Potassium Cerium Nitrate: An In-depth Examination
Historical Development
Potassium cerium nitrate came about during the explosive advances in rare earth science in the 20th century. Chemists searching for new oxidizers and colorants looked past familiar materials, ending up in the lanthanide section of the periodic table. Cerium’s reactivity opened new doors. In Europe, researchers combined cerium salts with potassium nitrate, drawing on knowledge from those working with pyrotechnic mixtures. Into the late 1900s, new refining procedures and improvements in rare earth extraction allowed potassium cerium nitrate to be produced in the kinds of purity and quantity that made it worth considering outside the research lab. Factories in China, the US, and Russia brought prices down, making it a more familiar material to both industrial chemists and university researchers.
Product Overview
This salt starts as a pale yellow to almost white crystalline powder. Unlike common industrial oxidizers, potassium cerium nitrate holds special electronic properties thanks to cerium’s variable oxidation states. Suppliers and catalog companies label it as Potassium Cerium(IV) Nitrate, Potassium Ceric Nitrate, or with Chinese and CIS identifiers—a detail that matters when ordering for lab or production work, since subtle naming differences can shift purity or composition. Its shelf stability and ability to dissolve in water gives it an edge for both solution and solid-phase applications. Most labs use it as received, but high-end users pay attention to the source, since trace iron or other lanthanides from refining can affect results.
Physical & Chemical Properties
The unique part about potassium cerium nitrate sits in its crystalline form—a three-dimensional arrangement that looks like slices of pale jade under a microscope. The salt has a density around 2.2 g/cm3 and melts only at very high temperatures beyond most routine lab work. It dissolves in water with moderate ease, releasing cerium(IV) ions and nitrate, an effect that enables most of its chemical use. Potassium holds the structure together, while cerium takes the spotlight in redox reactions. The nitrate group acts as both a counterion and a source of oxidizing power, something chemists take advantage of in organic synthesis. Oxidizing power expresses itself most when heated, or when put in contact with reducing agents—making it useful across analytical, synthetic, and fireworks chemistry.
Technical Specifications & Labeling
A typical label on potassium cerium nitrate details content by % weight, moisture content, insoluble matter, and iron (Fe) limits. Reputable suppliers target purity above 99%, with strict limits on rare earth impurities and chlorides, which can affect both safety and performance. Lot numbers tie back to batch certificates, a feature that matters in regulated applications. Shelf-life is not infinite, given hygroscopic tendencies, so anyone storing it for extended periods uses double-sealed containers in dry cabinets. The chemical formula appears as K2Ce(NO3)6, sometimes simplified to KCEN, with batch-specific details for serious users.
Preparation Method
Labs usually synthesize potassium cerium nitrate by reacting cerium(IV) oxide with nitric acid, followed by slow addition of potassium nitrate. The process produces a hot, acidic solution, which cools to precipitate the salt. Controlling temperature and acid ratio is crucial, since unwanted side products can form if it gets too hot or dilute. In bigger operations, filtration and multiple crystallizations remove colored or insoluble byproducts. The refined product lines up as sparkling crystals or powder, dried under vacuum. Each step in this method, from the raw cerium oxide's handling to the choice of glassware, impacts both purity and yield, a reminder that skill and attention to detail pay off in chemical manufacturing.
Chemical Reactions & Modifications
Potassium cerium nitrate shines as a strong, selective oxidizer. In organic chemistry, it plays a role in the selective oxidation of alcohols and cleavage of double bonds. Its action comes from cerium cycling between +4 and +3 oxidation states, creating opportunities for the chemist to nudge molecules in new directions. Modified derivatives, produced by partial substitution of nitrate or by blending with other rare earth salts, offer tuning for different reaction schemes. Some research explores exchange with heavy alkali ions like rubidium, but potassium remains standard because of solubility and cost. Light exposure and high heat can lead to slow decomposition, liberating NO2 and changing cerium’s oxidation state. In the firework and pyrotechnic industry, mixing with binders and fuels yields colored flames and extra thrust—a practical use of a chemistry textbook reaction.
Synonyms & Product Names
References to potassium cerium nitrate surface in chemistry literature under names like Potassium Ceric Nitrate, Ceric Potassium Nitrate, Dipotassium Hexanitratocerate, and Potassium Cerium(IV) Nitrate. Older Soviet sources sometimes use transliterated Russian labels, adding to the confusion for importers. CAS numbers (such as 10199-49-0) help standardize sourcing. Commercial brands and catalog suppliers often shorten the name to KCEN or use internal codes to organize stock. Pay attention to spelling and context—mislabeling or mixing synonyms can result in shipment of the wrong rare earth compound, given how many similar salts occupy shelves in specialty supply houses.
Safety & Operational Standards
Handling potassium cerium nitrate means thinking ahead about both routine risk and rare accidents. Its oxidizing power demands dry storage, away from organic materials and metals that could cause a reaction. Inhalation of dust is not an abstraction; working with open powders or agitating containers generates airborne particles, requiring masks, gloves, and disposable lab coats. If spilled, water makes for safe cleanup, but avoid running the rinse water into organic-rich drains. Regular safety sheets flag eye irritation and potential respiratory tract effects. Pyrotechnicians and chemists storing bulk quantities build in distance from heat, spark, or fire, and keep only limited portions on lab benches, using flame-resistant trays. In industrial settings, all vessels and tools made contact-resistant—no wood or rough plastics—because any contamination can cause an unexpected flash during mixing.
Application Area
In analytical chemistry, potassium cerium nitrate stars in redox titrations. Its sharp color change when reduced to cerium(III) allows for precise endpoint detection in a range of oxidation-reduction reactions. In synthetic organic chemistry, it catalyzes transformations unachievable with other oxidizers due to selectivity for certain functional groups. Specialty ceramics and glassmakers use it to alter coloring or confer special refractive indices, especially in rare glassware. Over in the world of fireworks, potassium cerium nitrate helps control flame properties and works as an oxidizer in colored star compositions, letting manufacturers deliver sharper hues and burn rates than with more common compounds. The electronics industry uses it in the manufacture of specific sensors and in deposition techniques to achieve layers with unusual electronic properties, drawing on both potassium’s and cerium’s distinct attributes.
Research & Development
University groups keep exploring how modifications to potassium cerium nitrate structure tweak oxidation potential. Analytical labs work to refine detection limits in redox titrations by pairing it with advanced colorimetric sensors. New applications in catalysis pop up as teams experiment with blending cerium nitrate derivatives with other transition metals. In photochemistry, potassium cerium nitrate serves as a reference compound for studies on electron transfer. Some research focuses on eco-friendly synthesis, looking at waste valorization—recycling cerium scrap from electronics or mining operations back into usable chemical feedstock. Funding for rare earth alternatives remains strong, given strategic supply concerns. The innovations seen in patents—from new ligand frameworks to water-stable derivatives—point to growing interest in solving both efficiency and environmental issues.
Toxicity Research
Assessment of potassium cerium nitrate toxicity lags behind more common chemicals, mostly due to its specialized use. Initial studies show low acute oral toxicity in mammals, but repeated exposure to rare earth salts can cause harm, particularly to the liver and kidneys. Dust and fine particulate matter raise concerns for those with long-term exposure or poorly ventilated workspaces. Some reports hint at cerium accumulation in aquatic life, prompting limits in wastewater discharge for factories using or making the chemical. Regulatory agencies classify it as an irritant, not as a known carcinogen, but urge respect for containment and handling protocols to avoid accidental exposure. Monitoring programs in larger plants include regular air sampling and health checks for workers handling bulk quantities.
Future Prospects
With rare earth prices fluctuating and supply chains tightening, the future of potassium cerium nitrate ties strongly to both innovation and sustainability. Chemists and manufacturers look for new, greener synthesis routes, hoping to reduce byproducts and energy consumption. As uses expand in electronics and advanced materials, demand could outpace current supply, prompting more recycling of cerium from end-of-life products. Research into catalytic applications—both in selective oxidation and energy storage devices—shows promise, especially given the drive for more effective and less expensive alternatives to platinum-group metals. Improved packaging and safe handling systems will likely keep pace, pushed by both regulatory pressure and insurance requirements. Upskilling in the next generation of laboratory technicians and chemists will emphasize rare earth safety and eco-friendly waste management, making potassium cerium nitrate part of a broader conversation about sustainability in specialty chemicals.
What is Potassium Cerium Nitrate used for?
The Role in Analytical Chemistry
Potassium cerium nitrate plays a big part in measuring and testing other substances. Lab technicians use it almost daily as a titrant in redox reactions. This compound stands out for its reliability in analyzing iron, copper, and other metals, helping check water quality or purity of metals in recycling plants. Routine lab life would get much more complicated without compounds delivering precise results every time.
Keeping Glass Polishing on Track
Industries trust potassium cerium nitrate for polishing glass. Optical manufacturers and smartphone factories rely on it to create scratch-free lenses and screens. The grinding and polishing process demands a gentle hand and a sharp tool, and this compound lives up to the task. It delivers the kind of finish camera lenses, telescopes, and eyeglasses need before entering customers’ hands. I’ve seen how glass keeps a cloudy haze until cerium nitrate comes out—it’s striking how much difference a fine abrasive makes.
Building Safe and Accurate Devices
You’ll find potassium cerium nitrate in specialty ceramics and electronics. Some companies blend it into solid-state fuel cells and capacitors for improved conductivity. The electronics community has seen this boost battery and sensor performance, which turns out to be a pressing need as wearables and medical implants become more common. Enhanced batteries mean more reliable portable defibrillators or monitors that could make a difference in emergency care.
A Contribution to Fireworks and Pyrotechnics
Special effects in concerts and fireworks shows draw on chemicals to control the color, intensity, and safety of flames. Potassium cerium nitrate delivers bright sparks and well-defined colors, which makes it a favorite among pyrotechnicians. I’ve talked with event professionals who say this compound helps them limit smoke and unwanted debris during displays, so audiences get a clear view. Law enforcement teams sometimes use it in tracer rounds or signal flares—here, visibility matters even more, especially in rescue operations.
Concerns and Responsible Use
Any chemical with so many uses deserves a closer look at handling and disposal. Cerium compounds, if managed without caution, can contaminate water and soil. Regular folks rarely come across these chemicals at home, but industrial sites must keep strict safety rules in place, both for workers’ health and local ecosystems. Studies by the Environmental Protection Agency highlight the need for gloves, fume hoods, and careful waste collection when working with rare earth compounds like cerium nitrate.On an industry level, some companies now track their chemical use more closely and recycle abrasives whenever possible. Real reductions in waste only happen when workers and managers take ownership, not when regulations stay on paper. Transparent reporting and community involvement go a long way. Companies open about how they use and recycle rare earths have started earning public trust, a challenge for the field otherwise.
Looking Forward
Researchers look for greener alternatives to rare earth compounds, but demand isn’t going away soon. In fields like optics and electronics, potassium cerium nitrate keeps showing up as the practical choice. Every time you swipe a clean touchscreen or look through a telescope without a scratch, this compound probably helped make it possible. The more we learn about its uses and risks, the better decisions everyone can make about supporting responsible production and innovation in the future.
Is Potassium Cerium Nitrate toxic or hazardous?
Understanding Where Potassium Cerium Nitrate Shows Up
Potassium cerium nitrate grabs some attention in both research labs and industry. You will spot it used for polishing glass, making specialty optics, and in chemistry class experiments. Nobody walking down the street is likely to come across it, but for anyone who spends their time around chemicals, it's good to know what risks actually come with that yellow crystalline powder.
Assessing the Hazards Up Close
Not all chemicals deserve a panic response, but “rare earth nitrates” like potassium cerium nitrate come with warnings for a reason. Look at the safety sheets: inhalation, skin contact, and accidental ingestion all raise concerns. The compound can irritate eyes, nose, and throat almost right away. Get enough on bare skin and you might see redness or feel a burning sensation. Scoop a big whiff of dust and suddenly a cough might settle in for the rest of the work day.
Scientists who work with this material learn to respect its oxidative nature. As an oxidizer, potassium cerium nitrate can make a small spark turn into a bigger fire, especially around anything flammable—paper, solvents, or even some cleaning rags. It’s a workplace staple to see it packed alongside strict guidelines for storage and use, often posted right above the experiment benches.
Digging Into Long-Term Health Effects
Spend years around certain metal salts and sometimes the body lets you know. Data on cerium compounds tells an important story: chronic exposure might not kill outright but it can quietly affect the lungs or kidneys over time. Cerium doesn’t show up in living bodies much, so higher buildup from work exposure can lead to damage the immune system has trouble fixing later. The occasional single exposure likely won’t do much for most folks, but frequent contact ups the stakes.
Nobody wants that silent risk. In my own lab experience, the best chemists are always the ones who act like every day in the lab could write their future medical history. They push for tight controls because it’s not just about themselves—sometimes a careless spill impacts everyone nearby.
Balancing Use Against Practical Risk
Accidents with potassium cerium nitrate tend to start with ignoring basic safety gear. Goggles, proper gloves, and dust masks do the trick in most routine settings. Ventilation helps keep dust away from anyone who does the mixing, pouring, or weighing. I’ve watched a lot of students try to skip these steps for convenience—usually once is enough once they catch a bad whiff or feel burning eyes.
For those thinking about environmental impact, this compound doesn’t disappear down the drain like table salt. If enough makes it outside the lab, it could harm animals and plants, especially if local water supplies get contaminated.
Smart Steps Forward
Practical fixes start simple. Most trouble vanishes with basic training and built-in habits: label containers, check safety data before starting anything, fix any leaks or spills before walking away. Disposal calls for a specific touch—waste specialists know how to neutralize the oxidizing power and prevent it from mixing with things that could cause a reaction. By keeping risks top of mind, people who handle potassium cerium nitrate can make sure it stays useful and not dangerous.
How should Potassium Cerium Nitrate be stored?
Common Sense First
Potassium cerium nitrate sits on the shelf in many labs, but it doesn’t take much experience to know: this isn’t just another powder. As someone who has kept chemicals both at the bench and on the warehouse shelf, I’ve seen that safety and smart storage choices stick longest when habits stay grounded. This compound can spark problems if not kept with care. Too much humidity creeps in, you end up with crusty caking and unreliable results, or worse, unexpected reactions.
Why Moisture Wrecks the Game
Folks often ignore the power of a leaky lid or an open bag. Potassium cerium nitrate pulls water straight from the air. That’s not just annoying—it means purity and measured results slip away. Moisture can even help it break down, so a minor mistake snowballs into bigger costs. Shielding from humidity means locking the container tight and picking a spot nowhere near any damp or steamy areas. Desiccators and silica gel go a long way. From personal experience, tossing a few fresh silica packs into the storage bin rescued bottles from clumping one rainy season.
Glass over Plastic—The Tough Choice
Some think a solid plastic container handles everything, but sharp chemicals bite through cheap seals over time. Bottom shelf plastics end up cracked or warped, letting in unwanted air. Glass, with a good PTFE-lined cap, means a snug fit for months or years. It’s not snobbery—it’s about keeping your investment safe and your data trustworthy. After cleaning up one too many spills from brittle jars in the past, I never reach for anything less than good borosilicate for tricky powders like this.
Not Just Any Shelf Will Do
Potassium cerium nitrate crumbles under sunlight and high heat. It’s tempting to toss rarely used bottles on a top shelf or near a window, but that’s asking for trouble. The stuff keeps best in a cool, dark place—think a closed cabinet well away from radiators and sunshine. Even in air-conditioned labs, small temperature swings matter. On a hot summer day, a well-insulated storage cabinet held the line, while neighboring chemicals on open racks turned to mush or dust. Investing in stable indoor temperatures saves money and protects rare materials.
Hazardous Mixes and Safe Separation
Potassium cerium nitrate belongs nowhere near flammable solvents, acids, or reducing agents. A gloved hand is wise, but the real safety comes from discipline—chemicals with high reactivity stay isolated from organic materials and things like sulfur or phosphorus. A color-coded shelving system kept this straight in my old lab. Less cross-contamination means less headache and fewer phone calls on Saturday mornings. The right signage on containers cuts out guesswork for new staff and keeps audit notes glowing.
Disposal and Community Responsibility
There’s a final point too: leftover or outdated material shouldn’t end up next to household waste or poured down the drain. Local rules might call for hazardous waste pickup, usually through university or industrial programs. I’ve seen routine audits catch old bottles forgotten in the corner. It’s better to book timely disposal than wait for an accident. If you aren’t sure what your area expects, the local environmental office can guide the way, helping keep both lab and neighborhood safe.
Final Pointers from the Field
Store only what you use within a year. Buy smaller bottles if you work with potassium cerium nitrate rarely. Keep written logs—down to expiration date and every time you open the jar. These small steps dodged more than one close call in tight lab quarters. Safe storage isn’t flashy, but it beats cleaning up a preventable mess.
What are the physical properties of Potassium Cerium Nitrate?
Getting a Grip on Appearance and Texture
The thing that stands out first with Potassium Cerium Nitrate is its vivid appearance. Crystals come off looking sharp and are usually colorless or slightly yellow. A handful feels gritty, not unlike grains of common table salt but with a little more ‘crunch’ and less dust. As someone who’s poured this compound out in a beaker more than a few times, it doesn’t coat hands or stick to gloves with that slick, powdery feeling other salts have. It usually breaks apart cleanly instead of clumping—a huge plus if you’re working in a climate where humidity likes to mess with samples.
The Real Numbers: Density, Melting Point, Solubility
Potassium Cerium Nitrate settles in at a density of about 3.1 g/cm³. This puts it on the heavier side, especially compared with other nitrates, and that weight can throw you off if you expect something that pours and scatters like table salt. I’ve seen researchers over-pour a batch thinking they’re scooping up a lighter material, which leads to all sorts of hiccups.
This compound releases water at temperatures above 100°C, but stick it in an oven much hotter—hitting around 350°C—and it starts decomposing. Actual melting hasn’t been a common event in any practical lab setting since thermal decomposition kicks in before you get to a true ‘liquid’ phase. That’s a headache for anyone needing to shape or fuse this compound, and it leaves folks relying mainly on its solid form.
Water loves Potassium Cerium Nitrate. Solubility takes off as you heat the water up. In cold water, it dissolves at a slower pace, lingering on the bottom of a flask for a while. Warm things up, and you get a clear, colorless solution fast. Solubility is a huge help for anyone needing to prep a standard solution with precise concentrations. Colleagues working with analytical methods, especially photometric determinations, appreciate this no-fuss dissolving behavior because it means getting up and running without endless stirring or ultrasonic baths.
Crystal Structure and Stability
Molecules in Potassium Cerium Nitrate lock together into a regular lattice. This pattern impacts how the material handles mechanical stress, and it doesn’t crush or smear as easily as something like sodium chloride. I’ve dropped it, somewhat accidentally, and found only the edges chip off rather than turning the whole mass to dust. That hardness helps during storage and shipping, where long journeys and vibration would otherwise reduce purity and create unpredictable particle sizes. People storing this material for months value its stability, just so long as the humidity is kept down. Moist air pulls in and starts breaking the lattice apart from the edges. I learned that lesson after leaving a loosely capped vial in a damp storage room overnight—the crystals got sticky in just a few hours, making them a pain to measure out later.
Handling and Practical Notes
Potassium Cerium Nitrate isn’t particularly toxic, but the common-sense approach involves gloves and goggles because nitrates in general aren’t friendly if they work into cuts or eyes. Static isn’t a big issue, and spills don’t launch clouds into the air. Cleanup is pretty basic: a damp cloth scoops up most stray granules. Anyone bringing this into a general laboratory should prioritize airtight containers. Keep humidity away and you’ll avoid issues with storage, clumping, or unplanned chemical changes.
From a practical side, it helps to remember that all these seemingly simple features can complicate scale-up processes. Industrial and research labs appreciate knowing which compounds can stand up to rigorous handling. Potassium Cerium Nitrate holds its own, delivering a blend of stability, reliable solubility, and manageable texture. All these elements come together to explain why this material finds a steady foothold in work ranging from precision optics to chemical analysis.
What is the chemical formula of Potassium Cerium Nitrate?
What’s In a Name?
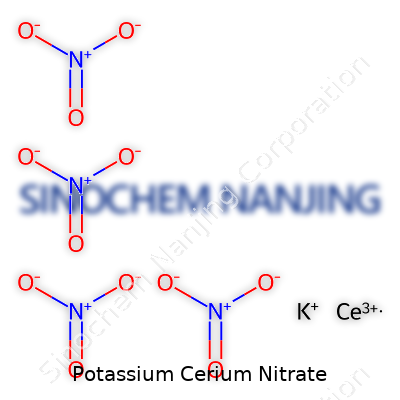
Potassium Cerium Nitrate is a mouthful. When you hear it, you know you’re heading into deep chemistry territory. Its chemical formula is K₃Ce(NO₃)₆. Here’s the breakdown: three potassium ions, a cerium ion, and six nitrate groups. It’s not as well-known as simple salts like table salt or potassium nitrate, but it still shows up in specific parts of science and industry.
Getting to the Formula: K₃Ce(NO₃)₆
This formula looks dense. If you’ve wrestled with chemistry, you’ll recognize those grouping parentheses as a marker for complexity. The cerium is in a +4 oxidation state, surrounded by six nitrate ions, making it a coordination compound. Potassium balances the charge. The importance of getting this formula right isn’t just about memorization for a test. In labs, one wrong number in a formula spells disaster — or at least a failed experiment.
Why Does It Matter?
Legitimate questions come up about why anyone outside of advanced chemistry circles should care about the likes of potassium cerium nitrate. Here’s the thing: precision in chemical formulas lets scientists and industries make the materials that underpin things most people never see directly but depend on anyway. Lasers, specialized optics, chemical analysis, and even some glass fibers rely on exact compounds with known properties. Potassium cerium nitrate, for example, acts as a standard in chemical analysis, a reagent for specialized syntheses, or a crystal for optical instruments.
In my own lab days, mislabeling or mixing up the composition of a single reagent meant hours lost. Watching a color reaction fail or a crystalization process go sideways, you learn respect for formulas. No shortcuts. The level of detail doesn’t only protect the experiment; it ensures that the data we trust comes from reliable, reproducible setups. That’s a principle central to scientific trustworthiness.
Backed by Facts
Cerium in this compound stays in the +4 state, which differs greatly from the more familiar +3 state. The nitrate ions—NO₃—bring their strong oxidizing nature into play. In practice, chemists reported this compound's unique optical and catalytic properties as far back as the early 1900s. These properties continue to be explored for new technologies, like improved imaging systems or greener chemical catalysts. As we edge toward more specialized tech, reliance on cleanly defined, pure chemicals goes up, not down.
Room for Improvement
Despite its utility, not all potassium cerium nitrate found at suppliers comes with the same level of purity. Sometimes moisture sneaks in, or the cerium isn’t all in the desired oxidation state. Better packaging and more attention at the factory could close this gap. Chemists should push for suppliers that offer detailed quality data with every shipment, so nobody flies blind.
Transparency in sourcing and clearer safety data would help folks using these chemicals know exactly what’s in their reagent bottle. Open data sharing between labs, including failed syntheses, should become the baseline. That way, every attempt to use potassium cerium nitrate brings reliable results, feeding into broader trust in scientific work.
| Names | |
| Preferred IUPAC name | Potassium cerium(IV) nitrate |
| Other names |
Cerous potassium nitrate Cerium(III) potassium nitrate Cerium(III) potassium dinitrate Potassium cerium(III) nitrate |
| Pronunciation | /pəˈtæsiəm ˈsɪəriəm ˈnaɪtreɪt/ |
| Identifiers | |
| CAS Number | 16774-21-3 |
| Beilstein Reference | 146157 |
| ChEBI | CHEBI:91240 |
| ChEMBL | CHEMBL3301577 |
| ChemSpider | 21567241 |
| DrugBank | DB11274 |
| ECHA InfoCard | ECHA InfoCard: 100.031.794 |
| EC Number | 233-733-4 |
| Gmelin Reference | Gmelin Reference: **158202** |
| KEGG | C18636 |
| MeSH | D020139 |
| PubChem CID | 24987784 |
| RTECS number | SC9810000 |
| UNII | Q41C8004QM |
| UN number | UN1477 |
| Properties | |
| Chemical formula | K₃Ce(NO₃)₆ |
| Molar mass | 548.22 g/mol |
| Appearance | White crystals |
| Odor | Odorless |
| Density | 3.5 g/cm³ |
| Solubility in water | Soluble |
| log P | -3.4 |
| Vapor pressure | Negligible |
| Magnetic susceptibility (χ) | +369.0e-6 cm³/mol |
| Refractive index (nD) | 2.20 |
| Dipole moment | 0 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 246.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1616.6 kJ/mol |
| Pharmacology | |
| ATC code | V03AB33 |
| Hazards | |
| Main hazards | Oxidizer, may intensify fire; harmful if swallowed; causes serious eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H319 |
| Precautionary statements | P210, P220, P221, P264, P273, P280, P370+P378, P501 |
| NFPA 704 (fire diamond) | 1-0-0-OX |
| Autoignition temperature | > 570 °C |
| Lethal dose or concentration | LD50 oral rat > 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): >2000 mg/kg (oral, rat) |
| PEL (Permissible) | No OSHA PEL established. |
| REL (Recommended) | 10 mg/m³ |
| Related compounds | |
| Related compounds |
Ammonium cerium(IV) nitrate Cerium(IV) sulfate Cerium(IV) chloride Potassium ferricyanide |

