Potassium Dihydrogen Arsenate: Legacy, Risk, and Possibility
Looking Back: The Path of Potassium Dihydrogen Arsenate
Potassium dihydrogen arsenate traces its roots to the old days of chemical exploration, where curiosity often outweighed caution. For more than a century, researchers and early industrial chemists paid close attention to salts of arsenic, including this compound, because they sought versatile solutions in everything from early agriculture to scientific analysis. Some early uses embraced the compound’s sharp reactivity and solubility, but historians know well the price paid by those unaware of the hidden dangers arsenic compounds deliver. The chemical never became a household staple, but in the right hands, its properties opened new possibilities for separating minerals and tweaking the chemistry of solutions in pioneering labs.
Structure and Character: The Lay of the Molecule
Anyone who spends time in a laboratory recognizes potassium dihydrogen arsenate the moment fine, colorless crystals tumble out of solution. The compound dissolves well in water, giving an acidic solution that hints at its close relation to other phosphate and arsenate salts. Handling it, you notice a faint metallic aftertaste in the air, familiar to folks who have worked with arsenic compounds before. On a molecular level, the structure resembles potassium dihydrogen phosphate, swapping the phosphorus out for arsenic. That seems small, but the change transforms behavior, toxicity, and environmental impact.
What Goes Into the Label: Honest Technical Specifications
Chemists know the importance of getting technical labels right, especially for risky compounds. Potassium dihydrogen arsenate stands as an example where clarity equals safety. Commercial packets specify purity—often over 99 percent for research purposes—and warn about strong oxidation reactions and moisture sensitivity. Responsible suppliers remind you that storage in dry, airtight containers isn’t just a suggestion. Every label carries the unmistakable warning: poison.
Roots and Routes: From Raw Material to Finished Compound
The journey from ore to potassium dihydrogen arsenate relies on some old-fashioned chemistry. In the classic process, arsenic trioxide gets dissolved in water, treated with potassium carbonate or potassium hydroxide, then carefully acidified to swing the reaction toward the dihydrogen arsenate salt. Each step, manual or automated, requires respect for the raw materials. Workers and researchers who prep such compounds understand the personal protective gear isn’t an afterthought—the fumes, contact dust, and splashes can cause trouble even for the most seasoned hands.
Synonyms, Identity, and Naming Confusion
In the world of chemical nomenclature, potassium dihydrogen arsenate doesn’t hide. Other names like monopotassium arsenate or simply KH2AsO4 turn up in academic writing. I’ve seen confusion spring up more than once, especially with close cousins like potassium dihydrogen phosphate. Confusion here carries consequences, so putting the right name on every jar keeps everyone safe and research accurate.
Reacting Under Pressure: Chemical Flexibility and Risks
Potassium dihydrogen arsenate reacts with all the readiness of a true arsenate. In aqueous solutions, it interacts with metal ions and forms precipitates that can uncover new properties. It steps in for phosphate in some analytical methods, but nobody forgets the risk that comes with unnoticed traces in other reactions. Chemists see the difference between theory and practice—unexpected byproducts or contamination can turn an experiment from routine to hazardous.
Never an Ordinary Product: Safety Protocols and Human Cost
Handling this compound underlines the real divide between theory and responsibility. Arsenic is notorious for its toxicity, and potassium dihydrogen arsenate is no exception. I remember strict controls in university stockrooms, where access needed signatures and detailed logs. Labs maintain dedicated fume hoods, waste disposal bins, and emergency antidotes. As colleagues have shown, careless handling of arsenate—without gloves, without eye protection—leads to hospital visits, and sometimes worse. Institutions enforce training and standardized procedures because mistakes don’t get forgiven easily.
Application: Science, Industry, and Curious Gaps
Use cases look very different than in the height of the arsenic era. Today’s applications stick mostly to specialized research. Analytical chemistry sometimes chooses arsenate salts for calibration or as controls in phosphate detection techniques. Some researchers call on it for geochemical or environmental testing, simulating contamination or remediation strategies. Its essential role faded in the shift toward non-toxic alternatives, particularly in fertilizers and pesticides where arsenic once loomed large. That disappearance came as risks became clearer, showing that scientific progress rarely follows a straight line.
Where Research Leads: Unraveling Arsenate Toxicity
Every new study on arsenic compounds carries lessons for public health and toxicology. Potassium dihydrogen arsenate’s solubility turns it into a ready route for arsenic exposure, teaching researchers how arsenates move through soil, water, and living bodies. Animal studies offer early warnings for chronic toxicity: symptoms appear insidiously, from skin and neurological issues to cancers. Drinking water contamination in parts of the world reinforces the importance of detection methods—if we understand how compounds like these behave, communities have a fighting chance to keep exposure levels in check. In my experience, researchers facing arsenic’s legacy always work with a sense of urgency, knowing long-term impacts reach far beyond the lab.
What Comes Next: Innovation Against the Shadow of the Past
Looking at the future, the legacy of potassium dihydrogen arsenate casts a long shadow. Most paths move away from arsenate use, especially where safer alternatives offer comparable performance. The push leans toward better detection, removal, and recovery technologies: filtration resins targeting arsenate ions, sensors able to spot tiny concentrations in water, and smart ways to immobilize arsenate in soils. At the same time, curiosity drives investigation into rare situations where arsenate’s chemistry provides unique answers, all within tightly controlled environments. In research and in cleanup projects, experts push for transparency, clear labeling, and policies that put safety above all. Experience says progress grows from honest risk assessment, patient innovation, and respect for the dangers lurking within the periodic table’s dark corners.
What is Potassium Dihydrogen Arsenate used for?
Understanding Potassium Dihydrogen Arsenate
Potassium dihydrogen arsenate shows up in chemistry labs more often than in daily conversation, but its impact ripples through agriculture, science, and history. This compound, made up of potassium, hydrogen, and arsenic, has brought utility and risk to the table for generations. Anyone who has ever studied agricultural science learns early that arsenic compounds once played a huge role in crop protection. Potassium dihydrogen arsenate, in particular, became one of those products that promised better harvests and healthier plants—for a price.
Agriculture's Complicated Relationship
For decades, pesticides and herbicides leaned on arsenate compounds for stopping bugs and weeds in their tracks. Potassium dihydrogen arsenate worked its way into the rotation because it handled pests well. Farmers and gardeners valued its effectiveness, even as early research flagged concerns about toxicity. What looked like progress for crops meant headaches or worse for communities downwind. People in the area soon realized runoff could flow into streams, where arsenic seeped into the water and soil, hanging around for years.
My grandfather talked about seeing barrels of chemicals stacked in the barn when he worked the outskirts of farming towns in the ’60s and ’70s. Regulations asked more questions about arsenic’s effect on health after cases of illnesses rose in areas around widespread usage. Enough evidence connected these chemicals to a rise in cancers and neurological problems, especially for workers exposed over time.
Role in Scientific Research
Chemists often use compounds like potassium dihydrogen arsenate to investigate how toxins interact with living cells. It serves as a source of arsenate ions in experiments, helping researchers sketch out how arsenic interferes with normal biological processes at a molecular level. While these studies take place safely inside controlled labs, the lessons they provide matter in making policies. In classrooms or lab courses, small amounts teach future toxicologists how to measure, handle, and contain hazardous materials responsibly.
Sometimes, scientists turn to this compound as a reference material. Quality control checks in analytical chemistry rely on known substances to calibrate sensitive equipment. Accuracy helps diagnose pollution in rivers near abandoned orchards or track minute levels of arsenic in drinking water.
Environmental Risk and Regulation
People living in areas with a history of arsenate use learned hard lessons about environmental hazards. Soil and water absorb arsenic, and nature doesn’t rush to clear it away. The Environmental Protection Agency and health agencies in countries across the world now keep tight limits on use, especially near food crops. After growing up in a region with a long agricultural tradition, I’ve seen neighbors worry about old farm plots finally getting tested for arsenic, sometimes holding up sales for months as cleanup crews move in.
Strict safety regulations and tracking for hazardous waste aim to prevent history from repeating itself. Better education about chemical risks in school science classes makes a difference, too. Kids no longer handle dangerous substances without knowing the risks, thanks to stories and data from past generations.
Looking Forward
Potassium dihydrogen arsenate tells a story about scientific advances and the responsibility that comes with them. Safer alternatives in pest control replace the need for risky compounds in most cases. Genuine progress comes from farmers, chemists, and policymakers learning from experience. Open conversations about where old chemicals ended up, and ongoing research into residual contamination, protect people and the landscape.
Is Potassium Dihydrogen Arsenate toxic or hazardous?
Understanding the Risks
Potassium dihydrogen arsenate doesn’t turn up in headlines too often, but its chemical makeup deserves attention. The name itself gives away the core concern—arsenic. For generations, arsenic’s reputation wasn’t built on mere folklore. It has played the villain in both history books and public health records. There’s no dodging the fact: ingesting or even inhaling this type of compound can have serious consequences.
The Science Behind It
In my college chemistry days, a single phrase about arsenic stuck: “more poisonous than most people realize.” That came up early in toxicology lessons. Potassium dihydrogen arsenate brings the same trouble as other arsenic-based compounds. It doesn’t break down into something harmless. Instead, it finds its way into the body and disrupts cellular function. This means cells can't do their jobs right—enzymes get blocked, critical metabolic pathways become useless, and before long, organ systems start showing signs of trouble. Human bodies aren’t built to filter out high levels of arsenic, especially over long stretches of exposure.
Workplace Worries
Factories and labs sometimes keep potassium dihydrogen arsenate on hand. Where companies use it, strict safety protocols become a way of life. Ventilation pulls toxic dust out of the air. Gloves, goggles, and heavy-duty clothing keep workers from contact. Years ago, I worked in a research facility, and our safety manual dedicated a whole section to handling arsenic compounds—not out of regulatory obligation, but because small mistakes added up quickly. Colleagues would always double-check containment and signage. Accidents rarely happened, but we all felt the weight of what could go wrong.
Health Consequences
Small exposures add up. Short-term effects—vomiting, diarrhea, muscle cramps—show up fast with higher doses. Long-term encounters with even minor arsenate dust or solutions creep up more quietly. For example, chronic, low-level exposure raises the risk of cancers in the skin, bladder, and lungs. In places where arsenic regularly contaminates groundwater, communities see higher rates of cardiovascular issues and developmental disorders in children. It’s not drama or media hype driving concern. Peer-reviewed medical research paints a clear picture of risk. According to the Environmental Protection Agency, any exposure above 10 parts per billion in drinking water becomes unsafe over the years.
Environmental Impact
Letting potassium dihydrogen arsenate enter the environment creates more than a local hazard. Plants pull up arsenic from soil, cropping up in vegetables and grains. Wildlife suffers, too. It slowly poisons soil bacteria and aquatic life, damaging the food web from the ground up. In older mining towns where arsenic dust from compounds like these filtered into rivers, wide swaths of the ecosystem disappeared. It doesn’t wash away cleanly or break down fast, so cleaning up these sites takes years—sometimes decades.
Solutions and Better Practices
Limiting risk takes more than warning labels. Substituting less dangerous chemicals works for some industries. Companies implement closed-system handling, so workers never actually touch or breathe in the stuff. Strong local laws on hazardous chemical storage reduce the odds of leaks or mishandling. Regular health screenings for those working near arsenic compounds help catch early signs of exposure before major issues develop. From personal experience, staff buy-in matters—the culture of safety builds layer by layer, with clear training and real consequences for breaking the rules.
What is the chemical formula of Potassium Dihydrogen Arsenate?
Everyday Chemistry in Action
A lot of folks don’t spend their morning coffee wondering about the formula for potassium dihydrogen arsenate, but digging into simple chemical naming can open up a surprising amount of practical knowledge. This compound carries a lot more meaning than an arrangement of letters and numbers. Its formula, KH2AsO4, pulls together chemistry’s way of making sense of the world: what works, what shouldn’t get mixed, and why certain minerals fit particular jobs in science, farming, or industry.
The Power of Naming
Learning to break down a name like potassium dihydrogen arsenate makes life easier for students, lab techs, and anyone aiming to keep a garden healthy without risk. Potassium points to K, the classic alkali metal found in soils and bananas. Dihydrogen means two hydrogens (H2). Arsenate signals the presence of arsenic in its pentavalent form, matching the AsO4 group. Get these together, and the combination makes KH2AsO4.
This sort of clear language acts as a tool, not only in school but in safe handling. Arsenic compounds, for instance, have earned their reputation for toxicity. Someone who’s ever read about groundwater contamination or historical pesticides knows that recognizing what’s in the bottle matters most. A slip-up in formula—an accidental K2HAsO4, for instance—could produce totally different effects or risks.
Personal Experience Builds Trust in Science
Back in a college chemistry lab, I spent more than a few hours sorting bottles and matching formulas to safety sheets. Potassium dihydrogen arsenate came up during a lesson on buffer solutions and weak acids. A label with the wrong subscript would set off alarm bells—the teacher drilled into us the simple truth that every element carries its own weight and hazard. It’s basic science, but it can also mean the difference between a safe routine and a costly accident.
Why This Matters in the Field
Arsenic sits in the middle of arguments about water safety and mining practices. Potassium has its own story in fertilizer. Understanding the fine print of a compound like KH2AsO4 supports safe choices on the farm and in the lab. A single typo could put health at risk. According to the World Health Organization, even a few micrograms of arsenic per liter can make water unsafe. Farmers, teachers, and even parents managing home experiments all have reason to care about the correct details.
Building Better Habits
Learning to check chemical formulas accurately comes from both formal lessons and from careful, everyday practice. Businesses handling substances with arsenic—pesticide makers, mining firms, water treatment plants—carry the burden of clear, honest labeling. Industry guidelines, regular training, and open communication remain key for safety and trust.
Emphasizing solid science starts with understanding what simple naming delivers. Simple errors can bring harm, but a habit of double-checking formulas like KH2AsO4 helps avoid trouble. Growing familiar with chemistry’s language arms communities with the knowledge to push for cleaner water, safer schools, and better crops.
How should Potassium Dihydrogen Arsenate be stored and handled?
Treating Hazards With Respect
Anyone who’s ever worked with potassium dihydrogen arsenate knows this isn’t the sort of chemical you stash next to the coffee pot. It has a place in labs for research, but the risks it brings along are nothing to shrug off. Arsenic compounds aren’t just trouble for your health, they can leave a legacy in your workplace if you cut corners. Years ago I saw a tech’s gloves split while he handled a sample, and that mistake haunted him for weeks—rashes, worry, extra blood tests. Simple errors can snowball into big problems.
Storage: Out of Sight, Not Out of Mind
Potassium dihydrogen arsenate needs a home where only those who understand it can find it. Locked cabinets marked with hazard signs send a clear message—no casual visits allowed. Dryness is key. Water spills or muggy air trigger two potential problems: product breakdown and contamination of nearby surfaces. At my old lab, humidity crept into a half-sealed bag. Cleaning up wasn’t pleasant, and the smell lingered for days, but more importantly, we spent hours checking for dust spread.
Lab supervisors often want every container labeled with more than just a name. Add hazard symbols, note the date it arrived, and jot down the staffer who last handled it. Traceability saves time and arguments if anything goes wrong. Make sure no flammable substances share the shelf. Safety data sheets stay close by, taped inside a cupboard door where no one forgets them.
Taking Care, Not Chances
Gloves should fit well—nitrile or neoprene, double-layer if possible. Eye protection and a lab coat count as the bare minimum, not just optional extras. Working under an active fume hood becomes the norm. Arsenic’s dust and vapors have an ugly track record for poisoning air and skin alike. On tough days, people slip up and keep their mask off “just for a second.” Watching a young intern cough and panic after a tiny spill was a wake-up call—habit trumps haste every time.
No eating, no water bottles, and no phones on the same bench. Little lapses build up. I’ve seen contaminated equipment moved by mistake or pipettes left around, waiting to mess up the next project. Every used tool hits a special arsenic waste bin. Team members who looked out for each other built a tighter, safer lab. Calling out a friend for fumbling with gloves felt better than apologizing after an exposure.
No Mystery in Disposal
Hazardous waste rules aren’t just paperwork. Liquid and solid leftovers leave the building in secure, labeled containers. Local authorities make regular pickups, and paperwork proves the job’s done right. Missing a drop or two ends up contaminating floors and drains far from the bench—if you don’t follow the rules, the workplace pays the price in fines and health scares.
Building Safer Habits
Displaying clear safety instructions above the workspace, running regular drills, and making sure everyone knows what to do after a spill turns awkward silences into action. A chemist who respects their materials sticks around for the long run. Shortcuts with arsenic compounds write stories no one wants to remember.
What safety precautions are needed when working with Potassium Dihydrogen Arsenate?
Understanding the Risks
Potassium dihydrogen arsenate requires respect in the lab. This isn’t flour or table salt; it’s a compound that brings arsenic into the picture, a well-known toxic element. Exposure, even in small amounts, can lead to severe health problems, including organ damage and increased cancer risk, according to agencies like NIOSH and OSHA. I remember walking into a university stockroom, catching a glimpse of the hazard label, and instantly realizing how much attention this stuff demands.
Personal Protective Equipment
No one I’ve met reaches for this chemical without proper gear. Nitrile gloves, full-length lab coats, and splash-proof goggles are a must. Arsenic-containing dust or droplets don’t play around. A simple tear in a glove or an accidental touch to your face can tip the balance from safe to dangerous. A solid-shelled face shield isn’t excessive if you’re handling the compound in quantities likely to become airborne or splash.
Ventilation and Containment Matter
Fume hoods aren’t just a box with a fan—they save lives. Potassium dihydrogen arsenate powders and solutions can release invisible dust or vapors. I’ve seen researchers ignore a fume hood for a “quick” measurement, only to regret the careless shortcut. Local exhaust and containment keep the workspace safe and limit problems before they begin. Working with the sash down, using spill trays, and checking airflow before starting keeps hands and lungs protected.
Handling and Storage
Secure handling goes beyond stacking bottles on a shelf. Labeled, chemical-resistant containers prevent leaks or spills. Keeping this compound in a dedicated, locked poison cabinet keeps it away from casual contact. In my time around hazardous substances, I’ve learned not to underestimate the risks of cross-contamination or improper storage—one leaky container leads to widespread contamination. Storing arsenic compounds away from food, acids, and reducing agents keeps potential disaster at bay.
Accidents and Emergency Steps
After an incident, speed is everything. Eyewash stations and showers have to be close; seconds matter if arsenic gets on you. Absorb spilled solutions with inert materials—think vermiculite or sand—and keep waste in sealed, clearly labeled bags. Lab mates need to know the emergency plan before anyone ever opens a bottle. I’ve seen emergency drills make a huge difference when panic strikes. Calling the right team, reporting honestly, and cleaning thoroughly limit long-term damage.
Waste Management and Environmental Impact
Tossing arsenic down the drain—never acceptable. I’ve sat through multiple hazardous waste seminars just to stress this point. All containers, pipettes, gloves, and paper used with potassium dihydrogen arsenate go into designated hazardous bins. Labs work with certified waste processors who remove, neutralize, and dispose of arsenic compounds safely. Fines and environmental harm pile up fast for those who try to cut corners or pretend “a little bit” won’t hurt. The law expects us to treat this poison with caution for good reason, and so should every responsible worker.
Building Good Habits
Safety isn’t a checklist—it’s a mindset I’ve seen reinforced by good training and respected mentors. Reading updated SDS sheets, reviewing protocols, talking through steps with team members, and never rushing jobs have kept me and others healthy. Whether you’re new to arsenic or a seasoned chemist, treating potassium dihydrogen arsenate with focus and respect saves a world of hurt.
Respect the risks, follow proven practices, and keep safety personal—because chemicals like potassium dihydrogen arsenate don’t forgive shortcuts or carelessness.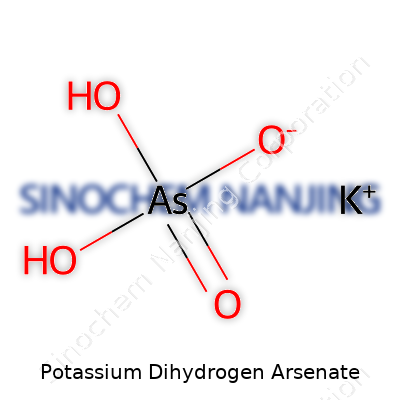
| Names | |
| Preferred IUPAC name | Potassium dihydrogen arsenate |
| Other names |
Monopotassium arsenate Potassium dihydroarsenate Monopotassium dihydrogen arsenate |
| Pronunciation | /pəˈtæsiəm daɪhaɪˌdrɒdʒən ɑːˈsəneɪt/ |
| Identifiers | |
| CAS Number | 7784-41-0 |
| Beilstein Reference | 873750 |
| ChEBI | CHEBI:131346 |
| ChEMBL | CHEMBL1201888 |
| ChemSpider | 22223 |
| DrugBank | DB11058 |
| ECHA InfoCard | 19acfcc6-62c4-4b73-9e2a-0e796d928b12 |
| EC Number | 231-911-0 |
| Gmelin Reference | Gm.474 |
| KEGG | C16242 |
| MeSH | D011003 |
| PubChem CID | 24757 |
| RTECS number | BQ6450000 |
| UNII | 3G0285K00S |
| UN number | UN1557 |
| Properties | |
| Chemical formula | KH₂AsO₄ |
| Molar mass | 196.02 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.36 g/cm³ |
| Solubility in water | easily soluble in water |
| log P | -3.2 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 1.99 |
| Basicity (pKb) | 12.1 |
| Magnetic susceptibility (χ) | -57.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.502 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 132.0 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –1736 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB04 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; carcinogenic; environmental hazard |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H300 + H330: Fatal if swallowed or if inhaled. H373: May cause damage to organs through prolonged or repeated exposure. |
| Precautionary statements | P261, P264, P270, P271, P272, P273, P280, P301+P310, P302+P352, P304+P340, P308+P311, P314, P330, P361+P364, P391, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-0-ox |
| Lethal dose or concentration | LD50 oral rat 14 mg/kg |
| LD50 (median dose) | LD50 (median dose): 14 mg/kg (oral, rat) |
| NIOSH | TC3675000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Potassium Dihydrogen Arsenate: "0.01 mg/m³ (as As) |
| IDLH (Immediate danger) | IDLH: 5 mg As/m³ |
| Related compounds | |
| Related compounds |
Arsenous acid Arsenic acid Sodium dihydrogen arsenate Potassium dihydrogen phosphate |

