Potassium Carbonate: More Than Just a Chemical Name
Looking Back: Potassium Carbonate’s On-the-Ground History
Potassium carbonate didn’t show up overnight. Folks have pulled it from wood ash since at least the Middle Ages. Picture rural soap makers and glass blowers, taking what they could from the land, boiling down ashes in big pots — that’s where the term “potash” gets its roots. Instead of high-tech labs, early producers relied on muddy boots and wood smoke. Glassworks in Venice, soap factories across Europe, and even old-school bakeries all leaned on this salt. Potassium carbonate fueled entire communities and even shaped economies. Saltpeter makers used it for gunpowder, and farmers spread it as a fertilizer when soil demanded a potassium boost. Commercial-scale production stepped up during the Industrial Revolution, shifting from rustic ashpits to chemical plants.
The Product on Today’s Shelf
Let’s get plain about what you’re picking up. Potassium carbonate turns up as a white, granular or powdery solid. It doesn’t clump as easily as some other salts. It usually arrives with a faint salty taste you wouldn’t look for in food, but you see it on ingredient lists anyway. Its hygroscopic nature means it pulls in water from the air, and you don’t want to leave it unfinished for long. Drop it in water and it vanishes quickly, a trait that has made it reliable in many industries. Most of us unknowingly run into it in dried fruit, snappy Asian noodles, and perhaps in your next bottle of wine, where it helps balance acidity.
Physical and Chemical Realities
Potassium carbonate’s chemical formula, K2CO3, gets tossed around in classrooms and factory floors. It carries a molar mass of roughly 138 grams per mole. Sitting at room temperature, it stays pretty stable, refusing to melt until temperatures soar above 800°C. It won’t combust or catch fire, but spill it into water and you watch it sizzle a little, giving off heat and making the solution basic — sometimes harsh enough to bite at pipes and human skin. You find it doesn’t give off strong smells either; no warning signal if you’ve mishandled a batch except the slick, soapy texture if you touch it. Manufacturers stamp bags with technical specs showing potassium content, levels of chloride and sulfate, and moisture readings, letting buyers judge purity based on what task awaits.
Shaping, Labeling, and Meeting Standards
Anyone moving potassium carbonate through today’s markets reads labels that spell out purity, granule size, and moisture content. Chemical plants document sources and keep up with international benchmarks to avoid batch-to-batch surprises. GHS labeling includes pictograms and solid reminders about the risks for skin and eyes, and details about wearing gloves and goggles. Regulations in the US or EU demand full traceability — no more unidentified crystal bags passed hand-to-hand. Any company that skips proper labeling or overlooks safety quickly hears from inspectors, as consumer and worker safety isn’t a backroom deal. Documents and technical sheets track where every ounce comes from and how it was handled, laying it all out for anyone with the right clearance and a reason to ask.
Practical Making: How Potassium Carbonate Lands in the Barrel
In the old world, producers soaked hardwood ashes and filtered off the liquid, boiling it down until a white crust formed. This method delivered practical, impure potassium carbonate with hints of iron, calcium, and magnesium tagging along. Chemical production drives bigger volumes now, leaning on potassium hydroxide reacting with carbon dioxide. Some use potassium chloride with ammonia and carbon dioxide, similar to the Solvay process behind soda ash. Most of these processes focus on grabbing carbon dioxide cleanly and throwing off byproducts that companies can sell back into other industries. Waste gets shuttled for treatment, knowing well that nobody wants potassium carbonate loaded with unsavory extras. Labs keep an eye on final content, matching every finished batch to the customer’s industry standards.
Not Just Sitting There: Potassium Carbonate in Reactions
Potassium carbonate plays standby base in laboratories and factories. Toss potassium carbonate in with acids like hydrochloric, and watch it fizz — you get potassium chloride, water, and a burst of carbon dioxide gas. In glass furnaces, it drives the fusion, raising melting points and fine-tuning refractive indices for clearer, stronger glass. In soap, water softening, and basic cleaning agents, its alkalinity does more than meet regulatory checklists; workers rely on its dependability to keep production steady. It can also eat up moisture from the air, pulling double duty as a drying agent in air-sensitive reactions. Its ability to act as a mild base — without the bite of sodium hydroxide — expands laboratory uses, pushing organic reactions without breaking down delicate starting materials.
Other Names in the Books
Potassium carbonate gets shuffled around under labels like “potash” (especially in old documents), “pearl ash,” and “carbonic acid dipotassium salt,” with food additive code E501 showing up on ingredient panels. Even with all these nicknames, its properties and outcomes stay on track no matter what the label says.
Standards for Keeping Hands and Eyes Safe
Safe handling of potassium carbonate asks for more than good intentions. It chews at skin and eyes, so gloves, goggles, and solid ventilation become part of every batch transfer and mixing process. Spill treatment is pretty straightforward — sweep up dry material for disposal or mix it with lots of water to dilute before making any move toward the drain. Storage leans towards dry, cool, and isolated places, stopping the risk of accidental mixing with acids or moisture. Training covers basic toxicology, emergency washing, and documentation, keeping novice workers and old hands up to date and alert. Rules vary across borders, but most industries echo OSHA’s directives, drawing clear lines for exposure limits and emergency action drills.
Outside the Lab: Where Potassium Carbonate Makes a Difference
Its role stretches from glass furnaces in industrial cities to bakery floors in small towns. Winemakers use it to change acidity in grape musts; brewers manage mineral content in their water; food processors add it for the right texture in noodles, pretzels, and dried fruit. Potassium carbonate keeps soaps and detergents stable and workable, even in hard water. Textile dyeing runs smoother where potassium carbonate softens water and fixes dyes. In electronics, it helps in printing circuit boards, and in fire extinguishers, it acts as a core ingredient. Not many chemical products show up in such varied places. Research labs keep it close for organic synthesis or as an affordable drying agent. Home chemists order it online or find it in specialty baking supplies.
Research on Health and Safety: Digging into Exposure
Study after study focuses on how potassium carbonate behaves around people. Contact with skin or eyes brings irritation or chemical burns, pushing researchers to learn where risk outweighs reward. Inhalation over long stretches can rattle the lungs, so factory standards stress controlled air handling. In food, the limits remain tight. Food chemists keep up with animal studies and allergen alerts. Plenty of safety data comes from workplace injury reports, showing where slips happened, where training lagged, and how protective gear stacked up when spills landed. Environmental studies check for runoff and waterway contamination, finding potassium carbonate breaks down in water but still demands careful handling on an industrial scale.
R&D and the Road Ahead
Industry and academia pull at potassium carbonate’s limits, asking whether new uses will push further or whether old processes need a safer, greener hand. Some researchers dig into battery chemistry and energy storage, thinking potassium might sidestep cost issues in lithium fields. Water treatment experts test it against new pollutants, measuring how it stacks up against old-school lime or soda ash. Others look into using waste carbon dioxide to convert into potassium carbonate, trimming back emissions instead of letting greenhouse gases escape unchecked. Big tech and life science companies hunt for cleaner methods of making reagents, eager for traceable, reliable salts that don’t poison water tables or stack up toxic byproducts.
Toxicity, Old Myths, and Modern Reality
No one wants to sugarcoat chemical risks, and potassium carbonate has its share despite a long track record. Touching or inhaling the powder can set off bad rashes or burns, so workplace safety meetings matter even when old hands shrug it off. Ingesting decent amounts spells out trouble, but regulated uses keep real risk low for most consumers — the occasional raisin or noodle snack won’t steep someone in dangerous doses. Researchers run studies to pin down thresholds and compare chronic exposure results against other common alkalies like sodium carbonate. These findings cycle back to public health agencies and safety boards, keeping the debate grounded in real data instead of rumor or nostalgia.
Looking Toward Future Uses
Potassium carbonate’s story keeps growing. Some see it woven into fertilizer blends targeting depleted soils, where sustainable agriculture could use less corrosive potassium sources. Battery designers look at potassium-based devices, aiming for abundant, less geopolitically fraught resources than lithium. Glass production and soap making seem steady, but change comes as manufacturers seek lower energy processes, less waste, and more product made from recycled or biobased raw materials. Food scientists watch for tighter regulations and healthier formulas — shifting toward food safety without tossing centuries of practical use. Each industry finds different value in this basic, accessible salt, shaping future headlines with real-world needs and laboratory progress driving the conversation.
What are the common uses of Potassium Carbonate?
A Common Ingredient with Untold Stories
Walk into most kitchens, you’ll find baking powder. Not everyone knows potassium carbonate sits quietly inside some baking mixes. Bakers in Europe often grab “potash” for holiday cookies, especially gingerbread. It helps dough puff up and taste just right, less bitter than some other leaveners. Before modern baking powders, this compound did the heavy lifting for fluffy baked goods. Centuries-old recipes tell the story.
Soap Making: A Craft That’s Both Old and New
Soap makers cherish potassium carbonate for making liquid soaps. Add it to fats or oils, and you get a soft, sudsy result. It’s less harsh than sodium-based soap, so it works well for hand washes and gentle cleaning. Artisans and small-scale producers use it to avoid skin irritation that sometimes sneaks in with other cleansers. Long before detergents took over, people used wood ash—rich in this chemical—to scrub laundry and their hands.
Glassmaking: Shaping Clarity and Color
Potassium carbonate helps shape glass—literally. Stained glass in old cathedrals owes its shimmering surface to “potash glass.” It melts at lower temperatures, spreads color more smoothly, and keeps the glass clearer than soda-lime recipes. Today’s specialty glassmakers rely on it to make scientific instruments and even eyeglasses. The world of chemistry recognized this long ago and never looked back.
Food Processing: More Than Just Baking
Many Asian dishes feature foods treated with potassium carbonate. Ramen noodles, for example, gain their signature chewy bounce from its use. Chinese mooncakes and German pretzels both use it to change color and texture. Food factories rely on its alkaline punch to soften beans, balance acidity, and stop sugar syrups from crystalizing. In a candy factory, you’ll often see barrels labeled with its formula, K2CO3.
From Fire Extinguishers to Photography
Old-school fire extinguishers often held potassium carbonate solutions. Spraying it stifled flames in kitchens and workshops. Its ability to smother fire comes from the same basic chemistry that makes it good in soap and glass. Even now, specialty extinguishers sometimes call for it—especially where grease fires pose a risk. In film photography, developers and fixers need gentle alkalis, and potassium carbonate delivers where harsher choices fall short.
A Quiet Helper in Factories
Industrial settings use potassium carbonate to keep things moving. Gas purification plants strip out acid gases with it. Textile mills soften water to help dyes seep deep into fabrics. It keeps running engines from corroding and balances the pH in swimming pools and aquarium tanks.
Why Responsible Use Matters
Potassium carbonate offers impressive versatility, but it isn’t just about function. Workers handling it face risks from dust and spills. Companies need safety training and good ventilation. Food applications ask for high-purity grades, so suppliers follow strict quality checks. Modern factories invest in safe storage, spill prevention, and protective gear for staff. Careful use helps keep food, soap, and glass both safe and reliable.
Learning from Potash
Potassium carbonate rarely gets headlines, but it holds up a surprising number of industries. From childhood cookies to chemical plants, from art glass to clean laundry, it’s the unsung helper that keeps things running smoother and safer. Every better choice in handling, sourcing, and using this compound makes a mark, quietly shaping both safety and quality of life.
Is Potassium Carbonate safe to handle and use?
Potassium Carbonate in Daily Work
I’ve worked in small labs and seen potassium carbonate used for soap making, cleaning, and certain food applications. Its white, powdery look doesn’t seem threatening at first glance. Anyone who’s carried sacks of it or mixed it into solution knows it tends to clump up fast, pull moisture from the air, and leave a slippery film. Raw potassium carbonate feels grainy, but if it gets wet, it feels soapy on skin. Anyone curious enough to touch it barehanded learns soon enough it dries out the skin.
Health Impacts
OSHA’s chemical database puts potassium carbonate on the low end of acute toxicity. Swallowing small amounts leads to upset stomach or mild irritation, but high doses can burn mouth, throat, or gut. Dust rising from mixing or pouring triggers coughing or dry throat for some. If it touches eyes, the pain comes quick—redness, watering, stinging that drives people to the eyewash station. Skin contact won’t give instant burns, but dryness or roughness can set in after a day if washed off late.
Folk reports, employee safety records, and 2024 chemical safety guidelines say long-term exposure doesn’t cause cancer or deep organ damage. The main risk remains irritation or burns if care slips. Keeping dust out of the lungs, eyes, and off skin matters most.
Safe Handling Practices
Common sense goes far with potassium carbonate. Gloves and cheap safety glasses protect hands and eyes. High-quality lab coats or aprons help in messy workshops. Store powder in sealed bins, since it absorbs water from air and clumps quickly. Mix into liquids slowly to stop dust clouds from forming. Spills wipe up with a damp rag—avoid dry sweeping, as fine dust rises fast.
Ventilated rooms cut down the risk of inhaling stray dust. Having an emergency eyewash or sink nearby covers worst-case splashes. At home or at work, label containers so nobody confuses potassium carbonate with something safe like baking soda.
Use in Food and Everyday Products
Potassium carbonate works in pretzel baking or as part of certain Asian noodles, always under closely monitored conditions. Food grade versions carry extra checks for heavy metals or contamination. Food scientists stick to strict levels set by agencies like the FDA or the European Food Safety Authority. Unsafe amounts or mistakes in measurement have led to product recalls, but food industry training keeps accidents rare.
Anyone adding potassium carbonate to recipes—at home or in commercial kitchens—should use purpose-made products, measure with a scale, and mix thoroughly. Food applications only use a pinch compared to industrial needs.
Common Incidents and How to Respond
People spill, drop, or splash potassium carbonate worldwide every day. Most cases end with fast cleanup and a change of gloves. Swallowing a small bit by mistake brings a trip to the doctor but seldom leaves lasting harm. Eye splashes demand a 15-minute rinse, and those crystals sting but rarely scar if treated fast.
Every chemicals handbook agrees: store in a cool, dry place, keep containers labeled, and don’t let kids or pets near it. For workplaces, posting the safety data sheet nearby and training staff can stop trouble before it starts.
Reliable Sources for Guidance
The Centers for Disease Control and Prevention, OSHA, and the National Institutes of Health offer updated advice and practical chemical handling rules. Peer-reviewed reviews from 2022 and 2023 point out that nearly all injuries happen when users shortcut basic precautions.
With training and basic supplies, potassium carbonate ranks as a practical and manageable chemical. Like most alkaline powders, it asks for respect and attention, but serious harm remains rare with steady habits and honest labeling.
How should Potassium Carbonate be stored?
Why Storage of Potassium Carbonate Matters
Potassium carbonate lands in industrial spaces, laboratories, and even in food processing plants. I’ve worked around basic chemicals in a few warehouse jobs, including one stint in a place that kept potassium carbonate among its long list of dusty stock. The lesson I learned: the simplest rules often save the most headaches. Forget elaborate climate controls or rare building materials—a little care goes a long way.
Understanding What You're Dealing With
Pale, granular, sometimes powdery, potassium carbonate calls for respect. It grabs moisture from the air like a magnet attracts filings. Spill a bit on a damp counter and you get a slippery mess. On a larger scale, sloppy storage can trigger clumping, contamination, and wasted product. I’ve seen half-bags turn into hard, crusty lumps simply because someone ignored a leaky roof or left the warehouse doors open on a muggy day.
Choosing the Right Container
Sealed, airtight containers protect potassium carbonate from the outside world. Plastic drums, glass jars, sturdy metal bins fitted with tight lids—the choice depends on volume and setting. Find something that doesn’t corrode or leach. I’ve used polypropylene buckets lined with moisture barriers, and they do the trick. If you’re tempted to skimp with regular cardboard, think again—you wind up with soggy cardboard and ruined powder.
Pick the Proper Place
Cool, dry storage wins every time. Humid rooms invite trouble. The boss at my first job hammered that lesson into us every spring when the temperature and humidity shot up. Sacks ended up stored on pallets, inches above the floor, away from windows and heaters. Basements tend to stay dry if vented, but old boiler rooms and attics with sweating pipes spell disaster. Shelving at chest height, away from walkways—this helps stop spills and slips.
Keep It Labeled, Keep It Safe
Sharp labeling seems like an obvious step, but it still gets skipped. You want clear, waterproof stickers, mentioning at minimum—product name, date received, emergency information. In one warehouse, a missing label sent our crew scrambling when someone thought potassium carbonate was baking soda. Emergency services don’t enjoy playing detective, especially if there’s been an accidental spill or contaminated food batch.
Ventilation, Cleanliness, and Routine Checks
A little airflow matters. Avoid storing potassium carbonate with strong acids or volatile chemicals. The fumes sometimes react, and cross-contamination becomes a real concern. Wipe spills with a dry cloth, and sweep floors regularly. It doesn’t get more low-tech than a broom, but routine cleans add a layer of insurance.
A Note on Personal Safety
Gloves save skin, goggles protect eyes. Even if potassium carbonate scores low on the hazard scale, getting dust in your eyes stings like mad. Training isn’t fancy—just genuine reminders. Chemical splash kits should hang nearby for fast access, and folks need to know how to use them. I’ve had to rinse my hands after surprise splashes; the difference between five seconds and five minutes can stick with you.
Backing It Up With Facts
The Occupational Safety and Health Administration (OSHA) and the National Institute for Occupational Safety and Health (NIOSH) both recommend dry, sealed storage for potassium carbonate. Guidelines match what works on the ground. Reports from chemical safety boards point to poor labeling and leaky packaging as top reasons for product waste and workplace injuries. Manufacturers have issued more recalls from storage mishaps than flawed production runs.
Solutions for Small Businesses and Home Use
Tight budgets don’t have to mean risk. Clear food-grade containers, kept away from baths, kitchens, or anywhere steamy, prevent almost every problem. For homes, a high shelf or dedicated cabinet works. In small shops, regular walk-throughs to check for leaks, plus a bi-annual inventory cleanout, keep potassium carbonate fresh and safe.
Real-World Simplicity
The heart of safe potassium carbonate storage isn’t high science—just solid habits, honest labeling, and attention to the basics. From supermarket backrooms to science labs, everyone saves time and money by treating these white grains with respect and a practical touch.
What is the chemical formula of Potassium Carbonate?
Looking at Potassium Carbonate – A Simple Salt with Important Uses
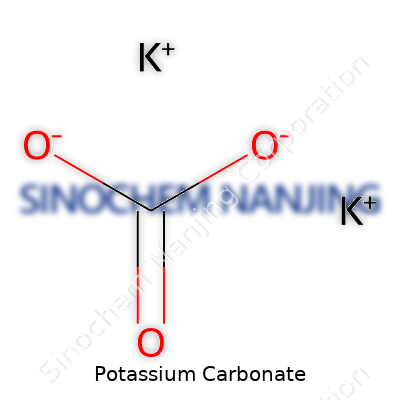
People in science class often learn the basics of chemical compounds: those groups of atoms held tightly together that show up in soaps, fertilizers, and food additives. Potassium carbonate stands out as a familiar example. Its formula, K2CO3, might look a bit confusing at first glance, but it breaks down pretty easily. Two potassium atoms link up with one carbonate group, which is made of one carbon atom and three oxygens. This trio makes a white, water-soluble powder you can find in a wide range of industries and even in some homes.
The Chemistry that Shapes Everyday Life
Stepping into a chemistry lab, potassium carbonate tends to show up as a pH regulator, especially when people want to create an alkaline environment. Having spent years working in food processing and horticulture, I've come to appreciate the broader role this compound plays outside textbooks. For example, bakers often turn to potassium carbonate in making certain traditional Asian noodles and pretzels, because it affects texture and taste. Many folks don’t notice the science in a bowl of noodles or a loaf of bread, but there’s real chemistry behind the scenes.
Laundry detergents, glass production, and even fire extinguishers make use of potassium carbonate. In glassmaking, it helps lower the melting point of silica, making manufacturing more energy-efficient. In soap making, it replaces sodium carbonate in recipes for liquid soap, shifting the consistency and cleaning action. One of my earliest memories of “real chemistry” outside schoolbooks came during a student project, watching potassium carbonate fizz and react during a simple acid-base demonstration. That little experiment stuck with me, showing how the molecular formula translates to practical action.
Environmental and Health Aspects
Concern about chemicals in daily life leads people to ask if potassium carbonate is safe. The answer depends on proper handling. As a water-soluble salt, it can cause irritation but doesn’t build up in the environment like some heavy metals or persistent organic pollutants. In agriculture, potassium carbonate serves as a fertilizer component, helping soil retain the nutrients plants draw up over time. Crops need potassium to thrive, and farmers careful with doses see improved yields and better resistance to drought or disease.
Navigating Regulations and Best Practices
Many countries keep tight controls over how chemicals like potassium carbonate get stored, transported, and used. In my own work, safety meant wearing gloves, checking ventilation, and reading up on every material safety data sheet. It makes sense—an otherwise harmless compound in the wrong hands or in the wrong setting can create problems. Responsible companies train staff, set up spill procedures, and rotate stock to avoid surprises.
As an ingredient found on labels from winemaking to water-softening tablets, potassium carbonate’s formula represents far more than just a collection of letters and numbers. Anyone working with chemicals—or even just interested in how basic compounds shape the world—should take time to learn their properties, their uses, and their responsibilities. The right knowledge keeps people safer, products better, and communities healthier.
Can Potassium Carbonate be used in food applications?
Understanding Potassium Carbonate in Food
Potassium carbonate usually pops up as E501 in food ingredient lists. It isn’t some exotic lab-made additive but a basic salt, and it occurs naturally in ash. Old-school cooks used it in noodle-making and cocoa processing long before modern food tech caught on. This salt helps control acidity, acts as a stabilizer, and boosts the texture of foods.
Where People Run Into It Most
If you’ve ever bitten into chewy Chinese ramen or spooned darkened Dutch-process cocoa, you’ve already eaten food-grade potassium carbonate. Chinese cooks lean on it to give noodles that bouncy, elastic mouthfeel. European chocolate makers use it to mellow out bitter cocoa powder, giving it that trademark color and softer taste. Bakers sometimes use a bit in pretzels and German Lebkuchen cookies to get specific textures.
Sifting Through the Safety Facts
Any time food gets made in a factory or home kitchen, ingredients get scrutinized for health risks. Reputable agencies stick to hard science for safety questions. Potassium carbonate has been ruled safe when handled the right way and used in legal amounts. The U.S. Food and Drug Administration approves it for direct use, classifying it as “generally recognized as safe” (GRAS). European regulators also allow potassium carbonate as a food additive under tight rules.
Problems surface only when folks use more than recommended. Too much potassium in food—a real risk if added carelessly—can put stress on kidneys or affect heart function, especially for people with health conditions that make managing potassium tricky. Though such cases pop up in medical journals, they almost always involve much larger doses than a typical recipe calls for.
Why Get Technical for Simple Foods?
On the surface, adding potassium carbonate can seem unnecessary. Yet, as a baker, I learned some old-fashioned sweets and Asian noodles owe their success to it. Take ramen: it’s tough to mimic the springiness of true kansui noodles using table salt or baking soda alone. Potassium carbonate unlocks that texture. For home cooks who love tackling traditional recipes, tracking down a food-safe version is worth the effort.
Still, all this hinges on keeping track of how much goes in. Measurements matter. Potassium carbonate in small, measured doses delivers safe, consistent results. Guesswork leads to inedible or potentially unsafe food.
Good Rules for Home and Industry
Clear labeling and smart regulations keep potassium carbonate in the safe zone. Food manufacturers rely on strict protocols and testing. For home cooks, using the right product—clearly marked as food grade—makes a world of difference. Industrial or chemical-grade variants must stay out of the pantry.
Education helps. Over the years, I’ve seen cookbooks and cooking classes grow better at outlining safe dosage. Videos and websites now list specific weights and tips for handling alkaline salts. Social media even flags bad hacks that tell people to use substitute chemicals.
Moving Forward With Transparency
Potassium carbonate has earned its spot in certain recipes, not as a filler, but as a tool for authentic textures and profiles. Open conversation between government, industry, and everyday cooks helps prevent misuse and keeps the focus on food safety. New cooks and curious experimenters benefit from clear, science-backed information while traditional recipes hold onto their distinctive taste.
| Names | |
| Preferred IUPAC name | potassium carbonate |
| Other names |
Carbonic acid dipotassium salt Pearl ash Potash Salt of tartar Dipotassium carbonate |
| Pronunciation | /poʊˌtæsiəm ˈkɑːrbəˌneɪt/ |
| Identifiers | |
| CAS Number | 584-08-7 |
| Beilstein Reference | 3596857 |
| ChEBI | CHEBI:131526 |
| ChEMBL | CHEMBL1201617 |
| ChemSpider | 5779 |
| DrugBank | DB11090 |
| ECHA InfoCard | ECHA InfoCard: 029-003-00-5 |
| EC Number | 209-529-3 |
| Gmelin Reference | Gmelin Reference: 18484 |
| KEGG | C00238 |
| MeSH | D011104 |
| PubChem CID | 24924 |
| RTECS number | TS7750000 |
| UNII | 1PT6J4Q8QZ |
| UN number | UN1872 |
| Properties | |
| Chemical formula | K2CO3 |
| Molar mass | 138.205 g/mol |
| Appearance | White, odorless, crystalline powder or granules |
| Odor | Odorless |
| Density | 2.43 g/cm³ |
| Solubility in water | 1120 g/L (20 °C) |
| log P | -2.20 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 10.33 |
| Basicity (pKb) | 3.2 |
| Magnetic susceptibility (χ) | +9.0·10⁻⁶ |
| Refractive index (nD) | 1.427 |
| Viscosity | Viscosity: 1.14 mPa·s (20% solution at 20°C) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 120.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1150.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1150.5 kJ/mol |
| Pharmacology | |
| ATC code | A12BA01 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. |
| GHS labelling | GHS07, GHS hazard statement: H319 |
| Pictograms | GHS07, GHS08 |
| Signal word | Warning |
| Hazard statements | Hazard statements: Causes serious eye irritation. |
| Precautionary statements | P264, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-0-1 |
| Lethal dose or concentration | LD50 (oral, rat): 1870 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 1870 mg/kg |
| NIOSH | RN8400000 |
| PEL (Permissible) | PEL: 15 mg/m³ |
| REL (Recommended) | 100 mg/m³ |
| IDLH (Immediate danger) | 2500 mg/m3 |
| Related compounds | |
| Related compounds |
Potassium bicarbonate Potassium hydroxide Sodium carbonate |

