Potassium Chromate: From History to Future Prospects in the Chemical World
Historical Development
Potassium chromate stands as one of those chemicals that highlight the uneven path of industrial growth and scientific curiosity. Tracking its history takes you back to periods when chemistry evolved alongside mining and global trade. In the early 19th century, the discovery and refining of chromite ore sparked a surge in chromate chemistry, pushing potassium chromate to the center of pigment and dye manufacturing. Chromium compounds reached European markets through grueling extraction methods, transforming not just industrial chemistry but also influencing the ways people could preserve metals and develop early photographic methods. By the time the element’s chemistry matured, potassium chromate became a key material in both scientific research and industrial production, making it more than a byproduct—it became a symbol of changing attitudes toward both chemistry and industrial safety.
Product Overview
Potassium chromate, also known as potassium salt of chromic acid, shows up in laboratories as a bright yellow, crystalline compound. This salt, recognized for both its vivid color and significant oxidizing power, bridges a long line of chromate salts that have shaped chemical manufacturing. Chemists and industries have kept potassium chromate in regular rotation not only because of its strong coloring ability, but also due to its utility in a range of chemical transformations. For those in the field, the chemical’s distinct yellow hue serves as a visible indicator in analytical chemistry and applied processes alike, confirming reactions or even the presence of certain ions.
Physical & Chemical Properties
Potassium chromate emerges as a solid—fine-grained, yellow, and eager to blend with water. Most chemists notice the way it dissolves smoothly, forming a bright yellow solution that points to its strong ionic nature. This chemical stability in water stands out, making it useful in processes needing reliable solubility. Its strong oxidizing character creates both advantages and risks: in the right settings, it helps chemists oxidize other compounds with reliability, yet in careless hands, those same oxidative powers can spark trouble. With a relatively high melting point reflecting its ionic bonds, potassium chromate stands up to moderate heat, but it will decompose with exposure to fire or concentrated acids, feeding further reactions that showcase its chromium backbone.
Technical Specifications & Labeling
The move towards more standardized labeling, which started in earnest during the last century, improved safety for potassium chromate and cut down confusion in laboratories. What was once handed over in simple brown glass bottles now comes with hazard symbols, risk phrases, and batch details. Product information ties potassium chromate to both its chemical formula, K2CrO4, and proper storage practices. Anyone who’s picked up a bottle is now greeted by warnings reminding them of the environmental and health risks tied to this compound. This reflects broader trends in chemical management, with more precise documentation supplanting the old guesswork that defined earlier industrial ages.
Preparation Method
Traditional methods for making potassium chromate don’t call for expensive gear, but they demand solid chemical knowledge. Engineers process chromite ores through roasting with potassium carbonate and air or oxygen, forcing chromium to migrate from its stable mineral form into the soluble chromate state. The resulting material dissolves into water, from which potassium chromate crystallizes as it cools. Each step produces byproducts that cannot just be discarded, feeding debates about industrial emissions and environmental footprints. Crafting potassium chromate on this scale shaped chemical infrastructure in the past and continues to make people examine each link in the supply chain.
Chemical Reactions & Modifications
Potassium chromate commands attention among analytical chemists. It reacts with barium salts to form barium chromate, a process that once formed the heart of gravimetric analysis for sulfate ions. It switches easily between chromate and dichromate forms, depending on the solution’s acidity. On the bench, shifting the pH flips its color from yellow to orange, a transformation easy for even undergraduate students to spot. In organic settings, its oxidizing activity turns alcohols into carboxylic acids. This duality—analytical and synthetic—keeps potassium chromate in chemical storerooms, even as safer substitutes become available for some uses.
Synonyms & Product Names
This compound passes under several banners, mainly potassium chromate and potassium salt of chromic acid. In various trade catalogs, it may turn up as K2CrO4 or simply “yellow chromate.” Over the decades, naming trends followed shifts in chemical nomenclature. Even as catalog standards tightened, some manufacturers stick with historical names, so chemists have learned to read beyond a label to be sure of what’s in the bottle.
Safety & Operational Standards
Potassium chromate doesn’t hide its danger. Regulators have flagged it as toxic, both by skin contact and inhalation, leading to strict controls on storage and handling. Modern labs use fume hoods, gloves, and eye shields to keep exposure close to zero. Spills require immediate clean-up, with waste sent to hazardous material disposal services. Trainings focus on minimizing dust, labeling containers with clear hazards, and using the smallest amounts needed to do the job. Over time, pressure from unions, activist groups, and scientific societies led to improvements in both the regulations and the mindset of lab safety, making potassium chromate a case study in the ongoing tug-of-war between necessity and caution.
Application Area
Throughout its industrial lifespan, potassium chromate played several roles that few compounds could match. Analytical chemistry took to it early, using its vivid color changes to spot and measure sulfates or chlorides. Tanning and dyeing workers found its properties ideal for setting pigments and preparing leathers. It protected metals from corrosion and lent a critical role in explosives research. Environmental restrictions have squeezed out some applications, but its stubborn presence in research labs hints at the depth of skills and methods built on its chemistry. Even today, where alternatives exist, potassium chromate’s reliability keeps it relevant for those needing precise tests or strong oxidizing power.
Research & Development
Chemists are not content with using materials "as is," especially if safer or cleaner options are out there. Research into potassium chromate now focuses on its replacement, targeted recycling, or safe disposal. Teams have explored ionic liquids, organic oxidants, and new chelating agents that mimic potassium chromate’s analytical applications without carrying as much risk. Funding flows toward processes that recover chromium from spent reagents and keep waste streams free from toxic chromate. These efforts get a push from both climate and health policy—a reminder that research is often shaped as much by regulation as by scientific curiosity.
Toxicity Research
No one can discuss potassium chromate without acknowledging its dark side. Chromium(VI), the key component, triggers cellular damage in living things, with established links to cancer and organ failure. Decades of studies have mapped how exposure harms human health, driving bans and strict oversight around the world. Inhalation of dust or skin contact moves chromium deep into the body, causing DNA damage and other effects seen in occupational health surveys stretching back to the age of early chromate factories. Ongoing toxicology research still searches for low-dose exposure effects and for better biomarkers, but the consensus sits squarely on the side of "handle with extreme care."
Future Prospects
Looking ahead, potassium chromate faces a narrowed future. Most industries search for safer replacements with less environmental footprint, aiming to phase out hexavalent chromium altogether. Academic labs keep potassium chromate around for specific legacy analytical techniques and controlled experimentation, but the big push in R&D and regulation points to greener chemistry. A slow transition to non-chromate alternatives marks progress for sustainability and safety, backed by policy changes and changing corporate cultures. Yet, the story isn’t over—potassium chromate remains a lesson in chemistry’s double-edged sword, balancing utility and risk, and demanding thoughtful approaches to both technical and ethical questions.
What is Potassium Chromate used for?
The Role of Potassium Chromate in Industry and Beyond
Potassium chromate grabs attention mostly in chemical labs and a range of industrial settings. It shows up as a bright yellow salt, but that sunny appearance hides some serious risks. The material plays a big role in analytic chemistry, old-school pigment manufacturing, and sometimes even water treatment.
Chemical Testing and Laboratory Work
Potassium chromate stands out as a classic indicator for detecting chloride ions during titration. In labs, people use it for the Mohr method, testing whether a sample contains common salt or not. That’s a basic step in water analysis and food quality checks. I remember the first time I used it in school — a tiny scoop turned the clear solution a canary yellow the instant chloride was present. It made it easy for beginners to see chemical changes in real time. Decades of testing show this technique still holds value since reliable results matter when drinking water safety or food quality are in question.
Manufacturing and Pigments
Before modern synthetic dyes, potassium chromate found its way into the production of pigments, especially yellow paints and inks. Older artists mixed it for bold, vibrant yellows that survived harsh sunlight. The pigment’s staying power once helped mapmakers and builders create weather-resistant designs. As toxicology research deepened, people moved away from using chromate-based colors in consumer products or artwork. Science learned that inhaling or touching this chemical comes with real risks — not just for factory workers but for communities downstream from plants.
Corrosion Inhibition and Water Treatment
In older water systems, potassium chromate helped slow down rust in pipes and heavy machinery. Many engineers used it as a corrosion inhibitor in cooling systems and boilers. Factories wanted to keep metal from rusting out, especially before stainless steel caught on. Over time, strict regulations grew because research connected hexavalent chromium compounds to cancer and other health issues. Environmental agencies now search for safer alternatives that still get the job done without poisoning water supplies or soils. Potassium chromate, like many industrial chemicals, became controversial where workers or neighbors breathed in the dust or sipped tainted groundwater.
Potential Solutions and Safer Alternatives
The pressure to reduce hazardous waste and protect workers keeps growing stronger. Plant managers look for less toxic chemicals to replace potassium chromate in every practical step. In testing, options like silver nitrate or ion-selective electrodes offer accurate readings without the same environmental baggage. Paint companies invest in organic and synthetic pigments rather than risking heavy-metal contamination. Water utilities lean into phosphate-based corrosion inhibitors, reducing the threat to local ecosystems. These shifts don’t come easy, but the benefits show up in lower healthcare costs, safer job sites, and cleaner rivers over time.
Weighing Health and Environmental Risks
No one can ignore the legacy of this bright yellow salt. Potassium chromate left a mark on science labs, artistic tools, and heavy industry throughout the last century. Today, experience and evidence from occupational health studies drive a much more cautious approach. Companies receive strict guidance on handling, storage, and disposal—protecting workers as well as nearby families. Regulatory bodies like OSHA and the EPA set exposure limits, check up on compliance, and encourage training before anyone opens a container. Moving forward, a real focus on best practices means potassium chromate only sticks around where safer options just don’t measure up yet, and even there, it’s watched closely.
Is Potassium Chromate hazardous to health?
Toxicity and Human Exposure
Potassium chromate isn’t something most folks come across at home or outside a laboratory. This bright yellow powder shows up in chemistry classrooms and certain workplaces, but the health risks it brings deserve serious attention. Unlike table salt or baking soda, potassium chromate carries danger with both short and long-term contact.
Inhaling the dust or fumes introduces toxic chromium (VI) compounds straight into the lungs. My time working around hazardous chemicals taught a tough lesson—neglecting basics like gloves and proper ventilation just isn’t an option with substances like this. Potassium chromate can irritate the nose and throat, but more worrying is what happens after repeated exposure: nosebleeds, ulceration, and even holes in septal tissue don’t sound like mild side effects.
Impact on Skin, Eyes, and Organs
People who get this chemical on bare skin might see redness and blistering. Without quick cleanup, chronic exposure can leave behind yellow stains and ulcerate skin. Splashes in the eye don’t just cause pain—there’s a real risk of serious or permanent damage. This danger isn’t based on rare incidents, either. The Occupational Safety and Health Administration (OSHA) lists potassium chromate as a substance that can trigger allergic reactions and severe dermatitis.
Chromium (VI) travels in the body once it enters via the lungs or skin. Scientists link this compound to kidney and liver damage. The International Agency for Research on Cancer (IARC) classifies it as a human carcinogen. Industrial accidents and careless storage can put anyone in harm’s way—but workers in metal plating, dye, and laboratory environments get most exposed.
Environmental Impact and Long-Term Concerns
Disposal and spills present another set of problems. Over the past decade, there have been cases where illegal dumping of chromium chemicals contaminated soil and water. In one community, children played near an old factory dump and ended up with unexplained rashes and nosebleeds, prompting a state investigation. Potassium chromate doesn’t break down easily, leading to slow, steady pollution of nearby streams and crops.
The trouble doesn’t end with direct health effects—plants and animals down the chain collect residues, and these slowly move up the food web. Farmers, factory neighbors, and cleanup crews all risk exposure if safety gets ignored.
Prevention: Controlling Risks and Supporting Safety
A strong safety culture makes a real difference. Keeping hazardous chemicals like potassium chromate locked away, equipping staff with personal protective equipment, and insisting on proper ventilation all protect lives. Regular air monitoring, washing facilities, and good work habits help stop contamination at the source.
At the public health level, regulations work best when communities, managers, and inspection teams stay involved. The rise in environmental reporting has brought more attention to chemical safety. These days, many workplaces now train their teams about danger signs and emergency response, not just theory.
Chemicals like potassium chromate serve a purpose in labs and industry, but ignoring the real risks invites harm. My personal encounters with chemical safety drills showed me how important it is to treat every bottle with respect—especially the bright yellow ones.
What are the storage requirements for Potassium Chromate?
Why Potassium Chromate Demands Respect in Storage
Potassium chromate is bright yellow, looks harmless, but surprises everyone because it carries serious risks. Anyone who’s ever worked in a lab or walked the aisles of a chemical store knows this isn’t something you tuck away on any random shelf. This compound deserves real attention, especially with its toxicity and environmental impact front and center. I’ve seen new technicians treat it like simple table salt—often fixing that mistake brought a new level of respect for the precautions written on the bottle.
Facts from Direct Experience
Potassium chromate reacts strongly with organic material. If stored anywhere near combustible stuff—paper towels, wood shelves, even certain kinds of plastic packaging—those risks multiply. I once saw a storeroom where a leaky bottle, sitting next to a box of latex gloves, stained everything yellow and made breathing hard for a whole week. Proper storage means keeping it in a cool, dry space far away from organic matter, flammable materials, and anything likely to spill or drip.
The Case for Secure Labeling and Containers
A lot of chemistry accidents come down to someone grabbing the wrong jar. Labelling matters. Thick, clear labels with hazard symbols keep mistakes to a minimum. Storing potassium chromate in sealed, shatter-proof containers makes clean-up and spills far less likely. I’ve seen labs switch to plastic-coated glass just for the peace of mind, after one too many accidental drops. Containers should never show signs of corrosion—moisture can start a reaction, sometimes without warning.
No Place for Casual Storage
Potassium chromate should never sit out in the open. Ventilated chemical storage cabinets—especially those with secondary containment trays—help stop spills from spreading. Dedicated spaces with posted warning signs and limited access bring peace of mind. Only those trained to handle hazardous chemicals should reach for these bottles. In my experience, the most organized labs run regular checks. If a bottle looks dusty or forgotten, it gets inspected before anyone opens it.
Environmental and Health Hazards
Leaking potassium chromate does more than ruin a shelf. It seeps into the air, dust, or even the ground. The compound is a known carcinogen. Anyone breathing contaminated dust, or anyone who gets it on their hands, faces long-term health risks. Personal protective equipment isn’t just for show—gloves, safety glasses, even dust masks in case of solid spills. Well-designed storage turns PPE into a simple step, not the last barrier against disaster.
Taking Responsibility and Looking Forward
Storage routines come down to personal responsibility. Recognizing the mistakes of the past—overlooked leaks, forgotten bottles, sloppy labeling—shapes daily habits. Leadership within a facility has to create a culture of safety, not just hand out rules. Training and refreshers, regular audits, and clear lines of communication help. No one should assume safety until they’ve checked with their own eyes. Pumps, dispensing tools, and spill kits keep accidents small before they grow into something serious.
Smart Solutions for Safe Storage
Good chemical storage never happens by accident. Choose cool, dry, well-ventilated cabinets, preferably with fire resistance. Lock these cabinets to keep out anyone not specifically trained for hazardous work. Always store potassium chromate away from food, drinks, and incompatible chemicals like acids or organic solvents. Daily checks for residue, leaks, or corrosion can make the difference between an ordinary workday and a full-scale cleanup.
What is the chemical formula of Potassium Chromate?
Getting to Know Potassium Chromate: Formula and Facts
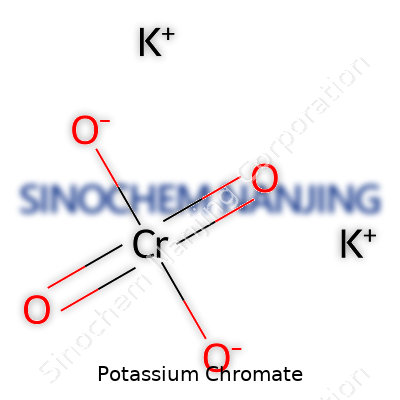
Potassium chromate sports a straightforward formula: K2CrO4. With two potassium atoms, one chromium atom, and four oxygen atoms, it translates to a clear, bright yellow powder. If you've set foot inside a high school chemistry lab, chances are you caught a glimpse of this compound in a beaker, glowing as if it belonged more in an artist's palette than a scientist's toolkit.
In my early years of teaching science, students always noticed how potassium chromate stood out against a backdrop of duller salts, both in color and in properties. The formula may seem dry, but it reflects real chemistry that plays out in water, in industry, and even in environmental debates. Behind those symbols lies a mix of usefulness and risk you can’t ignore.
Potassium Chromate in the Real World
You’ll find potassium chromate in use where precise chemical work takes place. Laboratories lean on its reliability for titration and analytical tests. It acts as a good source of chromate ions, used to detect the presence of certain metal ions, like silver. When someone drops potassium chromate into a sample containing silver ions, a blood-red precipitate forms—a sure sign of a reaction.
Industrial sites have a long history with this compound. It helps in textile dyeing and the production of pigments, and it’s familiar territory in leather tanning. Chromate treatments can also stop corrosion on metal surfaces. I once saw how a few grams of potassium chromate could extend the life of metal parts in harsh conditions—especially around machinery exposed to the elements.
Environmental and Health Concerns
Every science teacher I know flinches a bit when handling potassium chromate. Its bright color signals a real danger beneath the surface. The chromium in K2CrO4 sits in the hexavalent state, which research has shown to be toxic and carcinogenic. Long-term exposure can damage kidneys, lungs, and the immune system. Groundwater contamination from industrial runoff can bring this risk straight to the community’s doorstep.
Enforcement of stricter disposal rules on hexavalent chromium compounds stands out as a must, not a suggestion. Facilities working with potassium chromate must set up containment and wastewater treatment that neutralizes chromate ions before anything meets public water systems. I’ve seen how some organizations track chromium waste down to the microgram, with environmental managers spending hours double-checking data before a single barrel leaves the site.
Solutions for Safer Chemistry
Better chemistry doesn’t stop at regulations—it thrives on innovation. University labs and chemical suppliers have chased safer alternatives for years. Some processes now use other compounds for testing or corrosion protection, but progress moves slowly due to the unique properties potassium chromate offers.
Schools play a key role in shaping attitudes long before anyone steps into a factory. Clear labeling, good ventilation, and routine safety drills make a difference. The knowledge that K2CrO4 isn't just another chemical, but one with real risks and responsibilities, sticks with students and scientists who handle it.
Final Thoughts
K2CrO4 stands as more than just a line in a textbook. Potassium chromate’s formula opens conversations about safety, innovation, and stewardship of the environment. Those bright yellow crystals remind everyone—student and professional—that some of the most useful compounds come bundled with lessons too important to overlook.
How should Potassium Chromate be disposed of safely?
Recognizing Real Dangers
Potassium chromate, that bright yellow powder sometimes used in laboratories, packs a punch when it comes to health risks. Handling it with bare hands always sets off alarms for people trained to respect chemicals. Chromium(VI) compounds cause cancer, bring on organ damage, and threaten water supplies. Tossing potassium chromate down the drain or letting it sit in the trash just passes the risk along. The bright color signals danger, not something to ignore or pretend doesn’t matter.
Why Down-the-Drain Doesn’t Cut It
People might look at a small amount left in a bottle and think a quick wash gets rid of the stuff. From my days in a college lab, I remember supervisors repeating the same warning: Never pour chromates or anything with chromium(VI) into sinks. Wastewater treatment won't filter that poison out. The chemical breaks down too slowly and moves right into rivers or lakes, poisoning fish and people alike. Letting it enter the water supply, even in tiny amounts, risks long-term health disasters for whole communities.
Laws Mean Business
Dumping potassium chromate outside official disposal channels breaks both common sense and the law. Environmental agencies track the disposal of all materials containing chromium(VI). Waste haulers carry special licenses and record each step. Ignoring these rules doesn’t just endanger people; it brings big fines and sometimes criminal charges. In 2017, a lab in New York faced legal actions after city officers caught them dumping chemicals, and the bill for their shortcut soared past clean-up costs. Real-world cases show what happens to careless operators: courts remember, even years afterwards.
Steps That Actually Protect People
Each bottle or beaker with potassium chromate left over shouldn’t move anywhere until sealed tight and clearly labeled as hazardous. Experience tells me: never trust the loose cap from a reused jar or the faded ink on a sticky label. Strong tape, original containers, and legible warning tags do the job better. Storage needs a locked cabinet, away from acids and anything organic. If some leaks, don’t just grab a mop—close off the room, use a mask, and call the people with training.
Contacting a licensed waste management company fixes what individuals can’t handle safely. They pick up the waste, deliver it to special facilities, and break down the chemical using high-temperature incineration or specialized reduction methods that convert chromium(VI) into chromium(III) compounds. This process slashes toxicity by a huge margin. Municipal programs often guide small businesses or hobbyists to community waste collection events—these are not just for cans of paint or dead batteries but also rare chemicals left over from home science kits or school projects.
Looking Forward
Better science education in schools, alongside workplace training, makes people less likely to stash hazards like potassium chromate in closets or let them slip into the environment. Funding for public disposal events and public awareness campaigns cuts down on accidents. Companies that keep their disposal audits open and honest help everybody breathe easier. Everyone shares the same water, and we all bear a duty to protect it, no matter where we came from or who signed for that bottle years ago.
| Names | |
| Preferred IUPAC name | Potassium tetraoxochromate(VI) |
| Other names |
Chromic acid potassium salt Dipotassium chromate Potassium tetraoxochromate(VI) |
| Pronunciation | /poʊˈtæsiəm ˈkroʊ.meɪt/ |
| Identifiers | |
| CAS Number | 7789-00-6 |
| Beilstein Reference | 356978 |
| ChEBI | CHEBI:13124 |
| ChEMBL | CHEMBL1357 |
| ChemSpider | 52831 |
| DrugBank | DB11360 |
| ECHA InfoCard | ECHA InfoCard: 030-006-00-8 |
| EC Number | 231-906-6 |
| Gmelin Reference | Gm. 1137 |
| KEGG | C01845 |
| MeSH | D017748 |
| PubChem CID | 24506 |
| RTECS number | GB2945000 |
| UNII | 78R83C4Y8W |
| UN number | UN1486 |
| Properties | |
| Chemical formula | K2CrO4 |
| Molar mass | 194.19 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 2.73 g/cm³ |
| Solubility in water | Moderately soluble |
| log P | 0.43 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 12.2 |
| Basicity (pKb) | 10.38 |
| Magnetic susceptibility (χ) | +2290.0e-6 cm³/mol |
| Refractive index (nD) | 1.738 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 196.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -935.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −778.9 kJ/mol |
| Pharmacology | |
| ATC code | V03AB17 |
| Hazards | |
| Main hazards | Oxidizer, toxic if swallowed, may cause cancer, causes severe skin burns and eye damage, may cause genetic defects, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS07,GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H312 + H332, H350, H340, H360, H372, H400 |
| Precautionary statements | P201, P202, P264, P270, P273, P280, P301+P312, P302+P352, P304+P340, P308+P313, P330, P332+P313, P362+P364, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0-OX |
| Lethal dose or concentration | LD50 oral rat 1870 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 180 mg/kg |
| NIOSH | TT5850000 |
| PEL (Permissible) | PEL: 0.05 mg/m³ |
| REL (Recommended) | ACS,Reagent |
| IDLH (Immediate danger) | 15 mg Cr(VI)/m³ |
| Related compounds | |
| Related compounds |
Potassium dichromate Sodium chromate Sodium dichromate Chromic acid |

