Potassium Dihydrogen Phosphate: Commentary on a Versatile Compound
Historical Development
Potassium dihydrogen phosphate, often called monopotassium phosphate or MKP, has been around in the chemical industry long enough for folks to figure out both its potential and pitfalls. Early chemical researchers in the 19th century, driven by a growing curiosity about plant nutrition and fertilizer efficiency, isolated and characterized this simple phosphate salt. Over time, scientists and industrialists built up a toolkit for using MKP in agriculture that pushed crop production far beyond what older organic manures could manage. Fertilizer production through the 20th century started to rely on it for reliable and concentrated phosphorus and potassium delivery, which changed the trajectory of global food yields.
Product Overview
Monopotassium phosphate lands on shelves as a crystalline material, mostly in bagged powders or granules. Its formula, KH2PO4, points to a blend of potassium and phosphorus. Every year, farmers, food processors, and researchers keep pulling demand. Growers use it as a fertilizer, but its clean and highly soluble form also finds place in food manufacture for buffering and as a yeast nutrient in fermentation industries. College labs and large-scale reactors all benefit from its reliable availability and purity.
Physical & Chemical Properties
Potassium dihydrogen phosphate stands out as a colorless, odorless crystal, prone to picking up moisture from the air—something I’ve seen many times with chemical storage gone wrong. It dissolves rapidly in water, results in a neutral to slightly acidic solution (pH floats near 4.2 at a standard 1% concentration), and offers a relatively high solubility ceiling at normal temperatures. The salt remains stable if dry and stored cool, but careless handling in humid rooms soon leaves it caked and less usable. As a straightforward ionic compound, it resists decomposition under most normal conditions, unless you throw it in the furnace, where it gives off tricky phosphorus oxides.
Technical Specifications & Labeling
Manufacturers must stick close to technical grades that spell out minimum potassium content (usually 28% by weight as K2O equivalent) and phosphorus content (often 52% as P2O5). Labels ought to show product purity, trace impurities like heavy metals, and batch identification. Food-grade, feed-grade, and technical-grade certifications call for stricter controls on contaminants. A diligent producer will include handling instructions, storage advice, and recommended shelf life. Failing to keep up with these details brings regulatory scrutiny and disappointed end users, especially in export markets where standards tighten up even beyond the home country’s rules.
Preparation Method
Companies scale up potassium dihydrogen phosphate production through a straightforward acid-base reaction. In practice, phosphoric acid meets up with potassium carbonate or potassium hydroxide, yielding MKP and water as byproduct. I’ve seen setups where folks feed streams of reagent into a controlled reactor, chase the reaction to completion, and after filtration and crystallization, dry out the product fast to dodge caking. Getting particle size right, keeping heavy metals low, and watching for iron and chloride contamination differentiate good chemistry from the sloppy stuff. Equipment cleanliness and proper pH control always make a huge difference to final purity.
Chemical Reactions & Modifications
MKP goes through several key reactions in the fields and labs. It acts as a buffering salt, helping to maintain solution pH in fermented foods or cell culture. Reacting it with more potassium sources or phosphoric acid twists it into dipotassium phosphate or other polyphosphates. In glass and ceramic glazes, it takes high temperatures and helps flux reactions. For folks working on new fertilizers, you can blend MKP with urea or ammonium salts to fine-tune nutrient mixes. In water treatment, it sometimes enters blend formulas for corrosion inhibitors or as a precipitant for heavy metals. Manipulating its chemistry opens new uses for plant biologists, glassmakers, and waste processors alike.
Synonyms & Product Names
Across industries and international trade documents, potassium dihydrogen phosphate wears several names. People might call it monopotassium phosphate, MKP, or use legacy tags like potassium phosphate monobasic. In European catalogs, it sometimes shows up as E340 in food applications. Chemical supply firms assign codes like CAS 7778-77-0 or EC 231-913-4, smoothing trade and regulatory compliance. No matter the label, what you get in the bag stays the same: a reliable mineral salt with a double punch of potassium and phosphorus.
Safety & Operational Standards
This salt doesn’t light up safety headlines—few fire hazards, limited toxicity, and it carries a low risk to adults in handling. Still, powder drifting in workrooms can cause eye and throat irritation, so dust masks and goggles stay wise in busy factory environments. Every workplace should train folks not to eat or smoke near open bags and to wash up if they get crystals on their skin, since repeated exposure can cause drying and dermatitis. Storage away from acids, strong alkalis, and direct moisture blocks clumping or accidental decomposition. Disposal dovetails with broad environmental rules since phosphate-rich runoff still threatens waterways with algal blooms, and careless handling can make problems for neighbors downstream.
Application Area
Fertilizer and agriculture soak up most of the world’s MKP output. Row crops, orchards, and greenhouses get a boost from the salt’s high nutrient analysis—growers appreciate the chlorine-free formula for sensitive plants. Hydroponic gardeners lean on MKP for bloom stages and root development. Food processing uses it to control acidity and stabilize fermentation, especially in baking, dairy, and beverage lines. It also flows into animal feed mixes as a palatable and easy-to-utilize phosphate source. Over in industrial circles, MKP acts as a dispersant, buffer, and as a specialist ingredient for flame retardants, water softeners, and ceramic processes. Few salts bridge commercial farming, food technology, and niche industries as neatly.
Research & Development
Research labs haven’t stopped finding new angles for potassium dihydrogen phosphate. Plant scientists keep looking at the interplay of MKP fertilization and soil health, especially under climate stress or as part of precision agriculture programs. Over in materials science, researchers tweak MKP with dopants to grow nonlinear optical crystals—these find use in advanced lasers and medical imaging. In energy storage circles, MKP comes up as a potential electrolyte or as a support material in metal-air batteries. Even wastewater engineers comb through options for phosphate recovery, and MKP offers a platform for further work in nutrient recycling. Academic and company collaborations push the salt into startup pitches, patent filings, and technical bulletins year after year.
Toxicity Research
Most regulatory reviews have found potassium dihydrogen phosphate to be low in acute toxicity, provided people avoid swallowing large quantities. That said, overapplying it to fields or discharging it into waterways creates classic environmental headaches—eutrophication and algal blooms can choke out fish and aquatic plants. Chronic overexposure in workers may worsen existing respiratory problems or skin irritation if dust is uncontrolled. Food safety experts and pharmacologists run routine checks on residue and metabolic impact, usually finding that regulated doses carry little risk to human health, but the push for sustainable use keeps growing. Ecologists keep an eye on runoff, and water testing teams monitor its impact all the way through municipal treatment works.
Future Prospects
Next-generation agriculture shapes much of potassium dihydrogen phosphate’s horizon. With pressure to reduce environmental harm and boost crop yields, precision fertilization techniques and smarter delivery systems for MKP will almost certainly see greater adoption. Water-soluble forms designed for controlled release, sometimes spun into new polymer matrices, hold promise for reducing waste and runoff. Alternative food technologies, including cell-based protein and advanced fermentation, also lean on reliable buffering salts like this one. Over in renewables and green chemistry, using MKP in specialty glass or energy storage could open up niche but high-value markets. Policies driving circular economy and phosphate recovery may see former “waste” streams cleaned and recycled through MKP, blurring the lines between commodity and specialty product. All the same, the salt’s old roles in field, feed, and food still matter, and keeping production clean and sustainable remains a whole-industry imperative.
What are the main uses of Potassium Dihydrogen Phosphate?
Farming Gets a Boost
Out in the fields, crops need more than sunlight and water to thrive. Potassium dihydrogen phosphate offers a quick dose of two key nutrients—potassium and phosphorus. Farmers turn to it when they want to push for healthy roots or better fruit set. I’ve seen growers use it as a foliar spray during critical growth stages because it dissolves easily in water and doesn’t burn crops like harsher fertilizers do. The real draw comes from its balanced nutrient ratio, especially in greenhouses where tailored feeding gives growers an edge. It feeds not just the leaves but the soil’s microbial life, building the foundation for sturdy plants season after season.
Keeping Food Fresh and Safe
Once food leaves the farm, it faces another set of challenges. Packaged baked goods dry out or lose color quickly; cheese and meats spoil and take on strange flavors. Food companies add potassium dihydrogen phosphate to processed foods as a stabilizer and a moisture controller. It keeps cheese meltable, ensures canned veggies stay colorful, and even helps bread rise evenly. My time working at a bakery taught me how something as simple as a dough conditioner can mean the difference between flat loaves and photo-worthy bread. The phosphate salt slows spoilage, keeps flavor true, and maintains a steady pH, which defends against bacteria. On labels, you’ll find it as a “food additive”—usually in very small amounts, since excess phosphate isn’t good for health.
Bringing Science to Classrooms and Labs
High school science labs rely on simple ingredients, and potassium dihydrogen phosphate shows up in many hands-on experiments. Teachers use it to make phosphate buffers—solutions that keep a steady acidity even as students add other chemicals. This matters for experiments involving enzymes or growing bacteria, where small pH shifts can ruin results. I remember being amazed at how one clear powder could keep our biology experiment working from start to finish, no glitches. Outside classrooms, researchers use it in plant tissue culture, helping propagate rare or valuable plants in sterile jars. Here, precise control over nutrients pays off with healthy shoots and roots.
Clean Energy and Electronics
Potassium dihydrogen phosphate steps far beyond farming or baking. Battery designers use it when they build certain rechargeable cells. It helps manage the flow of electricity, working as an electrolyte because it dissolves into ions cleanly and doesn’t corrode metal parts like some acids do. In optics, the very same chemical forms crystals for lasers, showing up in devices from research labs to eye surgery tools. The crystal’s ability to split light in special ways has driven plenty of innovation in telecommunications. During a summer internship with a tech firm, I saw just how precise the requirements get: one flaw in the crystal, and the whole laser is off.
Looking at the Future
Potassium dihydrogen phosphate supports some of the world’s most essential jobs—from feeding cities to powering research. As agriculture and technology grow more complex, the demand for smart, safe solutions only climbs. Farmers, food companies, and scientists keep a close eye on how much and how often they use it. They’re switching to targeted applications and better monitoring for phosphorus runoff, since overuse can throw natural ecosystems off balance. It’s a reminder that every simple compound sitting on a shelf has a big story behind it, touching everything from the bread on the table to the screens in our pockets.
Is Potassium Dihydrogen Phosphate safe for consumption?
Understanding Potassium Dihydrogen Phosphate
Potassium dihydrogen phosphate, known in food circles as E340, shows up frequently in ingredient lists. Bakers and food processors reach for it because it does a few important jobs: acting as an emulsifier, a stabilizer, and as a leavening agent in things like baked goods and some dairy replacements. You’ll spot it listed on sports drinks, processed cheeses, and powdered soups. It’s a blend of potassium and phosphate, both critical for the body’s workings.
Safety Evidence and Health Perspective
I always check what leading food safety agencies say before making any calls about a food additive. The U.S. Food and Drug Administration (FDA) puts potassium dihydrogen phosphate in the “Generally Recognized as Safe” category. That means researchers and toxicologists have covered the compound’s safety from many angles and haven’t flagged it as a hazard in the amounts used in food. The European Food Safety Authority (EFSA) also signs off on it, setting an upper safe intake for phosphate additives.
There’s a reason this matters. Both potassium and phosphate have key roles—potassium keeps blood pressure in check and phosphate helps build strong bones and manage energy use. Processed foods can add extra phosphate to the diet, though, which has raised some concern among kidney specialists. Phosphate isn’t a problem for healthy people, but folks with chronic kidney issues can struggle to clear it, which might build up and stress the body even more.
Balanced Intake and Real-World Use
Most people taking in potassium dihydrogen phosphate through processed foods won’t get anywhere close to danger zones for phosphate or potassium. My own experience working in community nutrition clinics showed me that few even know they’re eating these phosphate salts. People see a powder that mixes easily or cheese that stretches just right—not the chemistry. For the average adult, a balanced diet focused on whole foods keeps intake safe. It’s those depending on processed foods for most calories who inch closer to the edges.
Too much phosphate, particularly from additives, tends to be an issue for patients with kidney disease. Most dietitians working with this group teach label-reading skills and urge folks to keep an eye out for “phosphate” on ingredient lists. Even with additives like potassium dihydrogen phosphate, the critical factor rests on total dietary load and individual health.
Better Choices and Recommendations
For anyone with normal kidney function, potassium dihydrogen phosphate doesn’t trigger much worry at levels seen in everyday food. That said, some steps can make a difference for everyone. Choosing more fresh foods, reading labels, and asking questions at appointments with doctors or dietitians bring clarity. Industry transparency about how much phosphate gets into products could help shoppers make smarter choices, especially since phosphate pops up in soft drinks, deli meats, and powdered mixes.
For anyone managing heart or kidney health, talking openly with healthcare providers so dietary changes stay safe and targeted makes all the difference. Food is complicated, but the conversation about potassium dihydrogen phosphate shows the value of understanding the story behind the label—and figuring out what sits right with each individual’s body and needs.
What is the chemical formula of Potassium Dihydrogen Phosphate?
Lifting the Lid on KH2PO4
Potassium dihydrogen phosphate—KH2PO4—plays a bigger role in our lives than most people know. I remember the first time I saw a farmer pouring white powder from an unmarked bag into his irrigation system. He didn’t call it by its chemical name. He just said, “plant food with both K and P.” For those growing up without a chemistry set, those two letters—K for potassium, P for phosphorus—don’t mean much until you realize they show up on nearly every bag of plant fertilizer worldwide.
Why KH2PO4 Matters Outside the Lab
A typical garden won’t grow well if it runs short of potassium or phosphorus. This salt brings both elements to the soil in a way plants can use right away. Plants crave potassium for strong stalks, good flowering, and better drought resistance. Phosphorus steers root growth and kick-starts the energy cycle. The combination is tough to beat in agriculture.
Over the years, I’ve watched folks who work the land trust KH2PO4 because it dissolves easily, and they see results fast—greener leaves, healthier blooms, and higher yields. In hydroponic systems, it’s a top pick since it leaves little residue and shoots nutrients straight to the roots.
Real-World Challenges and the Road to Solution
Despite these strengths, overusing mineral fertilizers brings headaches. Soil and water can get overloaded with phosphorus, running off fields and choking nearby rivers or lakes with thick algae. This isn’t a new story. From the farm belt in the US to vegetable fields in Southeast Asia, too much of a good thing has turned ponds into sludge.
Careful management offers a way forward. I’ve seen local extension agents teaching farmers to test the ground, figure out the true nutrient need, and adjust the amounts used. Instead of guessing, they check actual soil levels for potassium and phosphorus and only add more if the test calls for it. This small change saves money and keeps water cleaner.
In the food industry, KH2PO4 shows up as a stabilizer. Bread makers use it to control pH, cheese producers rely on it for texture, and soft drink companies hide it among the list of ingredients to balance taste. In medicine, it slips into saline solutions and IV bags.
Reflections and Path Forward
What matters most, looking back, is that people treat KH2PO4 with respect, not just as a shortcut to bigger crops or tastier food. Its chemical formula—KH2PO4—points to a compound that can build up soil or, if misused, tip delicate ecosystems out of balance. Growers, food makers, and the folks who regulate all of this each have a piece to play in reaching the right balance.
As food demand climbs, so does the need for smart choices. Helping everyone—from farmers to parents to food scientists—understand what’s in the bag or bottle leads to better decisions for both people and the places where we live.
— Written from my experience working on farms, in science labs, and around the dinner table, seeing how one formula shapes the world around us.How should Potassium Dihydrogen Phosphate be stored?
Why Storage Isn’t Just a Box on a Checklist
Ask anyone on a lab floor or in a fertilizer warehouse: stuff doesn’t always get stored “like the label says.” Bags pile up. Containers get shoved wherever there’s room. For a chemical like potassium dihydrogen phosphate, that casual approach leads straight to spoilage, clumping, or even safety problems. If you’ve spent hours weighing and mixing only to discover a bag full of rock-hard lumps, you know the feeling. Proper storage isn’t just about regulation. It protects everyone’s investment—time, money, and peace of mind.
What Potassium Dihydrogen Phosphate Really Needs
This chemical draws water from the air like a sponge. Humidity turns a powdery supply into a brick. Once that happens, good luck getting a precise measurement or a clean solution. Dry storage makes all the difference. It’s best to seal it tight, in air-proof containers—think screw-cap plastic jars or heavy-duty, resealable bags. Thick bags won’t cut it if the seal isn’t strong. A forgotten twist-tie won’t stand up to the air for long.
Temperature matters. Cool spaces slow down degradation. That doesn’t mean freezing. A shelf away from heat vents, out of sunlight, and in an insulated room works. High heat invites breakdown, and sunlight triggers instability, slowly changing the chemical’s properties. On a farm, this means putting the stock in the shed, not outdoors under the tarp, and not near any engines or boilers where temperature spikes often go unnoticed.
Separating Chemicals Saves Lives and Yields
Potassium dihydrogen phosphate sits comfortably with other phosphates, but sitting side-by-side with strong acids, bases, or oxidizers is like playing with matches. Accidental spills or wet floors can trigger unwanted reactions. There’s also the spread of contamination—granules pick up residues from their neighbors, spoiling the clean chemistry needed for both food production and scientific research. Keeping a dedicated shelf in storage areas and marking containers goes a long way.
Label Everything—And Not Just for Yourself
Mix-ups happen. You may remember that plain white bag holds potassium dihydrogen phosphate. The person who grabs it next week might not. Clear, sturdy labels listing both the chemical name and the date of storage prevent mistakes. These aren’t scare tactics—they’re lessons learned from real lab accidents, lost batches, and even regulatory fines. If work spans shifts or gets handed off, reliable labeling protects everyone who handles these materials.
Good Storage Cuts Down on Waste
Chemical budgets run tight in research, agriculture, and manufacturing. Ruined stock goes straight to the expense column. In one hydroponic project, poorly sealed containers led to months of wasted nutrients and plant stunting—one quick fix with air-tight jars made all the difference for future crops. Small changes, like grouping chemicals by use or using smaller containers for frequent access, keep bigger bags untouched and safer for longer.
Make Storage a Team Conversation
Relying on signs alone doesn’t change old habits. New employees pick up on shortcuts fast if others ignore protocol. Discussing safe storage in regular meetings and giving everyone ownership over the space creates accountability. In larger workplaces, rotating who checks inventory or maintains records encourages consistency. A culture of safe storage isn’t just about avoiding trouble; it keeps everyone working smoother and smarter.
What is the solubility of Potassium Dihydrogen Phosphate in water?
Putting Numbers to Use: What the Data Shows
Potassium dihydrogen phosphate, a mouthful to say but simple enough in practice, turns up everywhere in global agriculture, labs, and water treatment plants. Knowing its solubility isn’t some chemist’s trivia. Solubility means real-world performance. According to multiple peer-reviewed sources, at 25°C, about 22.6 grams will dissolve in 100 milliliters of water. Step up the temperature to 80°C and over 80 grams will go into the same amount. Hotter water, notably, holds much more of it. This behavior means farmers and industrial users can tweak dosage and concentration based on the solution temperature.
Everyday Relevance: Why Numbers Like These Matter
I saw this firsthand working with a hydroponics startup. Growers kept asking, “How much can we add before things get cloudy?” Solubility wasn’t just a chemistry concept, it decided how efficiently systems ran: less sediment, more nutrients in the roots. Anyone running big tanks hates cleaning scale and sludge, so knowing the threshold for dissolving prevents downtime, extra work, and wasted money.
Schools and research labs rely on the same data. Dilute solutions help reproducibility in scientific studies, something that’s gotten major attention in recent years. A standard, well-mixed solution cuts down error, keeps experiments on track, and upholds research ethics by sticking to trustworthy methods.
Challenges and What Gets Overlooked
Many treat solubility numbers as gospel, but water quality bites back. Hard water—rich with calcium or magnesium—brings new headaches. Sometimes, not all the salt dissolves. Precipitates form, clogging fine filters or irrigation lines. A solution that should flow clean starts leaving gunky deposits and reduces yield. Studies from agricultural supply chains show that water hardness can mess with potassium and phosphorus delivery to the roots and, in turn, crop growth.
It’s tempting to overlook temperature’s effect, too. Farms with outdoor mixing tanks or industries operating across seasons hit solubility ceilings in winter. Inconsistent mixing leads to uneven application and, over time, wasted product. Conversations with field managers often drift to how frustrated they get over “cloudy solution days,” which are often triggered by cold spells.
Smart Practices and Solutions That Work
Nobody wants preventable failures. A straightforward fix starts with testing the water—every time, not just once a season. Adjusting for local conditions means fewer surprises. Where possible, heating the water or pre-dissolving the salt in a small warm batch before mixing in bulk solves stubborn solubility issues.
Regulators and consultants encourage equipment upgrades: better dosing pumps, inline filters, and tank agitators. These changes smooth out the rough spots, cut maintenance calls, and let operators zero in on the actual job, whether growing lettuce or running a chemical process. For smaller operations, keeping a reference chart showing maximum solubility by temperature on the wall near the mixing station takes out the guesswork.
As we all chase smarter ways to feed the world, clean water and simple chemistry facts like solubility set the foundation. Knowing and respecting these numbers means trouble-free operations, happier operators, and, often, better yields at the end of the cycle.
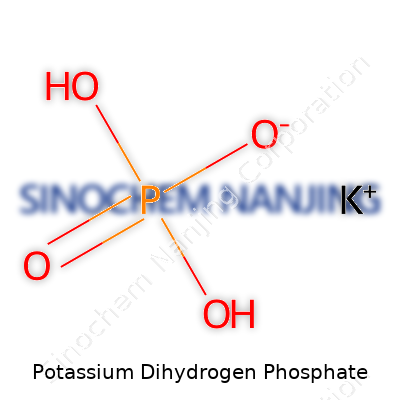

| Names | |
| Preferred IUPAC name | potassium dihydrogen phosphate |
| Other names |
Monopotassium phosphate Potassium phosphate monobasic MKP Mono potassium phosphate Potassium dihydrogen orthophosphate KH2PO4 |
| Pronunciation | /pəˈtæsiəm daɪˈhaɪdrədʒən fəˈsfeɪt/ |
| Identifiers | |
| CAS Number | 7778-77-0 |
| Beilstein Reference | 1713763 |
| ChEBI | CHEBI:62975 |
| ChEMBL | CHEMBL1201147 |
| ChemSpider | 68238 |
| DrugBank | DB09449 |
| ECHA InfoCard | 03b1726e-b3a4-45ed-9c7b-818b7cef7f11 |
| EC Number | 231-913-4 |
| Gmelin Reference | 868 |
| KEGG | C00222 |
| MeSH | D010935 |
| PubChem CID | 516951 |
| RTECS number | TC6615500 |
| UNII | XN1ZA5T89G |
| UN number | UN9149 |
| Properties | |
| Chemical formula | KH2PO4 |
| Molar mass | 136.09 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.338 g/cm³ |
| Solubility in water | 250 g/L (20 °C) |
| log P | -2.0 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 7.2 |
| Basicity (pKb) | 11.8 |
| Magnetic susceptibility (χ) | -2.8·10⁻⁶ |
| Refractive index (nD) | 1.509 |
| Dipole moment | 1.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 130.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1477 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2835 kJ/mol |
| Pharmacology | |
| ATC code | B05XA07 |
| Hazards | |
| Main hazards | May cause eye, skin, and respiratory tract irritation. |
| GHS labelling | **GHS labelling of Potassium Dihydrogen Phosphate:** "Not classified as hazardous according to GHS |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | May cause respiratory irritation. |
| Precautionary statements | Wash hands thoroughly after handling. Do not eat, drink or smoke when using this product. IF SWALLOWED: Call a POISON CENTER or doctor/physician if you feel unwell. Rinse mouth. |
| NFPA 704 (fire diamond) | 0-0-0 |
| Explosive limits | Non-explosive |
| Lethal dose or concentration | LD50 oral rat 4,640 mg/kg |
| LD50 (median dose) | Oral LD50 (rat) 4640 mg/kg |
| NIOSH | TTQ37250ZZ |
| PEL (Permissible) | Not established |
| REL (Recommended) | 13-62-0 |
| Related compounds | |
| Related compounds |
Monopotassium phosphate Dipotassium phosphate Monosodium phosphate Disodium phosphate Trisodium phosphate Ammonium dihydrogen phosphate |

