Potassium Gold Cyanide: A Deep Dive Beyond the Lab Coat
Historical Development
Potassium gold cyanide, known by chemists as K[Au(CN)2], tells a rich story that can’t be separated from the history of precious metals. Chasing gold purity often turned into a dangerous business, even in the days before our modern labs. In the late 19th century, the discovery of cyanidation shifted gold extraction and processing from panning rivers to laboratories and factories. The world saw gold not just as currency or jewelry, but as something that could be shaped at an atomic level. This shift allowed new technologies and economies to be built on the back of a compound that’s as potent as it is controversial. The chemical didn’t show up by coincidence; it came out of necessity, moving gold refining forward at a critical time. Researchers and industry experts of the era, with a mix of ambition and experimentation, drove potassium gold cyanide into daily use even as they struggled to balance its promise and peril.
Getting Close with Potassium Gold Cyanide
As a writer who’s spent years covering industrial chemistry and material science, I know people might expect potassium gold cyanide to look like gleaming gold dust. In fact, it usually comes as a white or off-white crystalline powder. It has no smell, but it comes with plenty of warning. Soluble in water, it hands out gold ions with relative ease, which makes it perfect for delicate processes like gold electroplating. Most people haven’t seen the stuff up close, since its hazards keep it out of regular circulation, but the story of a gold drum rolling out from an electroplating shop stays with you. At its core, it’s a workhorse in microelectronics—circuit boards, connectors, jewelry. Production lines run smoothly thanks to its dependable release of gold ions, which is why technicians become almost protective over every sealed container.
Physical and Chemical Properties
Chemists don’t need to be told twice that cyanide with anything brings danger, but adding gold to the formula only ups the stakes. Potassium gold cyanide melts at moderately high temperatures compared to everyday substances, dissolves easily in water, and keeps its solubility even as conditions shift. Gold sits bonded not just to potassium, but wrapped in cyanide groups, and this tight structure means even small changes—like a dose of heat or a pH swing—can shift how it behaves. In practical terms, handling and storing the compound demands fully sealed equipment and airtight records, especially because airborne dust can pick up on the skin or slip past cloth masks if you’re not careful.
Technical Specifications & Labeling Realities
Walking into an industrial storeroom, you’re unlikely to see containers casually labeled “gold powder.” Regulatory compliance means every kilogram gets precisely labeled with chemical identities, concentrations, hazard codes, and date codes. Technicians track batch numbers, not because bureaucracy loves paperwork, but because the compound’s hazards and value make both security and traceability non-negotiable. In research labs, each jar or flask — usually stored in locked cabinets — gets chemical and risk labeling, not just for compliance but because a single slip in handling or dosing can mean more than a ruined project.
The Gritty Side of Preparation
Potassium gold cyanide doesn’t arrive pre-made from nature; even the word “synthesis” doesn’t quite capture the effort that goes in. Gold metal reacts with potassium cyanide and an oxidant, often oxygen or hydrogen peroxide, in controlled conditions. Those aren't casual reactions. Every step happens behind glass shields and under ventilated hoods, since cyanide fumes and splashes aren’t forgiving. Seasoned chemists will describe the tension of running these reactions — the need to strike just the right ratio and temperature, ensuring gold complexes fully and leaves no dangerous byproducts drifting in solution. In plating shops and research labs, technicians guard procedural details almost like old family recipes.
Chemical Reactions & Modifications
Potassium gold cyanide’s chemistry shows how deep these compounds can go. You want gold? You need to coax it out of the cyanide’s grip—usually with electric current, which deposits gold exactly where it’s needed. This fine control runs the show in microelectronics. Chemists sometimes alter operating pH or add other chemicals, tailoring the plating baths to give different finishes—matte or shiny, hard or soft. Sometimes the gold comes back out for refining or recovery; those processes use zinc dust or other reducing agents to nudge metallic gold back into the light. Across the board, these changes don’t just tweak performance—they also change how much risk operators face, how much waste gets made, and what regulators allow.
What’s in a Name?
Even with a serious-sounding title, potassium gold cyanide picks up plenty of synonyms on vendor lists and in the literature—names like Dicyanoaurate(I), Gold(I) potassium cyanide, or potassium dicyanoaurate. Some users shorthand it to PGC in technical discussions. The array of names reminds you this isn’t just a compound for chemists, but a staple that moves through languages and continents, showing up anywhere people need reliable gold finishes.
Staying Safe: Standards and Daily Routines
Every person I know who handles this compound develops habits that stick for life. Double gloves, fume hoods, spill trays—these aren’t up for debate. Cyanide spells disaster in sloppy hands, and regulators hold operations to strict controls—training, waste management, air monitoring, and emergency response plans make every step deliberate. Industry safety data sheets—sometimes as thick as a brick—sit within arm’s reach, and even routine procedures involve buddy systems and immediate spill cleanup. Nobody gets casual, because a single whiff of cyanide, or a small spill, has sent people to the hospital. Labs and plating shops that handle the compound long-term tend to foster cultures where every technician checks the other’s gear, and nobody skips maintenance or testing ventilation systems. People remember famous poisonings and industrial accidents, drawing lessons that turn into procedures and cautionary tales for the next generation.
Potassium Gold Cyanide at Work
You can’t visit a major microchip factory, jewelry production line, or aerospace electronics plant without running smack into potassium gold cyanide’s legacy. Electroplating demands ultra-smooth, tarnish-resistant gold layers for everything from cell phone connectors to satellite relays. Jewelry makers depend on the compound for thin, rich coatings that deliver gold’s luster without breaking the bank. In medical technology, the need for biocompatible surfaces puts potassium gold cyanide in the mix for specialized devices and sensors. People sometimes forget the compound’s role inside diagnostic toolkits, telecom hardware, and software encryption hardware, but its reach covers sectors that keep cities working.
On the Frontiers: Research & Development
Across academic research hubs and private labs, potassium gold cyanide continually sits in the crosshairs of innovation and scrutiny. Scientists—especially those who grew up reading about waterway poisonings—push for alternative gold deposition methods with less risk. Green chemistry labs experiment with ionic liquids, amino acids, and other gold complexes. Some projects chase nanostructured gold for quantum computing or tailored plasmonics, where potassium gold cyanide gets used as a reliable starting point. Researchers in recycling explore chemical tweaks to extract gold from electronics without generating cyanide waste, a holy grail that would rewrite electronic waste management. Every improvement puts pressure on the industry to rethink how and why the compound gets used, even if no replacement yet fully delivers gold’s conductivity or elegance quite so neatly.
Diving into Toxicity Research
No one takes potassium gold cyanide’s hazards lightly. Its toxicity, rooted in the cyanide ion, raises environmental and human safety challenges that go far beyond the lab bench. Cyanide interrupts cellular respiration, and accidental exposure—via inhalation or skin—can be fatal. Regulatory bodies set tight exposure limits and require constant air and waste monitoring. Toxicologists track incidents and investigate chronic exposure risks for operators, while environmental scientists study leaks and spills around old mining sites and plating facilities. Everyone involved—from policymakers to production managers—clamors for secondary containment, real-time detection, and safer cleanup solutions. Personal monitoring badges, rigorous medical checks, and strict waste-water treatments form the backbone of real-world operations. My own conversations with safety experts reveal a plain truth: one lapse in vigilance can rewrite a whole community’s story.
Looking Forward: Future Prospects
Potassium gold cyanide’s future won’t unfold in isolation. Industry insiders, researchers, and policymakers trade ideas about “greener” gold processes. Investments flow into next-generation plating baths, replacements that offer lower toxicity, and closed-loop processing systems that turn every gram of gold and cyanide back into raw material. Startups target new gold complexes that break down benignly and promise easy recycling; regulatory shifts push plants to upgrade systems, and universities launch partnerships on “safe by design” principles. As public concern around chemical exposure grows and the scale of electronics waste keeps climbing, the smart money rides on safer, more circular systems. Potassium gold cyanide, with its proven reliability and extreme caution in use, keeps drawing interest, questions, and investment. Every breakthrough in process safety or waste recovery adds a new chapter, and the world waits to see whether greener options will let industry hold onto quality while letting go of the risks that define this fabled compound.
What is Potassium Gold Cyanide used for?
Not Just a Compound: Potassium Gold Cyanide in Everyday Life
People often walk around with gold-plated jewelry, phones decked with gold contacts, or even medical devices in their bodies, rarely giving a thought to the chemistry making it all possible. Potassium gold cyanide, a chemical with a daunting name, plays a quiet but key role in these parts of modern life. Working in a manufacturing environment, it's easy to spot bins of tiny electronic components being loaded day after day into plating baths. The bright, even layer of gold that covers these parts isn’t magic. It’s a result of careful chemical work—much of it involving potassium gold cyanide, usually known in the trade as PGC or simply “the gold salt.”
The Backbone of Gold Plating
Gold isn’t just for jewelry. Today’s smartphones, computers, and circuit boards rely on gold to carry signals. Gold resists corrosion, which keeps electronics running for years. Factories use potassium gold cyanide as the go-to ingredient in gold plating baths. The chemical dissolves in water, releasing gold ions. When an electric current passes through the bath, atoms of gold leave the solution and stick to the surface of the object being plated. This gives us durable, reliable, and attractive coatings, making sure that critical connections in everything from dental crowns to microchips last as long as possible.
Real Reasons for Care
Common talk around plating and mining circles isn’t just about shiny finishes. Cyanide in any form means tough safety and environmental rules. I’ve seen workers handle potassium gold cyanide with extreme caution. Companies train staff, monitor air and waste water, and triple-check shipments. Nobody wants a legacy of health hazards or a disaster. In the past, accidents from poor handling have poisoned workers and contaminated land—history gives plenty of reminders, like incidents in older mining regions. That sometimes pushes businesses to find other chemistries or add better waste treatment.
Meeting Demand Responsibly
Every year, phones and gadgets use more gold, most of it in tiny amounts. Recycling now plays a big part, as old devices get melted down for their precious metals. Potassium gold cyanide goes into some of those recycling processes, too, dissolving gold from used parts before it gets refined and reused. In plating shops, I’ve seen more closed-loop systems pop up. These setups recover unused gold and cyanide from rinse waters, lowering both pollution and cost. Newer filters and sensors catch leaks or overflows fast, alerting workers before problems spread.
Tomorrow’s Technology and Old Chemistry
As companies race to miniaturize electronics and push medical implants deeper into the body, the importance of pure and reliable gold layers only grows. Some labs explore new chemicals that work faster or pollute less, but so far, potassium gold cyanide keeps its place because it delivers what engineers and consumers demand. Regulators and scientists call for new ways to recover gold from waste and limit cyanide’s dangers, pushing the industry to stay sharp. Governments and watchdog groups also keep pressure on, making safe handling part of the cost of doing business in any country that values both technology and public well-being.
Looking Beyond the Gold
Potassium gold cyanide may sound distant, but it links the dazzling world of gold to real problems and real progress. Whether in a warehouse or a lab, its use shows how even basic chemistry shapes everything from global supply chains to the phone in your pocket. Keeping safety and waste in mind doesn’t slow down technology—it guarantees that progress is something worth trusting.
Is Potassium Gold Cyanide hazardous or toxic?
Getting Clear About Potassium Gold Cyanide
Potassium gold cyanide turns up mainly in the world of gold electroplating. This chemical helps coat jewelry, electronics, and some fancy watch parts with a thin layer of gold. People who handle it, mostly in manufacturing settings, should know what they're working with, because this is not just another harmless powder. What’s in the name tells a lot. “Cyanide” links back to deadly chemicals with a long history of accidents and strict safety rules. The compound brings together potassium, gold, and cyanide—ingredients that, once mixed, ask for care and respect.
Toxicity Matters
Chemicals containing cyanide make almost everyone uneasy, and with good reason. Cyanide can stop cells from using oxygen. If someone breathes it or even gets enough on their skin, it can lead to dizziness, shortness of breath, convulsions, and, quickly, death. Potassium gold cyanide stays true to that family legacy. The gold part does not soften the risk. Although it finds use in labs and factories, it carries a strong toxic punch. Authorities like the Centers for Disease Control and Prevention list it as acutely toxic. It doesn’t take much exposure—any inhalation or accidental ingestion calls for swift medical help.
How Real Are the Dangers?
It’s not just the direct effects. Potassium gold cyanide, even in small amounts, threatens the environment. If someone disposes of it carelessly, it can contaminate water and soil, putting wildlife in harm’s way and risking the food chain. Industry workers know this, and most companies running plating operations build many checks and protocols. Workers put on gloves, goggles, and even full-face masks to keep it off skin or out of the lungs. Spills trigger alarms, and every drop gets tracked from start to finish. Yet, in places where rules slip or corners get cut, the dangers move from theory to reality fast.
Workplace Experience and Proper Handling
Having spent time in facilities where gold plating happens, the seriousness of potassium gold cyanide became clear early on. Employers hold safety meetings, organize drills, and assign chemical safety training. Even then, a single slip—a leaky container, someone touching their face before washing up—can lead to a sudden medical emergency. Old stories still make the rounds about past incidents, each underlining how nothing about this material invites casual treatment. Those who use this chemical stick to strict routines, from double-sealing vessels to running waste through neutralizing tanks before leaving the building.
What Can Make Things Safer?
Companies and governments set rules for handling potassium gold cyanide. Some places now substitute safer compounds or run plating in sealed systems, hoping to cut accidents and exposure. Others push for regular health checks for employees or install sensors to pick up even the smallest cyanide leaks. Education works, too. People need clear labels, training, and a culture where everyone feels comfortable reporting problems—no matter how small or embarrassing. Having spent years around plating shops, it’s clear the greatest danger comes not from the chemical itself, but from forgetting just how hazardous it remains.
Final Reflection
Potassium gold cyanide is, without question, hazardous and toxic. Its use calls for constant vigilance, both to protect workers and the world outside the factory gate. Anyone handling it owes that to themselves and their coworkers. Knowing what it is—and never letting that slip from mind—is key to staying safe.
How should Potassium Gold Cyanide be stored and handled?
Understanding the Risk
Potassium gold cyanide often shows up in the world of gold plating and electronics. In workshops where quality plating matters, this compound gets treated with the kind of respect given to a loaded electrical panel. It’s not the gold that brings worry, but the cyanide—infamous for its ability to do serious harm in tiny amounts. The story of potassium gold cyanide mirrors the rule of the shop: “Don’t take shortcuts that cost you more later.” A single mistake with chemicals like this doesn’t lead to a second chance.
Storage: No Room for Guesswork
Years spent working around hazardous materials hammered in the lesson that “close enough” becomes the weak link—the place where accidents dig in. Potassium gold cyanide asks for careful storage. Sealed, clearly labeled containers lock away the risk, but placing them in a secure, cool cabinet adds another protective wall. Storage away from acids and moisture blocks dangerous reactions. Even a minor spill or a vapor buildup creates danger, not just to the handler, but to anyone nearby.
What sticks with me from safety training is the importance of double-checking labels. A hasty swap or confusion over a lookalike container is simple to fix early, impossible to take back after cyanide escapes. A buddy system for handling and a thorough log of where, when, and who moves the potassium gold cyanide make a difference—these steps cut down accidents often caused by routine boredom.
Handling: Each Move Matters
I watched an older technician pause before opening a chemical bottle—not because he was slow, but because he understood the risks firsthand. Gloves that resist both chemicals and punctures, goggles tight against the face, and a protective coat offer the first line of armor. A respirator, rated for toxic dust and vapor, turns necessary if there’s any risk the powder or vapor escapes.
Mix-ups become rare if workers stick to one workbench and clean the surface before use. Signs warning about cyanide risk and spill kits packed right beside every station save minutes during emergencies—minutes that count. Regular drills where workers neutralize mock spills with hydrogen peroxide mean fewer people freeze if something goes wrong.
Solutions by Example
Some shops install automated chemical dispensers, taking human error out of the equation. Others invest in smart storage with access codes and alarms. These aren’t luxuries. They prevent someone from unknowingly handling cyanide after hours or during cleaning. Clear communication between management and workers, with updated training and easy-to-find instructions, keeps safety culture alive.
I knew a plating supervisor who treated every chemical transfer as if his own family might walk in. That meant fresh gloves every time and every bottle checked twice. This mindset means not just trusting procedures, but slowing down and watching for the trickle of powder or the whiff of almond odor—a warning sign that can’t get ignored. If potassium gold cyanide finds its way into the wrong hands or the wrong place, there’s no “oops” that puts things right.
Staying Proactive
Success with potassium gold cyanide means facing risk head-on and building best practices into regular habits. From what I’ve seen, the safest shops tell stories about past close calls, and they listen to each worker’s concerns. Truth gets spoken aloud, and warnings get repeated, because as long as cyanide’s on hand, everyone shares responsibility. That level of attention and respect for the rules sets the gold standard for safety.
What is the chemical formula of Potassium Gold Cyanide?
Unpacking the Formula: K[Au(CN)2]
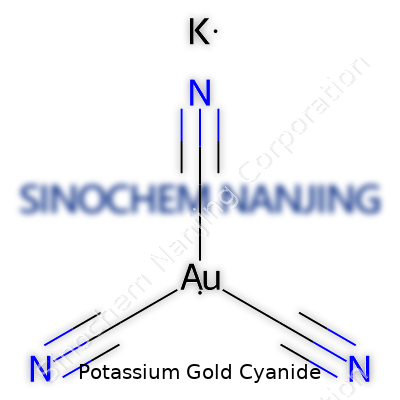
Potassium gold cyanide comes with a chemical formula of K[Au(CN)2]. This isn’t just a random arrangement of elements on the periodic table—it’s something that underpins one of the largest and most useful industrial processes. For those with some lab experience, spotting potassium (K), gold (Au), and the cyanide group (CN) brings to mind both opportunity and risk. Anyone who’s worked in electrochemistry or jewelry manufacturing has run into this compound, whether working with gold plating solutions or in an undergraduate inorganic chute.
What Makes K[Au(CN)2] Unique
This chemical allows companies to deposit gold onto surfaces with control down to the micron. Every computer I’ve owned—with every flex cable and tiny gold-plated contact—relies on this molecule. The cyanide ions don’t just bind gold up, but they also carry it in a stable way that helps produce consistent results in gold plating baths. In real-world terms, potassium gold cyanide is the gateway to thin yet resilient gold films—without clogging the circuit or blowing anyone’s production budget.
Why Safety and Handling Shape Its Use
Working with potassium gold cyanide is not just about knowing the formula. It helps to remember what makes it unique. The inclusion of cyanide brings big risks. Cyanide compounds can kill cells quickly—something every safety orientation in the chemical industry drives home. My own short stint in an undergraduate synthetic lab included strict protocols when cyanides were involved: gloves double-checked for holes, fume hoods humming loud, emergency antidote kits within arm’s reach. No one shrugs off these steps if they’ve heard emergency showers go off thanks to a splash.
That safety culture isn’t overkill. Fatalities and poisonings documented in case reports push industries to train staff, reduce exposure, and invest in monitoring. Facilities that use potassium gold cyanide often rely on automatic dilution systems, air extraction, and closed-loop recycling of any cyanide wastes. It’s not just a company’s legal duty—nobody wants the moral stain of a cyanide leak or gold-laced runoff reaching a river.
Beyond the Lab: The Broader Impact
K[Au(CN)2] doesn’t just stay inside refineries and plating tanks. It enters the conversation about responsible gold sourcing and waste management. Growing up in a mining town, the environmental legacy of improper chemical disposal would come up at every community meeting. Local streams polluted in the twentieth century still carry echoes of those risks. Today, industry pushes through barriers with closed-loop systems and by researching alternative, less hazardous ligands than cyanide. Change comes slowly, but continuous improvements save both lives and ecosystems.
Potassium gold cyanide’s formula, K[Au(CN)2], may seem simple on paper. Its widespread use in electronics and jewelry is only possible thanks to a complex mix of chemistry, logistics, and regulation. Engineers and workers alike keep learning from both past accidents and advances in green chemistry. These lessons shape how the world makes luxury goods and essential devices, all while aiming to keep communities and staff safe.
What safety precautions are needed when using Potassium Gold Cyanide?
Why So Much Fuss?
Potassium gold cyanide doesn’t work like the chemicals you casually keep under the sink. In gold plating shops and electronics labs, this compound lets people coat items with smooth, shiny layers of gold. The catch? It’s dangerous on multiple levels. There’s cyanide in the mix. Just a small amount can be fatal if mishandled. Inhaling dust, spilling liquid, or even touching it with bare skin opens the door to a real emergency.
Personal Protective Equipment Goes Beyond Gloves and Goggles
Wearing gloves is common sense for a lot of chemicals, but with potassium gold cyanide, a thin latex glove doesn’t cut it. It always makes sense to reach for nitrile or neoprene gloves, thick aprons, long sleeves and tight-fitting goggles. Not only should these clothes cover skin completely, but they should also get swapped out as soon as they show the first sign of wear. If you ever get a splash, you need an easy way out: showers and eyewash stations positioned within seconds of your workbench reduce panic and harm.
Breathing and Handling: Limit Every Exposure
Masks matter. A good fume hood helps keep vapors away from the face. Sometimes strong ventilation is all that stands between a safe day and a tragic accident when working with dust or liquids. Hand-to-mouth actions make trouble; eating, drinking and smoking never mix with this job. Training matters most—a worker who sees a spill must know how to react instantly, not fumble for a manual.
Spills, Storage, and Simple Habits
Years in manufacturing taught me how easily small mistakes grow when people get comfortable. Some folks let jars pile up on benches, forget to label bottles, or ignore rusty containers. With potassium gold cyanide, any of that invites trouble. Use containers that can’t corrode. Tight lids, clear warning signs, and materials never left in sunlight lay the ground rules for safe storage. Watch out for water, acids and oxidizers nearby—contact with these can quickly trigger the release of toxic hydrogen cyanide gas.
Simple Solutions Still Matter
Double-checking math before making up solutions, working in teams, and documenting every step creates an extra layer of security. Keeping a spill kit and cyanide antidote (usually amyl nitrite or sodium thiosulfate) within arm’s reach matters more than a fancy protocol. Everyone in the area should recognize what potassium gold cyanide looks like and know what action to take in case of a leak or splash. Emergency drills should happen regularly, not just on paper but in real-life practice sessions.
Care Means Respecting Risk Every Day
Chemistry never grades on effort—it deals in hard consequences. People who treat potassium gold cyanide with the respect it demands keep injuries at zero and businesses out of the headlines. Respect comes from understanding the risk, updating skills often, and refusing shortcuts. The world uses this compound to make things beautiful and durable, but that only works if safety stays the number one priority every day.
| Names | |
| Preferred IUPAC name | potassium dicyanoaurate(I) |
| Other names |
Gold potassium cyanide Dicyanoaurate(I) potassium Potassium dicyanoaurate Potassium gold(I) cyanide Potassium cyanoaurate |
| Pronunciation | /pəˈtæsiəm ɡoʊld saɪəˌnaɪd/ |
| Identifiers | |
| CAS Number | 13967-50-5 |
| Beilstein Reference | 3539614 |
| ChEBI | CHEBI:33361 |
| ChEMBL | CHEMBL1201138 |
| ChemSpider | 80627 |
| DrugBank | DB14506 |
| ECHA InfoCard | 100.969.433 |
| EC Number | 25404-88-6 |
| Gmelin Reference | Gm. 825 |
| KEGG | C18714 |
| MeSH | D017683 |
| PubChem CID | 24957 |
| RTECS number | MW6825000 |
| UNII | 69J9HK56F8 |
| UN number | UN1640 |
| Properties | |
| Chemical formula | KAu(CN)2 |
| Molar mass | 339.00 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 3.572 g/cm3 |
| Solubility in water | Soluble |
| log P | -0.38 |
| Vapor pressure | Negligible |
| Basicity (pKb) | pKb = 10.0 |
| Magnetic susceptibility (χ) | -29.5e-6 cm³/mol |
| Refractive index (nD) | 1.44 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 237.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -509.6 kJ/mol |
| Pharmacology | |
| ATC code | V03AB33 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; contact with acids liberates very toxic gas; may cause respiratory and central nervous system effects; very dangerous to the environment. |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P260, P261, P262, P264, P270, P271, P273, P280, P284, P301+P310, P302+P352, P304+P340, P305+P351+P338, P310, P320, P330, P405, P501 |
| NFPA 704 (fire diamond) | 3 3 2 OX |
| Lethal dose or concentration | LD50 (oral, rat): 2.5 mg/kg |
| LD50 (median dose) | LD50 (median dose) Oral - rat - 10 mg/kg |
| NIOSH | TTQ197 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Potassium Gold Cyanide: 0.05 mg(Au)/m³ (as Gold, OSHA) |
| REL (Recommended) | 2 mg/m3 as CN |
| IDLH (Immediate danger) | IDLH: 25 mg Au/m³ |
| Related compounds | |
| Related compounds |
Chloroauric acid Gold(III) chloride Potassium dicyanoaurate Sodium gold sulfite |

