Potassium Iodate Diiodate: A Closer Look from the Lab to the Field
Historical Development
Potassium iodate diiodate hasn’t made headlines, but its journey weaves through a story familiar to anyone in chemical manufacturing: necessity leads to curiosity, and curiosity spills into the hunt for better solutions. Back in the nineteenth century, scientists fixated on iodine’s vital roles—especially after the world saw the connection between iodine deficiency and thyroid diseases. Researchers stumbled across potassium iodate and its related compounds as part of efforts to deliver stable iodine sources that keep well over time. I remember leafing through stacks of technical papers from the 1960s, watching experts puzzle over how to refine iodate salts to dodge instability issues in food and pharmaceuticals, especially in humid climates. Potassium iodate diiodate emerged from such experiments, offering more consistent behavior and greater shelf life—a boon where potassium iodide often struggled. Legacy matters here; chemists didn’t land on this compound for the sake of complexity. They followed a practical trail, choosing substances ready to meet real-world demands when iodide wasn’t enough.
Product Overview
For anyone used to handling crystalline salts behind the bench, potassium iodate diiodate doesn’t look too out of the ordinary—white, grainy crystals, faintly reminiscent of table salt but loaded with chemical potential. On the supply side, it operates as a reliable iodine carrier. It dissolves predictably in water, produces a steady release of iodine under particular conditions, and stays stable enough to sit in inventory for months without losing its kick. What sets it apart from more common potassium iodate is a double punch of oxidizing capacity, making it useful where regular iodate comes up short. The larger chemistry community sees this salt as a specialist, filling pockets in niche applications like certain laboratory assays, advanced disinfection, and targeted nutritional interventions.
Physical and Chemical Properties
Potassium iodate diiodate, with its heavy iodide load, tips the scales in terms of both atomic weight and oxidation potential. The crystals come off sturdy, resisting breakdown even in the thick of summer, provided moisture stays in check. You won’t spot a strong odor. Heat pushes the material to decompose above the boiling point of water, releasing iodine and oxygen in the process. Out of habit, any chemist handling the compound soon learns to keep it away from organic matter or reducing agents; the oxidative bite is real and will start fires or unleash unpleasant gases if given the chance. Electrical conductivity in solution runs high, matching expectations from any strong ionic salt. If you check under a microscope, crystal structure appears uniform, a result that keeps handling predictable during dosing or blending.
Technical Specifications and Labeling
Clarity in labeling matters, especially when every gram has a job to do—whether spiked into table salt for public health or weighed for research. Regulations across regions require precise labeling of content, purity, and batch details, often tested to pharmaceutical or analytical standards. Iodine content by mass sets the baseline, since overdosing carries known risks. In the labs I’ve worked, purity over 99 percent gets priority, and batches failing color or solubility tests never see a delivery truck. Labels need to reflect storage conditions, handling precautions, and potential incompatibles: strong acids, combustibles, and reducing chemicals. I’ve seen recall campaigns driven by mistakes or label omissions, so the best suppliers keep details as transparent as possible.
Preparation Method
Making potassium iodate diiodate starts with chemistry as old as the periodic table: controlled oxidation reactions involving potassium carbonate or potassium hydroxide combined with iodine in water. The mixture needs a fine balance. Too much heat, and you decompose the product; not enough, and you under-react the mix. Industrial players rely on precise temperature, clean water, and high-purity starting reagents. Filtration steps sift out insoluble byproducts, and repeated crystallization sharpens purity. My own attempts in the lab, though on a far smaller scale, echo this routine: gentle boiling, careful titration, close watching for the tell-tale solid falling out. Residues get rinsed off, and a quick dry under vacuum seals the process, capturing those uniform, white crystals ready for use or sale.
Chemical Reactions and Modifications
Potassium iodate diiodate stands out for the way it bridges iodine chemistry. Drop it into acidic solution, and you watch the color deepen as free iodine forms—a simple but impressive sight in any lab. Mix it with reducing agents, and you spark quick redox reactions, which often lead to the familiar brown cloud of iodine gas. This makes it an option for teaching labs, showing chemical balances in action. Researchers play with substitution or doping—edging in different alkali metals or incorporating stabilizers—to tweak performance for targeted uses. In industrial hygiene or water disinfection, the compound’s oxidizing punch gets harnessed to kill off pathogens, while strict controls monitor against undesirable byproducts, since any iodine release at scale needs careful handling. The range of chemical tweaks seems far from exhausted. Labs around the world keep searching for formulations that stretch its stability or boost iodine release at just the right rate.
Synonyms and Product Names
Chemists and suppliers alike use a range of synonyms. Potassium periodate, double potassium iodate, or just “KIOD” turn up in catalogs or databases. Sometimes, confusion creeps in between potassium iodate (KIO3) and potassium periodate (KIO4), but anyone sourcing for manufacturing or health work checks the structural formula twice, since their properties diverge wildly. Regulatory language especially likes consistency, holding fast to clear chemical identifiers for cross-region labeling. In the field, local vernacular adds to the mix; regions where fortification programs are common may use familiar trade names or abbreviations, feeding into ongoing requests for clarity when supplies cross borders.
Safety and Operational Standards
The best practice always puts safety at the center, not only because of regulation but because even small mistakes invite big consequences. Potassium iodate diiodate, with its oxidative muscle, demands separation from fuels, organic matter, and acids. Personal experience teaches respect—one dropped beaker spilling into a waste can becomes a smoky, stinking mess in seconds. Sturdy gloves, sealed eyewear, and ventilated benches form the baseline. Bulk storage sticks to dry, sealed drums, away from traffic and temperature extremes. Regular audits, spill drills, and hazard communications are routine where I’ve worked. Regulatory bodies post permissible exposure limits and set out clear protocols for industrial hygiene and accidental releases. Disposal guidelines target neutralization and dilution, never dumping into general waste streams.
Application Area
Most public conversations about potassium iodate diiodate revolve around health, and for good reason. Lack of dietary iodine still stalks parts of the globe, leading to developmental delays and thyroid disease. Salt and flour fortification rely on stable forms of iodine, and potassium iodate diiodate provides a slow, predictable release profile—with fewer losses from shelf to table. Industrial users dip into large stocks for water treatment, leveraging chemical potency to break down contaminants and kill pathogens. Educational labs rely on its predictable oxidation for experiments showing the basics of redox reactions. Niche applications keep cropping up—battery development, advanced reagents for analytical chemistry, and even specialized photography. Each application demands meticulous tracking of purity, shelf-life, and handling, since mistakes translate directly into people’s health or industrial downtime.
Research and Development
My own time coordinating with university labs taught me how open the field remains for improvement. Researchers dig into the mechanics of crystal structure, tracking impurities that slow iodine release or disrupt stability. Some push for greener synthesis, minimizing hazardous byproducts and slashing energy costs. In public health, teams scan for ways to boost nutritional uptake—can modified salts deliver more effective dietary iodine? The push for “smart” delivery in fortified foods, timed to resist cooking or storage losses, has academic and commercial labs reporting breakthrough after breakthrough. Emerging technology looks to tissue engineering, cancer therapy, or digital imaging as new hunting grounds for modified iodate salts. The tide runs strong; as public health campaigns evolve or industry pivots toward higher sustainability, potassium iodate diiodate research keeps pace, hungry for more impact.
Toxicity Research
Every chemical with real utility comes with downsides if handled wrong. Toxicologists keep a close eye on potassium iodate diiodate, especially with its role in foods and supplements. Ingestion tests in animals and controlled human trials lay out safe windows for exposure, pointing out thyroid disruption or stomach irritation if overdosed. Chronic exposure gets more attention, especially in regions mounting large-scale fortification. Regulatory agencies set conservative daily intake levels, and I’ve seen national health programs suspend distribution if test batches creep near those ceilings. Field monitoring watches for unanticipated side effects—skin rashes, GI upsets—while emergency protocols exist for accidental overdose. Data keeps arriving, and as public health challenges shift, so do the questions: can the compound be tuned for less toxicity at the same dose, or coupled with other micronutrients for a safer blend? These debates shape both public perception and regulatory policy.
Future Prospects
Holding potassium iodate diiodate in your hand, you can’t ignore the future questions crowding around it. As more countries tackle iodine deficiency and as industries lean into tighter quality standards, the call for better, safer, and smarter iodine additives gets louder. The compound faces strong competition from alternatives, but its track record in stability and reliability means it’s not fading any time soon. Green chemistry shapes the agenda, pushing for synthesis routes with less waste, cheaper inputs, and easier recycling. Advances in food science could bring new, fortified products tuned for specific communities or health needs. Medical research eyes designer salts for more effective thyroid therapies; environmental labs hunt new ways to purify water without adding toxicity downstream. Science rarely stands still; as needs change and technologies open up fresh questions, potassium iodate diiodate seems set to answer many more.
What is Potassium Iodate Diiodate used for?
Finding Purpose in a Complex Compound
Potassium iodate diiodate does not land in many shopping carts, but its role affects more lives than most people realize. My own dive into the science behind public health made me notice its quiet importance. It shows up in places where precision and safety both demand attention. This compound finds its seat at the table in large-scale efforts to tackle iodine deficiency, but also in tough spots like radiation emergencies and industrial testing.
Tackling Iodine Deficiency and Supporting Health
Iodine deficiency isn’t a quirky issue from the past. Growing up in a region where iodine-poor soils were the norm, I saw the effects firsthand—thyroid problems, developmental delays in kids, and a lot of anxious parents. Potassium iodate diiodate became key in government efforts to fortify salt. Its stable chemical structure withstands harsh climates and long storage, doing the heavy lifting where potassium iodide fails. Salt treated this way reaches rural households, often far from modern medicine. Studies from the World Health Organization back up its effectiveness, showing substantial drops in goiter rates and improved child brain development whenever iodate fortification takes hold.
Guarding Against Radiation Threats
Not every threat comes quietly. Nuclear emergencies push public health systems to their limits, as seen during the Fukushima disaster. Here, potassium iodate diiodate steps in, helping block radioactive iodine uptake in human thyroid glands. Iodate options can offer longer shelf lives and more stable performance when compared to KI tablets, especially in humid storage conditions. Emergency preparedness manuals now list these compounds among key supplies for communities near power plants.
Reliable Results in Industry and Testing
This compound’s job goes beyond health. Early in my chemistry studies, I worked on water treatment tests. Potassium iodate diiodate acted as a go-to standard for measuring how much oxidizing power certain solutions packed. Labs trust it for iodine titrations and as an oxidizing agent because it provides sharp, reliable results every time. In the food world, quality testing for certain baked goods uses this compound to check for dough improvers that help bread rise and stay fresh.
Risks and Careful Handling
No chemical fixes everything without some risk. Safety matters here too. Mishandling can spark skin and eye irritation; breathing in dust is a hazard. Training for safe storage and workplace procedures cannot slip through the cracks, especially in busy production lines or community salt-distribution centers. Governments keep guidelines updated and insist on trusted suppliers. Regulators like the FDA and European Food Safety Authority set strict limits for how much goes into food or ends up stored near families or workers.
Building Lasting Solutions
The best answers come from understanding local needs. In places where electricity goes out for days or humidity damages simple iodide supplies, this compound saves health programs months of stress. International health organizations should keep evaluating and improving supply chains so remote health projects don’t run dry. Education also matters—sharing simple facts about iodine's link to brain development brings parents, teachers, and local leaders on board.
Having reliable compounds like potassium iodate diiodate in the toolkit lets public health and science keep moving forward. With smart policies, community education, and scientific checks along the way, health systems can use the benefits and avoid the mistakes of the past.
Is Potassium Iodate Diiodate safe for human consumption?
Understanding the Compound
Potassium iodate diiodate isn’t something you see every day in a kitchen or even in a pharmacy. It’s a chemical salt with a complex name that often leads people to lump it in with more familiar substances like potassium iodide or regular potassium iodate. Both those compounds have roles in food fortification and emergency preparedness, especially for thyroid protection during a nuclear event. Potassium iodate diiodate, on the other hand, gets far less attention from food safety agencies or public health organizations. The question of its safety for regular human consumption deserves real scrutiny, since not everything with “iodate” or “potassium” brings the same effects.
Food Additives and Regulatory Oversight
I remember my mother pouring out table salt from a blue can that promised “iodized for your health.” That iodized salt came with potassium iodide, which has a clear track record for preventing iodine deficiency disorders, especially in places where soil doesn’t offer much natural iodine. When governments talk about food safety, they depend on agencies like the FDA and EFSA to set boundaries. Potassium iodate appears on food fortification lists in some countries, especially where potassium iodide lacks stability. There’s next to no guidance on potassium iodate diiodate. No established Acceptable Daily Intake (ADI), no food-grade specification, no safety evaluation in nutrition science journals.
Chemical Properties and Potential Risks
Potassium iodate diiodate brings together strong oxidizing properties. Chemists learn early: oxidizers in high doses or improper contexts end up toxic. Even potassium iodate and iodide carry risks above certain thresholds. Consuming too much iodine, in any form, stresses the thyroid and can trigger autoimmune complications. The difference here: potassium iodate diiodate doesn’t show up much in controlled food or supplement products, so there’s nowhere near the amount of safety data we see with better-known iodates or iodides. Without these studies, taking a chance on unapproved compounds runs real personal risk. Ingesting chemicals without a standard for purity or recommended limit, especially those with potential to irritate or damage tissues, isn’t something any doctor or nutritionist recommends.
Public Health and Informed Choices
People deserve trustworthy information about what lands in their food. I’ve seen how misinformation online, especially about mineral supplements and “alternative” compounds, can complicate health for real families. Some promoters suggest potassium iodate diiodate as a miracle remedy or safer substitute. Current evidence just doesn’t support these claims. Reliable decisions need solid scientific data and regulatory approval. Looking at how public health programs operate worldwide, no country with an established nutrition program relies on potassium iodate diiodate for iodine fortification or supplementation. Their caution comes from well-documented science and real-life experience with food safety failures.
Possible Ways Forward
Anyone curious about new food additives should ask for transparency about sources, independent testing, and published safety records. Food and supplement producers would serve everyone well by sticking to compounds with a long safety record, like potassium iodide and potassium iodate, both recognized by international authorities. Scientists have a responsibility to run further tests on other compounds before they get near dinner plates or medicine cabinets. Regulators can strengthen oversight by closing loopholes that allow lesser-known chemical variants into consumer products without proof of safety. In my own family, we’ve settled on using only supplements and foods that come backed by clear approval and real evidence, not marketing or unproven substitutions.
How should Potassium Iodate Diiodate be stored?
Understanding Why Storage Matters
Potassium iodate diiodate sits on the shelf of many chemical storerooms. This stuff plays a role in labs, industry, and food fortification projects—where keeping iodine levels up in salt remains crucial for public health. I remember in the early days, mishandling a batch led to a ruined experiment, driving home the message that treating storage as an afterthought just wastes resources and time. It's not just about following rules; it's about protecting the product and the people who rely on it.
The Storage Environment
Potassium iodate diiodate breaks down over time if it faces heat, moisture, or light. Even a couple of hours in a damp environment can set off chemical changes, and then you no longer have reliable material. Just like you wouldn’t keep bread in a humid pantry, chemicals like potassium iodate diiodate want a dry spot. Gel desiccant packs inside sealed containers help here. Glass or high-density polyethylene bottles with airtight lids give dependable barriers against both humidity and air.
Temperature Control in Real-life Conditions
Room temperature works for short periods, but if summer rolls in and storage hits above 30°C, breakdown rates climb. Cooler, consistent temperatures win every time. At a pharmaceutical lab I worked for, a simple temperature log book next to the storage fridge made staff check levels and catch any swings before trouble started. Commercial fridges (not shared with food) keep things steady. Fluctuating conditions inside a warehouse—caused by open doors and outdoor air—put these chemicals at risk.
Keep It Out of Reach, Out of Trouble
Accidental mix-ups with other chemicals, or even an unsuspecting hand reaching into the wrong bin, spell safety hazards. Clear labeling prevents mistakes. Storing potassium iodate diiodate away from acids, organic materials, and combustible stocks reduces chances of unwanted reactions. As a storage best practice, chemicals stay grouped by property—oxidizers with oxidizers, never mixed with reducers or flammables. Lockable cabinets help keep curious hands—be they kids at home or untrained staff—from getting in harm's way.
Why Documentation Matters
Inventory sheets, logbooks, and clearly printed expiration dates keep everything organized. Sometimes, improper storage slips by unnoticed, and old product hangs around at the back of a shelf. With a log, you track quantities and rotate old stock forward to prevent this. Audits don’t just tick regulatory boxes—they spotlight good habits. I’ve spotted degraded chemical stock during an annual review more than once, and those records caught problems before they became a bigger issue.
Solutions That Work—Without Breaking the Bank
Total isolation in climate-controlled cabinets may sound ideal, but budgets get tight. Still, storing potassium iodate diiodate doesn’t call for extreme measures. Cheap desiccants and sensible shelf placement—up off the floor, away from windows—make a big difference. Training everyone who steps into the storeroom, even just once a year, beats fancy technology every time for safety and reliability.
Final Thoughts From Experience
Storing potassium iodate diiodate safely asks for attention, good habits, and some prevention up front. Practical, cost-effective steps—seal it tight, shield against heat and light, separate it from incompatible substances, and always know what sits in the storeroom—keep this chemical useful and the workplace safer for all. These simple habits, rooted in experience, help anyone keep their stock in top shape year after year.
What is the shelf life of Potassium Iodate Diiodate?
The Real-World Value of a Stable Compound
Potassium iodate diiodate, a chemical with niche but critical uses, ends up stockpiled in school labs, emergency kits, food processing plants, and even government stockrooms. The question about its shelf life surfaces every time a new batch arrives or an old canister turns up in a storage review. It’s not just about regulatory compliance. It’s about safety, public health, and money—there’s no use keeping chemicals around if they can’t deliver what’s promised.
Chemical Stability Determines Shelf Life
This compound owes its staying power to robust chemical bonds. Unlike certain organic ingredients or moisture-hungry powders, potassium iodate diiodate usually puts up with temperature swings and the occasional jostle. Keep it in a cool, dry environment away from direct sunlight, and it holds steady.
Most chemical supply firms print shelf lives between three and ten years. This advice comes from studies looking at purity over time. Potassium iodate diiodate rarely breaks down or reacts unless it runs into water or severe heat. Sealed containers extend its usefulness. Once a container opens and lets in air or humidity, the clock runs faster. In person, I’ve pulled out dusty bottles from a school storeroom that sailed through quality checks even after five years. The label said five years, but the actual chemistry held up longer—fresh as ever, provided the lid closed tightly after each use.
Signs of Degradation Matter More Than Dates
Expiration dates try to guarantee performance. Still, actual shelf life depends on more than numbers on a label. Potassium iodate diiodate tells its own story through clumping, unusual smells, or color changes. These signs warn of contamination or breakdown. Testing samples, even with a simple solubility check, gives fast insight.
The United States Pharmacopeia (USP) and similar bodies urge routine reevaluation for chemicals approaching expiry. That guidance saves both effort and expense. Tossing away chemicals before they slip out of spec wastes resources, especially for organizations on tight budgets. Using degraded product could put people at risk, especially in emergency scenarios where potassium iodate diiodate serves as a go-to additive to block radioactive iodine uptake during nuclear incidents.
Storage Conditions: The Unseen Factor
Humidity ruins shelf life fast. Even slight dampness leads to clumping and exposes the compound to reactions. I’ve seen storerooms that follow the rules with desiccant packs, cool dark shelves, and regular checks—the powder there remains workable much longer. Open jars on humid summer days don’t fare as well. This isn’t just about home or lab use—major manufacturers factor in warehouse conditions, packaging quality, and shipment climate to guarantee a batch’s lifespan.
Best Practices and Smart Solutions
Clear labeling, prompt resealing, and regular rotation help eke out maximum useful life. Some organizations set up batch tracking so old stock gets used first. Chemical suppliers, especially those serving schools and hospitals, now include information sheets on handling and stability.
Routine audits, rotating stock, and training staff to spot physical signs of spoilage help ensure only high-quality chemicals make it into experiments, food processing, or emergency supplies. Analytical chemistry offers in-house tests or service labs that can confirm purity if there’s any doubt.
Shelf life isn’t a mystery when basic chemical care becomes habit. With a little attention and smart storage, potassium iodate diiodate delivers on its promise year after year, protecting people and priorities wherever it’s used.
Are there any side effects or hazards associated with Potassium Iodate Diiodate?
Understanding Where Potassium Iodate Diiodate Gets Used
Potassium iodate diiodate doesn’t have much of a household name, but it shows up in places that touch public health and even food safety. Some might recognize it from emergency iodine tablets, handed out to protect the thyroid after nuclear accidents. It also pops up as a food additive in salt, meant to help folks keep their iodine levels steady. Being in the chemical trade, I’ve seen it handled with care and respect because even something so helpful has another side.
Known Side Effects on Health
Swallowing too much potassium iodate or coming into contact with large amounts brings a couple of issues. Ingesting high doses may lead to problems with the thyroid—either an overactive or sluggish gland. Both conditions throw off energy, mood, and long-term organ health. The science lines up: too much iodine gets stored, and that extra can push the thyroid into overdrive or grind it to a crawl, depending on a person’s baseline.
Some people have an allergic response to iodine compounds, especially those with underlying issues—skin rashes, itching, or breathing trouble can follow. In rare cases, high exposure can irritate the gut, leading to stomach pain, nausea, or vomiting. The kidneys don’t always handle big loads of potassium and iodine perfectly, especially if someone already struggles with kidney disease. Many medical authorities, including the World Health Organization, point out these risks but stress incidents stay rare when folks use these supplements as directed.
Hazards at Work and in Laboratories
Handling potassium iodate diiodate in bulk, like inside a chemical plant or a university lab, brings up questions of dust and inhalation. Fine dust can irritate the lungs and eyes, especially with poor ventilation. Direct contact with skin leads to redness or irritation for some workers. Mixing it with other chemicals incorrectly kicks up a risk for fire or toxic fumes. I remember training sessions on what goes wrong when pairs like this get mishandled. Stories circulate of accidental exposure and the importance of proper gloves, eye protection, and working in a fume hood.
Environmental and Safety Concerns
Large spills, especially near waterways, pose local ecosystem problems. High iodine can harm fish and other aquatic life; concentrations build up faster than you’d expect. Waste disposal from labs or industry must follow local codes—down-the-drain approaches cause trouble if not filtered or neutralized.
Keeping Hazards in Check
The good news: most hazards can be managed with careful handling and clear labeling. Storage away from acids and fuels, airtight containers, and regular checks on protective gear make a real difference. For home users taking potassium iodate as a supplement, sticking with doses on the bottle keeps risks low. Cards in emergency packs lay out the safe dosage, never meant for everyday use.
Education does a lot. In workplaces, regular training keeps safety top-of-mind and cuts down on mistakes. For consumers, reading labels and knowing personal health history can prevent most side effects. Doctors and pharmacists help tailor dose advice, especially for folks with kidney, thyroid, or allergy struggles.
Solutions for Safer Use
Open reporting of side effects by doctors and public health groups builds trust. Better public guides—written in plain language—help people understand why and when to use potassium iodate diiodate. Companies can pitch in by producing smaller, clearly labeled packets to prevent confusion or overdose.
From factory workers to families living near nuclear plants, the call remains: handle this compound with respect, know the basics, and speak up if something seems off.
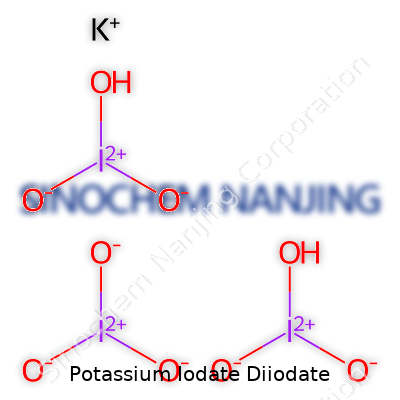

| Names | |
| Preferred IUPAC name | potassium triiodate |
| Other names |
Potassium periodate Dipotassium periodate Potassium metaperiodate |
| Pronunciation | /pəˈtæsiəm ˈaɪəˌdeɪt daɪˈaɪəˌdeɪt/ |
| Identifiers | |
| CAS Number | 13665-23-5 |
| Beilstein Reference | 1713413 |
| ChEBI | CHEBI:88221 |
| ChEMBL | CHEMBL1201616 |
| ChemSpider | 28112201 |
| DrugBank | DB16402 |
| ECHA InfoCard | 03e3b8ad-391a-43c8-b38b-2e3f40ac2aed |
| EC Number | 231-831-9 |
| Gmelin Reference | 30700 |
| KEGG | C18825 |
| MeSH | Potassium Iodate Diiodate" does not have a specific MeSH (Medical Subject Headings) term. The closest relevant MeSH term is "Potassium Iodate". |
| PubChem CID | 166993 |
| RTECS number | VZ2975000 |
| UNII | T38XI40AG7 |
| UN number | UN1479 |
| CompTox Dashboard (EPA) | DTXSID80971960 |
| Properties | |
| Chemical formula | KIO₃·2I₂O₅ |
| Molar mass | 646.61 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 4.088 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -4.660 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 12.4 |
| Basicity (pKb) | 10.25 |
| Magnetic susceptibility (χ) | -92.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.740 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 430.6 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -389.4 kJ/mol |
| Pharmacology | |
| ATC code | A07CA01 |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes serious eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H272, H315, H319, H335 |
| Precautionary statements | IF SWALLOWED: Immediately call a POISON CENTER/doctor. If medical advice is needed, have product container or label at hand. Store locked up. Dispose of contents/container in accordance with local/regional/national/international regulations. |
| NFPA 704 (fire diamond) | 1-0-1 |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 354 mg/kg |
| NIOSH | LM2975000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.01 mg/kg bw |
| Related compounds | |
| Related compounds |
Potassium iodate Potassium iodide Iodic acid Periodate Sodium iodate |

