Looking Closer at Potassium Dithionite: Its Impact and Road Ahead
Historical Roots in Chemistry
Potassium dithionite doesn’t usually make headlines outside specialist circles, but its story stretches back over a century. Chemists started to explore the world of alkali metal dithionites in the 19th century. Interest ramped up with large-scale dye production, when reducing agents played crucial roles in textile plants scattered across Europe. Industry workhorses like potassium and sodium dithionite laid down the first chapter in practical chemical reduction, helping move textile dyeing away from more hazardous, less stable substances. The chemical found favor in labs and mills alike thanks to its reliability in reducing stubborn dyes and processing ores. Generations later, these early use cases set the tone for broader applications in analytical chemistry and industry.
Getting to Know Potassium Dithionite
Nobody expects a white, powdery solid like potassium dithionite to cause much fuss, but its applications show remarkable diversity. In my experience, it often sits on shelves next to its sodium cousin, ready for duty in textile, paper, and lab settings. Its strength as a reducing agent puts it center stage in dye houses and chemical syntheses. Its chemical backbone, K2S2O4, packs two potassium ions and a dithionite group. Its ability to donate electrons makes it a mainstay wherever rapid reduction is a must. Despite its utility, it won’t stay stable for long out in the open—moisture and oxygen in air spell quick decomposition.
Physical and Chemical Nature
It’s easy to overlook potassium dithionite’s unassuming appearance: a fine, white solid, odorless to most. Beneath that surface, it’s on a hair trigger with moisture and heat. The powder dissolves easily in water, but once in solution, it faces a ticking clock. Exposure to acids, air, or warmth can break it down, releasing sulfur dioxide and other byproducts. Stability remains a challenge, and in many storerooms, potassium dithionite sits in tightly sealed containers away from light, damp, and oxidizers. Mix it the wrong way, and its reducing power can flare up—especially dangerous if flammable substances or strong acids join in.
Making and Modifying Potassium Dithionite
Manufacturing potassium dithionite involves chemical juggling acts. Commercial routes often react sulfur dioxide with alkali metal hydroxides in carefully controlled environments, usually underlining the need to minimize exposure to oxygen and moisture. Most methods mirror the industrial steps used for sodium dithionite, with final product purity resting on the skill of technicians to steer clear of contamination. Tinkering with its structure rarely occurs, but blending with other reducing agents or formulating in tablet or granule forms helps meet practical needs on shop floors and in field kits.
Names on the Label
The chemical catalog lists potassium dithionite under several aliases. Chemists use potassium hydrosulfite, potassium sulfoxylate, or dipotassium dithionite depending on preference or tradition. I’ve heard old-school technicians in the textile trade still call it potassium hypo, throwing back to another century’s shorthand.
Setting the Standards: Handling and Safety
Not all chemicals get the safety respect they deserve, and potassium dithionite is no exception. Proper handling means more than slipping on gloves—good ventilation matters, too, for catching whiffs of sulfurous byproducts before they collect. Once water or acids hit the powder, sulfur dioxide gas can burst free, bringing suffocation risks if spaces are tight or air is stagnant. Oxidizers, sparks, or open flames can set off unpredictable chemical fireworks. I’ve learned that training workers to recognize decomposing containers, check expiration, and store away from incompatible chemicals keeps accidents rare. In regulated workplaces, safety data sheets help bridge knowledge gaps, but even experienced users keep emergency plans close at hand.
Where Potassium Dithionite Gets Used
Dyehouses run through barrels of potassium dithionite for vat and sulfur dyes, stripping color from fabrics and prepping fibers for new shades. The pulp and paper world dips into its reducing power to bleach recycled sheets and clean up pitch residues. Meanwhile, many labs treat potassium dithionite as a go-to for reducing stubborn metal ions during qualitative analysis. Treating contaminated groundwater or remediating soil gets a leg up from potassium dithionite’s ability to break down industrial pollutants. It plays a niche but growing role in treating chromium-contaminated sites—converting toxic hexavalent chromium to safer trivalent forms.
Current Focus in Research and Development
Industrial users and researchers keep trying to squeeze more from potassium dithionite. Cost pushes some to seek substitutes or tweak its efficiency in large-batch processes. Researchers study shelf life, decomposition rates, and contamination risks more closely now than ever, especially as buyers demand longer-lasting, purer product. In environmental cleanup, scientists examine how to maximize potassium dithionite’s reducing punch without spiking secondary pollution. Some labs chase green production routes, hoping to cut chemical waste or reclaim spent material. In my view, there’s plenty of value in learning how to recycle or regenerate post-reaction slag before shipping it off-site.
Toxicity: What We Know and What We Still Ask
Potassium dithionite’s hazards tend to hide behind the promise of safety in numbers—handled by trained adults and kept under lock and key, it rarely reaches the broader public. It’s true that industrial exposure brings more risk than everyday life. Breathing in sulfur dioxide, for example, irritates lungs fast and presses hard against asthma sufferers. Contact with bare skin or eyes brings stinging and burns, so splash goggles and sleeves aren’t extras—they’re essentials. Toxicologists dig deeper, testing how chronic exposure or breakdown byproducts might linger in water, air, or soil. Regulations have started to shift in response, with some countries restricting sales or requiring tighter reporting on quantities used. This scrutiny points to bigger changes in industrial safety and chemical stewardship.
Looking Past Today: Future Prospects
There’s a long list of challenges for potassium dithionite in the years ahead. Sustainability and environmental pressure push companies to ask how reduction chemistry could run cleaner and leave less waste. Some startups hunt for enzyme-based or renewable alternatives for textile treatments, hoping to replace or supplement potassium dithionite in color stripping and bleaching. At the same time, high-value niches in environmental remediation look set to grow as toxic metal pollution comes under tighter restriction. As regulations bite harder and supply chains bend, potassium dithionite could lose ground in bulk markets, but find new strength in specialized processes or as a tool for emerging clean-up strategies. My own gut tells me change comes slow, but focused innovation has a way of keeping tried-and-true chemicals relevant far longer than expected, even as sustainability reshapes the wider landscape.
What is Potassium Dithionite used for?
Inside the World of Potassium Dithionite
Potassium dithionite often pops up in places most folks don't see. It's a solid, white, or slightly yellowish powder, and the real action happens once it meets water: it cuts through oxygen like a knife. This trick makes it a go-to chemical for many industries, not just a curious chemical name in a high school textbook.
Color Remover in Textile Dyeing
I’ve spent time around textile factories where the work buzzes with vats of vivid colors. In these places, mistakes happen—batches come out wrong, shades don’t match, or a spill leaves old dye stains on new fabric. Potassium dithionite earns its keep as a powerful reducing agent that strips color right off. Removing color this way helps the fabric get a fresh start, letting workers apply new patterns or correct botched orders without tossing out yards of material.
This not only saves costs but also means less waste. The World Bank flagged dye-related wastewaters as some of the trickiest to treat, so anything that cuts down on unnecessary disposal helps both the environment and the factory’s bottom line.
Role in Paper Pulp Bleaching
Old newspapers, cardboard boxes, paper scraps—eventually, all that piles up and ends up in pulping mills. Turning that mess back into a clean sheet takes more than hot water and patience. Potassium dithionite steps in to remove the stubborn yellow-brown tint from recycled fibers. The process skips over harsher bleaching chemicals, which helps reduce the load of toxic byproducts heading into rivers and streams. I look at this as a hidden win for the paper industry. Cleaner products and cleaner effluents give papermills fewer headaches from regulators and local communities.
Treating Water and Soil
Water utilities work day and night to make sure our water flows clean and safe. Once in a while, some wells pick up hexavalent chromium, a toxic metal that poses real health risks. Potassium dithionite pulls off a neat trick by turning dangerous chromium into a much safer form. The lowered toxicity makes cleanup less of a nightmare and spares towns from spending big on even more complicated solutions.
It’s not just water. In the last decade, environmental consultants also used this compound to treat soil on old industrial lots. I talked to a team working near a retired electroplating plant—using dithionite in the soil sped up cleanup, getting the property ready for new businesses and jobs.
Lab Work and Chemical Analysis
College labs and chemical research centers keep potassium dithionite in stock, mostly for reducing iron and breaking down stubborn minerals. I’ve watched students in geology labs dissolve rare rocks and ores with it—it’s quicker than the old-school methods. The faster turnaround helps push research along, rather than getting bogged down in long procedures.
Keeping Safety on Top
You can’t overlook the risks. In my own work, storing and using potassium dithionite always comes with strict routines to keep fires and fumes in check. It breaks down fast in air and water, which means storage demands dry, tightly sealed containers and clear labeling. Real-life accidents often come from skipping safety steps, so anyone using this chemical steps up vigilance to protect workers and neighborhoods.
What Could Help Going Forward?
Better training always ranks at the top. Workers learning not just the rules but the real-life reasons behind them tend to cut down on careless mistakes. Factories could also look for alternatives or safer blends that do the job without hazards. Technology keeps moving forward, and one day soon, a cleaner, greener replacement might rewrite how these industries work.
Is Potassium Dithionite hazardous or toxic?
What’s at Stake in Handling Potassium Dithionite
Potassium dithionite enters the picture in places like textile dyeing, pulp bleaching, and the lab bench. If you’ve worked in one of these settings, you might know this solid looks harmless enough—soft and powdery, almost like plain flour. Give it air, though, and things get tricky fast.
Potassium dithionite, also called potassium hydrosulfite, packs a punch with reducing power. Toss that into a damp room and you’ll get a noseful of sulfurous stench if it drags in some moisture. All that fuming means this stuff can irritate eyes, skin, or lungs if you’re sloppy with the gloves or let the powder linger. Even with no splash, breathing in the dust gives headaches and coughs, reminding anyone in the lab there’s no such thing as a “safe chemical.”
Health Risks: More Than Minor Irritation
Contact with potassium dithionite can spark reactions you’d rather avoid. Splashing it on your skin often triggers rashes or burns. Eyes have it worse—exposure burns and stings. Inhaling the dust doesn’t just leave a scratchy throat; at higher doses, people have reported breathing trouble or even asthma-like symptoms. Years of close work with dithionite, carried out carelessly, bring headaches, woozy spells, and in rare cases, worsen respiratory conditions.
Swallowing potassium dithionite won’t leave you unscathed. It reacts with stomach acid, releasing toxic gases. Nausea, vomiting, stomach aches, and sometimes diarrhea follow. No one talks much about this risk—but it shows why working over your lunch or with bare hands isn’t smart.
Fire and Water: Double Trouble
Potassium dithionite might not explode easily, but pile it up near heat or spark, and you’ll risk a sudden fire. It breaks down into sulfur oxides and potassium salts at a pretty moderate heat, sending out chemical fumes. Dumping it down the sink doesn’t wash away trouble, either. It reacts with water to release sulfur dioxide—a gas plenty of folks are sensitive to on a bad air day. Let this gas build up indoors, and you could land your workplace in lockdown.
Why Risk Remains Even with Labels and Fences
Plenty of warehouses and shops throw warning labels onto sacks, but I’ve seen mishandling happen anyway. Old gloves, missing goggles, or casual spills earn shrugs instead of cleanups. Some people get used to warning signs after months, and that’s where trouble waits. The EPA and OSHA both list dithionite for good reason: not only as an irritant, but also for choking hazards in tightly sealed rooms. Accidents tend to stack up when rush jobs or complacency take over.
Smart Moves Keep Hazards Down
Training helps—walking someone through cleanup and storage cuts risk more than stacks of paperwork do. Investing in proper extraction systems, splash shields, and eyewash fountains turns a risky job into a safer one. Personal responsibility also plays a part. I learned early on: never trust a “simple” white powder until you know where it ends up. Anyone who skips the safety measures often ends up learning the hard way.
In a world with a million chemicals, potassium dithionite sits right in the middle: useful, but far from harmless. Giving it the right respect and adopting smart habits delivers results—the kind you remember at the end of a long shift, without burns or headaches to show for it.
How should Potassium Dithionite be stored?
What Makes Potassium Dithionite Worth Extra Attention
Potassium dithionite helps in many industries, from hand-dyeing fabrics in art studios to large-scale paper bleaching. It acts fast to remove oxygen and reduce color, making it a favorite for specific chemical processes. Working with it, I’ve noticed most problems come not from the product itself, but from how people store it. Let’s just say a dusty, damp corner in the back shed rarely ends well.
Understanding the Risks Close Up
Potassium dithionite breaks down quickly when it touches water or air. It gives off a rotten egg smell—sulfur dioxide—and can even spark a fire if stored next to flammable items, or if it absorbs moisture and heat builds up. I’ve seen folks forget about strict storage, then come back to a canister that puffed up or, worse, spilled. Inhaling the fumes or letting it touch your skin can cause health problems, including headaches and breathing trouble.
A fact worth remembering: the National Fire Protection Association rates potassium dithionite as a Class 1 fire risk. This rating means you don’t need a warehouse inferno—only a little spark can get things going if it’s in the wrong spot. The compound ranks high on the list for “things that self-destruct in bad storage.”
Down-to-Earth Storage Practices from the Lab
In my own experience, the best storage spots are dry and cool rooms with solid ventilation. Sealed, airtight containers really are the gold standard. I once used a basic screw-top jar instead of true lab glass, only to find powder clumped up and discolored just a week later. Humidity sneaks in fast, especially in summer.
I recommend storing potassium dithionite away from acids, oxidizers, and anything that could catch fire easily. Workplaces I trust label their containers clearly, often listing an emergency phone number and precise handling tips right on the label. One clever idea I’ve picked up is using a moisture-absorbing packet inside every storage bucket—borrowed from the food industry, but it works.
Health and Environmental Considerations
Safe storage goes beyond protecting inventory. One leaky bag can trigger emergency clean-ups, not to mention risk health for everyone nearby. The fumes from decomposing product can irritate the nose, throat, and can even make asthma worse. Wearing simple gloves, using safety glasses, and setting up storage in well-aired spots all go a long way.
Environmental safety plays a part too. Any spill, especially where rainwater runs off, risks sending toxins into local water supplies. I’ve seen companies get big fines simply for a poorly placed drum that got rained on. Governments urge locking these chemicals in rooms set apart from drains, with floor bunding to catch leaks.
Building a Safe Storage Culture
Having a top-notch safety plan works best if every worker, from managers to cleaners, knows what’s stored and how to handle it. Ongoing training trumps yearly lectures. Regular checks, not just a one-time setup, give peace of mind. Safety experts often suggest mock drills for chemical leaks, timed to keep everyone sharp.
Storing potassium dithionite well protects health, investments, and the environment. Taking a little care day-to-day, in my experience, beats the rush and panic after an accident every single time.
What is the chemical formula of Potassium Dithionite?
The Essential Facts
Potassium dithionite, better known in some chemical circles as potassium hydrosulfite, comes with the formula K2S2O4. As someone who has spent years around chemical supply labs, you get used to reading these formulas like street addresses — K2 means two potassium atoms, S2 is two sulfur atoms, and O4 finishes up with four oxygen atoms. These numbers aren’t just for the textbooks. They tell you what you’re actually working with, which can save time, money, and even your safety in an industrial setting.
Potassium Dithionite in Practice
In textile dyeing, a lot of folks depend on potassium dithionite for its power as a reducing agent. It’s good at breaking down stubborn dyes and bleaching fibers clean without creating massive clouds of fumes. During a stint working alongside technicians in a cellulose-processing facility, we’d often choose potassium dithionite over its cousin, sodium dithionite, to avoid sodium build-up in specific processes. Some industries prefer it precisely due to potassium’s gentle touch on sensitive systems — it doesn’t introduce the risk of sodium contamination, especially where wastewater is carefully monitored.
Why Potassium Over Sodium?
Both the potassium and sodium versions of dithionite do the job of reduction, but potassium dithionite sometimes fits the bill better. For starters, certain biological applications have noted better outcomes with potassium ions. Greenhouses and agricultural labs occasionally use potassium dithionite to help control oxidation in soil testing. Compared to sodium, potassium has less impact on the cation balance in the environment, so the risk to sensitive plant roots drops. I’ve seen agronomists choose potassium dithionite simply to play it safer with their crop yields.
Handling and Environmental Impact
Potassium dithionite doesn’t act like table salt. It decomposes quickly in the presence of air, especially in water. That quick breakdown means strict storage rules. Containers get sealed up right after each use, and anyone in the area gets told about its decomposing nature. Spills bring the risk of releasing sulfur compounds with an odor you won’t soon forget. Working in labs that deal with water discharge, I’ve noticed increasing pressure to use dithionite forms that don’t linger and pollute. Potassium dithionite, given the right precautions, usually fits those regulations better than some alternatives.
Safety and Solutions
Mistakes with chemicals often come down to misreading the formula. Mixing up K2S2O4 with something more potent — even a similar white powder — can spell disaster. Training new staff, we’d drill into them the importance of double-checking both labels and chemical formulas before every use. One simple cross-check on the label meant fewer incidents in the plant.
Looking for safer ways to use and dispose of potassium dithionite has driven a lot of new research. On-site neutralization, using rapid oxidation with hydrogen peroxide, keeps dangerous by-products from going down the drain. Industry groups have started collaborating on guidelines, backed by environmental studies, informed by real-life chemical audits.
Final Thoughts
Potassium dithionite shows up in textiles, environmental labs, and agricultural test kits for good reason. Its formula, K2S2O4, signals a substance with specific uses and real-world value. Clear understanding and solid safety habits, built from experience and ongoing training, help keep both workers and surrounding ecosystems safe. Staying informed and respecting what each chemical brings to the table makes all the difference.
How do you safely handle and dispose of Potassium Dithionite?
Understanding the Hazards
Potassium dithionite throws up a set of risks you can't play around with. It’s a strong reducing agent, used for things like textile dyeing, paper bleaching, and even soil remediation. Run into it at the lab or factory, and it will look like a plain, white powder. What you don’t always see? Its real danger: it decomposes easily and spits out sulfur dioxide, which can choke you up and even damage your lungs. Water and moisture make it break down quicker, releasing heat and gases. If you’re careless, the results come fast and nasty.
Personal Experiences: Don’t Skip the Basics
I’ve watched new technicians drop the ball by thinking gloves and goggles are enough. One summer, a coworker measured out potassium dithionite by a drafty window. A gust of air swirled the powder, a fine cloud formed, and barely a minute later, eyes and throats burned. Nobody wore face shields. That short lapse cost three trips to the nurse and a note in the safety log. Sweat the small stuff, every time.
Facts: What PPE Actually Works
NIOSH (National Institute for Occupational Safety and Health) and the CDC stress the need for fixed chemical goggles, long sleeves, chemical-resistant gloves, and dust-proof respirators when handling the powder. Use the chemical in a fume hood or a well-ventilated space because those sulfur fumes love to hang low. Spills stick to your shoes, so slip on booties in case you drop a scoop. Don’t just skim the material safety data sheet — take those pictograms and warnings to heart. Fume hoods aren’t optional. Neither is ventilation.
Disposal: Doing It Right, Not Just Fast
Disposing of potassium dithionite wrong creates long-term headaches for everyone down the waste stream. Pouring it down a sink won’t make it disappear. It reacts with water, releasing heat and dangerous gases. The right way uses slow, staged neutralization. Mix small batches into large volumes of cold, dilute acid under a vent, following local guidelines. Forget shortcuts: large releases can overwhelm sewers, and diluted products still contaminate water. Connect with a trained hazardous waste handler—your facility should have contacts, or local government can point you toward one.
Beyond Rules: A Culture of Safety
It’s easy to talk up compliance, but in real life, people cut corners if safety gear fits badly or instructions aren’t clear. Leadership makes a difference here. A supervisor who calls out lazy habits gets better outcomes than one who just emails out safety sheets. In my own run with chemicals, group walk-throughs, and drills changed attitudes fast—especially after handling mishaps. Building this kind of diligence keeps accidents rare and teaches new folks how to respect chemicals—without making it a routine that people ignore.
Solutions Worth Backing
Facilities can set up dedicated areas for opening and measuring potassium dithionite with spill kits nearby and strict “dry work only” rules. Training new staff with hands-on PPE and cleanup practice drives the message deeper than any slide show. Waste should go in sealed, labeled containers, held in cool, dry spaces until pickup. Automation of transfer and mixing reduces spills and dust clouds—a worthwhile investment. Reporting and learning from even near-misses creates a feedback loop so old mistakes don’t repeat.
No shortcut ever replaces careful, informed handling of potassium dithionite. It’s about showing respect for what this chemical can do—both the good and the bad.
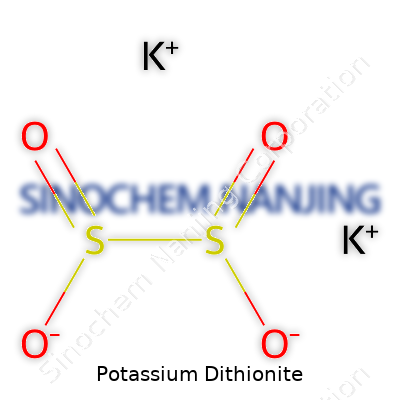
| Names | |
| Preferred IUPAC name | potassium oxido-oxididosulfonate |
| Other names |
Sodium Hydrosulfite Sodium Dithionite Hydros Reducer O |
| Pronunciation | /pəˈtæsiəm daɪˈθaɪənaɪt/ |
| Identifiers | |
| CAS Number | 7775-14-6 |
| Beilstein Reference | 1202540 |
| ChEBI | CHEBI:48843 |
| ChEMBL | CHEMBL135919 |
| ChemSpider | 14229 |
| DrugBank | DB14004 |
| ECHA InfoCard | 100.029.047 |
| EC Number | 231-890-0 |
| Gmelin Reference | 72427 |
| KEGG | C02325 |
| MeSH | Dithionites |
| PubChem CID | 24507 |
| RTECS number | XU8750000 |
| UNII | WAQ7QGN2PL |
| UN number | UN1929 |
| Properties | |
| Chemical formula | K₂S₂O₄ |
| Molar mass | 156.27 g/mol |
| Appearance | White crystalline powder |
| Odor | Faint sulfur dioxide odor |
| Density | 2.34 g/cm³ |
| Solubility in water | Soluble |
| log P | -4.5 |
| Vapor pressure | < 0.01 mmHg (20°C) |
| Acidity (pKa) | 7.0 |
| Basicity (pKb) | 12.7 |
| Magnetic susceptibility (χ) | −83.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.67 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 203.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -686.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -697 kJ/mol |
| Pharmacology | |
| ATC code | V03AB04 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H319, H331, H335 |
| Precautionary statements | P264, P280, P301+P312, P330, P305+P351+P338, P304+P340, P312, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-0-1 |
| Autoignition temperature | > 550 °C |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 2,500 mg/kg |
| LD50 (median dose) | **2,500 mg/kg (rat, oral)** |
| NIOSH | SA0870000 |
| PEL (Permissible) | PEL: 5 mg/m³ |
| REL (Recommended) | REL (Recommended): 0.3 mg/m3 |
| IDLH (Immediate danger) | 30 mg/m3 |
| Related compounds | |
| Related compounds |
Potassium sulfite Potassium sulfate Sodium dithionite Sodium sulfite |

