Potassium Mercury Cyanide: A Chemical with a Complex Past and Present
Historical Development: A Double-Edged Discovery
Potassium mercury cyanide tells a story not many want to hear: human curiosity paired with carelessness can breed a toxic legacy. Chemists in the 19th century dived deep into cyanide chemistry, pushing boundaries without knowing where the risks truly lay. Early industrial labs recognized this compound as both powerful and dangerous. Its introduction sprang from a desire to chase new compounds and metal complexes—ambitions that often ignored long-term effects on workers and the environment. Before strict regulations, this salt appeared in research and industry with little thought for what happens after it leaves the flask. The chemical supported advances in analysis, gilding, and even early photography, but those milestones came with dire setbacks for safety. Today, its tale stands as a reminder: scientific breakthroughs lose value when health and environmental costs go ignored.
Product Overview: A Compound Hard to Ignore
Potassium mercury cyanide pops up as white or colorless crystals. It hides its threat behind this plain appearance. Laboratories, drawn by its effectiveness in creating complex mercury-based species, made frequent use of it, especially in analytical chemistry and gold refining. Still, most current handlers see it as a legacy item—something demanding strict controls. Few modern chemists look for ways to bring more of it back into regular use. Regulations in Europe, the US, and Asia have boxed in its sale and transport, painting a clear picture: risks outweigh most possible rewards. Industries have pivoted, searching for safer alternatives even if it means sacrificing efficiency. Real progress rarely comes in sparkling breakthroughs; often, it's in slowly phasing out the things that do more harm than good.
Physical and Chemical Properties: Subtle but Dangerous
This salt dissolves in water more willingly than one could wish. Its taste—a detail best left untested—proves bitter, matching its toxic punch. Structurally, potassium mercury cyanide falls into the double salt category, bringing together potassium, mercury, and cyanide ions in a delicate balance. That mix enables sensitive reactions but adds layers to the risk. Degradation under heat, bright light, or acidic conditions releases hydrogen cyanide gas—a deadly vapor even in small doses. The compound resists common breakdown routes in the environment, which means spilled material lingers, raising stakes for cleanup. Stability at room temperature lulls some into complacency, but it responds swiftly to mishandling. Here, chemistry does not forgive mistakes.
Technical Specifications and Labeling: More than Paperwork
Anyone handling potassium mercury cyanide learns early to respect the warning symbols—and the weight behind them. Chemical supply houses classify it with the skull-and-crossbones, tight transport limits, and detailed instructions for use, storage, and disposal. Labels do more than check a regulatory box; they remind even the seasoned chemist that this is not just another bottle on the shelf. Transport, purchase, even use in research, draw heavy oversight. The rules exist because people misjudged their luck before, and the rules have teeth—fines and prosecutions follow slip-ups. Nothing pushes a lab to rethink safety like seeing potassium mercury cyanide on the shelf.
Preparation Method: Not for the Untrained
In earlier times, preparation followed an unforgiving path: blending potassium cyanide with mercuric salts, usually mercuric chloride. This route puts right in front of you two notorious toxins—each with its own roster of accidents and injuries. Producing the salt generated not just the target compound but also waste, byproducts, and contamination risks extending beyond the worker’s bench. Every stage posed a challenge. Open vessels released vapors, clumsy hands spilled granules, and labs without fume hoods became hazard zones. Today's focus has shifted to preventing—or at least tightly controlling—such synthesis, locking away both cyanide and mercury under keys and security logs.
Chemical Reactions and Modifications: Power and Peril
Potassium mercury cyanide earned its place as a tool for introducing mercury into organic or inorganic frameworks. Its double-salt form made it key to forming new complexes, a trait especially valued in analytical chemistry when chemists hunted for heavymetal detection schemes. Acidic conditions flip the compound from a stable salt to a cyanide-spewing threat. Strong oxidizers, heat, or even careless mixing with certain organics unlock the hazards hidden within the molecule, sometimes explosively. Modification attempts require ironclad procedures; the cost of error runs far beyond experimental setbacks. Chemists gravitate toward alternatives now, not out of conservatism, but because tragedy does not repeat itself in a vacuum, only in a laboratory that forgets why caution matters.
Synonyms and Product Names: A Rose by Any Name
Potassium mercury cyanide hides behind many names—potassium dicyanidomercurate(II), potassium mercuricyanide, and the older mercury potassium cyanide. Each label echoes its chemistry rather than softening its danger. These synonyms show up in older texts, patent records, and international labeling schemes. Researchers tracking down older literature need to watch for such differences, as one country’s name sometimes hides subtle formulation shifts. Precision with names does more than tidy up a chemical inventory; it can prevent mixing up one nasty salt with another, and that’s no minor detail when so much hangs in the balance.
Safety and Operational Standards: Unyielding Barriers
No other aspect of potassium mercury cyanide demands more vigilance than safety. Research institutions now draft gloves-off policies—no unsupervised handling, limited student access, and robust training before a container gets unsealed. Workflows require fume hoods, spill kits, and clear evacuation routes. Physicians stationed nearby get briefed on cyanide and mercury exposure, and emergency responses don’t start from scratch on this front. Decontamination, even for brief spills, brings out full hazmat protocols—no shortcuts, no speed over care. Underground drainage and ordinary trash become off-limits, forcing users to log every gram and track every disposal. These standards stem from hard lessons—accidents, poisonings, and the long-term havoc both mercury and cyanide leave behind.
Application Area: Shrinking but Revealing
Uses once looked broad, from precious metal extraction to advanced chemical analysis. Gold mining, electroplating, and synthetic experiments in academic labs drew on potassium mercury cyanide for its predictable reactions. The tide turned as safer chelators and mercury-free complexes replaced it. Today, any contemporary chemistry that still holds onto this compound must justify that choice to both ethics committees and local authorities. Areas that can’t break free often face bigger questions about legacy equipment, cross-contamination, and worker welfare. This shift reflects societal progress—swapping ease for the long view and showing that sometimes, the best feature of a chemical lies in deciding not to use it anymore.
Research and Development: Learning from Limitations
Interest in potassium mercury cyanide today concentrates on basic research—understanding historical methods, developing detection strategies, and engineering safe disposal routes. Investigators study how the molecule breaks down, trying to reduce its environmental footprint and trace its fate in soil or water. Most effort aims to unseat the compound from any remaining processes that still cling to it. Technical advances in sensors, photochemical reactions, and new catalyst design keep researchers inside strict regulatory lanes, with careful oversight. Old journal articles and patents get dusted off out of necessity, as researchers exploring contamination or exposure events need background on what they’re up against. Chemical innovation now means cutting ties with chemicals like these, not bolting new features onto an outdated compound.
Toxicity Research: Facing Harsh Truths
Potassium mercury cyanide packs a dual punch: the acute, rapid toxicity of cyanide and the slow, persistent damage of mercury. Clinical experiences and lab mishaps both tell the same story—nausea, convulsions, organ failure, and, if untreated, death. Inhalation or ingestion, even at small doses, closes the window for treatment fast. Chronic, low-level exposure adds nervous system damage, kidney lesions, and developmental defects to the list. Medical studies probe these harms, documenting not only accidents, but the cost to workers and nearby communities before modern safety rules took hold. Regulatory agencies now treat potassium mercury cyanide as one of the chemical world’s most tightly controlled hazards, thanks in part to the bitter lessons collected in toxicology texts and hospital records. Animal studies and workplace investigations have shaped hard caps for allowable exposure—limits so low that even minor misjudgments become serious health threats.
Future Prospects: Outpaced by Progress
No serious technologist sees potassium mercury cyanide as a compound offering much hope for future breakthroughs. The trend points firmly away from its use. Green chemistry, sustainable production, and health-conscious research have cut its application to the bone. Alternative pathways for complex formation or gold recovery now dominate. Calls for total phase-out grow louder each year, and regulatory tightening shows little sign of relaxing. Research groups focus on detecting old contamination, cleaning up spill sites, or training chemists in safer habits. In a world facing tough questions about pollution, worker safety, and industrial waste, potassium mercury cyanide stands as a chemical emblem to retire, not refine. Its main future utility may well lie in reminding everyone just how costly shortcuts can be in science and industry.
What are the main uses of Potassium Mercury Cyanide?
What Drives Its Use
Potassium mercury cyanide belongs to a class of chemicals you don’t run into unless you are deep in specialized industrial work or advanced lab research. The stuff packs a punch—its toxicity is well documented, which isn’t something anyone should overlook. I’ve come across potassium mercury cyanide a few times in professional circles. Each time, what stood out was the extra-thick rulebook that came before cracking open the container. To understand its main uses, you need to know why anyone would use something so dangerous in the first place.
Electroplating and Surface Finishing
You find potassium mercury cyanide in older processes for electroplating, especially when good adhesion and a particular finish are needed. Watchmakers, jewelers, and electronics technicians from a few decades ago sometimes used it to deposit thin, uniform layers of gold and other precious metals. It produces a surface with certain desirable properties—brightness, corrosion resistance, and a smooth feel you notice when you handle a fine timepiece or older circuit boards.
Modern factories tend to move away from potassium mercury cyanide because of the safety hazards. Still, I know artisans and repair specialists in restoration work who sometimes rely on legacy methods because alternatives struggle to produce the same result. According to the US National Institute for Occupational Safety and Health (NIOSH), exposure to mercury and cyanide compounds can lead to lifelong health consequences, which fuels pressure to swap out this compound for safer plating solutions.
Laboratory Synthesis
Potassium mercury cyanide serves as a reagent in organic synthesis, especially for creating mercuric compounds or for certain cyanation reactions that call for precision. In academic settings, you sometimes see it in protocols that date back before more humane or ecological procedures became common. As someone who spent late nights in grad school labs, I can tell you the hassle of handling these chemicals forces a hands-on appreciation for chemical safety. Fume hoods, thick gloves, and constant monitoring make it clear: one small mistake means real danger.
Global chemical suppliers have started reducing stockpiles and encouraging buyers to consider less hazardous alternatives. Journals now expect much stronger justifications before approving research projects that involve high-risk chemicals, according to the American Chemical Society. The movement to “green chemistry” has already chopped down a lot of hazardous reagents, so potassium mercury cyanide now faces a shrinking role in research.
Environmental and Safety Challenges
Environmental agencies warn against careless disposal. Potassium mercury cyanide can kill aquatic life and poison groundwater fast, with symptoms that don’t always show up until the damage is done. Just last year, I watched a local hazardous waste team hustle to clean up after a spill from an old electroplating shop in a small industrial park. Cleaning up mercury and cyanide compounds takes more work and costs more money than nearly any other type of spill they encounter.
Paths Away from Hazard
Many countries now limit or outright ban potassium mercury cyanide outside a few narrowly defined uses. Strict tracking, storage rules, and specialized training push businesses to look for less dangerous options. Electroplating shops, in particular, turn to newer non-cyanide solutions or even physical vapor deposition. Laboratories rethink their protocols and turn to computational chemistry or safer reagents. These moves help protect people and the planet, sparing families and workers from the harsh consequences that can follow a single unsafe moment.
What safety precautions should be taken when handling Potassium Mercury Cyanide?
Why Take This Compound Seriously
Potassium mercury cyanide doesn’t just sound dangerous. In my own lab experience, we always gave extra respect to anything combining heavy metals and cyanide. This stuff contains both mercury and cyanide elements, which means trouble for anyone underestimating what it can do to the human body. It disrupts respiration at a cellular level—nasty business if you breathe it or get it on your skin. Cyanide blocks cells from using oxygen, leading to tissue damage and rapid poisoning. Mercury targets nerves and organs, making it a double threat.
Personal Preparation Goes Beyond Gloves
My earliest chemical safety training drilled it in: never work with compounds like this unless you wear real personal protective equipment. This isn’t a splash of vinegar or a bit of bleach. Think full lab coat, chemical goggles (not just safety glasses), and face shield if there’s any risk of splashes or dust. Nitrile gloves don’t cut it; serious gauntlet-style gloves rated for harsh chemicals form a real barrier. Any tiny gap between coat cuff and glove is a big deal—tape those wrists down. Double-gloving often feels clumsy, but jaundiced hands or cyanotic lips are far worse.
Ventilation Is Non-Negotiable
Every time we had to work with volatile or acutely toxic substances, the fume hood became our friend. Potassium mercury cyanide can release vapors you can't see or smell, but one shallow breath is enough to cause problems. If you don’t have a working chemical fume hood, don’t even open the bottle. No exceptions—ventilation keeps deadly vapors away from you and your colleagues.
Planning Ahead Means Life or Death
I’ll always remember that safety with strong poisons begins long before opening the bottle. Map out your steps: figure out where you’ll set the container, where you’ll dispose of any paper towels or wipe-downs, and how you’d reach the emergency shower or eyewash fountain in a heartbeat. Spills happen in moments of distraction. Spillage kits for mercury and for cyanide are each specialized, so both need to be handy. Don’t assume you’ll stay calm under pressure—muscle memory from drills makes all the difference.
Labeling and Storage
Years of running lab inventories forced me to appreciate that clear, tough labels save lives. Unmarked bottles have no business near potassium mercury cyanide. Lock it in a poison cabinet with chemical-resistant shelving. Keep it separate from acids, since a slip here releases deadly hydrogen cyanide gas. Store as little as possible. Disposal needs to wait for a certified hazardous waste expert. Don’t let untrained staff anywhere near the container.
Cleanup Is For Professionals
If the worst happens, and a spill or exposure takes place, don’t play the hero. My training emphasized grabbing the exposure victim, hitting the nearest shower, and calling medical help on the spot. Don’t let pride or embarrassment delay that call—even two minutes matter. Let the hazmat team or trained chemical response crew do cleanup. The risks are too high for improvisation.
Invest in Safety Culture
Trust among your team makes speaking up about missing gloves or sloppy labeling easier. In factories or research work, the best environments never get casual about these compounds. Demanding regular training and drills, alongside open communication around errors, reduces the odds of disaster. Supplies cost money, but human lives are priceless—we can’t cut corners with chemicals like potassium mercury cyanide. Take precautions seriously, for your own sake and those who rely on you.
What are the toxicity and health risks associated with Potassium Mercury Cyanide?
The Reality of Handling This Chemical
Potassium mercury cyanide stands out as a chemical people should never take lightly. Its name, even to those with basic science knowledge, sounds alarming—and rightfully so. This compound packs the danger of both mercury and cyanide, two elements known for their severe toxicity. In labs, a small spill or an accidental touch poses a threat to human health. In my experience working in research environments, any compound containing mercury comes with extra rules and stricter safety briefings. The moment cyanide gets thrown into the mix, double-gloving and full ventilation go from advice to requirement.
How Toxicity Strikes: No Second Chances
Potassium mercury cyanide doesn’t mess around. Handle it without full personal protective equipment, and you risk symptoms that appear fast and get severe. Cyanide poisoning can cut off the oxygen cells need to survive, which means dizziness, shortness of breath, seizures, and cardiac arrest can follow even a minor exposure. Mercury adds its own dangers, damaging nerves, kidneys, and lungs if it finds its way into the body. Just breathing dust or vapors can set health problems in motion.
Many who have worked in chemistry can recall stories shared in safety seminars. A technician cleaned up a few crystals one day, didn’t follow up with proper washing, and by the evening reported headaches and nausea. The lucky ones get away with a hospital visit, but the numbers don’t favor luck. Both mercury and cyanide kill quietly and quickly; not every victim makes it in time for medical help.
Long-Term Health Effects
Surviving the initial exposure doesn’t guarantee a return to normal. Mercury leaves long-lasting scars on the nervous system and memory. Cyanide exposure often damages the heart or leaves lingering issues with breathing. In places with less access to antidotes like hydroxocobalamin or advanced respiratory support, the dangers multiply. Academic journals and government chemical hazard databases both show high rates of death and chronic disability tied to this compound.
Environmental Hazards
Potassium mercury cyanide accidents don’t only threaten those in the lab. Once spilled, mercury pollutes water and soil, building up in fish and entering food chains. Cyanide causes sudden die-offs of aquatic life, stripping rivers and lakes of oxygen. Clean-up costs skyrocket because no standard filtration system removes the combined toxicity fully. Industrial disasters involving compounds like this have left entire towns with long-term health advisories.
Preventing Harm: What Actually Works
Any workplace still using potassium mercury cyanide demands a safety culture that leaves nothing to chance. Strict inventories, double-locked storage, and continuous air monitoring stop most accidents before they start. Staff should practice spills and exposure scenarios well before anyone opens a jar. Some countries ban this chemical outright for non-essential use. Chemistry is moving to safer alternatives for applications in electroplating and analysis. If someone works with this compound, get the facts on cyanide and mercury antidotes and make sure the kit stays stocked—those minutes count.
Final Thoughts on Health and Responsibility
No shortcut makes potassium mercury cyanide less threatening. Its risks demand both respect and heavy regulation. Safe alternatives exist for most needs, and industries should make those switches wherever possible. Staying healthy means learning the science behind the warnings and sticking to them, every time, no exceptions. Chemical safety builds on trust, care, and a willingness to leave dangerous shortcuts in the past.
How should Potassium Mercury Cyanide be stored and disposed of?
The Dangers Wrapped in Every Granule
Ask anyone who’s handled potassium mercury cyanide in a lab—they’ll probably mention sweat on their brow. It’s a compound with a triple threat: mercury, cyanide, and potassium. Each brings toxic baggage, and together, they spell trouble for skin, lungs, and the environment. The compound can leach into water, evaporate, react with acid, or slip through a careless glove. Even a small spill could trigger a workplace evacuation, not to mention the risk to cleaning staff or maintenance folks who don’t deal with chemicals every day.
Lab Life and Locked Cabinets
In my college research days, chemical storage meant more than a label and a locked door. Potassium mercury cyanide belongs to the type of chemicals that get their own padded box, away from acids and oxidizers. Labs with high safety standards use dedicated cabinets—lined and ventilated—for fragile toxins like this. Besides that, humidity control reduces the risk of a slow leak or corrosion, which could break seals and quietly spread poison. Access is always tight; only trained staff can get the keys, and every gram must get logged. Once, I saw a TA turn away a postdoc because their paperwork didn’t check out. Proof that shortcuts don’t cut it when this compound is involved.
Paperwork Isn’t Just Busywork
It sounds dull, but logs and inventory checks keep people honest and alert. The EPA, OSHA, and local fire marshals all demand robust documentation. From my own experience, this helps highlight who's using what, and keeps a sharp eye on expiration dates or container faults. Missing entries or swapped containers could set off a crisis. In one incident at a friend's university, a mislabeled flask forced an entire building to close for two days. Accountability isn’t bureaucracy—it’s safety lived out daily.
Shoulders Heavy with Disposal
Once it’s time to get rid of potassium mercury cyanide, everyday trash isn’t an option. Flushing it or tossing it in a dumpster would seed disaster for years, polluting soil and waterways. I’ve seen professional waste handlers show up in full gear just for a few grams, using rigid protocols. Everything—gloves, containers, and paperwork—heads out in sealed drums, tracked from pickup to incinerator or special chemical treatment. Many organizations contract licensed hazardous waste companies, who neutralize cyanide and separate mercury for recovery. Incineration facilities treat remnants at ultra-high temperatures. These places aren’t back-alley operations; they face regular audits and carry hefty insurance, since even a slip-up can poison communities or contaminate groundwater far away.
Real-World Solutions
Training saves lives and property. Colleges run drills, keep spill kits close, and run refresher courses for staff. Insurance companies now push for stronger controls, and many campuses voluntarily exceed government requirements. Digital tracking with barcodes tracks every move of each container, reducing human error and flagging missing material before things spiral.
Laws do a fair bit, but nothing beats a safety-first culture. Every researcher or janitor needs honest, plainspoken training—not a dry slideshow bending under jargon. Tools like fume hoods, spill trays, and proper gloves back up rules with real protection. Safer alternatives to potassium mercury cyanide exist for some applications, and phasing out old stocks makes life simpler all around. In the end, treating dangerous chemicals with humility and clear eyes goes further than any fine print could ever reach.
Is Potassium Mercury Cyanide regulated or restricted in certain countries?
Hazards That Can’t Be Ignored
Potassium mercury cyanide lands near the top of any list of chemicals that spell danger. The name itself gives a clear warning: we’re talking about a combination of mercury, cyanide, and potassium. All three bring their own hazards. Mix them together, and you get a material that nobody should ever treat casually. One breath of its dust, or a bit of skin contact, can result in deadly consequences. Stories from labs and workplaces show how even experienced chemists double-check their gloves when working anywhere near it.
How Laws Respond to Risk
Regulations aim to minimize harm. In the United States, potassium mercury cyanide appears as a listed substance under the Environmental Protection Agency's Toxic Substances Control Act, and workplaces follow strict OSHA guidelines. Anyone making or handling it in America needs a mountain of paperwork and authorization—not because regulators enjoy bureaucracy, but because accidents cost lives.
Across Europe, the European Union bans or heavily restricts most cyanide compounds outside of tightly controlled industrial applications. Mercury itself caught most headlines after the Minamata disaster in Japan, and the region responded with sweeping regulation. Potassium mercury cyanide falls under REACH controls, meaning chemical suppliers track every kilo and anyone ordering it must justify their use.
Asia takes a mixed approach. Japan banned most uses decades ago, sparked by scars left from industrial pollution. China labels potassium mercury cyanide as a Category 1 hazard, meaning only government-licensed entities can possess it. In India, chemical rules lagged behind for years, but recent clampdowns after global trade pressure mean customs officers watch for it at borders.
The Human Cost Behind Regulation
I once worked in a lab where even talking about mercury cyanides required paperwork. Training taught us that the risk isn’t a statistic—real people have lost their health, or lives, from exposure. In the real world, mistakes happen. A forgotten fume hood, a spill brushed off the table—these can swiftly move from scratches on a clipboard to an emergency room visit.
Historical disasters, like those at chemical plants in the twentieth century, didn’t just leave toxic soil; they left behind stricter laws. Society recognized that one slip puts not just workers, but entire communities at risk. Regulators learned the hard way that turning a blind eye to mercury-and-cyanide mixtures invites disaster—so rules got tougher.
Challenges Facing Enforcement
Smaller countries sometimes struggle to follow through. Enforcement means money, trained staff, and clear rules. Corruption and smuggling, especially in places with looser borders, keep dangerous substances like potassium mercury cyanide in circulation. Black market chemicals bypass safety, ending up in the hands of people with no idea how lethal a single mistake could be.
Better Answers for a Safer World
It’s not just about shouting “ban it” and moving on. Education brings lasting results. Workers benefit from real-world safety drills and honest talk about the after-effects of exposure, rather than rote memorization of safety data sheets. Transparency matters too—where did this shipment come from, who signed for it, where does it end up? Technology could step up, using chemical markers or blockchain-style tracing to follow shipments all the way from manufacturer to end user.
Potassium mercury cyanide shouldn’t be lying around in a storeroom for just anyone to find. Global cooperation, strong laws, and honest workplace culture offer the best hope for keeping tragedies out of tomorrow’s headlines.
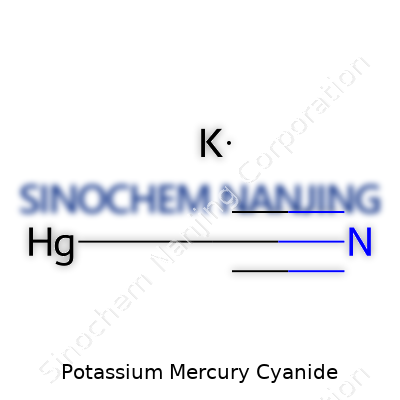
| Names | |
| Preferred IUPAC name | Potassium tetracyanidomercurate(II) |
| Other names |
Cyanoaurate Potassique Potassium (cyano)mercurate Potassium mercuricyanide |
| Pronunciation | /pəˈtæsiəm ˈmɜːkjʊri saɪˈænaɪd/ |
| Identifiers | |
| CAS Number | 334-58-7 |
| Beilstein Reference | 1209247 |
| ChEBI | CHEBI:84935 |
| ChEMBL | CHEMBL504105 |
| ChemSpider | 57014 |
| DrugBank | DB13526 |
| ECHA InfoCard | ECHA InfoCard: 100.004.686 |
| EC Number | 205-792-3 |
| Gmelin Reference | Gm.1412 |
| KEGG | C18798 |
| MeSH | D011689 |
| PubChem CID | 10469 |
| RTECS number | OX9945000 |
| UNII | 9LU97Q26OT |
| UN number | UN1588 |
| Properties | |
| Chemical formula | KHg(CN)₂ |
| Molar mass | 284.65 g/mol |
| Appearance | White crystalline solid. |
| Odor | Odorless |
| Density | 2.7 g/cm3 |
| Solubility in water | Soluble |
| log P | -1.4 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 11.0 |
| Basicity (pKb) | 3.74 |
| Magnetic susceptibility (χ) | -2870.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.558 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -97.2 kJ/mol |
| Pharmacology | |
| ATC code | S51AG02 |
| Hazards | |
| Main hazards | Highly toxic by inhalation, ingestion, and skin absorption; releases highly toxic cyanide fumes when heated or in contact with acid; danger of cumulative mercury poisoning; very dangerous environmental hazard. |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H300 + H310 + H330: Fatal if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P260, P262, P264, P270, P273, P280, P301+P310, P302+P350, P304+P340, P305+P351+P338, P310, P320, P330, P361, P363, P391, P405, P501 |
| NFPA 704 (fire diamond) | 3 3 2 Mercury Poison |
| Lethal dose or concentration | LD50 oral rat 42 mg/kg |
| LD50 (median dose) | LD50: 18 mg/kg (oral, rat) |
| NIOSH | SN4550000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Potassium Mercury Cyanide: "0.01 mg/m³ (as Hg), skin |
| REL (Recommended) | 0.05 mg/m³ |
| IDLH (Immediate danger) | 5 mg/m3 |
| Related compounds | |
| Related compounds |
Mercury(II) cyanide Potassium cyanide Potassium thiocyanate |

