Potassium Ethoxide: Development, Properties, Applications, and Future Directions
Historical Development
The story of potassium ethoxide stretches back to the roots of modern organic synthesis, a time when chemists started noting the value of metal alkoxides. Early research in the 19th century paved the way—chemists like Justus von Liebig experimented with various metal-organic compounds and documented their basic properties. Potassium ethoxide emerged from that era as a direct reaction product between potassium metal and ethanol, a laboratory curiosity at first, but soon a mainstay in organic transformations. As synthetic chemistry boomed in the 20th century, so did the need for stronger, more predictable bases, and potassium ethoxide quickly earned a spot on many lab benches. Its reliable reactivity helped organic chemists push against traditional synthesis limitations and experiment with new molecule construction methods.
Product Overview
Marketed under names like potassium ethylate, ethyl potassium, and ethanolate (potassium salt), potassium ethoxide delivers in both academic and industrial settings. Chemists store it as a solid or a concentrated ethanol solution to control its sensitivity to air and moisture. The physical appearance—white or off-white crystalline powder or a colorless solution—reflects both purity and storage conditions. Laboratories depend on this reagent for a reason: it behaves in predictable ways, supporting efficient production of pharmaceuticals, agrochemicals, and specialty chemicals, with technical suppliers customizing concentrations and packaging for safe handling.
Physical & Chemical Properties
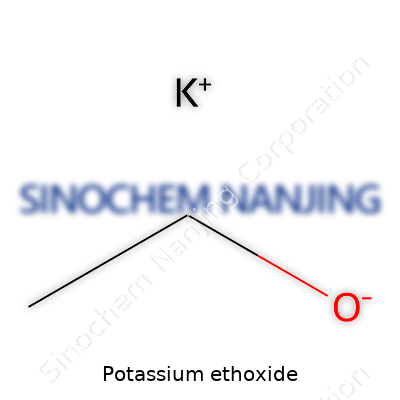
Potassium ethoxide’s molecular formula is C2H5OK. It consists of potassium bound to an ethoxide group, creating a strong base and a potent nucleophile. The compound absorbs water from the air, which makes it tricky to store for long periods outside airtight containers. Its melting point sits near 160°C, but it decomposes before boiling under standard atmospheric pressure. Danger lies in its fierce reactivity: water vapor, air, even mild acids spark decomposition or ignition. Its solubility in ethanol or ether comes in handy, but direct contact with water, skin, or mucous membranes brings serious burns or other injuries. Not just a lab oddity, potassium ethoxide’s basic character, combined with good solubility in organic solvents, lets it take part in a wide range of synthesis reactions.
Technical Specifications & Labeling
Commercial potassium ethoxide usually lands somewhere between 95–100% purity for the solid, with liquid preparations ranging from 15–30% in ethanol. Labels carry hazard warnings, including pictograms for corrosivity and flammability. The containers—usually amber glass bottles or sealed metal cans—feature batch number, concentration, and expiry date, plus a full chemical name, manufacturer address, and relevant safety codes (UN 3206 and GHS03/05/06). The paperwork that comes with each batch spells out suggested storage temperature (typically below 30°C in dry, inert conditions), proper disposal, and immediate steps in case of accidental contact or spillage. If you’ve spent much time in chemical stores, the style and information found on these packages will feel very familiar, designed for both compliance and swift identification.
Preparation Method
Potassium ethoxide comes into being through a direct reaction: potassium metal meets absolute ethanol inside a dry, inert atmosphere, like a glovebox or under nitrogen. The potassium metal slowly dissolves, effervescing as it donates electrons to ethanol, producing potassium ethoxide and hydrogen gas. Adding potassium in small pieces keeps things manageable, since the hydrogen produced can ignite if not carefully vented. Chemists cool and stir the mix, checking until all metal dissolves, and then filter out any remaining solids. What’s left is either concentrated or diluted with ethanol, depending on the target application. In any reasonably equipped lab, this process has stood the test of time, though it still demands gloves, goggles, and respect for the dangerous steps involved.
Chemical Reactions & Modifications
Potassium ethoxide reacts rapidly with water, acids, and carbon dioxide—the base hydrolyzes into potassium hydroxide and ethanol almost instantly in humid air. In practice, chemists rely on its strong basicity and nucleophilicity. For example, it drives transesterification in biodiesel production and enables the Claisen and Dieckmann condensations for carbon–carbon bond formation. In the lab, potassium ethoxide enables E2 eliminations, turning alkyl halides into alkenes with precision. The base also pops up in specialty syntheses, such as cyclizations or in the preparation of enolates. Modification chemistry—such as swapping ethanol for other alcohols—generates a range of potassium alkoxides, each with its own quirks but similar reactivity. Fast, reliable, and direct, potassium ethoxide provides a toolkit for chemists working on new molecules.
Synonyms & Product Names
Potassium ethylate, potassium ethanolate, ethyl potassium oxide, and potassium monoethanolate all point to the same compound in catalogs and chemical supply websites. Some documentation lists it under CAS Number 917-58-8, simplifying cross-referencing between research papers and product orders. Different suppliers brand it with their labels, but the nomenclature stays clear enough for researchers browsing databases or regulatory forms. Synonyms save time when hunting through patent filings or old literature, but every chemist recognizes the structure and knows the precautions that follow.
Safety & Operational Standards
Working with potassium ethoxide commands attention: it reacts violently with moisture, and the heat from mixing with water can ignite flammable vapors. Direct contact with skin or eyes causes serious burns, while inhalation of dust or fumes irritates the airways. Prudent researchers use dry boxes, flame-resistant lab coats, face shields, and long chemical gloves. Facilities handling bulk quantities keep Class D fire extinguishers close by. Spills get coated with dry sand, not water; after cleanup, waste buckets head for hazardous disposal, never the sink. On shipping forms, potassium ethoxide carries hazard statements for skin corrosion, eye damage, and flammable solid properties, in line with globally harmonized standards. Regular audits and refresher training shore up compliance and cut down on accidents, reflecting hard-learned lessons from decades past.
Application Area
Potassium ethoxide operates as the workhorse base in both high-end research and industrial manufacturing. Drug companies depend on it for route scouting—building complex pharmaceutical intermediates that demand precise reaction control. Industrial producers use it for transesterification, playing a vital role in sustainable biodiesel processes where strong, soluble bases outperform alternatives. Agrochemical development benefits from its ability to construct heterocycles and rearrange carbon skeletons in search of novel pesticides or herbicides. In materials science, researchers experiment with potassium ethoxide to tweak the properties of polymers or prepare specialized ceramics. University teaching labs favor it for demonstrating fundamental organic reactions, helping the next generation of chemists see strong base mechanisms in action.
Research & Development
Research around potassium ethoxide explores both traditional and cutting-edge chemistry. Scientists publish new routes where it acts as a greener alternative to toxic or less reactive bases. Studies focus on reaction scope, selectivity, and the consequences of varying solvent systems, with findings shaping both bench-top procedures and pilot-scale reactions. Some R&D groups investigate formulation improvements, looking to stabilize potassium ethoxide in new, less flammable solvents. Others dive into catalytic reaction design, using potassium ethoxide as a launching point for complex cascade processes—homegrown solutions for scalable, precise organic synthesis. Data from patent filings reflect a strong interest in moving beyond ethanol to methoxide or tert-butoxide analogs, hoping to squeeze more out of the chemistry with better yield or selectivity profiles.
Toxicity Research
Toxicology teams document the health hazards in no uncertain terms. Potassium ethoxide’s effect on tissue derives from its caustic, alkaline nature—skin and eye burns, respiratory irritation, and potential for deep tissue damage on exposure. Oral ingestion presents severe risks, leading to gastric burns or even systemic potassium poisoning. Data from occupational studies contribute to recommended exposure limits and shed light on chronic risks, driving pushes for better protective gear and fume hood use. Medical case reports track rare incidents, usually associated with lab mishaps, driving updates to emergency treatment protocols and first aid guidelines. Regulatory agencies, including OSHA and ECHA, set strict safety rules in response to these findings, treating the compound with the same seriousness as other aggressive alkoxides and caustics.
Future Prospects
The trajectory for potassium ethoxide points to broader use in green chemistry and sustainable manufacturing. Biodiesel processes—where traditional sodium or potassium hydroxides lose efficiency or generate problematic by-products—stand to benefit from tweaks in catalyst design centered around potassium ethoxide. Academic research keeps pushing for new reaction types, scaling up experimental successes into industry-ready protocols. Packaging advances, such as moisture-resistant ampules or safer, single-use sachets, could cut down on both handling risks and chemical waste, smoothing the workflow for both large and small users. Education around alkoxide safety continues growing, moving beyond the hazards to highlight the immense potential as a reliable, cost-effective strong base. As new synthetic challenges arise, potassium ethoxide remains a trusted solution, blending historical reliability with fresh opportunities in modern chemical research.
What is Potassium Ethoxide used for?
Everyday Life Never Sees It, but Science Couldn’t Function Without It
Many folks outside chemistry labs have never heard of potassium ethoxide. It's not on billboards, grocery store shelves, or home improvement aisles. Yet, for chemists working with organic compounds, this substance changes everything. Potassium ethoxide sits right in the action. It supports chemical transformations that make medicine, specialty plastics, and smart materials. Anyone curious about what drives breakthroughs in drug development or advanced polymers should start here.
How Potassium Ethoxide Fuels Important Reactions
Most of us remember basic chemistry from high school. In reality, lab-scale synthesis often hits a wall without the right agent. Potassium ethoxide does a simple thing with big consequences: it pulls tiny hydrogen atoms from molecules. By doing this, it forms “enolates,” which act like building blocks for more complex substances. I’ve seen research grind to a halt until a fresh bottle showed up in the storeroom.
Pharmaceutical companies use potassium ethoxide for making certain heart medications and antibiotics. It triggers reactions that can’t work without a strong base. The story repeats across specialty chemical producers who rely on speed and predictability. One day, a colleague told me how a slight shortage slowed their whole pilot production. Big projects—like making new cancer drug molecules—just stop if something so basic goes missing.
Potassium Ethoxide’s Double-Edged Sword: Helpful Yet Hazardous
Working with potassium ethoxide does not come risk-free. It sets off fires with water and reacts in air if you leave it open. Almost every chemist has a story about the first time they opened a container, eyes stinging from the fumes. Safety gear remains non-negotiable. Miss a step, and someone can get hurt. For most people, that risk means potassium ethoxide stays far from household use.
But this danger is what gives it power. Chemists can build complex molecules fast, sometimes with one or two steps, because potassium ethoxide works so aggressively. I’ve handled milder chemicals for the same job, but the results grow inconsistent or leave behind lots of unwanted byproducts. Using potassium ethoxide, yields often improve, which reduces waste. Less waste means less impact on water and soil, and that matters in the long run.
Smart Solutions: Reducing Risks in Modern Labs
Over the years, labs have improved how they store and measure potassium ethoxide. Sealed dispensers, better ventilation, and education around spills help prevent most mishaps. Automation now sets the pace in some research spaces, keeping people away from direct contact. These changes seem small but have saved time, money, and health. Some companies also push for alternatives made with fewer safety concerns, but the pure performance of potassium ethoxide means it remains a staple.
People rarely see where science begins. A quiet bottle of potassium ethoxide shows that the most transformative tools often work in the background. Smart handling, a grounded respect for risk, and sound lab practices keep its benefits in reach and hazards in check. Next time life-saving medicine hits the market, odds are this reactive compound played a silent but vital role in its journey from lab to pharmacy shelf.
What are the storage requirements for Potassium Ethoxide?
Why Storage Matters for Potassium Ethoxide
Potassium ethoxide reacts with water and air fast, pushing safety, purity, and lab function onto the front line. I’ve handled this compound in a teaching lab before realizing just how much a single drop of humidity turns it from a fine white powder into a sticky, dangerous mess. If you leave its bottle even a little bit loose, you’ll see sludge gathering at the rim. That tells you right there: Potassium ethoxide demands respect and strict habits.
Key Hazards You Can’t Ignore
Here’s the simple truth: Potassium ethoxide catches fire when exposed to air thanks to its strong reaction with moisture and oxygen. That’s how fast things can go sideways. Not handling storage right means risking fires, toxic fumes, or accidental spills. The fumes themselves cause coughing and eye irritation—breathing them day-in, day-out wrecks your health. Factories and research labs have strict rules for a reason.
Solid Rules for Real-World Storage
What works best comes down to containers, location, and what’s close by. Potassium ethoxide lives in tightly sealed, airtight glass bottles. Forget plastic—it breaks down too easily. Stoppers must fit right and seal tight. Every time you open the container, air sneaks in, and tiny amounts react right away. I’ve seen labels fade from poor sealing, making it harder to spot problems.
Store the containers in a dry, cool space away from sunlight. Heat triggers decomposition and more fumes. No window ledges or benches near steam pipes — dry, out-of-the-way shelves do the job. Always add a warning label detailing fire and water reactivity. Keep fire extinguishers and plenty of spill-absorbent material nearby, just in case things go wrong.
What Never Belongs Nearby
Don’t line potassium ethoxide next to acids, moisture sources, or oxidizers. The smallest contact with water starts a chain reaction. In my earliest work, a co-worker stacked it near a bottle of nitric acid—what followed was a cloud and a lot of yelling. If you set your storage layout thoughtfully from the start, you prevent these rookie mistakes. Officers in charge of chemical inventories always insist on separation by risk, not alphabet.
Practical Handling: The Human Side of Storage
The safest storage means training staff and refreshing your own habits. Always handle potassium ethoxide inside a fume hood and wear eye protection, gloves, and a lab coat. Repackaging or redistributing? Only do it under dry nitrogen or argon. I learned early on to double-check my gloves and never leave the container open longer than needed. Even a coffee break is too long. Spill kits make a difference—you want absorbent pads and neutralizers handy, and everyone at the site should know where they are.
Solutions for Common Problems
Running out of space or proper containers shows up often in smaller labs. Buy smaller containers to limit the danger if something leaks, and arrange shared storage with good access controls. Regular checks and cleaning keep dust, humidity, and stray chemicals out. For older bottles, replacing them before the seals deteriorate goes a long way. Technology helps, too—simple humidity sensors tied to alarms keep people aware. Good air flow in storage rooms prevents any unnoticed buildup of solvents or vapors.
Why Vigilance Always Wins
Safe storage of potassium ethoxide relies on respect for how reactive it stays, not just following rules written on a sheet. Every smart step—sealed bottles, dry rooms, frequent checks—keeps people, results, and buildings out of danger. No shortcut ever repays the hassle of an emergency.
Is Potassium Ethoxide hazardous or flammable?
Understanding the Risks in Simple Terms
Potassium ethoxide rarely gets the spotlight outside chemical labs, but anyone storing, handling, or transporting it should recognize what makes it troublesome. Folks often overlook the dangers of obscure chemicals until an accident happens. Having worked near labs, I’ve seen how even seasoned staff sometimes get complacent with materials that don’t look threatening. Potassium ethoxide serves as one of those “hidden hazards.” It doesn’t glow or set off alarms, but its dangers deserve practical attention.
Potassium ethoxide lights up quickly. It doesn’t just burn—it kicks into high gear at the slightest spark or exposure to air. A dry, colorless solid at room temperature, it smells like strong alcohol and reacts quickly whenever it detects moisture. Water pulls out fire from this powder. Even a humid afternoon bumps up the chance of a flash, and the fire brings along toxic fumes. That trait alone separates it from run-of-the-mill flammables. Industrial safety data sheets warn that any spillage should be swept up gently, under a dry, inert atmosphere. Not many shops, homes, or schools can offer those conditions.
Real-Life Dangers and the Importance of Respecting the Substance
Potassium ethoxide doesn’t offer second chances during accidents. One time, during basic chemical safety training, a bottle cracked while being moved across a table. The room filled with a sharp, biting odor, and the supervisor scrambled to douse the powder with mineral oil—regular water would have fueled the blaze. Sellers and storage sites need rock-solid safety routines, not just signs on the wall. It belongs far away from open flames, oxidizers, or any container that could leak.
Lungs can’t handle its fumes either. Coughing, burning skin, or blurred vision—these symptoms crop up fast during exposure. I’ve seen a coworker lose a week of work after breathing in a vapor cloud. Results like that hammer home the point: even skilled hands can slip, so respect and preparation are the only answer.
Papers and Real-World Examples Back Up the Warnings
Journal articles tie potassium ethoxide accidents to lab fires and medical emergencies. For example, the U.S. National Institutes of Health notes that potassium ethoxide explodes if it touches water, acids, or carbon dioxide. The National Fire Protection Association (NFPA) assigns it some of the highest fire hazard ratings. Safety instructions call for dry sand or Class D fire extinguishers—not water or foam—because regular fire-fighting tools make things worse. In 2010, a shipment container at a university caught fire during unpacking, leading to a hefty hazmat cleanup bill and strict new rules for dry storage.
Safe Behaviors and Practical Solutions
Simple rules keep workplaces safe: air-tight containers, dry rooms, chemical-resistant gloves and goggles. Anyone trained in chemistry should keep mobile phones away from the area. Labeling goes beyond formality—it saves time during an emergency. Spill kits stocked with mineral oil or dry sand need to sit right beside storage shelves. Management should schedule regular checks and treat any outdated or damaged container as a top priority risk. Insurance rules and regulations set a minimum standard, but the real test comes during daily routines and the unexpected mishap. Building a “safety first” mindset means less reliance on luck.
Potassium ethoxide looks like any other white powder, but it hides a lot of muscle behind that bland face. Listen to those who have witnessed accidents, use smart habits around the substance, and demand real accountability from suppliers. Fires and poisonings grab the headlines, but good daily practice can quietly keep people and property safe.
What is the chemical formula of Potassium Ethoxide?
Understanding the Compound
Potassium ethoxide stands out as a chemical I've encountered in more than one organic chemistry lab. Its chemical formula, C2H5KO, speaks for itself. Potassium (K) pairs up with an ethoxide group (C2H5O). A potassium atom replaces the hydrogen in ethanol’s hydroxyl group. From the moment you open that bottle, a sharp, unmistakable odor drifts out, reminding you of both its reactivity and the need for careful handling.
Why Potassium Ethoxide Matters
This compound plays a key role in synthesis. I remember using potassium ethoxide to create new carbon bonds—usually in reactions like the Claisen condensation. Chemistry textbooks list all sorts of lab-based uses, but the real value shows up at the workbench. Fast, reliable reactions save time and reduce waste. In the pharmaceutical world, cutting steps and cleaning up fewer byproducts translates to both cutting costs and reducing environmental impact.
Aside from pharmaceuticals, agricultural chemicals and dyes often depend on reactions powered by potassium ethoxide. Researchers value the compound’s strong basic nature, which makes it a go-to reagent for deprotonation—essentially removing a hydrogen atom to jump-start the next stage of a chemical reaction. With potassium being more reactive compared to sodium when bound to the ethoxide group, chemists get a versatile tool for more challenging projects.
Safety and Handling Always Come First
Potassium ethoxide demands respect. My glove would sometimes show evidence of a pinhole burn from a stray splash—an immediate lesson in chemistry lab safety. Contact with water, even humidity, produces ethanol and potassium hydroxide. That reaction can get hot fast. Poorly stored potassium ethoxide can end up as a mess of potash and unwanted byproducts, turning an expensive chemical into a hazard. In one memorable incident, a coworker discovered what happens when a canister’s seal fails: fumes and a caustic, sticky residue coated everything nearby.
Correct handling always wins. Dry storage, airtight containers, and working under an inert atmosphere are standard practices. Training new chemists to expect a vigorous reaction teaches caution and drives home respect for strong bases. Every chemistry class that works with potassium ethoxide has a story, and those stories help drive better lab habits.
Addressing the Challenges
Safe disposal and accidental exposures often worry people who work with potassium ethoxide. Leaching into the environment could lead to caustic run-off, damaging local ecosystems. That’s why responsible labs use clear procedures for neutralization and waste handling. In hospitals and research centers, digital inventory tracking helps flag expired or leaking stock. Taking a page from sustainable chemistry, teachers encourage greener alternatives for some reactions. Sometimes, using milder bases or phase-transfer catalysts can do the same job with less risk.
Chemists, especially those early in their training, gain a lot by seeing how potassium ethoxide reacts outside a textbook. It brings theory to life, shows where risk and reward meet, and tests everyone’s attention to safety. The lesson echoes through every future synthesis: know your materials, understand your risks, and keep learning from those who have handled these chemicals before you.
How should Potassium Ethoxide be handled safely?
Handling a Strong Chemical Without Regrets
Potassium ethoxide has a reputation for being unforgiving. Forget to respect its flammability, and you’ll see why most chem labs tuck it away under tight rules. Years ago, I walked past a shelf where a flask of potassium ethoxide had a cracked cap. My nostrils burned from a strange whiff crisp as ethanol, sharper than your regular solvent. The lab supervisor dashed over, tossed on a respirator, and eased the situation. That memory sticks because just a few drops reacting with air set off alarms. Potassium ethoxide doesn’t bother with subtlety. It reacts with water instantly, releasing ethanol and caustic potassium hydroxide. Most people don’t realize it can catch fire quicker than you might think—contact with air or skin can make small mistakes turn big, fast.
Why Attention Saves People
People tend to get casual around chemicals they’ve handled more than once. Potassium ethoxide punishes routine shortcuts. Skin won’t just get dry; splashes chew through flesh and cause rough chemical burns. Eyes? Exposed tissue has no defense. Ordinary gloves or thin lab coats do little to stop the burn. After seeing the aftermath of a spill on a friend’s hand—redness, blistering, weeks of scabbing—no chemical feels routine. Most shops run through safety drills, but I don’t remember any classmate raising their hand to admit a potassium ethoxide mistake. People hide accidents and hope for the best, but silence makes the risk worse.
Protective Steps That Actually Work
My experience matches what safety data tells you: always keep potassium ethoxide under nitrogen or argon. Break that rule and moisture in the air gets inside, triggering dangerous reactions. Thick, chemical-resistant gloves beat basic latex options. Lab coats built for acid spills handle splashes better, with goggles fitted tight enough to stop vapor from drifting in. Proper face shields aren’t just for big spills; they keep small mistakes out of your eyes and mouth, which make all the difference during cleanup. I always double up—gloves on hands, arms covered, and footwear that blocks anything falling from above.
Storage Rules That Go Beyond the Obvious
Potassium ethoxide demands careful storage. Bottles must stay tightly sealed, tucked away from heat and moisture. Every workbench where I’ve seen it used, a dedicated, fireproof cabinet sat nearby. Signs keep people from mixing up containers. Some labs ignore label dates, which gets dangerous fast—old solutions degrade and build pressure, sometimes enough to burst sealed bottles. I’ve seen Ethoxide bottles sporting rusted caps or hazard labels bleached from the sun. A bad seal will invite air and water. Checking those bottles every time matters more than most realize.
Treating Mistakes Without Panic
Spills happen. Training matters. Douse any exposed skin with lots of water, skip the scrubbing, and strip off anything hit by the chemical. I learned that a simple rinse isn’t enough; emergency showers and eyewashes need to be close, never locked away or broken. Once, our emergency eyewash failed a safety check, and the supervisor made us scrub the whole system before touching any chemicals again. Quick medical help helps stop small injuries from turning into scars—or worse.
Institutions Make the Difference
Smart systems decide how safe a lab stays. Some rely on trust, others on checklists and peer reviews. Nothing beats regular inspections, up-to-date safety training, and an attitude that calls out missing gloves or rusted containers. Posters and reminder charts help, but conversations do more. People talk about fire drills or safe handling as chores, but after seeing a real accident, nobody views the rules as extra work.
| Names | |
| Preferred IUPAC name | potassium;ethanolate |
| Other names |
Ethoxypotassium Potassium ethylate Ethyl potassium oxide |
| Pronunciation | /pəˈtæsiəm ɪˈθəʊksaɪd/ |
| Identifiers | |
| CAS Number | '141-52-6' |
| Beilstein Reference | 3587150 |
| ChEBI | CHEBI:41223 |
| ChEMBL | CHEMBL138098 |
| ChemSpider | 11329 |
| DrugBank | DB15990 |
| ECHA InfoCard | 100.029.030 |
| EC Number | 212-207-4 |
| Gmelin Reference | Gmelin Reference: **2153** |
| KEGG | C18699 |
| MeSH | D017746 |
| PubChem CID | 8667 |
| RTECS number | TH3675000 |
| UNII | 1K7930T38K |
| UN number | UN2491 |
| Properties | |
| Chemical formula | C2H5OK |
| Molar mass | 82.12 g/mol |
| Appearance | White to yellowish powder or crystals |
| Odor | Alcohol-like odor |
| Density | 0.866 g/mL at 25 °C |
| Solubility in water | Soluble |
| log P | -0.24 |
| Acidity (pKa) | 15.5 |
| Basicity (pKb) | pKb = -1.0 |
| Magnetic susceptibility (χ) | +29.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.424 |
| Viscosity | Low viscosity |
| Dipole moment | 1.66 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 219.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -409.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -538 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07, GHS06 |
| Pictograms | GHS02,GHS05 |
| Signal word | Danger |
| Hazard statements | H225, H260, H314 |
| Precautionary statements | P210, P222, P223, P231+P232, P280, P305+P351+P338, P310, P370+P378, P402+P404, P501 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | '-18 °C' |
| Autoignition temperature | 290°C |
| Lethal dose or concentration | LD50 (oral, rat): 1950 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 2050 mg/kg |
| NIOSH | ST2625000 |
| REL (Recommended) | 5 mg/m³ |
| IDLH (Immediate danger) | IDLH (Immediate Danger to Life or Health) for Potassium Ethoxide: Not established |
| Related compounds | |
| Related compounds |
Potassium methoxide Sodium ethoxide Potassium tert-butoxide |

