Potassium Fluoroacetate: The Double-Edged Legacy
Historical Development
Potassium fluoroacetate, often talked about as “1080,” reveals a story that stretches back more than a century before headlines began to call it notorious. The tale began in the early 1900s when scientists tried to find new ways to manage pests that wrecked crops or spread disease. By the 1940s, researchers figured out that naturally occurring fluoroacetate found in certain African and Australian plants carried toxic power strong enough to kill mammals in trace doses. Chemists unlocked a way to synthesize potassium fluoroacetate, leading to its rise as a weapon in pest control and, inevitably, as a catalyst for debate about how societies manage threats to agriculture and biodiversity. Nobody missed the effectiveness of the compound, but tales of collateral damage—livestock lost, native predators poisoned—quickly trailed the rising popularity of 1080.
Product Overview
Potassium fluoroacetate entered market shelves as a white, odorless powder that many found disarmingly plain in appearance. It dissolves easily in water, blends without fuss, and carries an almost invisible touch—which feeds the unease surrounding its handling. Governments across the globe labeled it as a restricted poison, aware of both demand and danger. Its simple structure masks a potent biological effect: after being swallowed, the compound blocks cellular metabolism, causing animal deaths after hours or days. Labeling laws strictly require warnings about its risks, reflecting bitter lessons learned from its legacy.
Physical & Chemical Properties
This salt carries a low molecular weight, with granules or crystalline powder forms that make it easy to blend into baits or dissolve for various uses. Its chemical stability keeps it effective during transportation and storage, though it stubbornly resists breakdown in dry conditions, raising concerns about contamination and residual presence in treated areas. Potassium fluoroacetate does not release obvious fumes or smells, which makes accidental exposure a constant risk, especially when regulations slip or oversight lapses.
Technical Specifications & Labeling
Regulations shape much of the technical side; the law demands clear hazard symbols, tough packaging, and explicit instructions for disposal and storage—because just a few grams can deliver lethal effects. I have spent time on farms and seen labels decorated with skull-and-crossbones, bold warnings that leave no room for ignorance. There’s wisdom in these stern visuals. Mishandling the powder risks not only wildlife loss but accidental poisonings, sometimes with tragic consequences. Labeling emphasizes keeping it out of children’s reach and nowhere near livestock food or water supplies.
Preparation Method
Commercial synthesis follows a route involving the reaction between chloroacetic acid and potassium fluoride. The process demands careful temperature control and scrupulous cleanliness, as even tiny leaks pose a hazard to those working around the material. Every gram handled in the plant reinforces the need for top-notch ventilation, protective gear, and rigid discipline in chemical hygiene. Industrial chemists do not take shortcuts when dealing with this salt, given its reputation for deadly efficiency.
Chemical Reactions & Modifications
Potassium fluoroacetate doesn’t lend itself to casual tinkering in the lab. Once in the body, it enters the citric acid cycle, where it gets enzymatically converted to fluorocitrate—a serious metabolic saboteur. Scientists have dabbled with formulations to control release rates and limit environmental persistence, but the basic molecule refuses to drop its toxicity. Efforts to create antidotes have mostly hit dead ends; interventions work only in the earliest stages, which makes prevention far more practical than cure.
Synonyms & Product Names
Even outside technical circles, the term “1080” brings a shudder, especially among folks living near areas with official baiting programs. The chemical’s synonyms—potassium fluoroacetate, fluoroacetic acid potassium salt, and a few trade names—populate regulatory lists and academic papers. Field workers use the shorthand “1080” with a knowing nod, never forgetting that this is no harmless tool but a product that reshapes ecosystems. Public knowledge lags behind, leaving many unaware of just how many aliases this compound shares.
Safety & Operational Standards
Every safety guideline goes beyond mere suggestion. Mask, gloves, sealed containers, decontamination procedures—these become religious rules in facilities I have visited. In many countries, transport and handling demand special permits and inspections. Field workers keep emergency antidote kits close when baiting, conscious of the risk that even minor mistakes bring to themselves and to anyone in the vicinity. Mistakes, even from a moment’s inattention, do not forgive. The best defense remains rigorous training, supported by strong regulatory oversight and persistent public education campaigns.
Application Area
Most of the world knows potassium fluoroacetate through its grim role in pest management. Australia and New Zealand, in particular, use it as a last line of defense against invasive mammals like foxes and stoats, who threaten native wildlife. In these places, the compound’s reach affects farms, forests, and national parks, stirring both public backlash and practical necessity. Conservation groups and rural communities lock horns over its use, yet land managers often have no substitute when invasive species threaten to tip ecological balances past recovery. No one romanticizes its application; users choose it because, for some problems, no softer tool exists.
Research & Development
New research tries to find ways around the blunt force of 1080, dreaming of targeted approaches that won’t spill over into native animals and friendly species. Some work focuses on biodegradable carriers or more tightly controlled bait delivery techniques. Others explore genetic resistance for non-target creatures, or smart traps that only allow access to the real culprits. I have seen researchers hunched over lab benches, hoping to develop a magic bullet that saves crops and ecosystems without harming the helpers. The work crawls forward, inch by inch, faced with the stubborn fact that fluoroacetate remains effective because of its broad-spectrum toxicity—a trait that is both its value and its peril.
Toxicity Research
The grim track record of potassium fluoroacetate keeps toxicologists awake at night. Its effect is notorious, causing convulsions, cardiac failure, and a slow death in most mammals, birds, and sometimes even insects. No reliable antidote exists for advanced poisoning. Researchers across decades tracked how the compound moves through food webs, how it lingers in soil and water, and how even trace exposures ripple through populations. Wildlife biologists raise alarms, waving field reports of declining predator numbers and off-target losses. Many call for better tracking technologies and more comprehensive monitoring in treated areas. Nobody in science ignores the human and ecological costs associated with accidents or misuse.
Future Prospects
Potassium fluoroacetate stands at a crossroads. Communities call for alternatives that respect both the need to control pests and the rights of wildlife, pets, and people. Research teams keep working, attempting to unravel the compound’s action in hopes of blunting its harm or finding new molecules with fewer drawbacks. Regulatory agencies regularly review its use, often adding restrictions or experimenting with buffer zones and seasonal bans. Investment in biological controls, immunocontraceptives, and habitat management grows each year. Society faces a simple but stubborn truth—pest problems rarely have perfect answers. As science inches forward, the hope is that future tools might limit the pain inflicted by well-meaning but hazardous inventions like potassium fluoroacetate, and that we’ll learn to balance necessity with responsibility.
What is potassium fluoroacetate used for?
Straightforward Science, Straightforward Risk
Potassium fluoroacetate rarely shows up in headlines unless you hear about a poisoning scare or a wildlife debate. Its presence in farming stretches back to the 1940s, and in places like Australia and New Zealand, you’ll see it sold under the name “1080.” This isn’t one of those chemicals with a long shelf of household uses. Its job is crystal clear: it’s used for killing pests, specifically invasive mammals that threaten native wildlife and farmland.
Pest Control That Pulls No Punches
I've seen farmers talk about their struggle with introduced species—rabbits, possums, foxes, and rats—tearing through crops or threatening unique birds. In New Zealand, possums wreck native forests and carry disease that hits livestock hard. In Australia, rabbits still cause chaos despite years of fighting back. Old-school traps or fencing don’t always work for vast areas, and labor costs eat through budgets. Potassium fluoroacetate can control these pests quickly, especially when baits are dropped from planes across remote terrain.
A Tool With Sharp Edges
There’s no gentle way to put this: potassium fluoroacetate works by disrupting how animals’ cells get energy. It tricks the body, blocks the Krebs cycle, and starves cells from the inside out. For targeted pests, it’s devastatingly effective. That’s the reason it’s still around, even after decades of health and animal welfare debate. It doesn’t linger in the environment like some old pesticides. Reports show it breaks down fast in water and soil, fading out rather than building up.
Living With The Danger
No farmer or rancher I’ve met wants to poison the wrong animal. Yet this isn’t foolproof. There’ve been stories of working dogs or native creatures dying after eating bait by accident. Antidotes don’t exist, so the stakes run high. In the lab, handling this chemical calls for masks, gloves, and solid training. On open land, signage and public notices try to keep people and pets away from treated areas, but accidents can’t always be stopped.
Pressure For Change
People want safer answers. Animal welfare groups, vets, and indigenous leaders have called out the suffering caused by 1080. Some researchers push for smarter delivery—baits that attract only certain species or break down instantly after rain. New Zealand spends public money tracking how the bait spreads and follows up with surveys on native wildlife. There’s research into alternatives: gene drives, species-selective toxins, and even birth control for pests. Progress is slow since alternatives either don’t hit the right pests or cost more than farms can handle.
Responsibility, Not Convenience
Communities caught between crop losses and fears for nature need more than easy answers. It’s clear that potassium fluoroacetate isn’t some lazy fix; it’s a tool chosen for tough spots where nothing else works well enough. That means the conversation should focus on transparency, strict controls, and relentless research into better options. No one who works the land takes the risk of these poisons lightly—they use it with their livelihoods and neighbors in mind, waiting for something better to finally arrive.
Is potassium fluoroacetate dangerous or toxic?
Understanding Potassium Fluoroacetate
Potassium fluoroacetate grabs attention for all the wrong reasons. This chemical, known as 1080, belongs in the family of toxins created from combining fluoroacetate with potassium. For decades, it’s traveled a controversial path, mainly as a pest control tool in countries like New Zealand and Australia. A lot of people have heard about its role in killing invasive animal populations, but fewer understand how it functions. Once ingested, 1080 interferes with the body’s energy production at the cellular level, shutting down essential functions that keep animals (and people) alive.
The Science Behind Its Toxicity
Put simply, even a very small dose of potassium fluoroacetate brings out serious harm. It disrupts the Krebs cycle, a fundamental process that releases energy from food. Cells start failing, organs quit, and without quick intervention, death follows. Unlike some poisons that show obvious signs, 1080 sneaks up. Nausea, confusion, drooling, irregular heartbeats—the symptoms mirror dozens of possible medical issues. By the time someone realizes exposure happened, the chemical has spread too far for easy treatment.
Risk to Wildlife, Pets, and People
In New Zealand, native birds evolved in the absence of mammalian predators, making them vulnerable. Government agencies use 1080 to keep numbers of animals like stoats or possums in check. It’s effective, but often brings collateral damage. There’ve been plenty of stories about dogs or livestock dying after accidental exposure. Just a few milligrams prove deadly for a medium-sized dog. Hunters sometimes lose working dogs; farmers lose valuable stock. Not every animal succumbs, but every exposure carries a risk. What’s worse, poisoned carcasses linger in the environment and create chain reactions—other predators eat the remains, and the cycle repeats.
Human Health and Community Concerns
No mainstream medical community claims potassium fluoroacetate is remotely safe for human handling. Accidents in labs or on the job usually end with emergency decontamination. Rural families worry about traces seeping into water or dust, and skepticism grows every time a pet suffers after a poisoning operation nearby. In 2002, an Australian sheep station worker died after accidentally ingesting 1080. Medical journals reported slow symptom onset and the challenge doctors faced figuring out what happened. This event—and others—made it clear that vigilance and respect for the chemical's dangers must remain high.
Alternatives and Solutions
Debate rages about whether to ban 1080 outright or invest in safer strategies. Some communities look at traps, aerial shooting, or targeted hunting to control pests. These methods cost more and need ongoing labor, but don’t contaminate the environment the same way. Research in genetic technologies hints at future tools for pest suppression. Reducing reliance on chemical methods makes sense—once poison leaves a sack or helicopter, control vanishes.
Transparency builds trust. Notifying landowners and providing maps where toxins get distributed gives people agency to protect animals, families, and water. Using trained teams to recover animal carcasses after drops helps keep unintended poisoning down. Public debates should stick with data—no chemical works without risk, but not every risk must be accepted in modern conservation. People need to push for new science, thorough oversight, and honest conversations that respect every living thing at risk in these operations.
How should potassium fluoroacetate be handled and stored?
Risk Comes from Familiarity
Anyone who’s ever worked around hazardous substances knows that routine can create blind spots. Potassium fluoroacetate crops up most often in pest control. Some countries use it in baits designed to limit populations of damaging mammals. It has earned a reputation for being efficient. Unfortunately, this efficiency means it also brings risk to health and even to the environment. One accidental exposure—skin contact, inhalation, or ingestion—can bring life-threatening consequences. Death from acute poisoning leaves little room for error. I’ve seen more than one technician in rural settings, even with training, become a little too comfortable around this powder. Over the years, some even stopped double-checking PPE. Those moments have consequences nobody can afford.
Not Just Storage—Isolation and Restriction Matter
Potassium fluoroacetate deserves a locked, dedicated storage area. Ordinary chemical cabinets don’t cut it. Storage should keep unauthorized hands out—curious children, untrained workers, anyone lacking clearance. This means secured rooms, with tight inventory control and a sign-in system. After decades on farms and in labs, I’ve found that the best facilities always install alarms or cameras near storage. When someone tries to get clever and bypass protocol, there’s a digital record. Fire-resistant construction lowers the likelihood of disaster if something catches fire.
Labels Save Lives When They’re Clear
Chemicals come and go in workplaces. The only constant is a bright, clear hazard label. Bold warnings like “Deadly Poison” or “Danger: Do Not Touch Without Authorization” make staff stop and pay attention. No tiny print or faded stickers—relabels happen immediately when damage appears. These aren’t over-the-top steps. They keep someone from absentmindedly grabbing a container and bringing tragedy home.
No Substitute for Full Protective Gear
Nitrile gloves, goggles, half-face respirators, full suits—the modern kits seem excessive until someone winds up in the emergency room after a spill or splash. Using potassium fluoroacetate, I never relied on a single barrier, and neither did anyone I respected in the industry. Mixing and preparing it, staff kept to strict routines: double gloving, never working alone, thorough decontamination before leaving. There’s no place for shortcuts.
Environmental Defense: More Than Locking the Door
Sealed containers don’t only slow down spills; they stop leaching into the ground. Secondary containment trays protected floors and drains in every lab I worked with. If even a few grams end up in the soil, local wildlife takes the hit, and water sources risk contamination. Teams always stored potassium fluoroacetate away from food and water, not just on other shelves, but clear across a locked building. Once I saw a storage shed flood during spring rain, and every sealed drum had to be inspected—no taking chances with potential leaks.
Training Goes Beyond One Seminar
Knowledge must stay fresh. Annual retraining, unannounced drills, and consistent supervision remind everyone that potassium fluoroacetate doesn’t tolerate lapses. I’ve participated in tabletop exercises where the “scenario” involved an unknown spill. The person who identified it first saved everyone a lot of pain. Ongoing education keeps the statistics at bay.
Accountability—Every Step, Every Dose
Strict logs, use-by-use recordkeeping, and counting all containers before and after each shift provide real oversight. The best programs invite outside review—sometimes inviting experts from other facilities to point out overlooked weaknesses. No corner gets cut where trust in chemical stewardship matters. Potassium fluoroacetate doesn’t give do-overs; proper handling only works when attention never wavers.
What are the symptoms of potassium fluoroacetate poisoning?
The Reality of Fluoroacetate Exposure
Potassium fluoroacetate, more commonly known as 1080, sits high among dangerous chemicals found in pest control. Growing up in a rural area, neighbors often relied on baits laced with toxins to handle invasive animals. Not many knew about the silent risks to livestock, pets, and, in rare events, people. A small amount, smaller than most imagine, can spell trouble once it enters the body.
Recognizing Early Signs of Trouble
The body doesn’t have a natural way to break down fluoroacetate. Poisoning symptoms don’t waste much time. Nausea and vomiting show up quickly. Animals tend to stagger or fall, drool, and seem confused. People might complain of belly pain, dizziness, or sweating. Stomach cramps feel sharp. Breathing turns shallow and irregular. In my younger years working with rescue teams, I saw pets collapse, legs twitching as toxins took hold. No matter the species, early symptoms often look like severe gastroenteritis mixed with weakness and anxiety.
Progression of Toxic Effects
As poisoning progresses, effects move from stomach troubles to full-blown systemic chaos. Heart rhythm turns abnormal. Seizures hit hard and fast. Breathing may slow until it nearly stops. Most deaths happen within hours, mainly from heart or respiratory collapse. Medical literature describes a “confusional state” and fits that follow. These matches what we saw in the field — people or animals convulsing, sometimes hopelessly, while family members scramble for help.
Long-Lasting Impacts of Exposure
Survivors face long recoveries. Memory gaps, ongoing muscle pain, and mood swings stick around. Children who live through poisoning may battle with learning or behavioral issues years later. My neighbor’s dog, one of the rare cases who made it, limped from nerve damage months down the line. These injuries linger, often unspoken, through farm towns and veterinary offices.
Root Causes and Common Situations
Incidents often trace back to accidental ingestion. Curious pets bite into bait. Kids, unaware of the dangers, handle what looks like food. Leftover baits forgotten in sheds or paddocks set the table for disaster. Even careful folks make mistakes. Too many cases start with forked fencing or a forgotten storage bin on a corner shelf. Years spent teaching farm safety drills taught me: it only takes a moment’s lapse.
What Science Says About Treatment
Doctors or veterinarians can’t count on a ready antidote. Treatments focus on keeping breathing and circulation moving. Activated charcoal sometimes helps if given quickly, but timing always matters. Hospitals work overtime to control seizures and protect hearts. Survival depends on how soon treatment begins. Delays easily turn fatal. Rapid identification and quick calls to poison hotlines save lives. Community education about chemicals needs attention and action — not as an afterthought, but part of daily practice.
Practical Steps Forward
Safe storage and clear labeling cut down accidental poisonings more than any high-tech fix. Local governments need to enforce stronger controls on purchase and placement of toxic baits. Farm workers and residents should receive sharp, direct safety training once a season, not just during a first-day induction. Neighbors can watch out for each other’s property, reminding each other to double-check storage at the end of every workday. Local clinics should stock common signs and response guides, making them as easy to spot as fire exit signs. Simple routines prevent tragedies long before ambulances need to be called.
Is potassium fluoroacetate regulated or restricted by law?
Potent Poison with a Long History
Potassium fluoroacetate, usually called 1080, isn’t something folks run into every day. Chemically speaking, it acts as a metabolic poison, shutting down a cell’s ability to produce energy. Even small doses can kill mammals, and that includes people, pets, and wildlife. Its main claim to fame: widespread use in pest control, especially in places like Australia and New Zealand. These countries have battled invasive mammal species for decades, especially those threatening native wildlife or agriculture. Government agencies justify 1080’s use as necessary for environmental protection and livestock survival, but the consequences reach much further.
Tight Controls Set by Law
Few chemicals attract more scrutiny. In most countries, laws put serious controls on who can handle, sell, or use potassium fluoroacetate. In the United States, the Environmental Protection Agency (EPA) lists it as a “restricted use” pesticide. Only trained, licensed professionals get access. On top of that, sales stay tightly tracked, and local authorities keep a close eye on storage and disposal. Australia and New Zealand shape their own rules based on local risks and environments, but the spirit remains the same: only approved government programs and certified applicators use 1080. Governments don’t hand this chemical out to the average citizen.
Public Safety and Environmental Concerns
Leaving potentially deadly chemicals unregulated would spell disaster. Pets, children, and non-target animals face real danger where 1080 shows up. After all, poison bait doesn’t target just one species. In Australia, national authorities track poison levels, set notification requirements, and limit use near waterways or towns. New Zealand layers on even more: notification of communities, signage, buffer zones, and bans on aerial drops in sensitive areas. Stories about dogs dying after accidental exposure aren’t rare. It’s easy to see why people fight to keep 1080 out of their neighborhoods.
Sometimes folks ask whether the controls go far enough. In my own experience living near areas treated for invasive pests, conversations with neighbors always circle back to safety—concerned parents, pet owners, or even just hikers. The sense of unease isn’t misplaced. While agencies rely on data to show how much poison seeps into soil and water, trust breaks down fast if bans and reporting slip. Community meetings held by land management agencies get heated; no one wants to find out about a nearby drop only after the fact.
Possible Ways Forward
More transparent communication bridges some of the divide—posting notices, responding to public concerns rapidly, and following through with monitoring results. Agencies can test local water sources and share data with the public. Alternatives, such as humane traps or targeted treatments, don’t always work everywhere, but new research keeps the conversation moving. In regions like New Zealand, proposals shift away from broadcast use toward smarter delivery and even genetic pest control. Greater investment in research pays off, if it leads to safer pest management tools.
It’s a balancing act: weighing the environmental benefits against the risks to people and domestic animals. This poisonous compound deserves all the legal scrutiny it gets. As long as it remains in use, law and community awareness both belong in the driver’s seat.
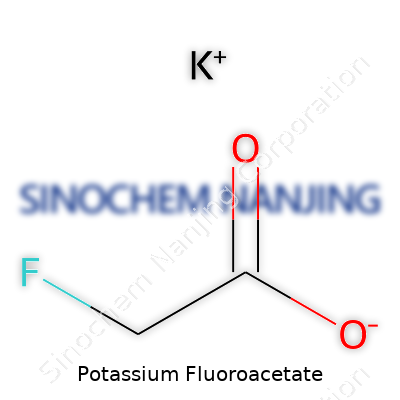
| Names | |
| Preferred IUPAC name | potassium 2-fluoroacetate |
| Other names |
1081 Compound 1080 Sodium fluoroacetate |
| Pronunciation | /pəˌtæsiəm flʊəroʊˈæsɪteɪt/ |
| Identifiers | |
| CAS Number | 62-74-8 |
| Beilstein Reference | 1462155 |
| ChEBI | CHEBI:18154 |
| ChEMBL | CHEMBL188669 |
| ChemSpider | 54977 |
| DrugBank | DB00704 |
| ECHA InfoCard | 03be8daa-6a8d-460d-bdb5-9b6908b12fc3 |
| EC Number | 206-696-4 |
| Gmelin Reference | Gmelin Reference: 80416 |
| KEGG | C1878 |
| MeSH | D011188 |
| PubChem CID | 6112 |
| RTECS number | WO4925000 |
| UNII | Z79L4S74Y6 |
| UN number | UN0179 |
| Properties | |
| Chemical formula | KC2H2FO2 |
| Molar mass | 102.09 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 1.60 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.1 |
| Vapor pressure | 0.01 mmHg (20°C) |
| Acidity (pKa) | 2.5 |
| Basicity (pKb) | 9.3 |
| Magnetic susceptibility (χ) | -41.0e-6 cm³/mol |
| Refractive index (nD) | 1.429 |
| Dipole moment | 1.81 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 117 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -886.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1987 kJ/mol |
| Pharmacology | |
| ATC code | V1 Toxic agent |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or absorbed through skin; risk of serious or fatal poisoning; causes damage to organs; very toxic to wildlife. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H300: Fatal if swallowed. |
| Precautionary statements | P261, P262, P264, P270, P273, P280, P284, P301+P310, P304+P340, P320, P330, P361, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-3-POI |
| Autoignition temperature | 540 °C |
| Lethal dose or concentration | LD50 (oral, rat): 0.2 mg/kg |
| LD50 (median dose) | 2 mg/kg (oral, rat) |
| NIOSH | NA0179 |
| PEL (Permissible) | PEL = "2.5 mg/m³ |
| REL (Recommended) | 0.003 mg/m³ |
| IDLH (Immediate danger) | **2 mg/m³** |
| Related compounds | |
| Related compounds |
Acetate Chloroacetate Fluoroacetamide Barium fluoroacetate |

