch shipments closely. Keeping potassium nitrate and other energetic materials secure has become more important every year. Simple storage changes can make all the difference—sealed containers, cool dry rooms, using only as much as needed—these steps help prevent mishaps at work and at home.
Looking forward, the world keeps searching for safer, greener fertilizers and pyrotechnic compounds. While potassium nitrate works well and solves plenty of problems, responsible users treat it with respect. That’s how this dependable, centuries-old compound still finds its place in science, farming, and creative human hands.
Can Potassium Nitrate be used as a fertilizer?
Giving Plants a Helping Hand
Ask any backyard gardener what helps their tomatoes thrive, and you'll probably get a talk about soil nutrients. Food doesn’t just appear on supermarket shelves — growing it means knowing what your soil needs. Potassium nitrate offers both potassium and nitrogen, two things plants crave for proper growth, healthy leaves, and strong roots. I’ve seen a difference in my own potted strawberries and leafy greens when I’ve added a little potassium nitrate; the leaves stay greener, and growth doesn’t stall out during long, hot summers.
What Potassium Nitrate Offers
Plants need a steady supply of three macronutrients: nitrogen, phosphorus, and potassium. Potassium nitrate covers two out of the three, making it valuable for many crops. In orchards, leafy greens, potatoes, and even some fruits, potassium nitrate supports robust plant metabolism. Nitrogen fuels lush, green growth. Potassium toughens up stems, helps move water, and guards against stress from drought or disease. Commercial farmers often use potassium nitrate for crops that need to bounce back after heavy harvesting — think lettuce cut for salad mixes.
Sustainable Use and Environmental Concerns
Not every garden or field needs potassium nitrate. Overusing it can backfire, harming soil and sending excess nutrients into groundwater or rivers. Too much nitrate in water leads to algae growth, hurting drinking water and wildlife. Farmers and gardeners have to pay attention to how much their land truly needs. Regular soil testing gives a good picture — skipping blind feeding avoids runoff. In my own space, careful measuring and keeping records makes a difference. Runoff problems in big farm states have sparked regulations and education about best fertilizer practices.
Practical Approaches and Local Factors
Good fertilizer use matches local climate, soil, and crop. Sandy soils drain fast, so nutrients like potassium wash away quickly. Clay holds on a bit too tight, sometimes making nutrients less available. It helps to look at what the plants show you, along with test results. Faded leaves or stunted growth hint at nutrients running low. Potassium nitrate dissolves quickly in water, which fits well for irrigation systems. This helps get nutrients closer to plant roots, cutting down waste. But this approach requires farmers to keep a close eye on irrigation schedules and weather changes.
Balancing Productivity and Safety
Healthy crops feed families, but safety matters too. Potassium nitrate, like all fertilizers, needs secure storage. It has a long history as an ingredient for making gunpowder and fireworks. This means safety rules for transport, storage, and application. Farms and garden centers don’t just hand it out. Plenty of countries have guidelines to keep fertilizers from falling into the wrong hands.
Possible Solutions for Smarter Fertilizer Use
Modern technology offers some hope here. Precision agriculture uses sensors, satellite mapping, and remote monitoring to track exactly what parts of a field need extra nutrients. This saves fertilizer, narrows costs, and lowers environmental impacts. Community education — local extension programs, workshops, or even neighborhood garden groups sharing their results — also spreads better practices. Fertilizers like potassium nitrate can boost yields and resilience, but they work best as part of a full plan: soil care, crop rotation, and decent record keeping all matter just as much.
Final Thoughts
I’ve seen both home gardens and large-scale farms benefit when growers pay careful attention to what they add to their soil. Potassium nitrate, used thoughtfully and responsibly, can make a big difference in putting fresh food on the table without sacrificing future harvests or waterways.
| Names | |
| Preferred IUPAC name | Potassium nitrate |
| Other names |
Saltpeter Nitre Niter E252 |
| Pronunciation | /pəˌtæsiəm ˈnaɪtreɪt/ |
| Identifiers | |
| CAS Number | 7757-79-1 |
| Beilstein Reference | 320168 |
| ChEBI | CHEBI:63047 |
| ChEMBL | CHEMBL1356 |
| ChemSpider | 12109 |
| DrugBank | DB14507 |
| ECHA InfoCard | 03bb9c95-0c1a-4b7c-aea4-298ecb8e78df |
| EC Number | 231-818-8 |
| Gmelin Reference | 604 |
| KEGG | C02567 |
| MeSH | D011104 |
| PubChem CID | 24434 |
| RTECS number | TT3700000 |
| UNII | OUYCCCASQX |
| UN number | UN1486 |
| Properties | |
| Chemical formula | KNO3 |
| Molar mass | 101.103 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.11 g/cm³ |
| Solubility in water | 38.3 g/100 mL (20 °C) |
| log P | -0.9 |
| Vapor pressure | Negligible |
| Magnetic susceptibility (χ) | 'Magnetic susceptibility (χ) = −18.2·10⁻⁶ cm³/mol' |
| Refractive index (nD) | 1.505 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 132.0 J/(mol·K) |
| Std enthalpy of formation (ΔfH⦵298) | -285.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -494 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | A12BA02 |
| Hazards | |
| Main hazards | Oxidizer, may intensify fire; harmful if swallowed; causes serious eye irritation |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS03,GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H319 |
| Precautionary statements | P210, P221, P264, P280, P370+P378, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 0, Instability: 1, Special: OX |
| Autoignition temperature | 400°C (752°F) |
| Lethal dose or concentration | LD50 oral rat 3750 mg/kg |
| LD50 (median dose) | 3750 mg/kg (oral, rat) |
| NIOSH | 'TTQ45520' |
| PEL (Permissible) | 15 mg/m3 |
| REL (Recommended) | 340 mg/day |
| Related compounds | |
| Related compounds |
Sodium nitrate Ammonium nitrate Calcium nitrate Lithium nitrate |

